2-Vinylpyridine: A Multifaceted Building Block in Modern Chemistry
Historical Development and Evolution
Tracing back to the mid-20th century, 2-vinylpyridine didn’t start out in a blaze of popularity. Chemists poked at the structure, wondering about potential beyond the classic aromatic. The initial draw centered on the unique electronic influence of the pyridine ring. Innovators during the synthetic polymer boom took a particular liking to vinylpyridines, seeking new monomers for robust copolymers. Scientists noticed its reactivity could lead to adhesives, specialty resins, and functional elastomers—often chasing performance other vinyl monomers just couldn’t deliver. By the 1970s, demand picked up, fueling large-scale processes and pushing suppliers to refine purification techniques. The journey since then has focused on tuning the compound for high-purity, keeping unwanted isomers and polymerization under control.
Product Overview
At a glance, 2-vinylpyridine looks modest, but researchers know there’s quite a range behind each drum. As a colorless to pale yellow liquid, the compound acts as a monomer and as a precursor for more intricate molecules. Specialty chemicals companies deliver it with assurances on purity: moisture and inhibitor content kept within strict limits. Production lots often carry a faint, characteristic aromatic odor—unmistakable in R&D labs. Most formulations land in volumes ranging from lab-scale bottles to large industrial totes. Glass containers with inert atmospheres guard against unwanted polymerization, especially for stock intended for high-value pharmaceutical or polymer work.
Physical & Chemical Properties
2-Vinylpyridine holds a molecular formula of C7H7N and tips the scale at 105.14 g/mol. It boils around 159 °C and freezes near −69 °C, reflecting its stable bond system and compact structure. It dissolves in most polar organic solvents, offering flexibility for formulation chemists. Its refractive index (about 1.544 - 1.547) and density (~0.98 g/cm3 at 20 °C) often come in handy for quick quality checks. The compound lights up the lab for another reason: it readily undergoes polymerization under radical initiation. Many folks working with this material know how easily it picks up moisture and oxygen, which can trigger unwanted side reactions. Mixed with acids, it forms salts, responding predictably, which helps during complex syntheses and product identifications.
Technical Specifications & Labeling
Quality matters here. Any bottle with “2-vinylpyridine” stamped on it usually comes marked with assay levels (upwards of 98.5%), water content under 0.1%, and a standard inhibitor (like tert-butylcatechol) kept between 100–200 ppm. Regulatory compliance flags sit on every shipment. Labels show proper shipping names according to international transport standards. Storage notes push for low temperatures and shielded from direct light, and suppliers add hazard pictograms covering flammability and acute toxicity. Many labs now require digital Material Safety Data Sheets with H-code and P-code labels for ease of integration with safety protocols.
Preparation Method
Industrial synthesis of 2-vinylpyridine continues to rely on the dehydrohalogenation of 2-(2-chloroethyl)pyridine, using strong bases like potassium hydroxide. Refined process steps minimize tars and reduce fouling by-products. Temperature control shapes the product yield, and vacuum distillation helps strip out residual starting material and stabilize the product. Some advanced routes tap into transition-metal catalysts, aiming to lower waste and boost atom economy. Research-grade samples sometimes run through an additional zone-refining step, providing even cleaner output for demanding applications.
Chemical Reactions & Modifications
The reactive double bond is where most chemists get creative. Free-radical polymerization gives rise to linear chains used in adhesives and coatings. The pyridine nitrogen stands ready for quaternization, allowing the molecule to turn into a cationic polymer or serve as a ligand for metal complexes. Sulfonation, halogenation, and oxidation open up access to a slew of derivatives, each with tailored properties. Cross-linking with butadiene or styrene forms tough copolymers for rubber compounds. Analytical chemists often use 2-vinylpyridine in modification strategies to introduce basic nitrogen atoms into polymer matrices, giving these materials new reactivity or tailored solubility.
Synonyms & Product Names
Over the years, industry and academia have volleyed around several alternative names. “2-Ethenylpyridine” pops up in some catalogs, matching IUPAC-conformant styles. Others stick to “o-vinylpyridine” or “alpha-vinylpyridine,” emphasizing the position of the vinyl group for clarity. Trade names vary according to supplier and region, with some vendors listing it under branded monomer series for use in specialty adhesives, coatings, or functional polymers. Each alias ties back to the same seven-carbon ring, though labeling quirks can confuse new entrants looking to compare sources.
Safety & Operational Standards
Nobody in the lab wants to cut corners with 2-vinylpyridine. Direct skin contact leads to irritation, and inhalation brings risk of respiratory discomfort. Standards developed by OSHA and international bodies require proper PPE: goggles, gloves, and fume hoods count as the norm. Storage areas stay cool and dry, kept far from oxidizers or acids. Many facilities run leak detectors, since the vapor carries occupational exposure limits. Spill clean-up kits wait on hand, paired with training for first aid and containment. Bulk handlers put measures in place to ground and bond all equipment, avoiding static sparks that could trigger a fire.
Application Area
Rubber chemists reach for 2-vinylpyridine to make adhesives that stick to tire cords—no other monomer brings both flexibility and bond strength at this level. Water treatment processes benefit from polymers that trap heavy metals, thanks to the basic nitrogen in the pyridine ring. Several pharmaceutical researchers pursue it for drug intermediates, using the compound’s nucleophilicity to craft molecules that withstand acidic or oxidative conditions. Electronics manufacturing leans on specialty resins for circuit encapsulation. Analytical chemists and surface scientists use derivatives to tailor sensor coatings, driving performance in detection and quantitation.
Research & Development
Academic labs and corporate R&D arms rarely pass up the chance to tweak 2-vinylpyridine derivatives. Development cycles stretch out as teams tune reactivity and search for greener, less wasteful synthetic routes. Computational chemists model the electron density of the pyridine ring, looking to predict new types of reactivity. Startups invest in novel polymer-blend technologies, aiming to upgrade coatings for extreme environments or selective membranes for resource recovery. The growing field of controlled/living polymerization brings out new block structures and turns 2-vinylpyridine into a building block for macromolecular engineering. Whenever teams aim for controlled release or complex functional interfaces, this monomer finds a place in the toolkit.
Toxicity Research
Toxicology studies keep scientists on their toes. Animal data reveal moderate acute toxicity, particularly after ingestion or inhalation. Chronic exposure led to liver and kidney changes in laboratory animals, sparking attention from regulatory agencies. Human data remain limited, but the material’s irritant potential means routine exposure can’t be overlooked. Laboratory testing continues to probe long-term risks, studying metabolic pathways and breakdown products for any carcinogenic or mutagenic hazards. Environmental scientists keep an eye on the fate of spills, given its moderate persistence and potential aquatic toxicity. All this feeds back into training, safe handling protocols, and workplace exposure limits.
Future Prospects
Chemists keep their eyes open for new opportunities with 2-vinylpyridine. Sustainable chemistry efforts nudge producers toward green synthesis and renewable feedstocks. Once-niche applications in smart polymers or medical delivery systems receive more funding as related technologies grow. Regulators push industry toward safer substitutes or improved hazard labeling, nudging both innovation and scrutiny. As demand for tailored electronics, advanced adhesives, and resource-efficient production rises, this monomer stands ready to bridge old-school performance with new-age challenges. Research teams experiment with bio-based reactions and recyclable derivatives while process engineers adapt plants to deliver higher yields with lower emissions. The compound’s long track record keeps it anchored in portfolios, but demand for cleaner, smarter chemistry ensures constant revision and rethinking.
What are the main applications of 2-Vinylpyridine?
2-Vinylpyridine: The Unsung Ingredient Behind Durable Materials
Step into any tire shop, walk down the aisle of home improvement stores, or glance at the labels of adhesives in a hardware store, and you brush shoulders with the handiwork of 2-vinylpyridine. This chemical rarely gets a headline, yet it holds down some of the toughest jobs in industry—jobs that touch daily life and global innovation alike.
Tires That Stick and Last
Synthetic rubber stands at the heart of modern tires. For car owners, the expectation today is miles of reliability and better grip on rainy days. 2-vinylpyridine plays its part in tire performance by helping form special latex copolymers. These copolymers work not as fillers, but as bridges between steel cords and the rubber matrix in radial tires. Without this chemical link, wires would pull loose over thousands of miles, leading to weaker tires and possible safety hazards. Tire recalls and accidents involving delamination have been traced to faulty bonding layers. The inclusion of 2-vinylpyridine reduces that risk and gives confidence to both manufacturers and drivers.
Modern Adhesives: Strength in Everyday Items
Open a tube of industrial-grade glue or check the formula for high-strength construction adhesives, and chemists have likely turned to 2-vinylpyridine at some step. This compound’s reactivity offers more bite, especially in situations where surfaces resist standard adhesives. Some multi-component glues use it to improve bonding between metals and plastics, which is essential in automobile assembly lines, electronics, and even some packaging processes. In my experience managing repairs in a busy fabrication shop, adhesives with these copolymers outperform basic glues for tough jobs like bonding dissimilar surfaces.
Specialty Coatings and Printing Inks
2-vinylpyridine steps beyond tires and into the world of specialty coatings. Take a look at printed electronics, magnetic tapes, and even some paints that promise better adhesion or resistance to corrosion. Copolymers using this molecule help coatings stick to tough surfaces, resist peeling, and keep crucial electronics safe from moisture. In printing inks, this character improves how pigments anchor to synthetic films like those used in food packaging, which is critical for legibility and durability.
Boosting Drug Manufacturing and Diagnostics
Away from the production floor, labs use 2-vinylpyridine for a different reason. In pharmaceuticals and biotechnology, it plays a role in resin manufacturing to help purify drugs or capture specific proteins. Diagnostics companies have also relied on polymers built from this molecule to coat plates and beads for analyzing blood or detecting infections. The result isn’t seen by the average patient, but fast, accurate testing often rests on advances in how these reagents are made.
Risks, Facts, and a Path Forward
Few chemicals act as versatile as 2-vinylpyridine, but it doesn’t come without risk. The compound can cause irritation or health effects during handling, so safety measures matter—protective gear, proper ventilation, and strict process controls create safer workplaces. Workers and community advocates have pushed for better monitoring, and both the US EPA and ECHA oversee its use to keep emissions and exposure within safe limits. As demand rises for safer, longer-lasting products, research into greener synthesis routes and lower-impact alternatives gains steam.
The world might not praise 2-vinylpyridine by name, but its role in delivering strong bonds, cleaner diagnostics, and longer-lasting products shows why this little molecule carries big weight in the background of modern manufacturing and science.
What are the storage and handling requirements for 2-Vinylpyridine?
Why Storage Rules Matter
2-Vinylpyridine isn’t just another chemical on the shelf. Years spent working around labs and warehouses have shown how the smallest slip in handling can trigger headaches, lawsuits, or worse. This chemical brings some real hazards to the table. Leaks or contact come with health risks, and the damage doesn’t stop there—poorly stored vinylpyridine breaks down, turning assets into liabilities. Workers can’t afford to take shortcuts and facilities can’t rely on good luck.
Common-Sense Steps for Handling
Anyone familiar with industrial environments has seen the cost of ignoring chemical management. Liquid 2-vinylpyridine reacts badly with the air’s moisture and oxygen, so let those bottles or drums breathe, and they’ll fight back. I’ve handled containers with swollen tops after a single misstep. Mistakes here have a habit of becoming ugly fast—no one likes dealing with an angry fume hood.
Every batch comes with a label to warn about its flammability, tendency to polymerize, and noxious effects. Once you get that warning, sturdy gloves, goggles, and solid ventilation aren’t optional. Fumes sting eyes and lungs, even from a minor spill. In my first year on the job, someone’s leaky funnel turned into an ER trip—no one forgot that lesson about face shields or using the right respirator again.
Don’t Cut Corners With Storage
Space matters. Dark, cool storage stretches the shelf life and keeps danger in check. Direct sunlight or warm rooms invite breakdown and pressure buildup. A steel or lined drum, tightly sealed, take care of most exposures. Friends in logistics often remind me: “You get what you pay for with cheap containers.” Investing in proper drums and regular inspections stops issues before they start.
Some older plants stretch their luck by letting up on maintenance or by storing reactive chemicals in mixed company. One careless neighbor, like a strong acid or oxidizer, and you’re courting a fire. I’ve worked in facilities where locked cages and segregated cabinets for vinylpyridine paid off, especially during audits and spot checks. Install alarms for leaks and ventilate storage rooms, or pay the price in lost chemicals and ruined reputations.
Keep a Paper Trail
Every movement should wind up in the logbook. I once saw a factory nearly lose an insurance payout because shipment dates weren’t tracked. Shelf-life monitoring and FIFO rotation keep expired stock from piling up. Safety culture grows from habits, not rules. Regular briefings on spill response, evacuation, and first-aid make a difference.
What Works—And What Could Work Better
Smart tech has moved the needle. Sensors that check for temperature spikes or VOCs give advance warning. Digital inventory and tracking help spot anything gone missing or close to expiry. Still, some plants rely too much on “the way we’ve always done it,” and that’s when things slip. Updates to regulations or fresh hazard training sessions shake off complacency and keep everyone sharp.
Safe storage and solid procedures aren’t just boxes for regulators to tick. They protect workers, investments, and the wider community. In all the places I’ve worked, teams that took storage and handling seriously avoided incidents—and kept their good names, too.
Is 2-Vinylpyridine hazardous, and what safety precautions should be taken?
Hazards of 2-Vinylpyridine
Many people working in labs or industrial settings run into chemicals that could cause real problems if handled without thought. 2-Vinylpyridine stands out on safety data sheets for a reason. According to its Material Safety Data Sheet (MSDS), 2-vinylpyridine can cause harm through inhalation, skin contact, or eye exposure. It doesn’t take long in a lab before you hear stories of someone getting burned or developing irritation because a glove or mask was skipped one day. This compound can irritate the respiratory tract, eyes, and skin. Extended exposure may even mean greater risks—such as sensitization or organ toxicity.
Studies from occupational health researchers and government organizations support the need for caution. For instance, the National Center for Biotechnology Information notes that 2-vinylpyridine carries a “toxic” label for a reason. At high concentrations, it can affect the nervous system and damage the liver or kidneys. Breathing in vapors over time simply isn’t something to brush off as “just part of the job.” Some people find that sensitivity builds, turning occasional headaches or dizziness into legitimate medical concerns.
Why Caution Matters
Years of hands-on lab work taught me that complacency breeds accidents. Even experienced chemists make mistakes in the rush to finish an experiment or keep up with production demands. Just thinking that “it won’t happen to me” doesn’t offer any protection. Failing to keep good habits—like swapping out gloves or using a fume hood—even for one task, can cost dearly. I’ve seen otherwise meticulous colleagues end up seeking medical attention after underestimating chemical fumes or splashes.
OSHA and similar agencies worldwide require clear labeling and regular training, but the real learning often happens from seeing up close what can go wrong. Hospital admissions and near-misses underline why chemical companies implement strict protocols. Toxic effects rarely show up instantly. Sometimes it’s repeated exposure that leads to bigger problems—or trouble only appears much later in life.
Personal Safety Precautions
The right gear and habits make all the difference. Anyone working with 2-vinylpyridine should wear a fitted lab coat, chemical-resistant gloves, and splash-proof goggles. Use of a face shield becomes essential if there’s any chance of a splash or spill.
Good ventilation ranks high on the priority list. Never rely on open windows or just cracking a door. A dedicated chemical fume hood traps vapors before workers inhale anything dangerous. Many labs also install continuous air monitors where volatile compounds get used often.
Spill kits and eyewash stations need to be close to where the chemical gets handled, not sitting in storage down the hall. All personnel, not just chemists or engineers, should know emergency procedures and how to use safety equipment.
Proper storage saves headaches, too. 2-vinylpyridine should stay in tightly sealed containers, stored away from strong oxidizers and sources of heat or ignition. Failing to control temperature and ventilation raises the risk of harmful reactions or even explosions.
Building a Culture of Safety
Conversations about safety work best face-to-face, sharing lessons learned from real experiences. Checking a box on safety training doesn’t protect anyone if the lessons stay theoretical. Look to promote a workplace where anyone can speak up about unsafe conditions without fear. Supervisors who model correct PPE and encourage slow, careful work lead by example; respect, not just rules, keeps everyone safer in the long run.
Planning ahead, double-checking storage and labeling, and staying alert make a world of difference. For communities relying on chemical work, that kind of vigilance and shared responsibility keeps people healthy, productive, and pain-free far into their careers.
What is the chemical structure and molecular weight of 2-Vinylpyridine?
Getting to Know 2-Vinylpyridine
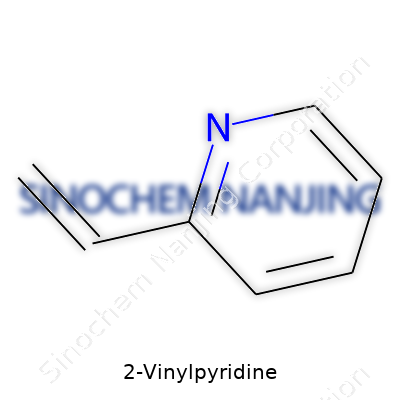
Every field has its unsung workhorses, and in chemistry, 2-vinylpyridine quietly fills an important role in research and industry. The structure of this molecule tells much about how it behaves. Picture the familiar ring of a pyridine, which comes from its six-membered ring containing five carbon atoms and one nitrogen. At the second carbon position, a vinyl group—a little two-carbon chain with a double bond—branches off, ready to reach out and react. Chemically, that’s C7H7N.
The details behind the structure matter. The molecular formula lines up as seven carbons, seven hydrogens, and a single nitrogen. To folks who work with polymers or specialty chemicals, that formula has muscle. The structure is as follows: a vinyl group (–CH=CH2) attached to the second position of the pyridine ring. Not every molecule gives away so much of its character just by its setup, but this one does. The molecular weight comes to 105.14 g/mol. Remembering that figure makes it easier to get calculations right on those days when every milligram counts, and I’ve had more of those than I care to admit.
Applications and Lessons
Now, what does all this mean in the real world? In my lab days, I ran into 2-vinylpyridine while looking for better adhesion between rubber and metal. Local industries use this compound in the production of adhesives, coatings, and specialty polymers. It’s a backbone ingredient for making latex that actually sticks to metal—an under-the-hood hero for tires and engine parts. In the laboratory, 2-vinylpyridine opens doors for making functional materials with a built-in reactivity, thanks to both its aromatic system and its vinyl group, which allow for further chemical tweaks.
Research shares that 2-vinylpyridine makes copolymers with styrene and butadiene, improving oil resistance and extending product lifespans. So, those handling greasy parts or sealing fuel tanks can thank the ingenuity behind this molecule. I remember looking at the molecular structure during study nights, realizing not every innovation needs to be glamorous—some just keep trucks moving.
The Safety Side
Safety doesn’t take a back seat with 2-vinylpyridine. Nobody wants to risk skin contact or inhale its vapors. It has an odor strong enough to get your attention and, in concentrated form, causes eye and respiratory irritation. According to the National Institute for Occupational Safety & Health (NIOSH), strict endpoints exist above which exposure becomes too risky. I saw one colleague veer too close to an open container and leave with eyes stinging and sinus issues—those lessons don’t fade fast. Good ventilation, gloves, and goggles were the minimum. Respect for the material meant the difference between a smooth day and a ruined shift.
Responsible Use and Future Possibilities
Responsible use means understanding both chemistry and context. At the manufacturing level, closed systems keep workers safe and reduce losses to the environment. Environmental impact is real for any compound, and advances are pushing for safer alternatives and better containment. For research labs, updated material safety protocols keep everyone on task and out of trouble. New technologies bring lighter, less hazardous alternatives, but the utility of 2-vinylpyridine lives on in many fields. The structure and weight might seem like dry details, yet they connect to practical outcomes: longer-lasting products, safer workplaces, and environmental responsibility. My experience says real value comes from these links between fundamental science and daily practice.
How should 2-Vinylpyridine be disposed of properly?
Understanding the Stakes
Ask anyone who’s spent time in a laboratory, especially one dealing with organic synthesis, and the reputation of 2-vinylpyridine quickly comes up. This stuff doesn’t just give off a funky odor—it’s also a headache for disposal. It’s a flammable liquid, can catch fire easily, carries toxicity risks, and spells trouble for water systems. The sort of mishandling that gets you regulatory visits, burned-out noses, or worse, damaged health and harmed ecosystems.
Risks Around the Corner
Pouring it down the drain might sound quick, but it’s about the worst thing you could do. Once in the water system, 2-vinylpyridine travels fast, binds to organic material and can enter the food chain. I know labs where a simple mistake led to a call from city waste authorities—a situation everyone wants to avoid. Fish and plant life pay the price for shortcuts like that, and so does your reputation.
Improper storage before disposal raises the risk even further. This liquid evaporates readily and those vapors catch light easily—meaning fire is a real possibility if someone leaves a bottle open or stores it near a heat source. I remember frustrating, smelly afternoons spent in overventilated fume hoods just to contain emissions.
How a Responsible Lab Handles It
Disposal starts the moment you open the bottle. I always kept a pre-labeled, tightly sealed waste container close, protected from direct sunlight and away from anything flammable. The container’s got to be compatible with 2-vinylpyridine—HDPE works well, but check for wear and keep it upright and undamaged. Label it specifically with the chemical’s full name; generic “solvent waste” won’t cut it.
Humidity control means a lot for 2-vinylpyridine. Water or acids kick off unwanted reactions, sometimes creating hazardous byproducts. Dry, cool storage in a chemical hood, or, even better, a ventilated flammable liquid cabinet, really lowers the risk. Every chemist in the lab should know the protocol—new interns especially benefit from a quick run-through before they ever touch the bottle.
The Legal Endgame
The EPA and local agencies view 2-vinylpyridine as hazardous waste under RCRA regulations. That places a duty on every user to ship it off to certified chemical waste handlers, and usually, no university or company gets exemptions. Skipping paperwork risks thousand-dollar fines and angry neighbors. I've spent hours completing manifests and arranging pickups, but every bit of that effort matters.
On-campus, or in midsized companies, the Environmental Health and Safety (EHS) team must be involved. Sure, it adds a step or two, but they handle communication with waste vendors who incinerate the material at special facilities. Incineration breaks down 2-vinylpyridine completely, stopping it from entering air or water. Incinerators reach the right temperatures, and trained operators make sure emissions are scrubbed and filtered.
Room for Improvement
Still, some places struggle with inconsistent training or skipped steps. Mandatory yearly refreshers, combined with unannounced safety checks, tend to raise the bar. Sharing disposal data openly among labs helps catch mistakes early. I also advocate for rooting out excess—don’t over-order, don’t let half-empty bottles linger on forgotten shelves, and promptly get rid of leftovers after projects wrap.
Safer substitutes can reduce risks further. In a few projects, using less toxic alternatives worked well; sometimes small process tweaks cut down overall use. Waste minimization strategies, like micro-scale reactions, add another layer of protection and save money. Experience shows real progress comes from both personal diligence and teamwork, backed by strict EHS support.
| Names | |
| Preferred IUPAC name | 3-Ethenylpyridine |
| Other names |
2-Vinylpyridine 2-Ethenylpyridine 2-Vinylpyridin 2-Vinyl-pyridine 2-Pyridylethylene |
| Pronunciation | /tuː vaɪˌnɪl pɪˈrɪdiːn/ |
| Identifiers | |
| CAS Number | 100-69-6 |
| Beilstein Reference | **605120** |
| ChEBI | CHEBI:52095 |
| ChEMBL | CHEMBL1085 |
| ChemSpider | 5377 |
| DrugBank | DB02852 |
| ECHA InfoCard | 100.057.837 |
| EC Number | 202-804-9 |
| Gmelin Reference | 6046 |
| KEGG | C02413 |
| MeSH | D014924 |
| PubChem CID | 7909 |
| RTECS number | UY0700000 |
| UNII | W8W8CQQ43H |
| UN number | UN3292 |
| Properties | |
| Chemical formula | C7H7N |
| Molar mass | 105.14 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | amine-like |
| Density | 0.975 g/mL at 25 °C(lit.) |
| Solubility in water | soluble |
| log P | 1.61 |
| Vapor pressure | 0.8 mmHg (20 °C) |
| Acidity (pKa) | 5.95 |
| Basicity (pKb) | 4.77 |
| Magnetic susceptibility (χ) | -58.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.510 |
| Viscosity | 1.06 mPa·s (25 °C) |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 105.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3274 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P403+P233 |
| NFPA 704 (fire diamond) | 3-2-2-Δ |
| Flash point | 68 °C |
| Autoignition temperature | 455 °C (851 °F; 728 K) |
| Explosive limits | 1.2–8.6% |
| Lethal dose or concentration | LD50 oral rat 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 200 mg/kg |
| NIOSH | NIOSH: UU4025000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2-Vinylpyridine is "1 ppm (4 mg/m³) TWA". |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
2-Ethylpyridine 2-Methylpyridine 3-Vinylpyridine 4-Vinylpyridine |

