2-Tert-Butyl-4,6-Dinitrophenol: A Deep Dive Into a Powerful Chemical
Historical Development
Historians of chemistry often find reference to dinitrophenol derivatives as far back as the late 19th century, when researchers first began searching for new energy sources and dyes. As industry boomed during the world wars, curiosity about energy metabolism led to exploration of many nitrophenol derivatives, including the elusive 2-tert-Butyl-4,6-Dinitrophenol (commonly called DNBP or Dinoseb with variant names). Early findings sparked both excitement and concern, since its bright yellow hue caught the eye as much as its potent metabolic effects. Lab journals from the 1920s and 30s describe how scientists hoped to harness its chemical potency without provoking the health scares that soon plagued workers in explosives factories and agricultural fields. The trail of DNBP, shaded by controversy, reminds us that science doesn’t just build on success—it stumbles, corrects, and learns from danger just as much as from curiosity.
Product Overview
DNBP stands as a well-characterized phenolic compound with two nitro groups at positions 4 and 6 of the aromatic ring, and a tert-butyl group at position 2. Its structure gives it a place among potent oxidants, redox agents, and industrial dyes. Chemical suppliers categorize it as a specialty intermediate, sometimes listing synonyms like 2-tert-Butyl-4,6-dinitrophenol, Dinoseb, or DNOC based on subtle structural or functional group differences. For those who have handled it, the sharp yellow crystalline solid tells its own story: a molecule crafted for power, not subtlety.
Physical & Chemical Properties
Walk into a lab storing DNBP, and the immediate impression comes from color—a deep yellow, crystalline substance. Its molecular weight is usually listed at about 242.2 g/mol. Melting points fall within the range of 92–93°C, much lower than related dinitrophenol compounds, likely because of the bulky tert-butyl group disrupting crystal packing. Solubility varies; in water it sits at trace levels, but organic solvents like ethanol and acetone dissolve it readily. This particular mix of hydrophobic bulk and hydrophilic nitro groups account for the potent activity across plant and mammal metabolism. Its UV absorption sits near 370 nm, giving it viability as a dye. In everyday work, this compound’s near-inexorable smell and dusting potential forces a level of respect that other phenols don’t require.
Technical Specifications & Labeling
Manufacturers typically deliver DNBP at purity levels above 98%, accompanied by batch-specific chromatography and spectroscopic data to confirm identity and purity. Product labels warn prominently of toxicity, especially inhalation and skin contact risks. UN numbers and DOT hazard designations highlight the explosive and environmental threats: UN 2761 signals an acute poison. Safe packaging relies on inner containment, strong sealants, and clear labeling with hazard pictograms. Those few times I saw it shipped, warnings bordered on overkill, but given the health impacts observed over decades, no margin for error exists.
Preparation Method
Lab manuals and industrial guides often reference nitration of 2-tert-butylphenol with a mix of concentrated nitric and sulfuric acids. Precise control of acid ratios, temperature, and reaction time shapes yield and purity. Out-of-control reactions risk runaway exotherms—hence the strict batch sizes and temperature monitoring. After reaction, the crude product settles, filtered with subsequent crystallization in ethanol to purify. Old texts show how early chemists groped for yield boosts, but often found themselves sidelined with hazardous byproducts like trinitrophenols. Over the years, safer batch reactors and closed-system handling reduced accidents, though the pungency never truly leaves the space where DNBP is made.
Chemical Reactions & Modifications
DNBP can take part in further transformations—not always predictable. The nitro groups invite reductions, often yielding amino derivatives suitable for dye chemistry or further coupling. The tert-butyl group gives backbone stability, yet harsh bases or acids risk cleavage, particularly at higher temperatures. Aromatic substitutions and coupling with diazonium salts extend its use to specialty dyes or analytical reagents, but workers know contamination with more toxic dioxins lurks if reaction conditions drift. Few compounds illustrate the blend of energetic chemistry and real biological hazard better than this one.
Synonyms & Product Names
Across chemical catalogs, DNBP goes by a range of names. Dinoseb, 2-tert-butyl-4,6-dinitrophenol, DNBP, and sometimes even “DNOC” in older European texts. CAS number 88-85-7 uniquely tags it in lab and regulatory paperwork. A chemist’s search across literature requires sifting through these synonyms, cross-checking data sheets and old patents where regional or era-based differences add confusion. Anyone researching DNBP quickly learns that paying attention to synonyms saves time and avoids dangerous substitutions.
Safety & Operational Standards
Operational risks with DNBP run higher than most specialty chemicals. Inhalation brings rapid onset symptoms: headache, sweating, increased heart rate, particularly for those who neglect respirator use. Skin absorption can cause systemic toxicity, aggravated by the molecule’s lipophilic tert-butyl group—standard nitrile gloves don’t always hold up. Waste handling falls under strict environmental and occupational hazards rules, and spills require both chemical neutralization and personal decontamination. Most industrial protocols demand negative-pressure hoods, chemical suits, and continuous air monitoring. These standards grew from tragic lessons over decades of agricultural and factory use—failures are well-documented, with debilitating long-term effects.
Application Area
DNBP once served as a key agrochemical, especially in the 20th century as an herbicide and pesticide under brand names like Dinoseb. Fields benefitted from rapid weed clearance—until reports of poisonings, reproductive harm, and residue buildup halted its use in more than 50 countries by the 1980s. Niche uses remain in dye synthesis and as a laboratory oxidative reagent, but most chemical firms restrict it to experienced handlers with comprehensive training. The tug-of-war between agricultural utility and public health traces strongly through regulatory archives; survivors in rural communities remember outbreaks tied to its misuse.
Research & Development
Earlier decades saw a rush to find new functions for DNBP, especially in crop protection and as a biochemical probe for mitochondrial function. Modern research shifted, focusing on environmental impacts, residues in water sources, and safer alternatives for similar oxidative processes. Green chemistry efforts redirected attention toward bio-based herbicides and selective enzyme inhibitors, leaving nitrophenols like DNBP to historical status. Academic researchers still study its metabolism for clues on energy coupling—by understanding its uncoupling action, we learn about basic biological processes and how modern drugs may affect energy metabolism. Chemical engineers in R&D rarely pursue new uses for DNBP but remain keen on developing remediation techniques for contaminated sites.
Toxicity Research
Few compounds have driven as much toxicological study. Acute exposure increases body temperature, depletes energy reserves, and triggers a cascade of organ damage. Scientists tracked its mitochondrial uncoupling effect, leading to hyperthermia and, in severe cases, death. Long-term studies linked exposure to reproductive toxicity, developmental delays in children, and significant cancer risks. Environmental science journals from the 1970s forward tracked residues in rivers and soil, showing how persistence outlived brief agricultural utility. Regulatory action followed reports of birth defects among farmers and factory workers. Recent years brought further research on soil and water cleanup, using advanced bioremediation and adsorption technology to tackle persistent environmental burdens. The sheer documentation of harm has locked DNBP away from most markets, a testament to how thorough toxicity research can drive industry and government response.
Future Prospects
Looking down the road, the future of DNBP seems shaped less by new product development and more by environmental cleanup. Chemical innovators focus instead on eco-friendly reagents with higher selectivity and lower toxicity. In the classroom or lab, DNBP remains a powerful case study in the unintended consequences of chemical innovation—balancing need with harm requires open eyes and a long memory. Emerging technologies may handle old DNBP stocks with more precise neutralization or destruction, and water engineers pursue techniques like advanced oxidation and carbon filtration, since nature alone handles degradation too slowly to keep pace with risk. In a world where greener chemistry drives investment, the story of 2-tert-butyl-4,6-dinitrophenol tells both a cautionary tale and a call to continual vigilance in chemical design and stewardship.
What are the main uses of 2-Tert-Butyl-4,6-Dinitrophenol?
What is This Chemical?
2-Tert-Butyl-4,6-Dinitrophenol doesn’t usually show up in headlines, but folks working in certain industries see the name enough. It’s a yellow crystalline compound, not something anyone would keep in a kitchen cabinet. Its real claim to fame comes from places that need strong and stable chemicals for purpose-built jobs. Anyone who has done work in pesticide or dye manufacturing might know it by its trade names or chemical shorthand.
Main Uses in the Field
Pesticide Manufacturing Has Deep Roots HereMany of the fields growing our food use pesticides to control bugs, fungus, or weeds. 2-Tert-Butyl-4,6-Dinitrophenol plays a tough role as an active ingredient or as an intermediate for other pesticide chemicals. Factories turn out lots of different compounds from this starting block. The compound’s molecular structure offers the potency and persistence that some pest control products need. This makes it valuable for agricultural chemical makers who don’t want half-measures.
Dyes for Industry and TextilesAnyone who’s shopped for colorful fabrics or worked in textiles understands synthetic dyes matter. Certain vibrant colors only show up with hard-to-replicate chemicals, and 2-Tert-Butyl-4,6-Dinitrophenol fits that bill for yellows and oranges. It gives color fastness, which means the color won’t fade after one or two washes. Even today, this chemical underpins some of the bold tones seen in plastics and textiles that have to stand up under sunlight or heat.
Potential Role in ExplosivesSome reports out of the academic world suggest this compound once played a side-part in explosives research. The nitrophenol structure appears in formulas for products intended to go boom, though 2-Tert-Butyl-4,6-Dinitrophenol itself no longer holds its place in modern commercial explosives factories. Manufacturing standards and new rules around safety have sidelined it in favor of other options, but you still find journal references and historical patents mentioning it.
Concerns and Looking Forward
Handling and Health RisksNo one wants dangerous posts in their workplace, but this chemical does pose a risk. Studies show it can harm the liver and kidneys and cause problems like skin irritation. Regulators in Europe and the US both list it on hazard inventories, with labels calling for gloves, masks, and tight control of airborne dust. In my time around chemical supply warehouses, managers always kept such substances behind clearly marked doors, not on open shelves.
Environmental ImpactFarmers might reach for pest control, but these compounds—if not used with care—drift into water and soil. The residues can stick around and affect bugs, fish, and sometimes mammal life. Environmental groups and agencies like the EPA keep a close watch, and several countries restrict use of products made with or from this chemical. There’s growing talk about safer, biodegradable alternatives that break down quicker and leave less hazard in farm runoff.
Finding Better Solutions
Some folks in research labs now work on making alternatives that do the job with less risk. Advances in green chemistry offer hope, and more manufacturers look for plant-based or microbe-derived compounds for both pesticides and dyes. Tighter rules on workplace exposure and disposal, along with new monitoring systems, also cut down on risks for workers and communities. Anyone responsible for purchasing or regulatory compliance needs updated knowledge and a direct line to environmental health specialists. Change won’t be simple, but the industry is moving in the right direction.
Is 2-Tert-Butyl-4,6-Dinitrophenol hazardous or toxic?
Facing Chemicals with Straight Talk
It’s not every day you run into a name like 2-tert-butyl-4,6-dinitrophenol outside a chemistry textbook. But if you work in a lab, an industrial plant, or even poke around specialized online shops, the odds go up. The real question people ask: is this stuff safe to work with, or does it carry serious risks?
Looking at the Science
This compound is a nitrophenol — compounds that, by their nature, often cause concern in the fields of chemistry and toxicology. Nitrophenols haven’t earned their reputation for being benign. Some hit lists for environmental pollution, others raise flags for health. For example, dinitrophenols have a notorious cousin: 2,4-dinitrophenol (DNP), which has a documented history as an industrial chemical and, unfortunately, as a dangerous weight loss substance. DNP’s dangers stem from its effect on cellular energy production. Overdose incidents caused fatal hyperthermia, organ failure, and long-term health problems. Facts like these matter when evaluating related chemicals.
2-tert-butyl-4,6-dinitrophenol shares chemical features with DNP. It contains reactive nitro groups and a bulky tert-butyl group. In practice, this structure means it could show similar toxic effects, though every compound brings unique quirks. Research highlights that dinitrophenols act as uncouplers of oxidative phosphorylation — science speak for “they mess with how cells make energy.” This action stresses cells and, in living organisms, stresses whole organ systems, especially the heart, liver, and kidneys.
Handling: Real Risks, Not Just Regulations
Practical experience tells me nothing good ever comes from ignoring safety data sheets. The fact that reputable suppliers lock away dinitrophenols behind extra paperwork and hazard warnings says a lot. Even a single accidental spill in a poorly ventilated lab can turn ugly. Skin contact can cause irritation or burns. Inhalation isn’t just unpleasant; it can mean headache, dizziness, or much worse with enough exposure. Swallowing a few grains of certain dinitrophenols ranks as a medical emergency.
Regulatory agencies in Europe and North America flagged these chemicals. The European Chemicals Agency classifies various dinitrophenols as hazardous — toxic if swallowed, cause damage to organs, and dangerous to aquatic life. No trusted health agency waves away their dangers. So even if a particular derivative isn’t as notorious as DNP itself, treating them all with caution makes sense.
Solutions: Rethinking Chemical Safety
Every workplace can take extra steps. I’ve seen labs prevent disaster by updating training, moving high-hazard chemicals to ventilated storage, and setting clear lines for who may access such compounds. Working with equal doses of respect and caution is the only way forward. Labeled containers, spill kits on hand, and a culture where no one gets shamed for speaking up about unsafe conditions makes everyone safer. On a wider scale, research into safer substitutes or formulations with reduced toxicity continues. Manufacturers and scientists can work together, pushing for greener alternatives where possible, so risks shrink with time and innovation.
In short, 2-tert-butyl-4,6-dinitrophenol shouldn’t be taken lightly. Instead, learn its risks, handle it with proper gear, and seek out safer, less toxic alternatives where you can. Safety in chemistry never comes from wishful thinking — it comes from respect, information, and a little healthy skepticism.
What are the safe storage requirements for 2-Tert-Butyl-4,6-Dinitrophenol?
Getting Real About Chemical Safety
2-Tert-Butyl-4,6-Dinitrophenol sounds like just another complicated name, but anyone working with it knows accidents aren’t picky. This compound, sometimes discussed in research or specialty manufacturing, doesn’t advertise its dangers, but the risk sneaks up. Without proper storage, sudden problems can mean property damage, injury, or even death. Over the years, I’ve seen well-meaning folks pay the price for taking shortcuts. That’s why safe storage matters more than probably anything else when you deal with 2-Tert-Butyl-4,6-Dinitrophenol.
Fire and Air: The Big Hazards
One tricky thing about this chemical comes from its potential to combust if things get too warm or it touches the wrong material. Too much heat or sunlight kicks off reactions nobody wants—smoke, fire, and toxic fumes. Its nitro groups make those risks real and not just a theoretical warning from a lab manual. Keeping it somewhere cool, dry, and out of direct light isn’t a detail; it’s wise practice. A flammable storage cabinet with a lock keeps honest folks honest and prevents mistakes on rushed days.
Why Labeling and Segregation Aren’t Optional
People sometimes store hazardous stuff “temporarily” in the wrong spot. That’s when trouble finds them. Always pick a container that won’t break down or react. Containers made from high-density polyethylene or similar don’t just last, they keep leaks in check. Clear, bold labeling with the full name and hazard warnings means even someone on a night shift knows what they’re handling, even if they’re tired or distracted.
Storing this chemical away from acids, strong oxidizers, and fuels isn’t paranoia. Nitrophenols don’t always play nice with other guests in the cabinet. I’ve seen costly lab shutdowns from careless storage—chemicals mixed by accident, a little spill, and then a lasting mess. Chemicals stored together should match in hazard class. Don’t count on memory; rely on routine and habit.
Control the Environment, Control the Risks
Ventilation in storage spaces plays a bigger role than most realize. Fumes will hang around in closed rooms, especially after a spill or break. A well-ventilated area, even just a thoughtfully installed exhaust fan, cuts that risk. Humidity control fights corrosion and keeps containers safe longer. No surprise, regulatory agencies like OSHA and NIH recommend specific ventilation measures with dinitrophenol compounds for good reason.
Training, Inventory, and Mindset Make the Difference
After years of working in and out of labs, I can say a big part of safety doesn’t come from hardware or fancy equipment. It’s about training people. Everyone in the storage area needs regular drills on what to do if there’s a fire or spill. Emergency eyewash and shower stations matter, but quick thinking saves time when seconds count. Inventory systems—old-fashioned or digital—catch missing or aging containers before disaster strikes. That awareness, more than any label or ventilation setup, keeps everyone safer.
Practical Solutions
Store 2-Tert-Butyl-4,6-Dinitrophenol in temperature-controlled cabinets that meet flammability and chemical resistance guidelines. Use tough, non-reactive containers with spill trays. Check inventory often and update records right away. Invest in clear hazard training, not just written rules nobody reads. Build habits, not just compliance.
No single measure handles every risk on its own. Real safety comes from routine, clear labeling, pay-attention training, and a willingness to ask when something feels off. In the end, these steps protect both people and property in ways shortcuts never do.
How should 2-Tert-Butyl-4,6-Dinitrophenol be handled in case of a spill?
What Spills Can Teach About Chemistry and Danger
Spill response tells a lot about a laboratory or warehouse. Every spill wakes up memories of training sessions and warning labels — but nothing can replace real experience. The stakes jump higher with hazardous chemicals. 2-Tert-Butyl-4,6-dinitrophenol, with its mix of explosive and toxic properties, brings out the seriousness required in chemical work. Simple carelessness can turn a small mess into a life-changing event.
Danger Doesn't Always Wear a Neon Sign
Some chemicals have obvious risks. Others only look harmless. 2-Tert-Butyl-4,6-dinitrophenol falls in that brittle middle ground. In powder or dust form, it burns and can blast violently if heated or struck. Skin contact causes rashes; breathing it triggers nausea, headaches, even worse. Once spread, it drifts into hard-to-reach corners, seeping into the air and clinging to work surfaces. Picking up a spill with bare hands? That story never ends well. In my first lab tech job, I fetched coffee for hours afterward, just trying to calm a coworker down after a spill. His skin glowed red for days.
Immediate Steps: Speed and Smarts, Not Panic
Once spilled, don’t wait around. Every second ticks toward more vapors, more spread. Grab a full set of gloves — not the thin ones for dishwashing, but chemical-resistant butyl or nitrile. Goggles and a snugly sealed mask keep toxic dust from eyes and lungs. Years ago, I watched a trainee skip this step. We all spent the afternoon in a negative-pressure room while he got checked out.
Work small. Dampen the powder with water to keep dust down — never sweep it dry, or the cloud could ignite. Scoop the mess with non-sparking tools into a rigid, sealable container. That means plastic scoops, not metal. Toss every disposable item used into the same container. Wipe down every surrounding area using wet cloths and place those cloths into waste as well. Skip bleach or strong oxidizers; some cleaning chemicals can trigger nasty reactions. If the spill seems bigger than a quick fix or splashes skin, stop the cleanup and get outside fast.
Waste Disposal: No Shortcuts Here
The cleanup pile counts as hazardous waste. Regular trash bins don’t cut it. All sealed material heads to a designated hazardous waste collection area. Never leave containers mislabeled. I once saw a box sit for weeks without a proper label, which created panic during the next collection cycle. Proper paperwork means faster, safer disposal. Local environmental regulations guide every removal, and waste contractors have their rules because lives and water quality hang in the balance.
Training, Not Just Warning Signs
Chemistry labs grow safer with real-world stories, not just laminated protocol sheets on a wall. Sharing mistakes, reviewing the details of each incident, and making time for hands-on drills transform the staff mindset. It becomes instinct to check for the right gear before approaching a mess. Emergency numbers, shower stations, and eye-wash setups must stay clear and marked. Supervisors should walk through these drills often, not just at new-hire orientation.
Prevention Means Respect
Stock only what’s essential and store dinitrophenol securely, well away from other incompatibles. Regular maintenance, not just during inspections, keeps shelves stable and clear. Old containers and leaky seals create situations that test everyone’s safety habits. The respect for chemicals never fades, no matter the career stage. Risk multiplies with routine; familiarity brings slips, and one mistake with dinitrophenol often sticks in memory far longer than any badge or paycheck.
What is the chemical structure of 2-Tert-Butyl-4,6-Dinitrophenol?
Digging Into the Heart of the Molecule
2-Tert-Butyl-4,6-Dinitrophenol—often abbreviated as Dinitro butyl phenol by chemists—catches the attention not only for its bold name but for the unique way its atoms arrange themselves. Structurally, this compound sports a benzene ring, that classic hexagon found in countless chemistry textbooks. Stuck onto that ring are two nitro groups, at positions 4 and 6, and a tert-butyl group hanging at position 2. A single hydroxyl group sits at the first position on the ring. It’s a deliberate arrangement, not just thrown together. Each piece brings its own set of properties, changing the behavior of the compound in practical use.
Why Structure Matters in Science and Beyond
Having spent years in chemistry labs, I’ve seen how even minor changes in molecular structure lead to massive shifts in behavior. The tert-butyl group, a bulky mass of carbon and hydrogen shaped like a mini three-pronged ball, adds real bulk to the molecule. This affects both its solubility and its ability to interact with other substances. The nitro groups, each made from a nitrogen and two oxygens, introduce electron-withdrawing power, which helps control how reactive the benzene core becomes. Companies making crop protection agents or industrial chemicals look for exact features like these to predict how a chemical will behave, both in a flask and in the environment.
Safety and Environmental Concerns
This isn’t just an exercise for the lab. Compounds like 2-Tert-Butyl-4,6-Dinitrophenol get used for legitimate purposes—including as pesticides, herbicides, and even in old-school weight loss drugs, though the latter carries huge health risks. The twin nitro groups, specifically, raise red flags. History has shown that nitro-substituted phenols sometimes act as uncouplers of oxidative phosphorylation in living cells. Accidental exposure, or worse, unregulated use, doesn’t always lead to good outcomes for people or the local ecology.
Handling such chemicals calls for strict controls. Gloves, goggles, proper ventilation—these aren’t just recommendations—they’re hard rules. People working with dinitrophenols learn quickly how skin contact or inhalation leads to harmful effects. Regular training and clear labelling help stop accidents before they start. Experience in chemical safety shows that strong policies plus regular monitoring can cut incidents to near zero.
Solutions for Better Stewardship
Solutions don’t emerge out of wishful thinking. Regulators need ongoing cooperation with manufacturers to trace the life cycle of chemicals like 2-Tert-Butyl-4,6-Dinitrophenol. Documented supply chains, stewardship at every step, and open reporting help keep things above-board. When environmental release does happen, remediation specialists need clear guidance and rapid-response methods tailored to nitro-phenol breakdown. Many industries now support research into greener alternatives—molecules that serve the same purpose without sticking around in soil or water for years. My own experience suggests progress is possible, but only with solid investment from both government and private sectors.
Final Reflections on Responsibility
Understanding the chemical structure of 2-Tert-Butyl-4,6-Dinitrophenol isn’t just an academic question. Every position, every functional group, carries impact—on performance, safety, and environmental fate. Continued transparency among chemists, regulators, and those living near production sites proves crucial. Real progress only shows up when safety embraces science, and environmental responsibility stands on equal footing with synthetic achievement.
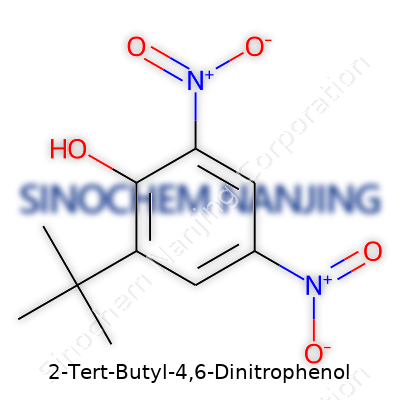
| Names | |
| Preferred IUPAC name | 2-tert-butyl-4,6-dinitrophenol |
| Other names |
Dinitro-ortho-tert-butylphenol Dinoterb DP-710 Dinitrobutylphenol DNBP Dinitrosec-Butylphenol Butilfenol 2-tert-Butyl-4,6-dinitrophenol |
| Pronunciation | /tuː tɜːrt ˈbɜːtaɪl fɔːr sɪks daɪˈnaɪtrəʊˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 88-85-7 |
| Beilstein Reference | 635542 |
| ChEBI | CHEBI:9441 |
| ChEMBL | CHEMBL64822 |
| ChemSpider | 21500 |
| DrugBank | DB02724 |
| ECHA InfoCard | 100.040.878 |
| EC Number | 215-696-3 |
| Gmelin Reference | 105844 |
| KEGG | C06537 |
| MeSH | D009606 |
| PubChem CID | 12321 |
| RTECS number | UC7875000 |
| UNII | 6Z9B1C58RH |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C10H12N2O5 |
| Molar mass | 258.21 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.34 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 2.83 |
| Vapor pressure | 1.2 x 10^-6 mm Hg (25°C) |
| Acidity (pKa) | 3.8 |
| Basicity (pKb) | 7.97 |
| Magnetic susceptibility (χ) | -47.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.597 |
| Viscosity | 14.7 cP (20°C) |
| Dipole moment | 3.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 332.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -308.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3177 kJ/mol |
| Pharmacology | |
| ATC code | N06AX10 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H351, H373 |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P312, P304+P340, P305+P351+P338, P308+P311, P312, P330, P337+P313, P405, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 1, Instability: 1, Special: |
| Flash point | Flash point: 185°C |
| Autoignition temperature | > 535 °C |
| Lethal dose or concentration | LD50 oral rat 340mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 320 mg/kg |
| NIOSH | GF9625000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 0.02 mg/m3 |
| Related compounds | |
| Related compounds |
2,4-Dinitrophenol 4,6-Dinitro-o-cresol Fenitrothion Dinoseb Picric acid |

