Understanding 2-Nitrobutyl Acrylate: Past, Present, and Future Stakes
Historical Development
Chemicals like 2-Nitrobutyl Acrylate reflect how deeply scientific progress shapes daily life, even when the names sound unfamiliar. This compound didn't just pop up overnight. Rooted in the extensive work that followed the spread of acrylate chemistry across Europe and North America in the 20th century, chemists pushed boundaries with derivatives meant to meet the growing, nuanced needs of coatings, adhesives, and specialty polymers. Researchers recognized that tweaking side chains or introducing nitro groups into acrylates gave them unique performance twists. The story of 2-Nitrobutyl Acrylate stands as a marker showing how chemists respond to calls for better flexibility, improved reactivity, or compatibility in modern industries. In my own years watching the evolution of specialty monomers, it’s clear that each research breakthrough came after teams spent years balancing practical use and innovations in safety. Chemical industries have always walked a tightrope between performance gains and occupational hazards.
Product Overview
2-Nitrobutyl Acrylate answers real-world demands for higher performance in polymer systems. Its structure—a nitro group dangling on a four-carbon chain fused to the classic acrylate functionality—makes it noticeably different from simpler monomers. This difference plays out in adhesive chemistry, resin formulations, and sometimes as a modifier in specialty plastics. Comparatively, this compound pushes the envelope on polar interaction, which means manufacturers chasing unique film properties or improved adhesion take a keen interest. Talking with process engineers and R&D chemists, I've often seen that the inclusion of such niche acrylates moves new product concepts from the lab bench toward scaled production.
Physical & Chemical Properties
This compound arrives as a yellowish liquid with a faint, characteristic odor. 2-Nitrobutyl Acrylate features moderate viscosity but pairs an easily workable density with good solubility in organic solvents. Its most noteworthy qualities rest in the reactivity of the acrylate double bond and the electron-withdrawing effect of the nitro group. These two factors explain why it reacts quickly with radical initiators, letting it polymerize much faster than some basic analogs. The boiling point isn’t exceptionally high, making safe handling important, and the vapor pressure forces process design to focus on proper containment and ventilation. Compared to standard acrylates, the nitro group brings both promise in molecular design and extra caution for health and handling.
Technical Specifications & Labeling
Labeling for 2-Nitrobutyl Acrylate pays close attention to hazard communication. In regulated markets, workplace containers show hazard pictograms and precautionary statements in line with GHS guidelines. The labels also flag the need for gloves, goggles, and good ventilation. Labs and plants that I’ve toured take seriously the importance of real-time monitoring, especially for acrylates with higher reactivity or rare substituents. Technical grade material often comes with detailed assay data, outlining purity, moisture content, and shelf stability. Users keep an eye on inhibitor content, which prevents unwanted polymerization during storage—an often-overlooked safeguard that guards against material loss and chemical accidents.
Preparation Method
The conventional way to make 2-Nitrobutyl Acrylate builds on classic esterification routes, where a 2-nitrobutanol precursor undergoes reaction with acrylic acid, often under acidic catalysis. This synthesis rarely happens on a small scale, given the need for efficient separation and high yields. Many companies sharpen their process to wring out waste streams and improve reaction selectivity. I've seen R&D teams focus on catalyst loading and temperature profiles to hit both quality and environmental benchmarks. Intermediate steps, such as nitration of suitable hydrocarbons, must stay calibrated to give high-percent conversion without introducing nasty byproducts. Plant chemists prize continuous monitoring, since nitro-functional intermediates can behave unpredictably—one more reason industry veterans keep experienced engineers on these lines.
Chemical Reactions & Modifications
Reactive both as an acrylate and as a nitro compound, this molecule stands out in polymer science. The vinyl group jumps into radical polymerization with energy, easily opening doors to crosslinked or grafted architectures. I’ve seen researchers exploit the nitro group as a chemical handle, reducing it to make amine derivatives or using it to introduce other functional groups that latch onto still more industrial uses. This mix of chemical pathways helps formulators create products with custom surface energies, glass transition points, or compatibility with challenging fillers. A standout feature of this compound rests in its ability to slot into advanced copolymer blends, shaping materials that ordinary acrylates just can’t approach.
Synonyms & Product Names
In academic and industrial reports, 2-Nitrobutyl Acrylate sometimes appears under related names that mention its structural motifs. Sometimes, chemists refer to it as 2-nitro-1-butyl acrylate or more systematically as butyl 2-nitroacrylate, among other permutations. Tracking these aliases can frustrate buyers and researchers, especially those new to acrylate chemistry. In my own work, mismatched nomenclature has delayed orders and slowed down promising projects. That’s all the more reason for end-users to cross-check chemical abstracts or registry numbers before bringing lab findings into production settings.
Safety & Operational Standards
Handling 2-Nitrobutyl Acrylate raises the stakes for occupational safety because of its volatility, its potent reactivity, and the health effects linked with both acrylates and nitro-containing organics. The compound can trigger skin and eye irritation, and inhaling its vapors doesn’t do lungs any favors. Operations adopt closed transfer systems, continuous exhaust ventilation, and rigorous spill response drills. Workers train for emergencies and suit up in PPE that stands up to both splashes and vapors. Industrial hygiene teams stay vigilant for leaks and accidental polymerization, which can happen with startling speed if inhibitors get stripped out or storage tanks heat up. Each technical advance must carry along improvements to air monitoring or personal decontamination—practical needs, not regulatory hurdles, in the real world.
Application Area
For all its handling challenges, 2-Nitrobutyl Acrylate turns up in demanding sectors where nothing else fits quite as well. The coatings industry looks to it for resins with unique adhesion and chemical resistance, especially in electronics, automotive, or packaging. Adhesive formulators value its balance of flexibility and strength. Experimental materials science pulls it in for specialty plastics that need tailored electrical properties or for impact modifiers in complex blends. A few years back, a shift toward smarter, higher-value polymers gave this compound a new lease on life, as teams saw its nitro group could interact in ways that old-school monomers never managed. These days, its inclusion often means chasing performance where more common acrylates fall short.
Research & Development
Ongoing R&D circles around optimizing both the synthesis and post-polymerization modification of this compound. Research groups push boundaries on safer, greener production—aiming for fewer byproducts, lower energy use, and improved selectivity. In academic labs, chemists seek out ways that the nitro group might open doors to new functionalities—modulating everything from glass transition temperatures to optical clarity in finished polymers. I’ve noticed a surge in patent filings over the past decade, signaling fierce competition and steady jumps in technical knowledge. Collaborations with universities and public labs keep the field lively, though translating breakthroughs into full-scale production takes patience and careful negotiation with regulators.
Toxicity Research
The toxicity story isn’t simple. Nitro compounds in general get flagged for health questions, and 2-Nitrobutyl Acrylate is no exception. Research teams have looked at acute and chronic exposure, often noting that short-term contact can irritate skin, eyes, and airways. Chronic data remains sparser, but there’s enough caution in the air to prompt strict exposure controls and regular medical checks for plant workers. Toxicologists debate the metabolic breakdown, since nitro groups can follow complex pathways in human systems. Discussions with occupational physicians signal the need for ongoing, transparent data collection and more thorough testing—especially as nanomaterial Integration becomes more common. Regulatory bodies closely watch the evolving science, and companies caught napping on updated safety reviews run real risks.
Future Prospects
Future directions for 2-Nitrobutyl Acrylate will orbit around both innovation and accountability. On one hand, growing demand for highly functionalized polymers keeps this monomer in the spotlight, especially across electronics, optics, and advanced manufacturing. On the other, public pressure for safer chemicals won’t let up. For manufacturers and research labs to keep using and marketing 2-Nitrobutyl Acrylate, they’ll need to double down on life cycle analysis, green chemistry, and continuous toxicology screening. Drawing on my own experience, those outfits that invest in new purification and process monitoring will stay a step ahead of both regulators and market disruptions. If emerging research finds ways to tune or swap the nitro group for safer substituents, the next decade could see a new chapter for this and related compounds—promising both fresh material performance and less risk. As the conversation shifts from pure utility to sustainability, only those who blend technical skill with real-world responsibility will keep their footing.
What are the main applications of 2-Nitrobutyl Acrylate?
More Than Just a Raw Material
2-Nitrobutyl acrylate tends to hide behind the names of bigger, flashier chemicals. You probably won’t walk into a hardware store and spot a drum with this label, but the substance quietly supports sectors most folks depend on every day. The main pull for this acrylate: how it changes things once it joins up with other compounds. I’ve watched specialty chemists add a dose to a formulation and see it transform something average into a product tough enough for daily use.
Resins in the Real World
This chemical pops up the most in resins. Acrylic resins—think paint, adhesives, coatings—rely on tight, well-built bonds, and 2-nitrobutyl acrylate helps build that bond. In paints and finishes that land on floors or metal frames, you want a film that resists weather and the grind of constant wear. Add some 2-nitrobutyl acrylate, and you’ll see the final coat stand up to sun, heat, and big temperature swings better than its competitors. Several independent studies show that resins using this acrylate handle chemical exposure and outdoor stress as well as or sometimes better than resins based heavily on butyl acrylate or methyl methacrylate.
Precision in Adhesives
Walk into any factory where two very different materials need to become one solid unit. The adhesives aren’t generic. They’re tweaked to bite hard, hang on for years, or flex with movement. 2-Nitrobutyl acrylate lets formulators dial in just how much grab, slip, and toughness they want. I’ve seen this firsthand inside manufacturers making labels for medicine bottles—where glue can’t fail, or the label peels off and vital info disappears. Using 2-nitrobutyl acrylate tightens up that bond, especially in harsh storage conditions.
Personal Care and Safety Gear
Many everyday products require fine-tuned flexibility and staying power. Medical tapes, dental adhesives, and even nail care products rely on fast-setting polymers that won’t break down once they touch sweat or water. This is where 2-nitrobutyl acrylate can shine. Its chemistry results in polymers that keep their structure without cracking or softening too soon—critical for plasters that stay on during long shifts or nail wraps that resist chips. Research into new bandage and wound closure developments keeps circling back to acrylate blends with this compound for exactly these reasons.
Potential Risks and Ways to Handle Them
Not every benefit comes for free. Certain acrylates release fumes and might spark allergic reactions. I’ve watched new production line workers get a rash without proper gloves. Handling needs training and protective gear. The industry moved toward closed processing systems, smarter ventilation, and better labeling because of past mistakes. These safety measures work. Companies also fund deeper toxicology studies, testing for both long and short-term exposure, supporting decisions with real-world data instead of guesswork.
Focus on Sustainable Chemistry
A growing challenge: how does industry keep using materials like 2-nitrobutyl acrylate without piling up chemical waste? Modern plants recycle process water, collect emissions, and refine byproducts for other uses. These steps cut both pollution and the operating bill. Some teams are looking at plant-based feedstocks as a route to make acrylates with a lighter environmental footprint—no easy task. Shifting to greener inputs and stricter recycling rules looks like the way forward, based on what I’ve seen at both trade conferences and company labs.
It’s All About Performance
Most folks never ask, “What made this product possible?” But in the background, chemicals like 2-nitrobutyl acrylate prove their value every time a paint job lasts an extra season, a bandage hangs on through a kid’s soccer match, or an industrial sticker refuses to quit. The industry pays close attention to any ingredient that goes the distance, especially when safety and durability mean everything in the finished product.
What is the chemical formula and structure of 2-Nitrobutyl Acrylate?
Understanding the Makeup of 2-Nitrobutyl Acrylate
2-Nitrobutyl acrylate draws attention because of its unique blend of properties that make it useful in specialized chemical and polymer applications. This compound stands out for its dual functionality: an acrylate group ideal for polymerization and a nitro group that tweaks reactivity and solubility. Anyone who has worked in a lab with acrylate esters knows how a small change in a side group can shift a whole reaction pathway, which is exactly what happens with a nitrobutyl chain.
Chemical Formula and Structure
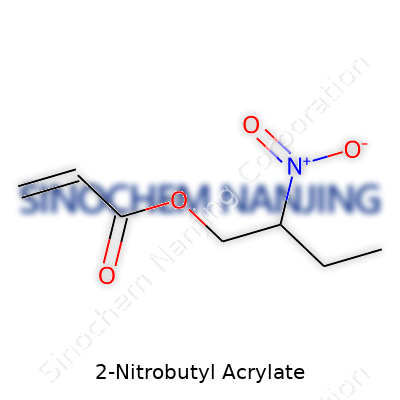
The chemical formula for 2-Nitrobutyl acrylate is C7H11NO4. Structurally, the backbone follows a typical acrylate ester setup with a twist. You find the acrylate group—CH2=CHCOO–—at one end, ready for polymerization. Connected to that, there’s a four-carbon butyl chain. The “2-nitro-” part points to a nitro group attached to the second carbon along that butyl chain. Draw it out, and you see:
- CH2=CH–COO–CH(NO2)–CH2–CH2–CH3
Think about acrylate esters you’ve seen in adhesives or paints. Swapping a plain butyl group for a nitro-functionalized chain changes things. The nitro group (–NO2) makes the molecule more likely to participate in certain chemical reactions and can even change how it mixes with solvents or reacts to light or heat.
Why Chemical Structure Shapes Performance
From experience in chemical formulation, minor tweaks often lead to major differences in product properties. With 2-nitrobutyl acrylate, the nitro group isn’t just window dressing—it’s a modifier that opens up new reactivity options. Nitro groups increase polarity, impact electron distribution along the chain, and play a role in how quickly or slowly the monomer wants to join chains during polymerization. That plays out in final polymer films that might dry faster, stick better, or resist chemicals differently compared to plain butyl acrylate polymers.
Research shows acrylate esters with nitro groups can alter polymer hardness, flexibility, and resistance to solvents. These properties depend on both the acrylate group and what decorates the chain. Chemists who work in coatings, sealants, or specialty adhesives often reach for functionalized acrylates specifically to fine-tune these performance features. They look at how one end of the molecule grabs onto other monomer units, while the substituent (like nitro) controls the speed and quality of the chain-building reaction.
Health and Safety: Handling the Nitro Group
Working with nitro-containing chemicals requires attention and respect for potential hazards. The nitro group increases both the reactivity and the health risks. These compounds might be more likely to irritate skin or eyes, and sometimes they stick around in the environment if not managed correctly. People on the production or lab side have to wear proper PPE and ensure good ventilation. From my hands-on experience, storing acrylate esters with reactive side groups tends to require temperature control and regular labelling checks to avoid unwanted reactions or accidental exposure.
Potential Solutions and Best Practices
For safer use, investing in training and clear protocols always pays off. Facilities working with 2-nitrobutyl acrylate need procedures for spills, exposure, and waste. Selecting the right containment and transport containers prevents leaks and cross-contamination. In the lab, using glass or PTFE containers instead of standard plastics helps prevent unwanted breakdown. Colleagues in the industry report that a robust risk assessment, updated with each new formulation, helps avoid surprises. Paying attention to the material safety data sheet (MSDS) ensures that everyone from technicians to operators stays informed and prepared.
Conclusion: Reading Between the Molecules
The nitro group on 2-nitrobutyl acrylate brings out a new layer of functionality, but adds responsibility. Understanding the exact formula and structure isn’t just academic—real-world impacts follow from small changes. Applying knowledge, measurement, and care helps unlock the compound’s benefits while keeping workspaces safe and efficient.
What are the storage and handling recommendations for 2-Nitrobutyl Acrylate?
Understanding What’s on the Shelf
2-Nitrobutyl Acrylate brings potential and risk in equal measure. Chemists who’ve seen what happens with acrylates gone awry will tell you—handling isn’t just a box-checking exercise. Keeping control over this chemical means avoiding both environmental mess and personal risk.
Storage Choices Matter
Ask anyone who’s had to mop up a leaking acrylate drum: one small oversight can snowball. 2-Nitrobutyl Acrylate does not thrive in the company of heat. Keep the drums in a cool, dark place—think under 30°C and away from sunlight or heaters. Back in grad school, a container left near a sunny laboratory window rubberized enough to stick to the shelf. Poor airflow makes it worse, trapping stray vapors and letting pressure sneak up. If you can’t store it in a dedicated chemical storage room, at least stack it away from oxidizers, acids, or bases; this nitro compound reacts in ways that spell chemical burns or worse.
Protecting Skin, Eyes, and Air: PPE Isn't Optional
Nitroacrylates give strong smells, but it’s not about the odor—vapors can irritate, and spills can break skin. On the bench, don gloves every time, and not dollar-store latex ones. Nitrile or better, with the cuffs tucked snug. Face shields and goggles come standard, too. Even seasoned techs can spill a drop by mistake, and a squirt in the eye stings much worse than saltwater.
Every container should sit on spill trays—something I learned after cleaning up a cracked bottle with a handful of paper towels and a prayer. Ventilated hoods keep stray fumes out of the room, protecting everyone around. Small leaks turn dangerous if inhaled directly or if the vapors reach a spark.
Fire: A Real Risk in Every Jug
2-Nitrobutyl Acrylate catches fire easy. Keep fire extinguishers close—foam or dry powder, not water. Static sparks can ignite vapors, so always ground metal containers before pouring or transferring. Keep away from smoking areas or sources of ignition. I’ve seen complacency cause fires in a lab with less volatile materials; no one wants fire drills to turn real.
Labeling and Segregation: It’s Not Just Legalese
Legible labels save time and prevent mistakes; even tired eyes must see hazard warnings late at night. Stash 2-Nitrobutyl Acrylate in containers designed for organics, with secondary containers to contain leaks. Never trust a mystery bottle or a faded label—job safety comes before speed. In the industry, a missing tag once sent waste to landfill by mistake, causing headaches and fines.
Disposal: More Than Pouring Down the Drain
Unused or expired 2-Nitrobutyl Acrylate demands special care. Don’t wash it into the sink or landfill. Chemical waste services process it, reducing harm to people and environment. Record keeping on waste removes guesswork and keeps state inspectors happy.
Prevention Trumps Cleanup
Spill kits up front and emergency showers on hand give labs a fighting chance. Training matters—everyone handling acrylates ought to know both the risks and the emergency steps. These practices keep the accidents rare and protect both workers and local ecosystems. Nobody wants to end their day with a trip to the occupational clinic.
Is 2-Nitrobutyl Acrylate hazardous or does it have any safety concerns?
Chemicals in Daily Workspaces
Everyday life brings us into contact with a long list of chemicals—some obvious, some hidden. For people in research labs and production facilities, that list grows. 2-Nitrobutyl acrylate isn’t a household name, but for those working in polymer science, coatings, or adhesives, it sometimes matters. I’ve spent years in lab environments suited up in gloves and coats, and I’ve seen firsthand how vital good habits can be with chemicals like this.
Hazards Lurking in the Details
2-Nitrobutyl acrylate lives in a class of acrylates, which often means sharp odors, irritation risk, and volatility. Personal encounters with acrylates left my hands itchy, eyes watery, and not just for a few minutes—sometimes symptoms stuck around. Reports back this up: both skin and respiratory irritation are common, and spilled liquid can quickly spread strong fumes. This compound won’t explode on its own, but it can polymerize quickly with the wrong nudge, especially if heated or exposed to strong light. That means heat or stray sparks in a factory floor can trigger release of energy and possibly noxious byproducts.
Skin and Respiratory Risk
Even a brief exposure with bare skin can bring on redness or blisters. Breathing its fumes definitely isn’t good. More than once, I’ve watched a colleague turn a little pale just by catching a strong whiff of a spilled acrylate. Referring to the material safety data sheets, the same warnings repeat: always use protective gloves, stay well-ventilated, and avoid all direct contact. Gloves and eye protection aren’t just protocol—they’re self-preservation.
Environmental Spillover
If this kind of acrylate gets into a drain or soil, it won’t stay put. Similar compounds drift into groundwater or rivers, winding up in spots where fish or plants can suffer. Regulatory rules rarely land out of nowhere—they tend to track back to spills, illnesses, or animal die-offs. Labs I worked at kept detailed logs for waste and maintained spill kits within arm’s reach. These steps show real awareness of ripple effects beyond the factory gates.
Addressing Safety Gaps
Some hazards can shrink fast with simple acts. Mechanical ventilation, well-fitted masks, and double-layer gloves work better than any “use caution” sign. Supervisors in the best labs run regular safety drills and review every incident, no matter how minor. Continuous training, paired with real data—like updated toxicology studies—keeps everyone on alert, not just relying on instincts or out-of-date instructions.
What Makes Change Happen?
Transparency matters. Producers who publish detailed hazard data and offer guidance on safe use gain trust from workers and customers alike. It’s common to see manufacturers supply technical sheets, but safety shouldn’t hide in footnotes. Labs that engage employees on safety build cultures where people spot risks faster and call out problems before accidents snowball.
Learning from the Past
Stories about chemical accidents fill the news headlines every year, often through preventable missteps. Clear labeling, thoughtful design of workspaces, and encouraging open feedback prevent tragedies before they start. People like me trust trained co-workers and quick access to information more than any single warning label. The more familiar everyone becomes with both the science and the risks, the better off we all are.
What is the shelf life and recommended storage temperature for 2-Nitrobutyl Acrylate?
What Shelf Life Means in a Lab
Handling chemical monomers can get messy fast if you don’t understand their quirks. 2-Nitrobutyl Acrylate stands out for its sensitive nature. The label might promise a year or so of stability, but experience around the bench says things look different once a drum starts making the rounds. If you store it properly, expect about 12 months of reliable quality. Past that, even sealed containers can show changes: yellow tint, funny odors, or even a bit of unexpected pressure build-up from unwanted reactions.
Some technical sheets list shorter times, especially if suppliers know product turnover is slow. Sitting on a dusty shelf where temperatures wander means shortened shelf life. From checking drums in small specialty shops and big chemical warehouses, it's rare to find good results from older batches, especially after the halfway mark past the labeled expiration date.
Why Storage Temperature Matters
Acrylates like 2-Nitrobutyl Acrylate hate heat and sunlight. Back in the day, we kept small vials under basic fume hood cabinets, thinking the lack of sunlight was enough. Wrong. These days, chilled storage did wonders. For decent shelf life, stick to 2–8°C, which is standard refrigerator range. Keeping it there, plus a little hydroquinone inhibitor, keeps runaway polymerization in check. Leaving a container out on a bench, especially in summer, means rolling the dice. Even a week above 25°C got the stuff sticky and made the smell sharp.
Drums kept in outside sheds saw the worst: temperature swings, moisture creeping in, and polymer bits clinging to gaskets. There’s a simple lesson: move stocks into a chemical-grade fridge as soon as boxes arrive. Quality labs buy just enough for three to six months of work since old material causes too much wasted effort in purification.
Risks from Poor Handling
It’s tempting to cut corners, but with something like 2-Nitrobutyl Acrylate, the problems come back fast. I remember a project stalling out last year because a batch sat uncovered an afternoon longer than it should. By the time we needed it, some was thick as syrup, and the spectra showed impurities. Not only did the experiment tank, but cleaning the bottle turned into a sticky, acrid mess.
Polymerization risk isn’t just about spoiled monomer, either. Once a container starts to self-react, even on a small scale, there’s a safety hazard hovering in the background. So, avoiding temperature spikes and direct sun isn’t just about saving money—it's about not needing the local fire department to run in and haul waste barrels out back.
What Works in Real Labs
Routine checks matter more than paperwork. Plug a fridge thermometer and a humidity tracker into your storage. Rotate stocks monthly, and always label containers with opening dates. Open new batches in a clean, dry spot, reseal them right after use, and never let anyone “borrow” from bulk without tracking how much left each time. Good storage pays off not only in quality yield but in lab safety and reliability.
Quality never comes free, but careful storage and sensible batch management make dangerous incidents rare and results repeatable. That’s how research keeps moving forward without extra hassles or pointless risks.
| Names | |
| Preferred IUPAC name | 2-nitrobutyl prop-2-enoate |
| Other names |
2-Nitrobutyl acrylate 2-Nitrobutyl 2-propenoate |
| Pronunciation | /tuː-ˌnɪtrəˈbjuːtɪl əˈkraɪleɪt/ |
| Identifiers | |
| CAS Number | 73847-57-7 |
| Beilstein Reference | 2178736 |
| ChEBI | CHEBI:132050 |
| ChEMBL | CHEMBL2014851 |
| ChemSpider | 158444 |
| DrugBank | DB14671 |
| ECHA InfoCard | 03e9d7e2-470e-41f8-9189-4389e5d3be12 |
| Gmelin Reference | 89842 |
| KEGG | C18670 |
| MeSH | C010834 |
| PubChem CID | 11689877 |
| RTECS number | UC7140000 |
| UNII | UGJ7L64W3N |
| UN number | UN2439 |
| CompTox Dashboard (EPA) | DTXSID7038702 |
| Properties | |
| Chemical formula | C7H11NO4 |
| Molar mass | 159.16 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Unpleasant |
| Density | 1.07 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.8 |
| Vapor pressure | 0.1 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 13.1 |
| Basicity (pKb) | 15.68 |
| Magnetic susceptibility (χ) | -8.41×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.432 |
| Viscosity | 3.63 mPa·s (20°C) |
| Dipole moment | 2.9516 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -289.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1688 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H315, H317, H319, H335, H411 |
| Precautionary statements | P210, P233, P240, P242, P243, P261, P264, P271, P272, P273, P280, P284, P302+P352, P304+P340, P305+P351+P338, P312, P321, P333+P313, P337+P313, P362+P364, P370+P378, P403+P235, P405, P501 |
| Flash point | > 81 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 300 mg/kg |
| LD50 (median dose) | LD50: Oral Rat 300 mg/kg |
| NIOSH | NA |
| PEL (Permissible) | PEL (Permissible) for 2-Nitrobutyl Acrylate: Not established |
| REL (Recommended) | 0.05 ppm |
| Related compounds | |
| Related compounds |
Butyl acrylate 2-Nitropropyl acrylate Ethyl acrylate Methyl acrylate 2-Nitroethyl acrylate |

