2-Nitrobutane: A Closer Look
Historical Development
Chemistry seldom stops with the basics. Decades ago, researchers began searching for efficient nitro compounds to support new synthetic challenges. The story of 2-nitrobutane developed as people moved past more familiar nitroalkanes. The compound didn’t catch as much limelight as its three-carbon cousin, 2-nitropropane, yet chemists in the 1930s and 1940s realized 2-nitrobutane offered its own set of advantages, especially for selective alkylation and as a base material in laboratory synthesis. Over time, manufacturers refined industrial production, finding improved nitration methods and working with regional regulatory bodies on safe work environments. As interest in green chemistry grew, fine-tuning the production steps to minimize waste and limit byproduct formation became a growing part of the conversation.
Product Overview
2-Nitrobutane stands as a clear, mobile liquid, best described as a mid-chain nitroalkane with the formula C4H9NO2. It brings a distinctive sweetness to its odor, which any synthetic chemist will recognize straight out of the flask. Its use focuses more on specialized synthesis or solvent functions than broad-based consumer applications. Most of the time, producers ship this chemical in drums intended for careful industrial use, always respecting the hazards tied to nitroalkanes. Labs working with 2-nitrobutane seek its particular reactivity profile. In some cases, it serves as a stepping stone toward novel heterocycles or as a reactant when other nitro compounds just can’t deliver the right selectivity.
Physical & Chemical Properties
2-Nitrobutane boils at roughly 117 °C and melts deep below room temperature. With a density near 1.01 g/cm³ at 20 °C, it feels heavier than many common solvents. Tucking away in the cupboards, you’ll notice the colorless, sometimes pale-yellow appearance. It dissolves modestly in water, interacts more willingly with organic solvents, and remains stable under dry, cool storage. Its vapor may irritate, so engineers secure tight systems and draw off fumes using proper ventilation. Chemically, 2-nitrobutane carries both acidic hydrogens at the alpha carbon and potential sites for reduction—unique levers for designers of syntheses or manufacturers scaling up a target molecule. Its balancing act between stability and reactivity marks its reputation in the toolbox of the organic chemist.
Technical Specifications & Labeling
Handling this chemical, users expect technical data that supports reliability and safety. Purity commonly surpasses 97%, with lower moisture content and minimal byproducts like 1-nitrobutane, which can complicate downstream reactions. Drums bear GHS hazard pictograms and carry UN numbers, providing a visual cue for flammability and toxicity. Quality-assurance teams run IR and GC analyses to confirm the product before shipping. Packing meets rigorous standards: high-density polyethylene barrels, corrosion-resistant steel containers, or, for research-scale operations, amber glass bottles. Clear lot numbers and expiration dates support tracking through inventory and regulatory audits, helping customers connect their batches back to full analytical documentation.
Preparation Method
Making 2-nitrobutane at scale starts by mixing butane or 2-butanol with a nitrating agent, throwing in nitric acid under pressure. Early research looked for ways to avoid dangerous byproduct explosions, so today’s engineers choose milder temperatures and control the reaction with careful monitoring. The process generates a mix of isomers—separation by fractional distillation or selective crystallization sorts out 2-nitrobutane from its more reactive cousin, 1-nitrobutane. Small labs sometimes favor tailored syntheses using alternate oxidants or even flow reactors, winding up with a safer, more predictable process. Advances in reaction engineering and continuous monitoring, like in-line gas chromatography, keep modern production far safer and more consistent than pioneers in the twentieth century could have imagined.
Chemical Reactions & Modifications
As a practical matter, chemists turn to 2-nitrobutane for its reactivity in both nucleophilic and electrophilic reactions. The alpha hydrogen, sandwiched near the nitro group, provides access to enolate chemistry, opening the door to C-alkylation or condensation reactions. In research, folks often explore reduction steps to crank the nitro compound down to primary amines, using hydrogen and nickel or catalytic transfer hydrogenation. The molecule acts as a bountiful platform for modification: halogenation at the beta position, substitution at the alpha carbon, or coupling with aldehydes to build more complex frameworks. 2-Nitrobutane also participates in select Michael-type additions and has value when someone needs an intermediate en route to pharmaceutical building blocks or new catalysts.
Synonyms & Product Names
In chemical catalogs, 2-nitrobutane pops up as sec-nitrobutane, 1-methyl-1-nitropropane, or more rarely by its systematic IUPAC name. CAS number 610-25-9 shows up in most procurement pages and regulatory filings. Some literature swaps out local names depending on region or historical context, but these core identifiers give buyers and regulators confidence they’re talking about the same compound each time.
Safety & Operational Standards
No one approaches nitro compounds lightly—2-nitrobutane included. Working with it, technicians focus on preventing inhalation and direct contact: gloves, goggles, and a working fume hood serve as the front line. Although it’s less volatile than other light nitroalkanes, its vapors can irritate airways and eyes. Safety data points to a low flash point, and experienced engineers practice careful drum handling and grounding to avoid static discharges. Spill response plans include absorbent materials, chemical-resistant disposal bags, and eyewash stations close by. Regulatory frameworks like OSHA and REACH guide workplace exposure limits and require reporting of storage volumes. Regular safety drills and updated training on chemical-specific fire and health hazards help keep manufacturing and research labs on the right side of both compliance and safety.
Application Area
2-Nitrobutane matters most in finely tuned chemical synthesis. Its use as a solvent brings low reactivity with many reagents yet a proven ability to dissolve diverse organic products. Some industries use it as a medium for extracting or processing sensitive compounds. In pharmaceuticals, the molecule’s chemistry supports synthesizing complex intermediates, sometimes providing a nitro group that will become an amine or another functional group after reduction. Paints, coatings, and specialty polymers occasionally start with nitro compounds like 2-nitrobutane before further transformation. In research labs, it provides a testbed for studies on nitro group behavior or for developing new green methodologies in organic synthesis.
Research & Development
Current R&D efforts branch out into greener nitration processes and new approaches to handling waste or boosting atom efficiency in production. Teams investigate safer catalysts and explore online analytics to cut down on off-spec batches and reduce energy consumption. From a synthetic standpoint, researchers chase ways to lengthen the carbon chain, attach new groups, or prepare novel heterocyclic compounds by leveraging the unique reactivity of the nitro moiety next to a butyl backbone. In pharmaceutical research, the potential for enantioselective alkylation opens up doors to new active pharmaceutical ingredients (APIs). Some academic groups tinker with modifications that allow 2-nitrobutane to participate in tandem or cascade reactions, shortening total synthesis times and lowering overall operational risks.
Toxicity Research
The health profile of 2-nitrobutane still leaves open questions. Studies show acute inhalation can irritate mucous membranes, but long-term exposure data remain scarce compared to more common industrial solvents. Animal trials suggest a profile similar to other small nitroalkanes, spurring regulatory authorities to press for more chronic and reproductive toxicity data. Workplaces lean on engineering controls, air monitoring, and personal protective equipment, backed up by medical surveillance for those regularly exposed. Ongoing tox studies focus on metabolic byproducts and routes of excretion in both animals and human cell models, hoping to better predict occupational and environmental risk with a higher level of confidence.
Future Prospects
As chemical producers face tighter regulations and growing pressure to shrink environmental impact, 2-nitrobutane sits at a crossroads. New synthesis routes emphasizing lower emissions, minimal hazardous byproducts, and renewable feedstocks hold a strong appeal. Researchers eye expansion into fields where selective nitroalkane chemistry or clean reduction to amines fills technical gaps that older chemicals can’t address. Looking ahead, application opportunities may grow in pharmaceutical building blocks, advanced coatings, or as intermediates for green chemical manufacturing. Better toxicity data and continued advances in process safety will decide how central a role 2-nitrobutane takes in the evolving world of organic synthesis.
What is 2-Nitrobutane used for?
Digging Deeper than Labels
2-Nitrobutane might not stand out at first glance. Many have trouble placing it outside of a chemistry set. The truth is, hidden behind this technical name is a substance that plays a real part in the chemical industry and beyond. Most folks don't realize how often their daily lives brush up against compounds like this—often invisibly.
The Chemistry You Don’t See
I remember the early days of my own work in a university lab, scraping labels off brown bottles and puzzling at names. 2-Nitrobutane caught my eye only because I kept noticing it on different shelves. It’s not as well-known as solvents like acetone, but it shows up quietly in several big-ticket industries.
Factories use 2-Nitrobutane as an intermediate—meaning it helps build other chemicals. Companies won't ship it as a finished product that lands in your home, but they depend on it to make substances that do. Its main job comes in as a building block for specialty chemicals, particularly in manufacturing pharmaceuticals, rubber chemicals, and dyes. Examining old chemical reaction logs, I kept seeing this compound’s name as a starting point before compounds picked up new atoms or got split into smaller pieces.
Behind the Scenes of Production
Not everyone enjoys the scent or presence of chemical plants outside town, but there’s a whole lot of planning behind the scenes before 2-Nitrobutane even leaves its drum. In manufacturing plants, engineers rely on its reactivity. Take the rubber industry, for example: 2-Nitrobutane gets mixed in to create antioxidants, which prevent tires and industrial rubber products from crumbling away under heat or stress. In pharma, it supports the making of complex drug molecules. It’s one of those invisible helpers—rarely named on ingredient lists, but present upstream in the supply chain.
Risk, Responsibility, and Future Choices
Working with nitro compounds demands care. People who have worked in small-scale chemical companies can share stories about the rigorous safety rules that follow anything labeled "nitro." 2-Nitrobutane shares this reputation, bringing the chance of fire, explosion, or health effects if handled poorly. Exposure can irritate the lungs, so companies put energy into training workers and supplying proper gear.
I’ve learned through conversations with plant managers and lab supervisors that environmental safety is also on their radar—waste from these processes can threaten local waterways. The push for green chemistry means looking for better substitutes that do the same job while being safer for workers and the earth. Efforts to tighten storage regulations and encourage the use of digital monitoring systems aim to catch leaks or spills before they hit the ground.
Is Substitution on the Horizon?
Some researchers want safer routes to the compounds 2-Nitrobutane currently provides, since reactive nitro substances pose risks. Academic labs are looking for alternative pathways that use fewer hazardous intermediates, which could lower both safety concerns and disposal challenges. Enzyme-based reactions or new catalysts might one day cut this compound out of some supply chains altogether. For now, folks in the know keep asking hard questions about necessity, risk, and safety practices—pushing the industry to balance production with health and the environment in mind.
What are the safety precautions when handling 2-Nitrobutane?
A Closer Look at the Risks
2-Nitrobutane isn’t a household name, but it pops up in many labs and some factories. People handling it soon notice the sharp, sweet odor that hints at trouble. Exposure can burn the nose and lungs, and even a tiny splash burns the skin or eyes. The lungs, in particular, don’t forgive much: vapors can spark coughing spells and, with enough contact, set off headaches or dizziness.
I once visited a small specialty chemicals factory and watched as a rookie poured a nitrobutane solution. He wore gloves, but skipped the face shield. One drop near his brow, and he learned the hard way: flushing chemicals from your face feels terrifying, and the threat of eye damage is real enough to change anyone’s practices. Workers who use it share a respect for proper shields, goggles, and nitrile or neoprene gloves—latex doesn’t cut it.
Why Ventilation Is Non-Negotiable
A poorly ventilated space becomes a trap for nitrobutane vapor. Since symptoms sneak up, nobody notices someone getting woozy until it’s too late. I’ve seen engineers retrofit their hoods with bigger exhaust fans just to keep these vapors in check. Even with minor tasks, no fume hood means no work. A splash-proof apron and disposable sleeve guards also help when pouring or transferring the chemical.
Fire is another fear. Nitrobutane starts to break down and release toxic gases in a flame. Static sparks or cigarettes can turn spills into emergencies. One chemist told me his old supervisor confiscated every lighter and match before a shipment arrived—no one argued with that call. Only spark-proof tools belong in these rooms, and every spill kit includes absorbent pads made for chemicals, not rags or sawdust.
Storage Isn't a Side Note
Unsealed drums raise alarms. Vapors leak, and even a little can crank up the risk of fire and accidental inhalation. All containers get double-checked and relabeled regularly. Good practice keeps them out of sunlight, away from heat, and stacked nowhere near oxidizers or acids. I’ve watched teams train on what to do if one falls—step away, alert safety staff, and handle nothing without the right respirator.
Labels from suppliers spell out every hazard. The best places display pictograms, so workers don’t gamble on memory. Sharing stories of mistakes—without shaming anyone—helps new hires take these dangers seriously. Lab managers encourage reporting even minor leaks or headaches; everyone trusts that getting help won’t get anyone fired for “wasting time.”
Emergency Readiness Matters
Reality check: showers, eyewash stations, and clear exits must stay accessible. Nobody moves barriers or wheels carts in front of them “just for now.” During annual drills, every worker must put on a respirator, not just talk about it. Well-designed signage reminds even veterans to wear every piece of gear, every time.
Companies with a good safety culture invest in ongoing education. They hold briefings on new research or regulatory updates, and keep quick-reference guides near each station. A few dollars on training can prevent a catastrophe nobody wants on their record. Safety with 2-nitrobutane never comes down to luck—and for those who work with it, that lesson sticks.
What is the chemical formula of 2-Nitrobutane?
Decoding 2-Nitrobutane: More Than Just a Name
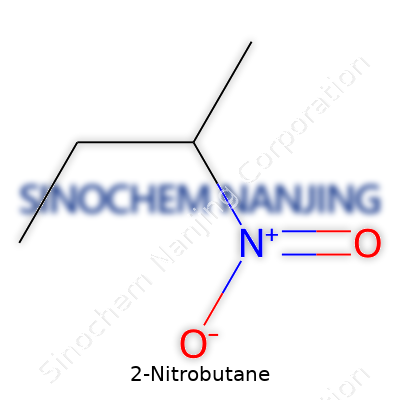
You don’t need a PhD in chemistry to recognize that every chemical we use carries a set of risks and benefits. 2-Nitrobutane is one of those names you might come across if you spend much time working near chemistry labs or read labels on industrial solvents. Its formula is C4H9NO2. Looking at the formula, you see four carbon atoms, nine hydrogens, one nitrogen, two oxygens. This arrangement points to a molecule where a butane backbone carries a nitro group on the second carbon.
Why Understanding This Formula Actually Matters
People sometimes overlook why chemical formulas are more than trivia. From my years teaching high school chemistry, I know there’s a practical value here. With this simple string of letters and numbers, chemists see how atoms are connected. That tells them how a molecule might react, what safety gear they’ll need, what kind of waste it might turn into, and how it should be stored. For 2-nitrobutane, the structure affects everything – from the risks it brings into a workspace, to how it should be cleaned up after a spill.
Often, 2-nitrobutane turns up as a solvent or as a building block in making other chemicals. Its formula isn’t just a set of rules for classroom quizzes; it shows how replacing a hydrogen atom on butane with a nitro group changes the compound’s properties. That nitro group (NO2) means you’re dealing with something more reactive, and not as simple to handle as plain old butane.
Real-Life Risks and the Role of Chemistry Knowledge
Working with chemicals like 2-nitrobutane, chemical engineers and lab managers don’t just memorize formulas for fun. The nitro group can make a molecule more prone to blowing up under the wrong conditions, so the formula becomes a warning sign. It’s not just about mixing chemicals together. It’s about understanding how every atom matters when you try to control risks and create products safely.
People often ask: “Why do we care about something like 2-nitrobutane?” The answer leans on public health and environmental stewardship. Mistakes in identifying or handling a compound, errors in reading a safety sheet, lack of understanding about how the structure pushes a molecule to be a fire hazard or a source of toxic fumes—those are the cracks where accidents start. That’s not fearmongering. Records from chemical safety agencies carry more than enough stories of what happens when formulas are ignored.
Common-Sense Solutions: From Label to Laboratory
Labs and factories can do simple things to prevent bigger problems. Staff need clear labeling and updated material safety data sheets. Rules for proper storage, personal protective equipment, and emergency spill plans make a difference. Bringing chemical know-how into workplace culture isn’t about chasing perfection, it’s about turning basics, like understanding the formula of 2-nitrobutane, into a habit. I’ve seen schools and small companies skip safety steps with “lesser-known” chemicals. Getting foundational facts right, such as C4H9NO2, anchors everything else that makes science both powerful and safe.
Is 2-Nitrobutane hazardous to human health?
Getting to Know 2-Nitrobutane
2-Nitrobutane doesn’t crop up much in everyday conversations. In reality, it’s a chemical that serves some particular uses in industry, mostly as a solvent or an intermediate in making other chemicals. You probably won’t find a bottle of it at the local hardware store, but there’s a good chance it ends up somewhere in the supply chain for paints or pharmaceuticals.
Health Risks: What We Know
Many folks get uneasy the moment ‘nitro’ appears in a chemical name, and for good reason. 2-Nitrobutane has earned attention from toxicologists, industrial hygienists, and organizations like the Occupational Safety and Health Administration (OSHA) due to its effects on health. Breathing its vapors or getting it on your skin brings risks. According to research seen in the International Agency for Research on Cancer (IARC), 2-nitrobutane can irritate the eyes, throat, and lungs. Headaches, dizziness, and nausea sometimes follow exposure. It can get absorbed through the skin, too, which adds another route for harm. Long-term animal studies raise concerns about cancer risk, so this isn’t a chemical you want floating around your living room.
Some labs report that high enough doses can damage the liver and kidneys. These organs work overtime to process foreign substances, so it’s not surprising they take a hit. Research into chronic exposure is still ongoing, but the evidence shows grounds for serious concern — especially for workers who handle this compound daily.
Straight Talk about Regulation
Both the Environmental Protection Agency in the United States and similar agencies around the globe set limits for workplace air concentrations of chemicals like 2-nitrobutane. The logic here is simple: it’s easier to prevent harm by keeping exposures low than to fix problems once they show up. For most people outside of industry, risk remains very limited—unless they live next to a facility that regularly uses or produces this compound.
Protecting Workers and the Public
Companies using 2-nitrobutane have a responsibility to keep workers safe. That includes proper ventilation, installing sensors that warn about vapor leaks, providing protective gloves and goggles, and training everyone in safe handling. Not every company is as diligent as it should be, so watchdogs and regular audits matter. The Material Safety Data Sheet (MSDS) offers clear steps to minimize risk, but having rules written on paper doesn’t always match what happens on the shop floor. That’s where unions and health advocates can step up, making sure workers know their rights and can speak up without fear of job loss.
Room for Safer Alternatives
Science keeps moving, and many organizations now push hard to replace hazardous solvents with greener, less toxic compounds. It’s not always easy or cheap, but nothing worth doing ever is. Some industrial chemists swap out 2-nitrobutane for less risky chemicals as regulations tighten and public expectations rise. The push for green chemistry, which favors substances that do less damage, offers hope that hazards like these won’t last forever in supply chains.
Looking Forward
Transparency about chemical hazards empowers people. If a factory works with substances like 2-nitrobutane, clear labels, regular training, safety improvements, and community outreach can tip the balance from risk to responsibility. No one wants surprise illnesses — especially for workers just doing their jobs. With enough focus on health, safety, and finding better replacements, the risk associated with 2-nitrobutane doesn’t have to be a life sentence for anyone.
How should 2-Nitrobutane be stored?
Understanding the Risks Around 2-Nitrobutane
Working in a lab, you come across all sorts of chemicals, and 2-nitrobutane isn’t one for taking chances. This compound gives off toxic vapors and has the potential to ignite under the wrong circumstances. Anyone who has spent their nights worrying if the fume hood's sash was down remembers why careful storage goes beyond following a checklist. Two decades dealing with nitro compounds taught me: vigilance is not optional.
Setting Up the Right Storage Space
Flammables always deserve a spot in secure, well-ventilated cabinets built for fire resistance—far from heat sources and incompatible substances. I remember seeing a cracked glass bottle after someone kept nitro compounds next to an old Bunsen burner. That incident ended with lab shutdown and a scolding from the safety officer. Oak or pine might sound sturdy, but only steel or specialized fiberglass shelving is truly up for this job.
Control Over Temperature and Light
A lesson learned: storing 2-nitrobutane at room temperature in a sunlit corner risks trouble. Even indirect sun can raise temps past safe limits, pushing containers closer to rupture or chemical decay. Chemically speaking, heat speeds up reactivity, and exposure to strong ultraviolet rays may promote breakdown or create unwanted byproducts. If you manage chemical storage, always keep compounds like this in a dark place or use opaque containment. A regular thermometer isn’t enough—digital monitoring helps catch unsafe spikes before damage happens.
Choosing Containers That Won’t Fail
Only tried-and-true glass or metal containers with tightly sealed lids should house volatile organics. I once saw a project run into disaster after a plastic vessel dissolved. 2-nitrobutane’s solvents chew through weak plastics, sometimes before you notice a slow drip. Reputable suppliers label compatible containers clearly for a reason—no one wants a leak spreading through the storage room.
Don’t Overlook Segregation
Chemistry isn’t a place for shortcuts. Acids, bases, strong oxidizers, and reducing agents have no business near 2-nitrobutane. One container slips into the wrong bin, and cleanup turns into an emergency. Paint bright stripes or use colored bins to create visual cues, so even someone in a hurry keeps chemicals apart. Simple barriers save real headaches down the line.
Labeling Saves Lives
Clear labels beat memory every time. Say you step away and someone else comes to grab a bottle, a good label with the chemical name, hazard warning, and emergency contact keeps confusion to a minimum. I've watched new lab techs scan shelves, picking up containers with only partial tags—one mistake can cause a ripple effect throughout the workplace.
Regular Checks and Training
Storage routines lose value if no one inspects the shelves. Monthly walkthroughs and quick checks on container condition and inventory catch small leaks before they become big problems. In-house training means everyone—interns to veterans—knows how to handle spills or move a leaking bottle fast and safely.
The Reality of Responsible Storage
Safety culture doesn’t come from rules; it comes from habits. Once I started treating every bottle the same way I’d want others to handle chemicals near me, accidents dropped off. 2-nitrobutane isn’t forgiving to mistakes. Only real discipline—built into how storage, labeling, and checks happen every day—prevents emergencies.
| Names | |
| Preferred IUPAC name | 2-Nitrobutane |
| Other names |
2-Nitrobutane sec-Butyl nitrite 1-Methyl-1-nitropropane |
| Pronunciation | /ˌtuːˌnaɪ.trəʊˈbjuː.teɪn/ |
| Identifiers | |
| CAS Number | 610-25-9 |
| Beilstein Reference | 1209248 |
| ChEBI | CHEBI:35806 |
| ChEMBL | CHEMBL1507651 |
| ChemSpider | 11710 |
| DrugBank | DB02157 |
| ECHA InfoCard | ECHA InfoCard: 100.008.772 |
| EC Number | 206-405-3 |
| Gmelin Reference | 82138 |
| KEGG | C08325 |
| MeSH | D009636 |
| PubChem CID | 8097 |
| RTECS number | EB5425000 |
| UNII | 02Z8C3J4FN |
| UN number | UN2608 |
| Properties | |
| Chemical formula | C4H9NO2 |
| Molar mass | 89.09 g/mol |
| Appearance | Colorless liquid |
| Odor | Unpleasant |
| Density | 0.883 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 0.97 |
| Vapor pressure | 2.79 mmHg (at 25 °C) |
| Acidity (pKa) | 11.0 |
| Basicity (pKb) | 15.20 |
| Magnetic susceptibility (χ) | -12.10 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.401 |
| Viscosity | 1.35 mPa·s (20 °C) |
| Dipole moment | 4.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −127.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1682 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P304+P340, P312, P403+P233 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 40 °C (104 °F; 313 K) - closed cup |
| Autoignition temperature | 474 °C |
| Explosive limits | 1.8–8.2% |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 640 mg/kg (oral, rat) |
| NIOSH | NIOSH: EO1400000 |
| PEL (Permissible) | PEL: 100 ppm (350 mg/m³) |
| REL (Recommended) | 16°C |
| IDLH (Immediate danger) | 140 ppm |
| Related compounds | |
| Related compounds |
Nitromethane Nitroethane 1-Nitropropane 2-Nitropropane 1-Nitrobutane |

