2-Trifluoromethylaniline: Unveiling its Place in Modern Chemistry
Historical Development
Chemists first realized the unique potential of fluorinated aromatics in the middle of the twentieth century, exploring their stability and reactivity drives. 2-Trifluoromethylaniline came into focus as researchers sought to fine-tune anilines for pharmaceutical and agrochemical uses. The trifluoromethyl group, with its electron-withdrawing nature, caught the interest of synthetic chemists who were looking to break the mold of traditional aniline chemistry. Over time, improvements in fluorination techniques and aromatic substitution pathways let labs produce 2-Trifluoromethylaniline in ways that would have been costly or impractical decades ago.
Product Overview
2-Trifluoromethylaniline stands apart from more common anilines because of its robust chemical characteristics. The presence of a trifluoromethyl group at the ortho-position shifts key properties compared to unsubstituted aniline. This molecular change gives it new reactivity, impacting everything from basic synthesis to its impact in chemical manufacturing. The compound belongs to the wide family of fluorinated aromatic amines—a niche, but one that continues growing as industries pursue molecules that blend stability, reactivity, and selectivity for demanding environments.
Physical & Chemical Properties
This aromatic amine brings with it a distinct physical and chemical footprint. The trifluoromethyl group adds bulk and increases hydrophobicity, while also making the compound less likely to participate in certain types of oxidation reactions. In practical terms, this translates to lower basicity than simple aniline, and oftentimes a higher boiling point due to fluorine’s strong effects. Synthetically, the electron-withdrawing character of the trifluoromethyl group can make substitutions on the aromatic ring less straightforward but opens new doors for selective transformations.
Technical Specifications & Labeling
Buyers and lab managers who handle 2-Trifluoromethylaniline know that clarity on technical details is vital. Researchers can’t afford ambiguity about purity, identification, and storage conditions. Standard practice involves clear labeling with the molecular formula and recognized hazard warnings, as mishandling such organofluorine species can introduce real risks. Clear communication in labeling also ensures that researchers down the road can track and compare outcomes based on precise batch information.
Preparation Method
Synthesis of 2-Trifluoromethylaniline often relies on seasoned aromatic chemistry. Early routes might have centered on direct substitution, but these days many labs choose targeted strategies for position-selective introduction of the trifluoromethyl group. For example, Sandmeyer-type reactions enable reliable amination, and nucleophilic substitutions often serve to install the necessary groups on pre-fluorinated aromatic structures. The evolution in preparation reflects broader trends: a push towards efficiency, controllability, and reduced hazardous waste.
Chemical Reactions & Modifications
Anyone who works with this compound quickly learns that its trifluoromethyl substituent influences nearly every reaction. Electrophilic aromatic substitution usually faces greater resistance due to the electron-withdrawing effect, which makes the ring less reactive at positions near the fluorine group. But for chemists seeking selective reactivity or tuning for downstream coupling reactions, this property presents an appealing puzzle. N-acylations, halogenations, and even cross-coupling using Suzuki or Buchwald–Hartwig approaches show distinct patterns versus non-fluorinated analogs. It’s a playground for synthetic specialists trying to teach old reactions new tricks.
Synonyms & Product Names
Looking for 2-Trifluoromethylaniline? Anyone in a chemical library or supplier database will also encounter synonyms like o-(Trifluoromethyl)aniline and 2-Amino-α,α,α-trifluorotoluene. These alternate names recall the compound’s history of classification and highlight conventions in chemical nomenclature. Such diversity in naming calls for vigilance—it’s easy for a mislabeling to lead to costly errors or safety missteps when swapping between suppliers or reference materials.
Safety & Operational Standards
Safety can never play second fiddle in any lab handling fluorinated amines. 2-Trifluoromethylaniline, like its analogs, can present inhalation and dermal risks, and presents the sort of hazards that show up long after the experiment wraps up—the lingering residues, the volatile emissions, and the potential for environmental persistence. Researchers need to lean on established personal protective equipment protocols, fume hoods, and proper disposal methods. With regulatory landscapes tightening worldwide, compliance is no longer just a box to tick—it’s the minimum expected for responsible discovery and production.
Application Area
Industry demand for trifluoromethylanilines bubbles up from pharmaceutical discovery, agrochemicals, and increasingly from materials science. Medicinal chemists keep turning to these fluorinated amines to build next-generation molecules that dodge rapid metabolic degradation. In pesticides and fungicides, their stability in the field and resistance to breakdown offers longer action windows. Polymers and specialty coatings also benefit, with finished products resisting harsh solvents or thermal extremes due to the trifluoromethyl influence.
Research & Development
The world of drug discovery holds a special fascination for this compound. Building on decades of evidence, researchers have shown time and again that strategic use of fluorine shifts bioavailability, metabolic stability, and sometimes even receptor selectivity for drugs. Chemists tinker with the basic aniline skeleton, adding or swapping functional groups based on hits or leads from early screens. With the global focus on efficient and sustainable syntheses, novel catalytic pathways and greener solvent systems keep getting the spotlight. Labs aim to cut down on toxic byproducts or excessive solvent use—a sensible approach from both a business and sustainability view.
Toxicity Research
Toxicology reviews for fluorinated aromatics like 2-Trifluoromethylaniline build on a mix of experience, direct study, and lessons learned from structurally similar compounds. Direct exposure may provoke classic aromatic amine effects on skin, eyes, and airways. Longer-term questions revolve around organofluorine impacts, because such groups often lead to persistent, sometimes bioaccumulative, environmental residues. This ingredient sits on the regulatory radar for exactly these reasons, though definitive conclusions await larger population studies. The research challenge sits in balancing knowledge from animal studies and in vitro screens with the real-world use of the compound in tightly-regulated facilities.
Future Prospects
Chemists and policy-makers face a crossroads as global demand for customized building blocks in pharmaceuticals and materials only increases. Technical challenges—like greener synthesis and tighter process controls—keep driving innovation in handling and applying compounds such as 2-Trifluoromethylaniline. The promise of new drug candidates, combined with regulatory pushback on hazardous waste and emissions, sets the stage for collaboration between industry, academia, and regulatory agencies. Continued investment in research promises to unlock next-gen synthetic methods, while digital tools such as AI-driven reaction optimization may soon change how chemists approach even staple intermediates. As a result, such compounds feel less like obscure specialty chemicals and more like vital contributors to modern industry’s need for precision, efficiency, and responsibility.
What is 2-Trifluoromethylaniline used for?
A Look Into Its Role In The Lab
2-Trifluoromethylaniline stands out for chemists who focus on making new drugs and developing advanced materials. This molecule doesn’t jump out at you on paper, but anybody who’s spent hours hunched over fume hoods knows certain building blocks really unlock new tricks. This compound—an aromatic amine with a trifluoromethyl group—finds its way into many synthetic routes that shape what ends up in clinical studies, specialty polymers, and next-gen dyes.
Molecular Building Blocks in Pharmaceuticals
Start with drug discovery. Medicinal chemists look for ways to tweak lead compounds so they interact more strongly or precisely with biological targets. The trifluoromethyl group on this aniline changes both the chemical’s shape and how it behaves in living systems. Adding fluorine can make a molecule stick around in the body longer or dodge metabolic breakdown. Several modern cancer therapies, anti-inflammatories, and drugs that treat central nervous system disorders trace some part of their design thinking back to swapping in groups like this one.
Many times, a researcher picks 2-trifluoromethylaniline as a starting piece in so-called “library synthesis,” where dozens or even hundreds of candidate molecules spring up from a common scaffold. By changing the position or number of fluorine atoms, labs can quickly see how these swaps affect potential treatments. The goal isn’t just to make something novel. It’s to find new medicines that actually help people without causing unexpected complications in the bloodstream or organs.
Powering Modern Agrochemistry
Chemists on the industrial side work with 2-trifluoromethylaniline for making new agrochemicals. The fluorinated side chain can help block pests or fungi from breaking down key ingredients in crop protection products. This means farmers can use less spray on their fields, or get better results over longer growing seasons. A big concern in agriculture involves environmental buildup, so the balance between durability and safety stays under constant review. Researchers have to run many rounds of environmental impact testing and field evaluation before anything makes it out of the lab and onto the market.
Specialty Materials and Color Science
The reach for fluorinated anilines doesn’t stop at healthcare and food. These ingredients show up in niche coatings and dyes too. Adding trifluoromethyl groups changes how a material resists heat, ultraviolet radiation, or chemical corrosion. Some fabric dye makers use this molecule to get brighter, longer-lasting colors that don’t fade or wash out as quickly. Electronics and display designers want coatings that resist fingerprints, water, and solvents. Engineered molecules derived from 2-trifluoromethylaniline help meet those demands.
Safety and Research Mindset
Any chemical this reactive demands respect. Handling protocols keep lab staff and factory workers protected from possible toxicity or irritation. Researchers use validated safety gear, extractors, and disposal routes. Green chemistry methods push teams to explore alternatives that work just as well without causing additional hazards or pollution. Each shift in regulation or public demand nudges scientists to rethink old formulas and test new ways to achieve the same results with lower risk.
Steering Toward Smarter Chemistry
From the lab bench to industrial reactors, 2-trifluoromethylaniline helps innovators solve real-world problems. Sharper medicines, safer foods, and more durable everyday materials all benefit from the science that happens around this molecule. That sense of purpose keeps researchers motivated: chasing better answers, stronger evidence, and smart ways to move discoveries from theory into something everyone can use safely.
What is the molecular formula of 2-Trifluoromethylaniline?
Understanding the Structure
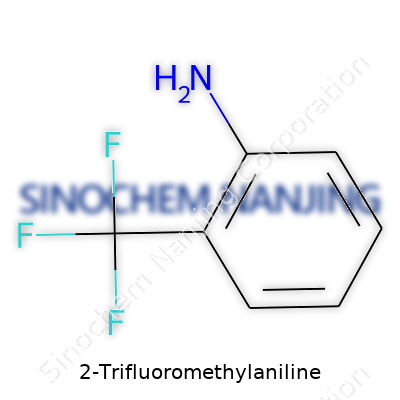
Talking chemistry may not spark excitement every day, but pinpointing the molecular formula for compounds like 2-Trifluoromethylaniline matters beyond laboratories or classrooms. In the case of 2-Trifluoromethylaniline, the name alone gives hints. The "aniline" part means we’re starting with a benzene ring that carries an amino group (NH2), and "2-trifluoromethyl" refers to a trifluoromethyl group (CF3) attached to the second position on that ring. With this in mind, we're looking at a molecule built of carbon, hydrogen, nitrogen, and fluorine atoms. After counting everything, the actual molecular formula stands as C7H6F3N.
Why Pinpointing Formulas Matters
No one likes confusion, especially in chemistry labs or industries where accuracy saves time, money, and even lives. Chemical research won’t function without precise molecular formulas. Having a solid answer for what sits inside a flask or beaker can dictate what’s safe, what might cause danger, and which next step works best.
Anyone building pharmaceuticals, agrochemicals, or tweaking specialty materials checks the basics before going deeper. Companies face strict regulations. Even a minor mislabeling of a chemical can shut down a lab or pull products off shelves. The molecular formula serves as the first checkpoint along the long road from research to real-world use.
Personal Experience: Seeing the Risks
Years ago, I worked in a lab where one batch of a chemical was off by just a fluorine atom. What seemed like a tiny detail led to a failed reaction, wasted days, and a big bill for disposal. I learned right then: the devil is in the details. Any small error in a molecular formula can derail entire projects, or even spark health and safety issues others might never see coming until it’s too late.
Health and Environmental Impact
Chemicals with trifluoromethyl groups, like this compound, present their own set of challenges. Fluorine atoms show up strong; they tweak how molecules behave inside living things and the environment. Some fluorinated compounds stick around for years, raising concerns over long-term exposure. Regulators in the US and Europe have raised flags about persistent fluorinated chemicals turning up in soil and water supplies, pushing scientists and companies to track formulas with eagle eyes.
Solutions and Better Practice
Chemists turn to reliable databases, careful lab notebooks, and cross-checking with trusted sources before calling a formula finished. Open databases—like PubChem and ChemSpider—back up textbook knowledge and bring in voices from across the globe. Companies build strict verification steps into routine work. Education programs don’t just push rote memorization; they work through drawing molecules, building models, and tracking the tiniest change in structure.
On a larger scale, international guidelines set by groups like IUPAC standardize names, structures, and formulas so anyone, anywhere, can pick up a bottle and know exactly what’s inside. That kind of transparency not only powers good science, it helps protect everyday people.
The Takeaway
The formula C7H6F3N describes more than a handful of atoms—it holds the key to safety, innovation, and trust in many fields. From firsthand lab mishaps to global environmental debates, having the correct information shapes not just experiments, but lives and futures.
What are the safety precautions for handling 2-Trifluoromethylaniline?
Understanding the Hazards
Working in a lab or plant brings all sorts of chemicals, some easier to handle than others. 2-Trifluoromethylaniline comes with its own set of risks—splashes, fumes, and accidental spills. Its strong amine odor signals that exposure can strike through inhalation or direct skin contact. I remember one late shift when a careless pour left a minor chemical burn reminding me that even small mistakes can end painfully.
Personal Protective Gear
Any time I step near substances like this, I reach for a pair of nitrile gloves, long-sleeve lab coats, and tight, splash-proof goggles. Cotton and latex won’t cut it; the small molecules in 2-Trifluoromethylaniline break right through them, sometimes without warning. For anyone with sensitive skin, double-gloving cuts down the chance of accidental absorption. Closed-toe shoes keep drips off my feet. Eye protection gets overlooked but cleaning up a spill without goggles can land you in an emergency room with a chemical splash in your eye, something no one wants to face.
Air and Ventilation
Good airflow goes a long way. Inhaling vapors risks headaches, dizziness, and respiratory irritation—so a chemical fume hood should always get used, even when handling what seems like “just a little.” Some labs try to rely on open windows or basic fans, but these don’t trap hazardous vapors. On hot days in the lab, fumes hang lower, making ventilation even more important. Years back, a drafty side window convinced a coworker they were safe, but it really just spread vapors across the room. Airflow should carry it out and away, not just circulate it around your head.
Storage and Labeling
Storing chemicals the right way prevents accidents down the line. I keep 2-Trifluoromethylaniline containers tightly sealed, away from heat sources, sunlight, and incompatible chemicals like acids or strong oxidizers. The label details hazard warnings, date received, and concentration—no shortcuts. Storing bottles at bench height keeps them out of reach of accidental kicks or drops, and spill trays underneath make cleanup faster if something tips over. Old, corroded containers often leak even if they look intact from the outside, so regular checks find problems before anything gets out.
Dealing with Spills and Waste
Accidents happen even with layers of protection. I always keep absorbent pads, neutralizing powders, and disposable cleanup materials nearby. Hydrochloric acid or bleach should never clean this compound—mixing them creates dangerous byproducts. After a spill, I double-bag all waste, label it, and put it in the proper hazardous waste bin. Pouring this stuff down the drain damages pipes and harms water systems downstream. A few years ago, local fish kills near a university traced back to improper disposal of aromatic amines—another harsh reminder to stay responsible.
Training and Vigilance
Even a careful scientist can get sloppy if distracted or rushed. Running drills on emergency showers and eyewash stations cements what to do in a crisis. Good habits include not working alone, cleaning surfaces often, and following standard operating procedures every single time. Solid training and honest communication between team members help root out bad practices before someone gets hurt.
Room for Improvement
Better safety starts with culture, not just equipment. Spreading stories of near-misses and sharing tips helps everyone learn. Automation can help, but nothing replaces paying attention at every stage—from ordering to disposal. Everyone in the lab plays a role in keeping things safe around 2-Trifluoromethylaniline—no move is too small when your health is on the line.
What is the boiling point of 2-Trifluoromethylaniline?
Understanding a Key Physical Property
2-Trifluoromethylaniline isn’t exactly a household name, but it shows up in research labs more often than you’d think, especially in the world of organic synthesis and pharmaceutical development. For the folks working with this compound, one of the first things needed is a clear idea of its boiling point. It clocks in at roughly 183 to 185 degrees Celsius, depending on lab purity and atmospheric pressure. This number might feel a bit dry, but try running a large-scale reaction without it. The difference between a controlled distillation and a chemical mess often comes down to knowing when the pot will boil.
Real-World Impact
Boiling point isn’t just trivia. It’s a safeguard for researchers, guiding choices around the kind of glassware used, setting expectations during purification by distillation, and sometimes influencing which chemicals get picked for a project in the first place. Having firsthand experience handling aromatic amines, that boiling point means avoiding the headaches of pressure build-up, nasty odors, or leaks. Once I tried to use a cheaper distillation setup for a similarly volatile compound, but the lack of proper head control led straight to wasted sample and a room full of sharp odors that lingered until the next shift.
The trifluoromethyl group changes a lot about this molecule compared to plain aniline. It pushes up the boiling point, thanks to higher molecular weight and stronger intermolecular interactions. Other compounds with similar backbone structures reflect this trend. That reminder helps chemists select the right temperatures for reactions and separations.
Safety in the Lab
Anyone working at elevated temperatures needs to care about more than just reaching a number on the thermometer. At 183–185°C, safety protocols become non-negotiable. Regular exposure to high temperatures raises concerns about glassware failing or vapors escaping. An aromatic amine with a trifluoromethyl group adds to the toxicity profile, so solid fume extraction and chemical splash precautions keep everyone safe. Industry standards and published safety data hammer this point home, not out of bureaucracy, but hard-learned lessons from past accidents.
Turning to Quality Data
Reliable information about physical properties like boiling point should never come from a quick web search or unchecked datasheet. Trusted databases—like those maintained by the American Chemical Society or peer-reviewed journals—give numbers that have backing evidence and citations. I remember being misled by an out-of-date material safety sheet, costing an afternoon to correct an experiment. Since then, I always cross-reference at least two reputable sources before accepting a boiling point at face value.
What Chemists Can Do
Accurate data in the hands of careful workers means fewer mishaps. Students and young researchers should learn the value of verifying these numbers and understanding what they mean in a practical sense. If a lab needs to distill 2-Trifluoromethylaniline, invest in proper temperature control, double-check the apparatus seals, and set expectations upfront by reviewing the material’s boiling point and potential decomposition. Established chemists can pass down these habits and give new colleagues extra confidence at the bench.
Building Safer Workflows
No single physical property tells the whole story, but boiling point often acts as the first red flag for risk and technical challenge. Taking a few minutes to show others why this matters can prevent accidents and wasted time. It’s the difference between a smooth prep and a late-night lab cleanup duty—a lesson that needs repeating as new faces join the lab each semester.
Is 2-Trifluoromethylaniline soluble in water?
Understanding the Nature of 2-Trifluoromethylaniline
2-Trifluoromethylaniline belongs to a group of chemicals where an aniline ring meets a strongly electronegative trifluoromethyl group. Chemists and students alike recognize this structure for its unique balance of hydrophobic and hydrophilic forces. In practice, you often see trifluoromethyl groups added to tweak the behavior of molecules for pharmaceuticals or agricultural chemicals. The strong pull of the trifluoromethyl group often means the whole molecule leans away from water, shifting properties toward oil-like tendencies.
Looking at Solubility in Real Life
Many expect organic compounds with just a touch of polarity to mix into water. After all, regular aniline will dissolve a little—about 3.6 grams in 100 milliliters of water at room temperature. Toss a trifluoromethyl group onto the aromatic ring, and things change. This group is bulky, packed with fluorines, and strongly resists the cast of water molecules trying to pull the compound into solution. The result? 2-Trifluoromethylaniline shows very low water solubility.
Those working in the lab often find this firsthand. The compound will hover and separate, forming a distinct layer instead of mixing in. Tech sheets from chemical suppliers list it as sparingly soluble or practically insoluble in water. This matches personal observations: try adding a dash to a beaker of water and give it a stir. You’ll see beads of the chemical or perhaps a thin film, but no homogenous solution.
Why Water Solubility Matters
People outside chemistry might wonder why this matters. Everyday safety and environmental issues sit at the core. Poorly soluble compounds move more slowly in aquatic environments. If an incident releases 2-trifluoromethylaniline into groundwater, it won’t spread effortlessly. Yet “not dissolving” doesn’t mean “not dangerous.” Even low concentrations in water can matter for toxicity—both to humans and aquatic life. Studies of similar substances point out that organic molecules with trifluoromethyl attachments often carry persistence, bioaccumulate, and resist breakdown.
In the lab, water solubility impacts how you handle, clean up, and dispose of the substance. Workers need reliable information so they can avoid mistakes, like flushing unused chemicals down the drain or assuming easy filtration will catch everything. Researchers often choose organic solvents, such as ether or chloroform, for dissolving or extracting 2-trifluoromethylaniline. Anyone organizing chemical storage must group such compounds away from water-reactive or water-soluble materials, limiting cross-contamination risks.
Safer Handling and Future Choices
This leads to broader questions about safer substitutes and greener chemistry. When designing chemicals for new medicines or crop treatments, considering water solubility early on can help sidestep costly missteps. Tools like quantitative structure–activity relationship models allow chemists to estimate solubility from the drawing board, narrowing down candidates before making a batch in real life. Supporting responsible research and development—and learning from past handling of persistent pollutants—drives progress toward safer, more manageable molecules.
For now, knowing that 2-trifluoromethylaniline avoids dissolving in water gives a starting point for better decisions on its use, disposal, and environmental risk. This blend of hands-on experience and tested scientific insight supports safer practices wherever this molecule shows up.
| Names | |
| Preferred IUPAC name | 2-(Trifluoromethyl)aniline |
| Other names |
2-(Trifluoromethyl)aniline o-(Trifluoromethyl)aniline 2-Aminobenzotrifluoride o-Aminobenzotrifluoride 2-Trifluoromethylbenzenamine |
| Pronunciation | /tuː-traɪˌfluːəroʊˌmɛθɪlˈænɪliːn/ |
| Identifiers | |
| CAS Number | 2955-76-0 |
| Beilstein Reference | 85934 |
| ChEBI | CHEBI:131650 |
| ChEMBL | CHEMBL14000 |
| ChemSpider | 56501 |
| DrugBank | DB08314 |
| ECHA InfoCard | 103249 |
| EC Number | 205-629-0 |
| Gmelin Reference | 8038 |
| KEGG | C14393 |
| MeSH | D065077 |
| PubChem CID | 69785 |
| RTECS number | BY5425000 |
| UNII | 0T0PKM4WQX |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C7H6F3N |
| Molar mass | 145.13 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Aromatic |
| Density | 1.326 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 1.97 |
| Vapor pressure | 1 mmHg (at 25 °C) |
| Acidity (pKa) | pKa = 3.72 |
| Basicity (pKb) | 10.03 |
| Magnetic susceptibility (χ) | -58.0 × 10^-6 cm³/mol |
| Refractive index (nD) | 1.4560 |
| Viscosity | 1.32 cP (25°C) |
| Dipole moment | 1.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −167.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1291.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Toxic to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H315, H319, H332 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P305+P351+P338, P312, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-2-0-☢ |
| Flash point | 72 °C |
| Autoignition temperature | 630°C |
| Explosive limits | Lower: 1.2%, Upper: 7.0% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1880 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1770 mg/kg (oral, rat) |
| NIOSH | NA4810000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Aniline 2-Nitroaniline 2-Chloroaniline 2-Bromoaniline 2-Iodoaniline 2-(Trifluoromethyl)nitrobenzene 3-Trifluoromethylaniline 4-Trifluoromethylaniline |

