2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate: A Deep Dive into Its Journey and Role in Modern Chemistry
Historical Development
Chemists have been coaxing new molecules out of old building blocks for over a hundred years, but 2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate brings together several threads that run through the 20th century’s growth in nitroarene chemistry and esterification techniques. It doesn’t belong to the earliest classes of nitro compounds—those long predate organics with this level of structural complexity—but its nitrophenyl backbone recalls dyes, explosives, and agricultural chemicals developed just before and after the World Wars. These areas pushed researchers to craft more specialized, less volatile nitro derivatives, giving rise to methods now used to build colorful, stable esters like this one. The journey of this compound draws a line from classic dye intermediates to selective molecular tools tailored for specific targets, reflecting the evolution from basic functional groups to engineered reactivity.
Product Overview
2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate stands out with a dense aromatic core, two electron-withdrawing nitro groups, and a sec-butyl appendage—all tethered to a branched methylbutenoyl ester. This gives chemists a molecule with noticeable steric and electronic effects that can’t be ignored during handling or application. In laboratories, this compound usually appears as deep-yellow crystals, a visual cue familiar to anyone who’s worked with nitrophenyl esters. Rarely found in consumer goods, it surfaces more often in specialized research fields, from targeted synthesis to niche biochemical probes.
Physical & Chemical Properties
The molecule carves out its niche with a melting point often quoted near 110-115°C (depending on purity and formulation), and it dissolves in a range of polar organic solvents but resists dissolution in water. This balance of solubility broadens its reach in organic synthesis while making waste treatment slightly trickier. Nitro groups on the phenyl ring soak up electrons, increasing stability towards slow oxidation, but at the cost of higher toxicity risk and greater sensitivity to reducing agents. In everyday practice, the molecule’s high density, low volatility, and vibrant yellow hue give decisive clues during purification and compound verification.
Technical Specifications & Labeling
Suppliers package this compound under strict dry conditions, warning against moisture and light exposure. Labels call out the chemical structure, batch number, purity (often >97% for research uses), and safety pictograms. Even on a crowded storage shelf, that combination of labeling and color helps avoid mix-ups—a lesson all too clear to anyone who has ever grabbed the wrong pale yellow bottle at the back of a cold room. Specifications also stress storage temperature—ambient or below—and stress the need for tight sealing to squelch hydrolysis. Batch-level certificates break down contaminants, trace metal content, and residual solvents, with reputable distributors offering full documentation to help researchers meet regulatory and reporting standards.
Preparation Method
Making this compound takes patience, a few intermediate purifications, and a steady hand—no shortcuts with crowded molecules like this. Synthesis kicks off with nitration of a sec-butyl-substituted phenol, typically using a controlled mix of sulfuric and nitric acids under chilled conditions to keep explosive side reactions at bay. The dinitro product, once dried and isolated, undergoes an esterification reaction with 3-methylbut-2-enoic acid chloride, often invoked with a non-nucleophilic base in an inert solvent like dichloromethane. The workup involves careful washing and drying, since leftover acid or traces of unreacted nitroarene can muddy the final product and compromise activity, especially for those putting it to work in precise catalytic or probe applications.
Chemical Reactions & Modifications
Chemists value this molecule for its blend of reactivity. The nitro groups on the aromatic ring open doors to nucleophilic substitution—a classic for adding other groups or making more complex derivatives. Reductive conditions reduce the nitro functions to amines, providing an anchor for dyes or sensors with near-site specificity. The ester linkage, though stable under mild conditions, can be hydrolyzed or swapped out under more basic or acidic treatments, giving flexibility when tailoring for new targets. While sturdy enough for most storage, the molecule’s conjugated system and steric bulk also allow it to withstand moderate heat, letting researchers push for reaction conditions that would shatter more delicate esters or nitroaromatics.
Synonyms & Product Names
Trade names rarely float far from the mouthful that is “2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate,” though you’ll see “Sec-Butyl Dinitrophenyl Methacrylate Ester” or simple abbreviations in catalogs and publications. Alternate names reference the dinitrophenyl core or “DNP” shorthand alongside esters. For those sorting through research papers, identifying the compound sometimes means combing supplementary materials for structural diagrams rather than relying on truncated acronyms that can blur with unrelated nitroesters. Either way, the chemical identifiers in catalogs and papers help prevent dangerous mix-ups—keeping researchers and technicians out of harm’s way.
Safety & Operational Standards
Safety officers don’t take compounds with two nitro groups lightly, and neither should anyone at the bench. Material Safety Data Sheets (MSDS) spell out toxicity risks—skin, eye, and lung hazards—with strong recommendations for full PPE in handling. Fume hoods form a non-negotiable part of any operation using or modifying this molecule. Like many nitrophenyl esters, the compound brings fire and explosion risks in dust or concentrated powder form, especially if stored near oxidizers or strong acids. Laboratories dealing with larger volumes keep dedicated spill kits and neutralizing agents nearby, and train technicians to use gloves, lab coats, and eye shields at all times. Disposal needs special attention: traces, even in wash solvents, must be tagged for hazardous waste pickup and never dumped in regular drains.
Application Area
Most applications of 2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate cluster around specialized organic synthesis and analytical probing. In biochemistry, the molecule acts as a tagging agent, conjugating with proteins or peptides for tracking and analysis—using the nitro groups for easy detection under UV light or specific colorimetric reactions. In catalysis, researchers leverage the crowded ring and bulky ester to model reaction pathways or create controlled reactivity traps in larger molecular constructs. Some groups have even experimented with this molecule in the development of smart polymers, exploiting its layered reactivity for cross-linking and sensing in advanced material designs. These uses draw on documented reactivity and visible signature rather than bulk use or low-cost production.
Research & Development
The world of bench chemistry and applied materials research sees new avenues for this ester almost every year. Research papers keep uncovering new routes to functionalize the arene core, pushing deeper into electron transfer and controlled reaction cascades. Multi-step syntheses involving this molecule often underpin efforts in chemical biology, where attached probes help illuminate enzyme behavior or cell signaling pathways. Teams seeking more sensitive detection methods have begun to build new sensors from this scaffold, benefiting from that combination of electron-rich and electron-poor pockets. My own experience tracking nitrophenyl-tagged intermediates underlines how a visible, reliable label reduces bench error and speeds up method validation, especially with new students or less experienced chemists.
Toxicity Research
No compound with two nitro groups and an aromatic ring escapes toxicity research, and this one is no exception. The molecule’s robust aromaticity and lipophilic nature give it a troubling tendency to stick around in biological tissues, risking bioaccumulation if mishandled. Studies on related compounds have shown red blood cell disruption with repeated or acute exposure—mostly through uncoupling of oxidative phosphorylation in living cells. Animal studies, admittedly rare due to the cost and risk, show tissue inflammation and organ toxicity after high-dose administration. Regulatory guidance for occupational exposure draws from broader nitrophenyl ester databases, urging aggressive limits and mandatory ventilation in enclosed spaces. To anyone looking for everyday use outside a tightly controlled lab, those results ring alarm bells that should never be ignored.
Future Prospects
Industries probing for smarter, more selective molecular tools keep circling back to nitroarene esters like this one. The next few years will likely see researchers engineer new derivatives with safety tweaks—swapping out more toxic nitro groups for safer alternatives or building ester links that can be cleaved under gentler conditions. Environmental pressure on persistent organic pollutants calls for biodegradable analogues, a growing thread in recent grant proposals. In research circles, this molecule’s visible, chemical, and physical signatures make it a go-to for method development and educational demonstrations, teaching new generations the intersection of structure, reactivity, and safety. As oversight grows and chemical safety expectations tighten, only robust documentation, steady supplies of high-purity batches, and strong stewardship in labs will keep valuable compounds like this in effective, ethical, and forward-thinking use.
What are the main applications of 2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate?
Finding Value Through Specialized Chemistry
Many people glance at a chemical name like 2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate and wonder if it ever matters outside of a laboratory. The honest truth is that compounds with complex names often turn out to drive change in sectors regular folks rarely think about—coatings, agriculture, and filtration being just a handful. I remember a conversation with a colleague who worked in crop protection; seeing these compounds in action, not just in theory, left a mark on me. It’s easy to overlook how a chemical, only a few people can pronounce, might influence food on the table or a quieter water treatment plant across town.
Plant Protection Brings It to the Fields
Chemicals like this one have found places in helping protect crops. Growers want solutions that interfere precisely with weeds or pests but not with their actual harvest. 2-Sec-Butyl-4,6-Dinitrophenyl 3-Methylbut-2-Enoate shows up in herbicide research for this reason. Its particular chemical structure helps target weeds by disrupting growth at specific points in their cycle, sparing other species from unnecessary stress. Farmers benefit by lowering the chance of chemical drift, protecting soil health, and even achieving better yields with less chemical input. Organizations like the FAO have highlighted how targeted herbicides, compared to the shotgun approach of decades ago, create safer conditions for consumers and workers alike.
Industrial Roles Behind the Scenes
Work in pigment and dye manufacturing taps into specialized molecules like this one, too. The nitrophenyl part suggests strong coloration properties. In practice, that turns into vivid, stable dyes for textiles or plastics. I once visited a dye production facility where the chemist explained, plain and simple, that only certain molecules can give rich colors that last through repeated washes and sun exposure. This compound’s structure lets it act as a base point for further chemical tweaks, extending its usefulness into custom colors and even specialty inks. The textile sector spent years searching for dyes that resist fading; molecules in this family made a dent by sticking to fibers longer than old-school alternatives.
Environmental Remediation Steps Forward
Detoxifying polluted water often comes up in urban planning meetings, and those discussions almost always involve hunting for compounds that latch onto pollutants. Researchers using phenyl-based esters such as this have studied their ability to bind heavy metals and organic toxins. Drawing from my own time in wastewater analysis, I’ve seen how certain charged molecules break apart toxic chains or trap them for removal. While not the only option, this family of chemistry can be engineered into filtration materials or reagents. That makes for less hazardous discharge into rivers or groundwater—which pays off by improving community health and keeping clean-up costs from spiraling later on.
Paving a Smarter Road in Chemical Safety
No real progress happens without keeping safety at the center. Regulatory agencies like the EPA keep a close eye on any compound entering farms or factories. Metrics like biodegradability, chronic toxicity, or risk to non-target wildlife come into play. Demanding transparency in sourcing and production keeps environmental impact lower and builds public trust. Back when I sat in a product stewardship meeting, the conversation always circled back to knowing exactly where the risks lie—not just hoping they don’t show up. Practicing open communication about safety data, supporting green chemistry initiatives, and working closely with local communities helps everyone get the benefits with fewer surprises down the road.
What are the storage and handling conditions for this compound?
Why Storage Matters
Every lab worker knows a spill catches up with you, and nobody wants to explain a hazmat suit to the neighbors. Safe storage rules keep more than just paperwork in order—they can protect your lungs, your research, and the building’s peace of mind. Most chemicals, including the meaner organic compounds and moody inorganics, ask for cool, dry shelves away from heat, direct sun, and sources of ignition. Leave a solvent near a faulty outlet, you invite a classroom warning straight into your lab.
Temperature and Humidity: No Room for Assumptions
Too many labs treat temperature as a line on a label, not as the difference between stable storage and a surprise fire. Refrigerators with locks and separated shelves stop cross-contamination and mistakes. Humidity can quietly turn a powder into a sticky mess or ruin years of careful synthesis, so those silica packs tossed aside at delivery should not go straight into the trash. In my time working with reagents for enzymatic studies, a careless intern’s open vial once brought condensation and lost data—no research fund wants to pay twice for careless hands.
Containers: Picking More Than a Lid
Glass works for acids. Plastic handles bases and some flammable liquids. The point is, the wrong container turns a secure compound into a leaking danger. Tight-fitting lids stop evaporation and vapor build-up, but anyone who’s had to mop up spilled benzene knows the real struggle: matching the container not just to the label, but to how people in the lab work. Tethered labels, unbroken seals, and regular checks save hours hunting down vague odors.
Labeling and Segregation: The Cheapest Fixes
A label that says only “Solution A” means little to the overnight crew or anyone new in the lab. Full names, hazard symbols, preparation dates, and who opened the bottle last—all of this info takes seconds to jot down. Separate acids from bases, oxidizers from organics. These rules came from emergencies, not bureaucracy. In chemical distribution, I watched a rushed shipment mix oxidizers and reducers—luckily, someone caught it. One mistake would have led to injuries and lost contracts.
Personal Protection: It’s Not Just About Gloves
Rules about wearing gloves, goggles, and coats exist because skin absorbs faster than you realize. Even the best chemist skips a step now and then and pays the price with a ruined shirt or worse. Training goes beyond telling people where the emergency shower is. It’s about real drills and easy access to spill kits. My old supervisor repeated one lesson: “Your protection is the first line, not the last resort.”
Disposal: The Hidden Threat
Disposal rules aren’t glamorous, but they decide whether your workspace runs smoothly or gets flagged by an inspector. Old solvents, unstable mixtures, and unknown leftovers invite fines and safety reviews. Work with a reputable waste handler, use logbooks, and never mix waste streams. More than once, cleared-out stockrooms revealed containers with faded labels and unknown histories. These discoveries turn into expensive, risky cleanups.
Takeaway: Routine Over Routine
Safe habits, not just inspectable storage, keep people healthy and research moving. Every time a bottle returns to the right place, safety makes it through another day. These steps grow into trust, better results, and fewer accidents, even if they never make the news.
Is there any safety or hazard information associated with this chemical?
Everyday Chemicals, Everyday Risks
Most people run into chemicals without thinking twice. I remember walking into my garden shed as a kid and smelling something sharp. I had no idea bleach and fertilizer—so common at home—could react and cause problems if mixed. In today’s world, chemicals have made life easier—from cleaning products to those found in the workplace—but this comfort sometimes blinds us to hidden hazards.
What’s Often Overlooked
Past jobs taught me that safety information isn’t just for chemists or factory managers. People need the facts before anything goes near their skin, eyes, or lungs. Simple contact can trigger allergies or burns. Breathing fumes could cause headaches or, over time, damage a person’s organs. A well-known case involved kids exposed to methylene chloride in bathtub refinishing. Even with warnings on the label, those who didn’t read the instructions or lacked protective gear landed in the emergency room.
Some chemicals, like ammonia or chlorine, draw quick reactions from the body—a cough, watery eyes, maybe dizziness. Others, like benzene, slip in quietly, and chronic exposure builds risk over years. In spite of this, people skim over instructions or skip using goggles and gloves because “it’s only for a minute.” Stories like these remind me no warning is too obvious.
Real-World Impacts—At Home and Work
Research from the CDC links chemical accidents with everything from minor rashes to serious breathing problems. Poison control centers pick up thousands of calls each year from folks handling household cleaners, paint removers, and garden products. At work, the stakes can be higher. One slip-up in storing two reactive chemicals together has cost businesses thousands—and, in tragic cases, cost lives.
I’ve walked through factories where even well-trained staff let safety habits slide. A missing label or unclear safety sheet invites trouble. Sometimes the information is there, but it feels buried under technical jargon. Daily routines make dangers blend into the background. That misplaced trust in routine hurts more people than freak accidents ever will.
Where We Can Do Better
Solutions aren’t complicated. For starters, clear communication stands out. Labels should use plain language and big symbols. Short videos or infographics placed near storage areas could keep safety reminders fresh. Anyone who works with chemicals, even at home, benefits from a quick read of the safety instructions—especially looking for fire hazards, toxic exposure, and first aid steps.
Every chemical shipment to a school, business, or community group should come with safety data sheets that don’t require an advanced degree to understand. Local classes or training events show people how to spot warning signs and act fast if things go wrong. In homes, keeping products locked away from curious kids or pets saves trips to the ER.
Regulators and companies should double-check their instructions and training—then ask people who aren’t experts if the information makes sense. If you’ve ever puzzled over a label or wondered, “Is this safe?” you’re not alone. What I’ve learned is simple—speaking up and asking questions beats learning about safety the hard way. After all, just because a chemical is handy doesn’t mean it’s harmless.
What is the molecular weight and chemical formula of the product?
Understanding the Product at its Core
Chemistry fills our lives, from medicine to cleaning agents. Every product on a shelf comes with a chemical identity, and two pieces of information matter more than most: molecular weight and chemical formula. Those terms show up on safety data sheets, lab reports, and sometimes even on product labels. But they’re more than fine print — they shape research, production, logistics, and above all, safety.
Why Knowing the Chemical Formula Matters
A chemical formula isn’t just a string of letters and numbers. It tells us what atoms join together to build the chemical in question. For example, water carries the simple formula H₂O; for caffeine, you’d write C₈H₁₀N₄O₂. These symbols let professionals verify the substance, assess whether it fits a project, and trust what they’re handling.
No formula means no clarity. Imagine a chemist trying to reproduce a result without full information — even a single atom out of place makes the difference between breakthrough and disappointment. In pharmaceuticals, giving the wrong formula could pose real dangers. In my lab days, checking formulas wasn’t bureaucracy; it was how we protected ourselves and got reliable results.
Molecular Weight: The Hidden Cost of Every Gram
Molecular weight offers a bridge between research and production. It measures the sum of the atomic weights in a molecule. One number, but it sets the stage for dosing, mixing, transporting, and reacting chemicals safely. If you’re in a factory, it means knowing how much raw material delivers the right amount of active ingredient. In the doctor’s office, accurate weights mean the correct dose.
Missing or wrong molecular weights can lead to miscalculations. In agriculture, a fertilizer blend only works if proportions match the real chemistry. During college, I saw a batch of soap fail because one team misread the molecular weight of sodium hydroxide. It clumped, wasted material, and taught a tough lesson: always check the numbers and labels before mixing.
Supporting Fact-Based Choices
Trust in products builds only when customers and professionals feel confident about what’s inside. Regulators demand those numbers for every new product, not out of red tape, but so downstream users can calculate safe handling limits and ensure compatibility with equipment. Researchers need them for recipes, and logistics teams rely on them for safe storage. Globally, chemical shipping requires correct weights for transport safety and customs compliance.
Improving Transparency: A Work in Progress
The push for clearer information doesn’t end with rules. Open-access chemical databases make formulas and weights available for anyone to check. Education in reading and using this data should be standard—not just for chemists, but anyone in agriculture, medicine, or industry. More manufacturers are including QR codes or links with product details, bridging knowledge gaps for buyers and handlers.
Simple measures work. Industry and laboratories benefit most when chemical formulas and molecular weights are displayed in plain view: on bottles, in catalogs, and inside manuals. Mistakes shrink, output improves, and users feel safer and more informed. My experience shows that sharing accurate, reliable data makes every part of the chain work better, from the research stage all the way to the final user.
What is the purity level and available packaging for purchase?
The Value of Purity
Purity stands out as a dealbreaker for anyone deciding what and where to buy. In the world of chemicals and specialized goods, purity tells you how much of the product you’re actually getting. If you see “99% pure,” you know almost the whole container holds what you need, with leftovers being minor impurities that come with the territory. Purity’s not just a lab worry. It affects safety, results, and sometimes the final cost.
Imagine baking bread with flour that’s mixed with dust or sand; the result’s never predictable. The same logic follows in more technical lines like pharmaceuticals or food production. Higher purity drops the risk of side effects or surprise reactions and makes sure people get reliable results over and over. These standards give a level of reassurance. For instance, pharmaceutical grade usually lands around 99% or better, while technical grade, used outside medicine or food, might settle at 95% or so.
Packaging: More Than Just a Shell
Packaging isn’t just a box or a bottle. It affects shelf life, transportation costs, and even user safety. At first glance, a one-kilogram plastic jug and a 50-gram glass vial seem like throwaway details, but they say a lot about what the supplier expects their buyers to do. Food producers working with huge batches look for big bags or drums. Researchers piecing together a new experiment look for small, tightly sealed ampoules, since they’re working with smaller amounts and need accuracy.
Choices generally fall across a few standard weights or measures. Everyday buyers might notice 100g, 500g, and 1kg stand out at the smaller end. Bigger players—factories, bottling lines—lean toward multi-kilogram drums, sometimes up to 25kg or 50kg. Liquid goods might come in liters, gallons, or even specialized containers that help with dosing precise amounts. Bulk packaging keeps costs down and cuts out unneeded waste. Smaller, precision packaging does the opposite: it aims for convenience, accuracy, and safety.
The Real-World Trade-Offs
It’s not always about aiming for the highest purity or the biggest container. Risk and need both play a part. Food-safe packaging helps keep things uncontaminated and comes with detailed labeling to help with traceability. Chemical packaging sometimes includes tamper seals or child-resistant tops. The higher the purity, the more it costs—not only to make, but also to keep clean and deliver. If a process doesn’t require 99.9% purity, there’s little sense paying for it. On the other hand, skimping on purity where it matters can have lasting safety or quality consequences.
From my own experience in research, small vials of 99% pure reagent have saved projects from contamination headaches. In contrast, ordering in bulk from industrial suppliers better fits manufacturing needs, but always spurs questions about handling, storage, and cost per use.
How Suppliers Build Trust
Trust builds from clear labeling, transparency about purity levels, and thoughtful packaging choices. Reputable suppliers share certificates of analysis for every lot. They list both purity and any known side reactants or potential contaminants. Packages arrive sealed, labeled with the contents, date, and batch number, which helps buyers double-check everything from regulatory compliance to shelf life. Good suppliers won’t shy away from questions about purity testing. In turn, smart buyers keep these details on hand for audits or troubleshooting.
Knowing what’s inside the box, and what isn’t, turns one-time buyers into regulars. Clean, honest packaging and reliable purity are not just selling points—they’re necessities for anyone who cares about quality.
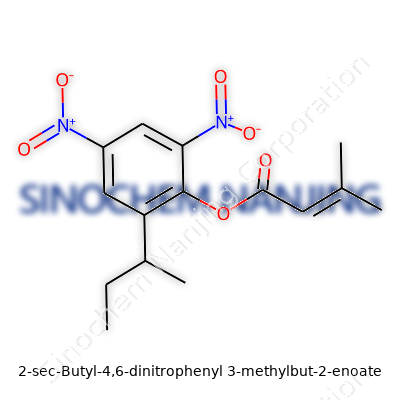
| Names | |
| Preferred IUPAC name | (2-sec-butyl-4,6-dinitrophenyl) 3-methylbut-2-enoate |
| Other names |
Dinoctec Dinoterb acetate Dinoseb acetate DNOC acetate Dinoseb acetic acid ester |
| Pronunciation | /tuː-sɛk-ˈbjuːtɪl fɔː siːks daɪˈnaɪtrəʊˈfiːnɪl θriː ˈmɛθɪlˌbʌt tuː ˈɛnəʊeɪt/ |
| Identifiers | |
| CAS Number | 88237-88-7 |
| 3D model (JSmol) | `/iso/expand__C=CC(C)C(=O)Oc1cc([N+](=O)[O-])c([N+](=O)[O-])cc1C(C)C` |
| Beilstein Reference | Beilstein Reference: 2216393 |
| ChEBI | CHEBI:131127 |
| ChEMBL | CHEMBL4108871 |
| ChemSpider | 18741581 |
| DrugBank | DB08329 |
| ECHA InfoCard | That product’s ECHA InfoCard string is: **"03afedfa-3706-4857-8adf-7366090d97a6"** |
| Gmelin Reference | Gmelin Reference: 90608 |
| KEGG | C18904 |
| MeSH | D009564 |
| PubChem CID | 22635924 |
| RTECS number | DA8750000 |
| UNII | QEL2K7C8U4 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C15H16N2O6 |
| Molar mass | 329.30 g/mol |
| Appearance | Yellow solid |
| Odor | Odor: odorless |
| Density | 1.24 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 2.87 |
| Vapor pressure | 7.56E-9 mmHg at 25°C |
| Acidity (pKa) | 10.72 |
| Basicity (pKb) | 11.03 |
| Magnetic susceptibility (χ) | -0.0006 |
| Refractive index (nD) | 1.589 |
| Viscosity | Viscosity: 18.3 cP (25 °C) |
| Dipole moment | 2.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 507.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -7417.2 kJ/mol |
| Hazards | |
| Main hazards | H302 + H315 + H319 + H335 |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS02, GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H317, H319, H335, H410 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | 122.9 °C |
| Lethal dose or concentration | LD50 oral rat 3200mg/kg |
| LD50 (median dose) | LD50 (median dose): rat oral 2650mg/kg |
| NIOSH | DD8575000 |
| PEL (Permissible) | No PEL established. |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
4,6-Dinitro-o-cresol Dinoseb Dinoterb DNOC Dinitroaniline Nitrofen |

