2-Nitroiodobenzene: A Deep Dive into Its Role in Modern Chemistry
Historical Development
2-Nitroiodobenzene didn't turn up out of nowhere. Its roots trace back to early aromatic chemistry research in the nineteenth century, when chemists were just starting to explore the combination of iodine with nitroaromatic systems. During that era, simple methods dominated synthesis, often involving harsh and inefficient conditions. Iodinated aromatics like 2-nitroiodobenzene served as valuable test cases for understanding substitution patterns and the special influence of large halogens in electrophilic aromatic substitution. Academic labs in Europe documented the distinct reactivity of iodinated benzenes, highlighting how nitro groups change the game in reactivity and selectivity. This groundwork paved the way for today's more complex functionalization and cross-coupling techniques that rely on substrates like 2-nitroiodobenzene.
Product Overview
2-Nitroiodobenzene counts among those building-block intermediates that synthetic chemists respect. With an iodine atom on the benzene ring beside a nitro group, this compound stands out in both research and industrial contexts. It pops up in the preparation of pharmaceuticals, specialty polymers, and even some crop protection agents. Its structure opens up a pathway for selective metalation, cross-coupling, and further substitution, making it a popular choice for constructing more elaborate aromatic scaffolds. Traders and procurement teams often keep options from several suppliers on hand, balancing between purity, batch size, and regulatory paperwork.
Physical & Chemical Properties
In the lab, anyone working with 2-nitroiodobenzene soon gets familiar with its bright yellow crystalline form, a testament to the nitro group’s impact on aromatic compounds. It generally melts at about 74-77°C and stays solid at room temperature. Its molecular weight tips the scale above many simple aromatic compounds, thanks mostly to that heavy iodine atom. The molecule carries a faint but sharp chemical odor. Solvent-wise, it likes organic choices—toluene, ether, acetone, or chloroform—yet doesn’t dissolve much in water. Reactivity boils down to its ortho structure: the nitro group activates the ring toward further substitution, although the big iodine draws attention for cross-coupling chemistry.
Technical Specifications & Labeling
Most chemical suppliers list 2-nitroiodobenzene with a minimum assay above 98 percent, sometimes bumping that higher for research-grade inventory. Detailed Certificates of Analysis tell the story, including moisture levels, melting point characteristics, and traces of residual solvents. Packaging runs the gamut from amber glass bottles for a few grams up to lined steel drums for bulk customers. Safety data gets printed front and center, usually highlighting hazard codes tied to irritation and environmental risk.
Preparation Method
Chemists tend to rely on electrophilic aromatic substitution for making 2-nitroiodobenzene. The most common route involves starting with iodobenzene, treating it with a mixture of nitric and sulfuric acid—classic nitration conditions—under temperature control. Some labs flip the script, starting with nitrobenzene and introducing the iodine with an oxidizing halogenation step. Yields depend on tight control of reaction time, temperature, and purity of starting materials. Side products can gum up the mixture, making purification a real event, so many synthetic teams refine their process with columns or recrystallization.
Chemical Reactions & Modifications
This compound joins the list of premium starting points for transition-metal catalyzed reactions, particularly palladium-catalyzed cross-couplings like Suzuki or Sonogashira couplings. The iodine atom pops off with remarkable ease under these conditions, letting chemists plug in a wild variety of groups—alkynes, boronic acids, or even amines—at that position. The nitro group refuses to sit quietly, enabling reductions down to anilines or further transformations into azo dyes, nitroso compounds, or heterocycles. Both academic and industrial chemists exploit these modifications to build complexity and generate structure-activity relationships in drug discovery.
Synonyms & Product Names
Depending on retailer or region, 2-nitroiodobenzene also gets billed as o-nitroiodobenzene, 1-iodo-2-nitrobenzene, or 2-iodonitrobenzene. Catalogs sometimes refer to it by its registry number, but the structural description remains clear from any angle—iodine on carbon one, nitro on carbon two of the benzene ring.
Safety & Operational Standards
Lab safety protocols call for gloves, goggles, and a solid fume hood while handling 2-nitroiodobenzene. Skin contact or inhalation can kick up irritation, and the powder easily becomes airborne during transfers. Disposal pulls no punches; leftover samples or waste should meet requirements for halogenated organics with nitro substituents. Local and international standards such as REACH and TSCA regulate supplier declarations and restrict unauthorized use. Automatic fire suppression systems go hand-in-hand with proper chemical storage, since concentrated samples present a real combustion hazard above a certain temperature.
Application Area
A chemist who wants to introduce structural complexity in aromatic scaffolds tends to grab for 2-nitroiodobenzene. Drug discovery teams put it to work in screens for kinase inhibitors, building molecules that bind tight to metabolic enzymes. Material scientists explore it in the design of conductive polymers, exploiting its ortho-disubstituted ring to control electronic shifts along the chain. Crop scientists look for analogs that might target pests, taking full advantage of its substitution-ready skeleton for offshoot synthesis. Even specialty dye manufacturers use derivatives for boosting colorfastness and fluorescence stability.
Research & Development
The ongoing refinement of cross-coupling protocols in academic labs drives constant demand for high-quality 2-nitroiodobenzene. Recently, methods that use greener solvents and lower catalyst loads have cut waste and costs. These advances open the door for even smaller research groups to tinker with its structure. Multi-component assembly lines in medicinal chemistry hinge upon reliable supplies of ortho-nitroiodo aromatics, pushing R&D chemists to seek new, scalable syntheses and examine the limits of its reactivity. Some researchers aim to further reduce environmental impact by developing biocatalytic or electrochemical methods for direct ortho-iodination of nitrobenzenes, which would cut down hazardous reagents and improve atom economy.
Toxicity Research
Most of the available toxicity data for 2-nitroiodobenzene rests on acute exposure tests, showing moderate toxicity by inhalation or ingestion. Animal models suggest that metabolic breakdown liberates free iodine and nitro metabolites, each raising their own flags for organ toxicity. Chronic exposure hasn’t received as much thorough study, but the consensus points toward caution, especially for workers in the pharmaceutical supply pipeline or those in downstream processing plants. Waste management and effluent treatment receive extra scrutiny, given the compound’s potential to leach into groundwater and affect aquatic systems. A push for more transparent reporting of long-term exposure outcomes continues inside regulatory circles, aiming to protect both frontline workers and end consumers.
Future Prospects
Shifts in pharma and advanced materials bring both hype and caution to 2-nitroiodobenzene. With global demand for novel small molecules on the rise, suppliers look for more sustainable sourcing and recycling options for iodine-containing aromatic feedstocks. Green chemistry initiatives grow louder, pressing for alternative nitration techniques and low-impact manufacturing cycles. Intellectual property battles flare up in the arena of cross-coupling, pushing smaller players to innovate with patent-free routes and robust downstream purification schemes. In the long term, synthetic chemists expect the niches for ortho-nitroiodobenzene derivatives to keep expanding, fed by curiosity-driven research and applied demand for high-performance aromatics.
What is the chemical formula of 2-Nitroiodobenzene?
A Closer Look at 2-Nitroiodobenzene
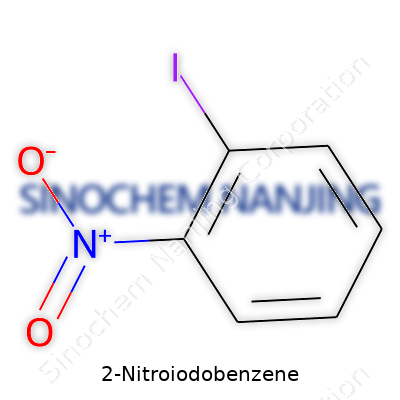
2-Nitroiodobenzene often finds a spot in research labs and organic synthesis projects. The formula, C6H4INO2, might not seem thrilling on first glance, but beneath those letters and numbers lies a chemical with a personality and purpose. On one end, the benzene ring gives it the aromatic stability, and on the other, nitro and iodo groups bring reactivity to the table.
Understanding the Formula: C6H4INO2
The structure sounds complex, but it breaks down pretty simply. Six carbons and four hydrogens form the classic benzene ring. Iodine steps in at the second carbon position, nitro (NO2) jumps onto the same ring right next to it.
Real chemistry nerds see this arrangement and know a couple of things: iodine is big and heavy, which changes how the molecule reacts compared to plain nitrobenzene. The nitro group, meanwhile, pulls electrons away, making the ring less eager for extra electrons. That influences everything from its color to the way it reacts when chemists want to build something bigger and more complicated.
Why 2-Nitroiodobenzene Matters in the Real World
Take it from anyone who’s handled coupling reactions or tried to make complex pharmaceuticals: a halogen and a nitro group working together open up real possibilities. In pharmaceuticals, new compounds often start from building blocks like this. It’s not rare to encounter a case where changing even one atom in a molecule can throw off a reaction, give an unexpected byproduct, or make the whole process sing.
In the classroom, students sometimes glaze over the difference between 2-nitroiodobenzene and its cousins, but real chemistry punishes that sort of laziness. Reactivity, solubility, and toxicity—all change with this one molecular switch. That matters in industries tracking everything from water safety to drug discovery.
Handling Risks, Using Smarts
What keeps me up at night is not just the elegance of a chemical formula, but what happens if people skip over safety. Nitro compounds can pack a punch if handled carelessly, and iodine brings its own challenges. Working with substances like this calls for solid planning—a fume hood, protective gloves, eye protection, and clear protocols. Always knowing what’s going into that flask makes life simpler. It keeps risks in check and delivers better science.
Improving Access and Sharing Knowledge
In my experience, newcomers often stumble on the basics before even tweaking conditions for a better yield. It helps to connect with others who have tried and failed before. Open science resources, shared databases, and community forums can level the playing field for everyone—no more hidden errors or overlooked hazards. The more openly scientists talk about the quirks of molecules like 2-nitroiodobenzene, the safer and more reliable chemistry becomes.
Looking Ahead in Chemical Research
Chemists innovate by building on solid groundwork. A clear formula like C6H4INO2 anchors a project, but then comes the art—finding new reactions, building smarter synthesis routes, and thinking critically about the impact on health and the environment. Every little insight stacks up. Choosing the right chemicals, treating them with respect, and sharing know-how—these habits fuel real progress in the lab and beyond.
What are the primary applications of 2-Nitroiodobenzene?
Understanding 2-Nitroiodobenzene in Day-to-Day Research
2-Nitroiodobenzene might sound like just another mouthful from a chemistry textbook, but it plays a noticeable part in labs that focus on developing new materials and medicines. It helps chemists piece together countless other molecules, acting as an important starting ingredient in larger synthetic puzzles. I’ve seen it pulled off shelves to kickstart complex experiments more times than I can count, and for good reason. With both a nitro group and an iodine atom stuck onto a benzene ring, it opens up doors for a range of chemical tricks. This isn’t just about filling test tubes—this work paves the way for real solutions in pharmaceuticals, materials design, and innovation in organic chemistry.
Direct Route to Key Pharmaceuticals
Medicinal chemistry turns to 2-nitroiodobenzene for its ability to add precision in building drug candidates. Chemists can swap out the iodine atom for other useful fragments, through reactions like Suzuki or Sonogashira couplings, and sometimes this tiny change leads to breakthroughs in cancer medicine, antibiotics, or central nervous system drugs. Companies have been able to develop building blocks for anti-inflammatory agents and antiviral compounds using building stones like this one.
Direct experience working with researchers has shown me that the versatility of 2-nitroiodobenzene keeps projects moving ahead when other chemicals hit a dead end. A well-placed iodine atom makes new bond-forming techniques easier, bringing new therapies closer to reality faster. This streamlines discovery and even helps keep costs in check for innovative treatments.
Supporting Advanced Materials and Electronics
Materials science teams keep 2-nitroiodobenzene on their list when looking to design organic electronics, dyes, and specialty polymers. With its useful combination of nitro and iodine, researchers can introduce new functions into polymers or organic semiconductors. Friendly rivalry between teams often pushes innovation, where a slight tweak in a material’s structure impacts performance in solar cells or sensors. This compound supports those tweaks and lets researchers push boundaries, whether the goal is brighter OLED screens or better thermal sensors. These changes start small, but ripple out into better gadgets and more efficient devices in our homes and workplaces.
Solving Bottlenecks in Synthetic Chemistry
Those of us working in synthetic chemistry appreciate tools that can get us over hurdles—2-nitroiodobenzene ticks this box. The iodine atom is a springboard for creating complex aromatic systems, which end up in everything from liquid crystals to agrochemicals. Chemists lean on it for protecting delicate molecules, upgrading reaction yields, and streamlining complicated reaction steps.
Of course, the wider world barely notices these unsung developments. Still, the impact surfaces indirectly—cost-effective herbicides, stronger plastics, and more stable dyes all tie back to these carefully chosen intermediates. Anyone searching for greener or more efficient chemistry will look for ways to swap in 2-nitroiodobenzene to cut waste and improve selectivity in reactions.
Looking Ahead: Safer and Smarter Applications
Every chemical has risk, and 2-nitroiodobenzene asks for respect in handling. Lab veterans stress strict protocols here, since its reactivity can pose headaches for the careless. That said, researchers keep searching for safer workarounds and new catalyst systems focused on sustainability and safety. Pushing for alternatives in metal-catalyzed reactions helps trim hazardous waste and protect those at the bench. If the chemistry community keeps insisting on greener reactions, this compound could play a starring role in cleaner, more responsible processes.
What safety precautions should be taken when handling 2-Nitroiodobenzene?
Understanding the Substance
Handling 2-Nitroiodobenzene means taking chemistry out of textbooks and putting it right onto the laboratory bench. Here’s the thing: this compound comes packed with real hazards. It mixes the reactivity of iodobenzene with the punch of a nitro group. Anyone who’s spent long hours in a lab knows that these features signal a need for sharp attention—especially when personal safety is at stake.
The Real Risks on the Table
Eye and skin contact top the list of risks. Nitro compounds and organoiodides have a reputation for causing irritation, and some can deliver harmful systemic effects if absorbed or inhaled. Spilled powder or broken glassware can shift a routine experiment into an emergency. Everyone on the team should treat ingesting or inhaling the dust as a genuine danger.
The Basics: Gloves, Goggles, Fume Hood
A thick pair of nitrile gloves doesn’t just keep hands clean; it protects the skin from direct exposure. Iodinated organics can sneak past ordinary latex, so skipping out on proper gloves risks more than a mild rash. Eye protection isn’t optional, either. Even a tiny bit of dust in the eye can bring searing pain and the threat of real damage.
The fume hood is more than a fancy cabinet. It’s your main line of defense against inhaling vapors or dust. Many researchers love to stretch lab space, but keeping open containers inside the hood gives peace of mind. The general air in a lab won’t clear away reactive fumes fast enough.
Solid Lab Habits Keep Trouble at Bay
Spill management kits, fresh lab coats, and regular housekeeping turn safety into a habit. If powder spills, keeping cleanup gear in arm’s reach beats a wild scramble for paper towels. I recall one incident: negligence with a clean bench led to a small powder spill that cost three hours of work and a restless night worrying about exposure. A simple wet wipe and a quick check for spills after every use have saved countless headaches since.
Waste Disposal—Don’t Cut Corners
Disposing of iodo-organics isn’t just a recommendation—it’s a rule that plays for keeps. Tossing this stuff in general waste risks exposing everyone in the lab. Dedicated, clearly marked waste containers for nitro and halogen compounds keep things safe, simple, and within the law. Experienced chemists triple-check waste lines, never assuming that the bottle labeled “Halogenated Waste” hasn’t been mixed up by someone in a hurry.
Label Everything. Then Double-Check.
Clear labeling doesn’t seem glamorous, but it’s crucial. Accidental grabs from an unlabeled bottle can trigger everything from minor mix-ups to full evacuations. Simple steps like dating the bottle and marking down hazards flag the dangers for new hands and veterans alike. Double-checking before use has stopped more than one preventable incident.
Emergency Readiness Can’t Wait Until Trouble Hits
Reviewing the safety data sheet before every order is more than paperwork—it forms the backbone of good lab culture. Anyone who’s scrambled for an emergency eyewash station understands the need to check the path, ensure the station works, and confirm that exits aren’t blocked.
Carrying out a practice drill, even just mentally, helps everyone react fast when it counts. Fast response can mean sparing a colleague a trip to the hospital.
Building Safety as a Mindset
Every individual in a lab faces the same expectation: watch out for yourself, and look out for your coworkers. Treat every container as if it’s as dangerous as the most toxic vial you’ve ever seen. Routine, careful handling doesn’t slow science down—it sustains it.
What is the proper storage condition for 2-Nitroiodobenzene?
Safer Labs Start with Smart Storage
Any chemist who’s handled 2-nitroiodobenzene a few times recognizes its quirky nature. This pale yellow, crystalline compound shows up in all sorts of organic synthesis work, particularly for folks who want to introduce iodine into aromatic systems. Because we’re dealing with an iodine compound, storage conditions play a bigger role than many learners realize.
What You’re Dealing With
Chemically, 2-nitroiodobenzene comes packed with both an iodine atom and a nitro group. That combination means you face more risks than with typical lab reagents. Both its sensitivity to light and tendency to decompose under the wrong conditions deserve respect. A look through the chemical’s safety data sheets and practical guides gives one clear picture: complacency leads to headaches or worse.
Risks Lurking in the Bottle
Let a bottle of 2-nitroiodobenzene sit on the edge of a hot, sunny windowsill, and you will soon get clumped solid and possible breakdown. Volatile organic chemistry projects stall not because the bottles leak, but because yellowed, decomposed solids ruin yields and wreak havoc in the waste bottle. Safety matters too. Iodinated compounds generally bring toxicity concerns, especially in unventilated storage. Many colleagues have stories of the one time someone left their bottle open on a bench, filling the workspace with an odd, acrid aroma.
Setting Up for Longevity
After years in a teaching lab and working with research groups, one rule stays true: for 2-nitroiodobenzene, cool and dark conditions always win. That usually means a sealed, airtight glass container, preferably an amber bottle, tucked into a chemical refrigerator. Room temperature can work for short-term stints, but if your schedule looks like most graduate students’, the bottle lingers longer.
Refrigeration isn’t about dropping things next to your lunch; you need chemical-grade refrigeration, kept dry and away from sources of ignition. Avoid keeping sources of acids and bases in the same fridge. Even vapor-phase cross-talk can start breakdown reactions. Desiccants help control moisture—silica gel packs tucked beside the bottle can prevent caking and water uptake.
No Corners to Cut with Security
Just as important as physical storage is labeling and documentation. Sharpie marks fade, so printed, solvent-safe labels with clear hazard pictograms prevent mishaps. Transfer operations get overlooked too. Pouring from one bottle to another in ambient light and humid air rapidly degrades the solid. Shield both your work area and the compound from direct light—portable shields help in crowded labs.
Solutions in a Busy World
Realistically, few labs have unlimited cold storage space or steam-cleaned darkrooms. Rotating stock and buying only what gets used in a semester or a project cycle prevents keeping old stock that’s more risk than value. Sharing cold storage among groups, posting reminders above storage lockers, and digital inventory alerts all encourage best practices.
If solid starts to look off—soft, clumpy, or oddly discolored—do not trust it in critical syntheses. Safely dispose according to your internal hazardous waste procedures. With the right routines, those headaches from degraded stocks and surprise safety audits don’t need to happen.
Final Thoughts
Smart 2-nitroiodobenzene storage shows up in every successful lab: cool, dark, dry, labeled, and frequently checked. The payoff is fewer failed reactions, safer workdays, and more reliable research.
How do you dispose of 2-Nitroiodobenzene waste?
Hard Truths About Laboratory and Industrial Waste
Most people never handle chemicals like 2-nitroiodobenzene. Out of sight, out of mind. For anyone who has spent time in a lab or on a production line, though, waste isn’t just trash. Each bottle, drum, or barrel tells a story about responsibility. It’s frustrating to see chemical waste treated with the same casualness as paper towels and coffee cups. Cutting corners can haunt us—physically, financially, and environmentally.
2-Nitroiodobenzene isn’t just some obscure molecule. It’s an aromatic compound with a heavy iodine atom carrying a nitro group. This stuff isn’t friendly to skin, lungs, or the water table. When I handled aromatic nitro compounds, I always worried about toxicity, persistence, and the long tail of liability. Keeping the waste isolated became second nature, because one careless splash can cost a lab days of work and put people at risk.
Downstream Effects We Can’t Ignore
Regulations exist for good reason. In the United States, the Environmental Protection Agency holds labs and factories to strict standards for hazardous waste like this. States with a stronger focus on environmental health, such as California, raise the bar even higher. Any 2-nitroiodobenzene leftover counts as hazardous without exception: you can’t dilute it, dump it, or toss it out with general lab debris. Mixing it down the drain might not sound like a big deal for an ounce of waste, but it’s a ticket to both groundwater contamination and lawsuits. Many nitroaromatics resist breakdown, building up in ways that nobody wants to explain to neighbors or children a decade down the road.
In my experience, talking to environmental safety officers and hazmat teams always brought one message: chain-of-custody matters as much as chemicals themselves. Waste that isn’t tracked puts the whole operation at risk, especially when dangerous substances are involved. If your safety protocol ends at the fume hood, you’re on thin ice.
Better Ways Forward: Containers, Contracts, and Courage
Dealing with 2-nitroiodobenzene waste means more than checking off boxes. Every bit needs containment in compatible, closed containers—no leaks, no shared space with incompatible chemicals. Using glass or HDPE plastic, sealed tightly and clearly labeled with all the right warnings, turns out to be the first line of defense. In my time, the best labs set aside a specific satellite accumulation area, away from clutter or heat, making sure nothing sits around long enough to become a mystery bottle.
The chain of responsibility doesn’t end at the lab door. Hazardous waste disposal contractors bring experience and infrastructure that most companies can’t match in-house. These firms handle segregation, transportation, and incineration or chemical processing, following federal and state rules every step of the way. Specialist incineration at high temperatures can break these molecules apart, reducing the worst risks. The cost of disposal often feels steep, but compared to the scale of potential fines or cleanups, it lands as a bargain.
Staff training saves more trouble than any spill kit. Seeing the real consequences of mishandled waste—whether in site tours, in news reports, or in regulatory citations—motivates everyone to follow protocols and respect warning labels. In my years with new lab technicians, walking them through hazardous waste logs and closing containers right every time set habits that stuck with them throughout their careers.
Prevention and Substitution: Smarter Chemistry on the Horizon
True responsibility includes looking upstream. Green chemistry practices—like using less hazardous reagents or scaling down experiments—cut down waste before it ever appears. I’ve seen researchers rethink entire syntheses to avoid stubborn, persistent pollutants. Substitution rarely feels quick or easy, but over time, it takes more pressure off the whole system.
2-Nitroiodobenzene isn’t just a test of compliance, but a reminder that every experiment leaves a footprint. Science can’t dodge its waste problem, so clear procedures, honest risk assessment, and steady vigilance form the backbone of any real solution.
| Names | |
| Preferred IUPAC name | 1-iodo-2-nitrobenzene |
| Other names |
2-Iodonitrobenzene o-Iodonitrobenzene o-Nitroiodobenzene |
| Pronunciation | /tuː ˌnaɪtroʊˌaɪədoʊˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 609-72-3 |
| Beilstein Reference | 1638758 |
| ChEBI | CHEBI:52244 |
| ChEMBL | CHEMBL3348925 |
| ChemSpider | 13078 |
| DrugBank | DB08325 |
| ECHA InfoCard | 100.014.234 |
| EC Number | 610-018-2 |
| Gmelin Reference | 823247 |
| KEGG | C19227 |
| MeSH | D016674 |
| PubChem CID | 69219 |
| RTECS number | QV7350000 |
| UNII | 0NJF8H9V2R |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C6H4INO2 |
| Molar mass | 233.02 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.96 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.9 |
| Vapor pressure | 0.00144 mmHg (25°C) |
| Acidity (pKa) | 12.08 |
| Basicity (pKb) | 11.10 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.673 |
| Viscosity | Viscosity: 2.21 mPa·s (25 °C) |
| Dipole moment | 2.25 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -5.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7.07E+03 kJ/mol |
| Pharmacology | |
| ATC code | null |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | Precautionary statements: P280, P261, P305+P351+P338, P301+P312, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-0-NA |
| Flash point | 113 °C |
| Autoignition temperature | 250 °C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 2-Nitroiodobenzene: "LD50 Oral rat 398 mg/kg |
| NIOSH | NA0450000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 16°C |
| Related compounds | |
| Related compounds |
1-Iodo-2-nitrobenzene 2-Fluoronitrobenzene 2-Chloronitrobenzene 2-Bromonitrobenzene o-Nitroaniline |

