2-Sec-Butyl-4,6-Dinitrophenol: A Practical Look at Its Past, Present, and Future
Historical Development
Chemistry tells human stories better than it’s often given credit for. 2-Sec-Butyl-4,6-Dinitrophenol, known in labs and factories by several nicknames, comes from the tradition of nitrophenol research that stretches back more than a hundred years. Original work on dinitrophenol compounds grew in tandem with the boom in dyes and explosives, two fields that pushed organic synthesis to new extremes. During the early 20th century, German and British chemists experimented with ways to tweak basic phenol rings with different nitro groups and secondary butyl chains. This hands-on exploration didn’t happen in a vacuum; the chemicals found a place in muscle-building, metabolic research, and eventually in industrial applications. Use as a herbicide and metabolic agent got a lot of attention. With greater understanding of toxicity and a drive for safety, regulators eventually forced a slowdown in widespread use. Today, 2-Sec-Butyl-4,6-Dinitrophenol doesn’t get the headlines, but it still claims a history tied up with progress in organic chemistry.
Product Overview
Chemically, this compound stands as an aromatic nitrophenol. Laboratories order it in powder or granular form, usually with a yellow to orange color. Its crystalline structure reveals the nitro groups at positions four and six and a sec-butyl chain at the second carbon. Industrial suppliers might label it under different trade names, but the backbone stays consistent. Researchers use it primarily as an intermediate in organic synthesis, but it also appears in select analytical chemistry protocols and sometimes as a standard in toxicity studies. The product enters through a secure supply chain, monitored for impurities due to environmental and health regulations, which helps define the ground rules for safe adoption anywhere in production.
Physical and Chemical Properties
Anyone who’s spent hours over a reagent bench will notice the sharp odor and feel a cautionary respect for aromatic nitrophenols. This compound melts around the 110–115°C mark, with a decent thermal stability until pushed to decomposition. Density tops out between 1.3 and 1.5 g/cm³, and the powder dissolves moderately in common organic solvents like ethanol and acetone but not so well in water. Under the microscope or bench lamp, it glows with that familiar yellow tint common to many nitro compounds. Its low vapor pressure keeps inhalation risk down in normal settings, although dust control remains necessary. Acidic hydrogen hangs on the phenolic group, making the compound reactive in a range of organic transformations. The nitro attachments on the phenyl ring prove stubborn, resisting most degradation outside highly reducing environments.
Technical Specifications and Labeling
Strict technical specs define its quality. Purity often exceeds 98%, with impurity checks for unreacted phenols and other nitroaromatic byproducts. Product labels highlight melting point ranges, recommended storage temperatures (usually below 25°C, away from direct sunlight), and batch traceability numbers. Identification numbers such as CAS registry and EC numbers help clarify what’s in the drum, box, or vial. Packaging standards avoid static buildup and moisture ingress, so shipping departments lean toward high-density polyethylene or glass containers. Weight and lot size should match the end-user’s protocol, whether that means research quantities of a few grams or industrial scales in kilograms.
Preparation Method
Most synthesis routes fall back on nitration of sec-butylphenol as a starting point. Nitrating agents such as a blend of nitric and sulfuric acid do the heavy lifting, working under controlled temperatures around 0–10°C to avoid runaway reactions and poor yields. Reaction work-up demands good separation, with ice-cold water driving crystallization and filtration. Techs follow several cleansing steps: washing with dilute alkali removes acidic byproducts, while successive water and alcohol washes limit contamination. Larger facilities with experience treat waste streams with care, knowing that nitrophenol residues can cause havoc if they reach public water systems. Newer process tweaks try to temper environmental impact, using less hazardous oxidizers or adopting continuous-flow systems to maintain safer, more consistent yields.
Chemical Reactions and Modifications
Chemists appreciate flexibility, and this compound can give multiple ways in. Both nitro groups show promise for selective reductions—think metal-acid combinations yielding either amines or hydroxylamines. The sec-butyl side-chain offers another access point, responding to oxidation or halogenation under the right conditions. Conjugation with other aromatic systems emerges in dye chemistry, and leaving groups at the phenolic oxygen site let researchers tack on new functionalities. Academic papers in the last decade have outlined novel coupling reactions or bioactive derivative syntheses starting from this nitrophenol. Some teams even attach short peptides, chasing potential in drug metabolism and delivery research.
Synonyms and Product Names
Names stack up quickly in chemistry catalogs: 2-(Butan-2-yl)-4,6-dinitrophenol, sec-Butyl DNP, and a handful of less common IUPAC variants. Some legacy materials might show old-fashioned trade designations, reflecting earlier periods of use. It saves headaches for everyone—chemist to safety inspector—when every order matches by CAS number and, where possible, the formula: C10H12N2O5.
Safety and Operational Standards
Decades of lab safety rules boil down to knowing the risks before popping a bottle open. 2-Sec-Butyl-4,6-Dinitrophenol poses real dangers: skin and eye irritant, serious respiratory hazard if dust floats unchecked, and above all, a toxic effect on the central nervous system and metabolism. Repeated or intense exposure can lead to headaches, rapid breathing, sweating, and, in the worst cases, systemic poisoning. Strict gloves-and-goggles protocol is non-negotiable, alongside good ventilation and dust extraction. Companies must meet the Workplace Hazardous Materials Information System (WHMIS) and the Globally Harmonized System (GHS) standards. Waste streams travel for high-temperature incineration or specialized treatment, never down the drain. These habits reflect not just company policy but a scientific community learning from past mistakes in mishandling related nitroaromatics.
Application Area
Today, the main uses turn up in organic synthesis, especially where researchers need an electron-deficient aromatic platform. That translates into intermediates for agrochemical synthesis, specialty dyes, or selective metabolic probes. Analytical labs also run reference standards for toxicity and metabolism studies. The shadow of historical misuses lingers—early metabolic studies led to nonmedical abuse, which brings reminders of past overreaches and underscores why modern controls feel so strict. Even with tighter regulations, demand continues in advanced chemistry, where no simple substitutes hit all the same marks for reactivity and selectivity.
Research and Development
Recent research pushes into two main directions: safer production and new derivatization techniques. Sustainability experts look for cleaner nitration, using greener solvents or catalysts that shrink waste and risk. Investigators develop derivatives with altered toxicity or targeted biological action, hoping to unlock value in controlled applications. I think about the work going into catalysis—using this molecule as a launch point for fine-tuned aromatic transformations, or as a scaffold for bioactive molecule discovery. Journals report progress, but risk assessments stay front and center, sometimes slowing down the transition from lab bench to full-scale production.
Toxicity Research
No discussion of dinitrophenols can leave out their impact on human health. Classic studies tie nitrophenol ingestion to acute poisoning, with symptoms ranging from nausea to fatal hyperthermia. Even low-level, chronic exposure risks metabolic disruption and organ damage. Rodent models confirm systemic toxicity, and environmental studies reveal bioaccumulation that outpaces natural degradation in aquatic habitats. Regulatory agencies like the US EPA and ECHA collect and publish detailed risk profiles, cautioning against casual disposal or lax handling. Evidence drives modern rules, including exposure limits at the workplace and requirements for training, monitoring, and personal protection.
Future Prospects
Looking ahead, demand for specialty nitrophenols will probably keep trending up in research, while commercial use remains tightly restricted. Opportunities show up in green synthesis and in drug discovery, as chemists keep hunting for nitroaromatic compounds that balance reactivity with lower toxicity. Process chemists chase new techniques for safer scale-up, from continuous flow to biodegradable solvents. Regulators and public health experts keep the pressure on, forcing the industry to document every risk and prove every process. There’s a quiet optimism in seeing ongoing research reframe old chemicals for new uses, provided everyone steers clear of repeating old mistakes. In the end, real value will depend on honest safety standards and a hands-on mindset at every stage from production to application.
What is 2-Sec-Butyl-4,6-Dinitrophenol used for?
Chemicals Like This Shape the Modern Farm
Factories and labs pump out all sorts of compounds with odd names, but 2-Sec-Butyl-4,6-Dinitrophenol stands out for its role in the world of agriculture. Known to a few as Dinosulfon, this yellowish compound has marked its territory as a herbicide and pesticide. Farmers have relied on chemicals like this one for decades, not just to keep their fields clear, but to make harvesting possible on a bigger scale. We see cheap, plentiful wheat and corn at the grocery store because weed killers cut down competition in the soil.
Benefits Keep Crop Yields High
Life on a farm involves a constant battle with invasive plants. I remember long rows of soybeans lost to weeds during a rainy season, before the field got sprayed. Dinitrophenols step in to scorch unwanted growth, letting cash crops stretch out and drink in the sun. That chemical precision helps feed millions. This is no sales pitch—it’s the backbone of modern food supply chains.
Risks and Drawbacks From Field to Table
There’s no denying chemicals like 2-Sec-Butyl-4,6-Dinitrophenol get the job done, but the trade-offs raise tough questions. Toxicity isn’t just a word buried in a safety data sheet. According to public health research, dinitrophenol derivatives affect mitochondria, disrupting how cells handle energy. The risks extend past the users. Runoff can reach rivers, groundwater, and food webs, impacting fish and frogs and the water people drink.
In the 1930s, a related compound—plain dinitrophenol—was sometimes misused as a “slimming” agent, leading to toxic poisonings and deaths. Regulations grew stronger, but mistakes and illegal uses still surface. Today’s herbicides come with strict label warnings, and it’s still easy to find headlines about improper disposal or exposure, showing that the margin for error remains slim.
Regulation and Responsible Use Matter
Government agencies like the EPA review the safety data and decide what gets approved. Many countries restrict or ban dinitrophenol compounds. Even where they remain legal, applicators need certification and training. Some rely on closed transfer systems, personal protective equipment, and efforts to contain runoff. These aren’t just check-the-box steps. I’ve seen the aftermath of chemical drift—dead fish, brown patches in woods, and sick cattle. Mistakes don’t just stay on one property.
Where Do We Go Next?
There’s pressure to phase out risky herbicides, but the alternatives aren't always ready or affordable. Organic methods ask for more time and labor, and aren’t a match for every farm. Getting creative means blending cover crops, targeted spraying, and smarter equipment. Universities and startups are testing biopesticides and robots to cut down on the old-school chemicals. Stronger monitoring programs spot problems early, and more farmers check soil and water after spraying. Even regular folks play a part, by asking about food origins or supporting sustainable practices.
Weighing Outcomes With Open Eyes
Every chemical in the field leaves a mark, as clear as the crop rows that stretch toward the horizon. The promise of bigger harvests keeps people fed, while hidden costs pile up in the groundwater and food chain. Solutions take time, but ignoring the stakes doesn’t work. Food isn’t just about what makes it to the plate, but how it gets there.
What are the safety precautions for handling 2-Sec-Butyl-4,6-Dinitrophenol?
Why Getting Safety Right Matters
Anyone dealing with 2-Sec-Butyl-4,6-Dinitrophenol in a laboratory picks up pretty quickly that this chemical means business. Researchers don’t just use this compound for its intricate chemistry—it comes with hazards that can change a day from routine to disastrous. Over time, I’ve seen that most accidents happen not with dramatic explosions but with small mistakes that build up: a glove left torn, a fume hood sash left a few inches too high, a bottle not quite sealed. This isn’t a compound to treat like table salt. It demands full respect and attention.
Knowing the Risks: More Than Just “Danger” Labels
2-Sec-Butyl-4,6-Dinitrophenol acts as a skin and eye irritant. Breathing in even small dust particles can irritate airways or trigger worse effects if exposure drags on. Nitrophenol compounds have a reputation for hitting the body’s cells where it hurts, interfering with energy production and possibly causing toxic buildup in the organs. There’s no badge of honor in “toughing out” exposure—the real skill lies in keeping your body safe in the first place.
Setting Up a Safe Workspace
A good safety routine kicks in way before that first bottle opens. Storage makes a real difference: a cool, dry spot, away from incompatible chemicals (especially reducing agents and combustibles), keeps things predictable. If moisture drifts into the bottle, you run the risk of unwanted reactions and slow decomposition, so seal containers firmly and check the integrity of the packaging. I remember the comfort of seeing tidy shelves with bottles lined up safely—there’s no substitute for visual cues that all’s in order.
Personal Protective Gear—Non-Negotiable
Goggles, a well-fitted lab coat, and sturdy nitrile gloves give a minimum layer of defense. I always preferred using a face shield during weighing or transfers, since dust from 2-Sec-Butyl-4,6-Dinitrophenol floats with a mind of its own. After peeling off gloves, I never skipped washing hands, even if I thought they stayed clean. High-quality gloves cost a bit more, but I found they save hours or days recovering from chemical burns or rashes.
Ventilation: The Unsung Hero
No trick in the book beats a good fume hood. Even a small spill or puff of dust can mean trouble, and relying on open windows is wishful thinking. In my experience, checking the hood’s airflow gauge every session stopped countless headaches and throat burns before they started. Poor air movement turns minor exposure into a lingering health issue.
Clever Handling and Housekeeping
Transfers and weighing should run like a practiced drill—steady hands, spill trays, and clean tools. I once had a close call when a scoopula left on a countertop dusted the next experiment with yellow powder, and I learned fast to clean as I go. Dedicated waste containers, labeled boldly, keep used gloves, contaminated wipes, and any accidental spills from spreading. If a spill happens, damp cloths (not dry sweeping) pull up dust without kicking particles into the air.
Training and Emergency Prep
Regular drills made safety second nature for my old crew. Everyone learned to wash chemical splashes from their skin at the eyewash station, not just grab a towel. Spill kits sat right where anyone could spot them. I can’t count how many new researchers thanked me later after practicing spill response. Calling for help isn’t about failure—it’s about protecting everyone in the building.
Knowledge Turns Caution Into Confidence
Facts underpin good safety: the OSHA guidelines, reagent datasheets, and past mishap reports turn guesswork into clear steps. Following protocol leads to fewer surprises and more peace of mind. Safe habits didn’t slow my experiments—they made the outcomes reliable, and took a load off my mind at the end of every shift.
What is the chemical structure and molecular formula of 2-Sec-Butyl-4,6-Dinitrophenol?
What Does 2-Sec-Butyl-4,6-Dinitrophenol Look Like?
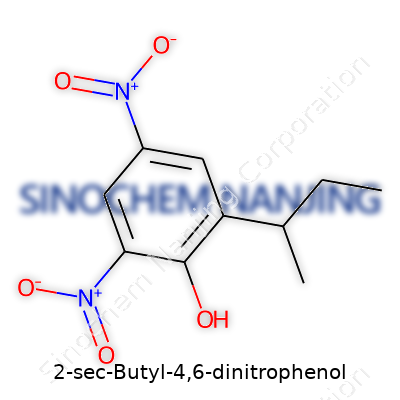
Anyone curious about the makeup of 2-Sec-Butyl-4,6-Dinitrophenol is diving right into the heart of organic chemistry. The name itself gives away the essentials. There’s a benzene ring, two nitro groups, and a sec-butyl chain hooked onto specific locations. The molecular formula stands as C10H12N2O5. Draw it out, and you see the nitro groups bonded to the fourth and sixth carbon atoms on the ring. Over on the second carbon, there’s a sec-butyl group sprawling out—a four-carbon side chain forked right at the base. Each group changes how the molecule behaves, both in the lab and the environment.
Chemical Structure:
- Benzene core acting as the backbone
- Nitro groups (-NO2) on carbon positions 4 and 6
- Secondary butyl group (–CH(CH3)CH2CH3) attached at position 2
Why These Features Matter
To someone tinkering in the lab, the nitro groups do what they always do: pull electrons away from the ring. That little sec-butyl group, on the other hand, changes the molecule’s solubility and stability. Discussing dinitrophenol families brings up history—early uses as dyes, explosives, weight-loss agents, even pesticides. Dinitrophenols have a notorious reputation for being potent and, at times, hazardous. Their chemical structure lays the groundwork for their high reactivity, particularly because those nitro groups make the molecule much more likely to participate in chemical changes, not always for the better.
Why Accuracy in Naming and Structure Counts
My years sifting through chemical databases and sizing up old bottles in labs taught me the importance of precision—one carbon in the wrong spot, and a compound can shift from harmless to dangerous. Take this specific molecule: just swapping the sec-butyl group to another place or flipping the nitro groups would change its profile entirely. Errors in names led to confusion, wasted time, and even hazardous mix-ups in the past. Lab safety depends on this accuracy, especially since phenols and nitro compounds carry health risks, from skin irritation to toxic effects on the metabolism.
Real-World Impact
It helps to remember: compounds like 2-Sec-Butyl-4,6-Dinitrophenol didn’t wind up as obsolete curiosities for no reason. Dinitrophenols like this one pop up in discussions about environmental safety—they don’t break down easily and can cause long-lasting issues in water and soil. Certain regulations restrict their manufacture and use for exactly that reason. Having the exact structure and formula means regulators, chemists, and industries avoid missteps that could impact ecosystems or health. A single careless label or missing group might end up polluting groundwater or endangering workers.
Better Chemistry Through Diligence
I once saw a colleague triple-check their labels after a near-miss with mislabeled dinitrophenols. Tiny molecular differences shape entire safety plans. In smaller labs, keeping proper references and labeling practices helps avoid accidents with compounds like these. Companies need to back up their chemical purchases with reliable data sheets. Regulatory bodies should share standardized identifiers like CAS numbers and have clear communication between suppliers, end-users, and environmental agencies. Knowledge, not shortcuts, keeps everyone safer around rarely-used but occasionally dangerous compounds.
How should 2-Sec-Butyl-4,6-Dinitrophenol be stored?
Why Safe Storage Demands Attention
Working around chemicals, every seasoned lab technician knows a casual approach sparks risks that nobody wants. Just reading “2-Sec-Butyl-4,6-Dinitrophenol” sets off alarms in the minds of anyone who has handled energetic compounds or substances with strong oxidizing potential. Unlike a simple bottle of household cleaners, this stuff calls for an extra level of respect. A small mishap—say a dropped container, a too-warm room, or a cracked seal—can land people in the hospital or lead to legal headaches. Chemical safety isn’t just a set of rules on a shelf; it saves jobs, businesses, and sometimes, lives.
Knowing the Hazards Makes a Difference
In labs I’ve worked in, there’s a big poster reminding staff: “DNOC and its analogs: Keep them cool, dry, locked away.” 2-Sec-Butyl-4,6-Dinitrophenol falls into this category. Explosive dust, toxic by skin contact, and dangerous if airborne—this compound earned its reputation. Literature from Chemwatch and PubChem paints a sharper picture: prolonged exposure damages health, accidental mixing with incompatible materials starts fires or toxic reactions, and container corrosion can endanger everyone in the building. Hearing about warehouse fires and lab accidents that start with minor leaks or poor labeling only reinforces how crucial proper storage is.
Practical Steps You Can Take
Don’t complicate things. Segregate this chemical from everything organic and especially from anything combustible. Its nitro groups don’t play nice with reducers or strong bases. Inside a controlled access cabinet, there’s a dedicated spot built for energetic materials. I always check the temperature, aiming for below room temp—typically below 20°C (68°F). Humidity control stops clumping and potential hydrolysis, which can trigger a runaway reaction. Desiccators work well for small amounts, and for bulk, good ventilation and environmental monitoring go hand in hand.
Sturdy Containers and Honest Labeling
Cheap plastic or rusty tins spell disaster. I learned the hard way that glass isn’t always best—if there’s even a little pressure build-up, glass pops. High-density polyethylene (HDPE) works, but only if the manufacturer confirms compatibility. Every bottle gets a clear, date-stamped label, and I keep a log for every move, even just to check inventory. Spills or mix-ups cost much more than the price of proper containers and ink.
Safety Culture Over Shortcuts
Veteran chemists repeat this advice often enough: never trust memory alone. Double-check the location listed in the chemical inventory database, and log all removals or transfers—even if it’s just for a gram. In a pinch, never store with solvents (especially not ethers or alcohols), never near acids, and keep off metal shelving that can corrode or spark. Lab supervisors insist on PPE: gloves, goggles, and lab coats never gather dust. The “it won’t happen to me” attitude got more than one promising chemist removed from the bench.
Why Vigilance Pays Off
Over the years, I realized small routines separate safe labs from risky ones. Rotating stock, regular hazard reviews, and swift removal of expired material make sure every ounce of 2-Sec-Butyl-4,6-Dinitrophenol remains a tool, not a threat. Relying on protocols, using real evidence from chemical safety boards, and working as a team keeps everyone safe. It’s the difference between reading about chemical incidents in the news and living through one.
Is 2-Sec-Butyl-4,6-Dinitrophenol hazardous to health or the environment?
Recognizing the Chemical
2-Sec-Butyl-4,6-Dinitrophenol pops up in technical circles as a type of dinitrophenol compound, a name that rang a bell for me following stories about industrial accidents. This chemical lurks in some pesticides and has ties to certain manufacturing processes. With a yellow tinge and crystal-like form, it falls among substances known for their reactivity, which should always set off alarm bells.
Health Hazards
Decades of toxicology research show dinitrophenols can pack a punch, and 2-Sec-Butyl-4,6-Dinitrophenol falls under that same tough reputation. Exposure—even at low levels—can irritate the eyes, skin, and respiratory tract. More to the point, dinitrophenol compounds impact how our bodies handle energy, uncoupling mitochondria from their regular job of producing usable fuel. Workers in facilities where this chemical gets handled without proper controls often report headaches, muscle pain, fever, and some cases reach acute toxicity.
In severe cases, people have suffered from confusion, dehydration, and organ stress. At higher doses, many related compounds became infamous for causing fatal hyperthermia, where the body overheats and critical functions break down. Some health authorities, like the US National Institute for Occupational Safety and Health, list these chemicals as having "immediate danger to life or health" limits because effects turn dangerous fast.
Environmental Fallout
It’s not just about personal health. Dumping or careless spills have a way of echoing out into rivers, wetlands, or even groundwater. Dinitrophenols persist in the environment because they break down slowly, especially when sunlight or bacteria don’t get a clear shot at them. This gives them enough time to move through water or soil, reaching places the original user never intended.
Wildlife faces the brunt of this stubbornness. Fish and aquatic insects exposed to dinitrophenols tend to show reduced activity, stunted growth, or even death at higher levels. I recall reading about particular marsh clean-ups where dinitrophenol contamination wiped out insects and frogs for miles, making clear that nontarget critters bear the cost for years beyond a single spill. Plants exposed to contaminated water often yellow and wilt, further shrinking habitats.
Real Solutions and Safer Alternatives
Risk control for this chemical comes down to limiting direct contact, upgrading personal protective equipment, and stopping releases into the environment. Chemical substitutes, especially ones with lower persistence or toxicity, have started to replace dinitrophenol-based compounds in some sectors. Strict storage and handling rules, along with frequent safety training, help lower the odds of accidents. Anyone who’s worked around hazardous chemicals can tell you: regular monitoring of air, soil, and water works as an early warning before real damage occurs.
Regulatory bodies, including the EPA and ECHA, push for tighter restrictions and regular reporting for chemicals like 2-Sec-Butyl-4,6-Dinitrophenol. I’ve seen companies move quickly on alternatives once the process got re-evaluated, especially after seeing long-term health data. Communities living near processing plants raise concerns early through local watchdog groups, pushing for cleanup and accountability. Real progress tends to track with transparency about risks and a willingness to retire dangerous substances for smarter choices.
| Names | |
| Preferred IUPAC name | 4,6-dinitro-2-(butan-2-yl)phenol |
| Other names |
Dinoseb DNOC Dinitrosecbutylphenol 2-(1-Methylpropyl)-4,6-dinitrophenol 4,6-Dinitro-2-sec-butylphenol |
| Pronunciation | /tuː-sɛk-ˈbjuːtɪl ˈfɔːr sɪks daɪˈnaɪtrəˌfiːnɒl/ |
| Identifiers | |
| CAS Number | 88-18-6 |
| Beilstein Reference | 1916190 |
| ChEBI | CHEBI:34475 |
| ChEMBL | CHEMBL1230434 |
| ChemSpider | 20013382 |
| DrugBank | DB04569 |
| ECHA InfoCard | ECHA InfoCard: 100.006.413 |
| EC Number | 2078-54-8 |
| Gmelin Reference | 79287 |
| KEGG | C18606 |
| MeSH | Dinitrophenols |
| PubChem CID | 7006 |
| RTECS number | SJ8925000 |
| UNII | 08EP56P4JP |
| UN number | UN3449 |
| Properties | |
| Chemical formula | C10H12N2O5 |
| Molar mass | 228.19 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.31 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.97 |
| Vapor pressure | 3.13E-6 mmHg at 25°C |
| Acidity (pKa) | 8.09 |
| Basicity (pKb) | 10.27 |
| Magnetic susceptibility (χ) | -78.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.589 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 385.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -372.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3825.6 kJ/mol |
| Pharmacology | |
| ATC code | N06BX08 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H318, H400 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-4-Explod |
| Flash point | 69 °C (156 °F) |
| Autoignition temperature | The autoignition temperature of 2-Sec-Butyl-4,6-Dinitrophenol is **515°C**. |
| Lethal dose or concentration | Lethal dose or concentration: Oral rat LD50 = 250 mg/kg |
| LD50 (median dose) | LD50 (median dose): 220 mg/kg (oral, rat) |
| NIOSH | KN1450000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 mg/m³ |
| Related compounds | |
| Related compounds |
Dinoseb Dinoterb DNOC Picric acid |

