2-Nitrobiphenyl: Legacy, Utility, and Challenges
Historical Development
Chemists stumbled across biphenyl chemistry during the mad dash of nineteenth-century dyestuff research. Researchers first made 2-nitrobiphenyl by nitrating biphenyl itself, finding a place for it among the arms race of synthetic intermediates. While coal tar chemistry drove early motivation—Britain and Germany both hoping to outdo one another in colorants—nitroaromatics like this unlocked a deep well for researchers to draw on in pharmaceuticals and advanced materials. Scientists soon realized these tiny tweaks to structural orientation, like nailing a nitro group at the ortho position of biphenyl, didn’t just change the molecule’s color. Instead, it flipped its chemical temperament, offering a springboard for hundreds of reaction schemes.
Product Overview
2-Nitrobiphenyl stands out as a pale yellow, crystalline powder. At first glance, the simplicity of its structure hides that this compound sits among the cornerstones of modern synthetic organic chemistry. Chemists count on it not just as an intermediate, but as a reference molecule to explore aromatic substitution patterns, and as a source material for more elaborate heterocyclic rings found in today's pharmaceuticals and pigments. Manufacturers consider both technical grade and higher-purity research samples for different purposes, underpinning their selection with concern for contaminants that could tangle up sensitive downstream chemistry.
Physical & Chemical Properties
The compound’s melting point usually registers between 68 and 70°C, which makes for easy handling in most lab benches. It dissolves only sparingly in water, but goes into a range of organic solvents—especially those that handle aromatic compounds well. Structurally, the nitro group at the 2-position of the biphenyl backbone takes its toll on electron distribution, giving the molecule a unique reactivity compared to its isomers or simple biphenyl. This influences not only reactivity in further transformations, but also how the compound partitions between different phases, a critical detail for both process design and environmental fate considerations.
Technical Specifications & Labeling
Producers typically sell 2-nitrobiphenyl with a stated assay of at least 98%, though research labs can score material at even higher levels of purity. The packaging directs handling—airtight containers guard against unnecessary exposure or degradation. Each shipment gets labeled with batch numbers, hazard statements, and the required GHS symbols. Chemists expect access to detailed safety data sheets and certificates of analysis, which detail not only purity but also byproduct content, heavy metal traces, and residual solvents according to region-specific regulations like REACH or TSCA.
Preparation Method
Traditional synthesis of 2-nitrobiphenyl turns to the nitration of biphenyl, using a nitrating mixture—typically a blend of concentrated nitric and sulfuric acids—at carefully controlled temperatures to tame selectivity. Modern methods may switch to milder nitrating agents or tweak solvent systems to boost yield and minimize environmental impact from acid waste. Scale-up developers want greener options, so researchers weigh catalytic and electrochemical alternatives that generate less hazardous waste and operate under milder conditions. The challenge: balancing selectivity for the ortho-nitrated product amid the temptation to make unwanted isomers or tar solids.
Chemical Reactions & Modifications
The nitro group, fixed at the ortho position, readies the molecule for further chemistry. Chemists treat this scaffold as a springboard for reductions to 2-aminobiphenyl or as a precursor for heterocycle formation via cyclization. The compound’s reactivity pattern means that organometallic substitutions and metal-catalyzed cross-coupling reactions run cleanly, especially since the nitro group both directs ortho-substitution and behaves as a good leaving group under certain transformations. For materials scientists, this property opens doors to new ligands, dyes, and custom-built organic frameworks. The legacy continues in drug discovery: simple chemical tweaks to 2-nitrobiphenyl shape molecules that find use in anti-inflammatory or anticancer leads.
Synonyms & Product Names
Folks in the lab may know 2-nitrobiphenyl by its synonyms like “2-nitro-1,1’-biphenyl” or “o-nitrobiphenyl.” Trade literature and chemical catalogs sometimes list older names or refer to registry numbers like CAS 86-52-2. Academic scientists, industry buyers, and compliance officers all juggle these names, which can add confusion, so listing each alias on safety documentation curbs mix-ups—a small detail, but one that prevents costly mistakes in procurement and storage.
Safety & Operational Standards
Nobody should downplay chemical safety—the nitro group brings real risk. Experience in the lab quickly teaches that improper handling of nitroaromatics causes trouble, both for acute exposure and long-term risks. 2-nitrobiphenyl may absorb through skin, hit the nervous system, or damage the liver and kidneys with chronic exposure. Inhalation of dust or contact with eyes require quick, practiced response. Engineering controls like fume hoods, gloves, and splash protection are non-negotiable. OSHA and EPA provide regulatory frameworks, but in practice, the most powerful safety tool remains continuous training, routine monitoring, and a culture that values proactive reporting over silence or bravado. Any process using 2-nitrobiphenyl gets built with secondary containment, spill control measures, and clearly marked emergency protocols.
Application Area
2-Nitrobiphenyl’s chemical personality makes it valuable well beyond the basic research world. The dye industry tuned it for decades to fine-tune azo pigment properties. In pharmaceuticals, the compound steps in as a modular intermediate, lending itself to quick transformation into bioactive compounds. Chemical engineers turn to it in developing custom ligands for metal complexation or in building advanced organic electronic materials. These days, specialty polymers, sensor development, and organic light-emitting diodes (OLEDs) provide new runs on the ladder for this unassuming molecule. Researchers also use it as a model substrate when probing new catalysts or mechanistic pathways for aromatic substitution and reduction.
Research & Development
Academic groups and industrial R&D alike still turn to 2-nitrobiphenyl when sharpening their methods for selective reductions, directing group-mediated transformations, and cross-coupling innovation. It appears in dozens of studies focused on the Suzuki–Miyaura cross-coupling, C–H activation, and metal-free coupling methods. Companies value it especially when optimizing greener synthetic routes, reducing waste, or shifting away from precious metal reagents. Forward-looking researchers also tap this compound in computational modeling of aromatic reactivity, using it as a benchmark for predicting selectivity and new reaction development.
Toxicity Research
Safety evaluations reveal sobering risks: animal model studies and occupational medicine literature stack up considerable toxicological concerns. Repeated contact or ingestion brings risks of methemoglobinemia and organ system toxicity, and while 2-nitrobiphenyl itself has not been widely classified as a high-level carcinogen, related nitroaromatics show mutagenic potential. Ongoing research tracks biological breakdown pathways, the enzymes involved in nitroreduction, and the fate of this compound in soil and water. Ecologists keep an eye on environmental persistence, and regulators consider thresholds for allowable exposure, especially for workers in manufacturing or bulk processing. The move to greener chemistry doesn’t just mean better yields or less waste—it also pushes for safer alternatives, or at least mitigation of long-term toxicological risk.
Future Prospects
Even after a century, new uses for 2-nitrobiphenyl surface every year. As demand for specialty chemicals, fine electronics, and engineered pharmaceuticals climbs, the molecule finds roles both as a feedstock and as a proving ground for safer, more efficient chemical technology. Synthetic chemists keep whittling away at process intensification—using continuous flow, less hazardous nitration strategies, and improved waste recovery—to keep the economic and environmental footprint in check. Researchers in the fields of green chemistry and sustainable materials watch 2-nitrobiphenyl as both a challenge and an opportunity. They look for biodegradable derivatives, functional polymers with new electronic properties, and health-protective measures rooted in a century’s worth of toxicology data and experience. The hope is that smart engineering, open data, and cross-disciplinary collaboration will keep the promise of this classic intermediate alive, without losing sight of the hard-learned lessons tucked in every safety data sheet and incident report.
What is 2-Nitrobiphenyl used for?
A Closer Look at 2-Nitrobiphenyl’s Purpose
Growing up in a family full of scientists, I learned early on that strange-sounding chemicals often crop up in places most people never expect. Take 2-Nitrobiphenyl, for example. With its long name and chemical structure that seems straight out of a textbook, this compound plays a role that runs deeper than many realize. Folks in the chemical industry or environmental health fields may recognize it as part of a wider universe where industrial chemistry meets real day-to-day influence.
Industrial Uses That Raise Questions
Most of the use of 2-Nitrobiphenyl links back to how it helps make other chemicals. In the manufacturing world, you find it working as an intermediate—meaning it sits in the middle of chemical processes, not as a final product but rather as a step toward something else. It helps build dyes, which end up on the clothes you wear or the books you read every day. Dye manufacturing relies on strong and consistent chemical reactions, and 2-Nitrobiphenyl provides that solid foundation.
Besides dyes, some labs use it to bring about specific organic reactions because its structure makes certain chemical changes easier to control. By acting as a building block, it influences what kind of products get made, especially in the world of specialty chemicals. You also see a footprint in research. Scientists working to understand how various biological systems respond to chemical stress have used it in studies tied to toxicity.
Why Health and Safety Can’t Be Ignored
Learning about the uses of a compound doesn’t mean ignoring its downsides. For example, the International Agency for Research on Cancer (IARC) lists 2-Nitrobiphenyl as a possible human carcinogen. Back in the 1980s, when my aunt worked in an environmental lab, she’d tell stories about strict safety rules around handling nitro-compounds. There’s good reason for that. Workers who used to make dyes a few generations ago paid the price in health problems before anyone sounded the alarm.
Today, industries take a much more careful approach. Factory floors rely on closed systems and air controls that keep exposure far below what workers suffered a century ago. Studies show that careful engineering and good protective equipment keep risks in check, even if complete elimination stays out of reach. Responsible companies now track how even low levels might affect both people and the planet, aware that nothing operates in a vacuum.
Finding Smarter Answers
It’s never enough to just talk about safe use—it takes effort. This means investing in research for less toxic alternatives, and using advances in chemical engineering to limit waste and exposure. Modern labs often run simulations before they put a new ingredient into full-scale production. Government agencies push companies to submit thorough safety data, and some demand tracking right up the supply chain.
Local communities benefit when information flows both ways. Sharing environmental data helps build trust and lets folks decide how close is too close for comfort. Fewer surprises mean better public health outcomes in the long run. Schools and training programs now expect students to understand not just how to use chemicals like 2-Nitrobiphenyl, but why the safest road remains the best one—even if it takes more time or money up front.
Looking Toward Tomorrow
It’s easy to focus on what something is used for and lose sight of its broader impact. The story of 2-Nitrobiphenyl keeps evolving as new technology arrives, making it possible to both harness industrial power and avoid the worst side effects. From safer workplaces to greener labs, real progress comes from folks who ask tough questions—and insist on better answers.
What are the safety precautions when handling 2-Nitrobiphenyl?
Why Take 2-Nitrobiphenyl Seriously?
Chemicals like 2-Nitrobiphenyl might look harmless on paper, but anyone who’s spent time in a lab knows how easily things can go sideways. This compound isn’t one of those substances you can shrug off—there’s research linking it to cancer risk, and chronic exposure can be a nightmare for your health. The scientific facts alone make a strong case: animal studies and occupational reports tie it to bladder cancer, and even small spills can pack a punch if you’re unprotected. There’s no room for improv when your safety’s on the line.
Protecting Yourself Starts with Proper Gear
Cotton lab coats won’t cut it with 2-Nitrobiphenyl. Choose a chemical-resistant lab coat or apron, and keep your sleeves pulled down. Splash-proof goggles go over your eyes—even a tiny drop can do damage. Nitrile or neoprene gloves work well against penetration, and double-gloving adds an extra layer if you’re nervous about a tear. Always toss gloves after a session; reusing puts your health on the line.
Anything more than a whiff? You need a mask rated for chemical vapors. If you notice any spill or suspicious powder, immediately grab a proper respirator—don’t just turn your face away.
Keep It in the Fume Hood
I've learned not to trust open benches for anything volatile or dusty. Run all transfers and mixing inside a certified fume hood. Make sure the airflow’s up to spec before starting your experiment. Even if you’re only working with a few milligrams, fumes or dust escaping into the room can quietly add up. Tape down the sash and check local exhaust rules—cut corners here, and you’re risking more than your own health.
Spill Response Isn’t Just About Cleaning Quick
Spills set off alarms for a reason. If something escapes, evacuate everyone not wearing the right gear. Use absorbent pads or powder made for chemical spills. Keep a dedicated bin for contaminated material and tools, and never throw cleanup waste in regular trash. Alert your supervisor and follow up with the environmental health office. Even the smallest mishap deserves a full write-up, so lessons get learned and repeated mistakes get curbed.
Your Body Knows—Listen Early
It’s not only about what happens after big exposures. Long-term effects sneak up when people ignore small symptoms: headaches, rashes, dizziness. If you feel off, stop working and get checked—trust your instincts, not just safety data sheets. My own experience around hazardous chemicals taught me that nagging coughs or headaches can mean the start of something worse.
Wash any exposed skin with soap and lots of water. Don’t touch your face or snacks until you’re certain your hands are clean. Shower and change out of work clothes before heading home, so you don’t bring anything nasty back to your family.
Training: The Most Overlooked Precaution
Everyone likes to think they’re careful, but formal training beats caution alone. I learned more from hands-on safety demonstrations than from reading the manual. Practice those monthly emergency drills, and run “what-if” scenarios with your team—if you stumble in practice, better there than in real life. Report near-misses. Share practical tips and keep the dialogue open: safety belongs to everyone in the room.
Finding Solutions for a Safer Workspace
Sometimes facilities rely on the bare minimum—a hood, gloves, instructions taped to the wall. Build a culture where people actually care. Install up-to-date safety showers, eye wash stations, and invest in modern PPE. Set up routine air quality monitoring. Work with credible suppliers who guarantee purity and labeling, since accidents happen far more often with poorly labeled samples.
Handling 2-Nitrobiphenyl is ultimately about respect—for yourself, your coworkers, and the science. Real safety means not settling for shortcuts or assuming it will “be fine this time.”
What is the molecular formula and weight of 2-Nitrobiphenyl?
The Core Details
2-Nitrobiphenyl stands out in both research circles and industrial labs. Its molecular formula—C12H9NO2—lays the foundation for all further exploration. This molecule weighs in at 199.21 g/mol. Looking at that formula, you get two benzene rings, fused with a nitro group tucked into the second position. That's more than trivia for organic chemists; it sets up everything from reactivity to environmental fate.
Why Knowing the Formula Matters
Experience in the lab underscores the headache that follows confusion between isomers. Swapping a substituent from one ring to another might seem minor on paper but transforms properties, hazards, and regulatory status. 2-Nitrobiphenyl, for instance, is not just another biphenyl derivative. Put that nitro group in the para or meta positions, and you shift reactivity and toxicity profiles. Molecular weight and formula serve as anchor points for everything downstream: reaction planning, safety data collection, and even procurement protocols.
Real-World Impact
Factories and research groups use 2-Nitrobiphenyl in dye production, pharmaceutical syntheses, and as a reference standard. In my own work with aromatic compounds, small changes—sometimes as minor as a single atom, sometimes just its position—turn a familiar molecule from benign to toxic. Regulators track these structures closely. 2-Nitrobiphenyl’s chemical relatives, including certain isomers, have flagged associations with carcinogenic risk, prompting strict controls in workplaces across the globe. This isn’t paranoia—health studies have found links that can’t be ignored once you dig deep into the toxicology literature.
Data-Driven Attention and Solutions
Reliable facts keep people safe. The National Institute for Occupational Safety and Health has flagged nitrobiphenyls as potential hazards, drawing attention to necessary air quality measurements and protective practices. Safety data sheets depend on accurate molecular details, right down to the decimal on that weight measurement. A misplaced figure can lead to poor ventilation setups or insufficient personal protective equipment.
My time working in chemical inventory systems made one thing clear: databases need constant housekeeping. Scanning barcodes won’t help if the underlying molecular weights or formulas were punched in wrong a decade ago. Cross-referencing trusted resources like PubChem or peer-reviewed literature, and teaching team members the difference between isomer names, clears up confusion that can ripple through a company for years.
Looking Ahead
Mistakes from ignoring the fundamentals—mixed-up isomers, rounded molecular weights, missing hazard alerts—won’t just slow research down; they can put lives at risk. Clear education around nomenclature, practical training on how structures influence properties, and good habits in double-checking formulas set organizations up for success. Technology now offers automatic cross-checks and flagging systems, but nothing replaces a culture that trusts science enough to sweat the small stuff.
How should 2-Nitrobiphenyl be stored?
Understanding the Real Dangers
Anyone who’s spent time working in a laboratory knows safety rules are written in blood—usually metaphorically, sometimes not. 2-Nitrobiphenyl comes with its own set of risks. The compound isn’t just another yellowish powder sitting on a shelf. Research shows long-term exposure links to cancer and unintended inhalation can cause headaches or liver trouble. Back in school, we learned to respect toxics: don’t touch, don’t breathe, and definitely don’t eat. That simple attitude still applies.
Why Location Matters
Proper storage starts with picking the right spot. Direct sunlight warms packaged chemicals faster than most realize—raise the temperature, and you risk breaking down 2-Nitrobiphenyl or, worse, causing a small fire. Sticking the chemical in a dark, stable environment prevents these problems. My old chemistry teacher locked nitro compounds inside a climate-controlled cabinet, no air currents, and clear labels plastered everywhere. Simple and effective.
Keep it Sealed, Keep it Contained
Once, while interning at a university, I saw the mess made by a cracked lid. Powder drifted across a shelf, and cleanup took half the afternoon. 2-Nitrobiphenyl needs glass containers—polyethylene can crack, and certain plastics don’t play well with nitro groups. Always check the lids. If the seal fails, vapors escape, bringing all the associated health risks. Chemical-resistant tape over the seal gives extra reassurance. If kids or unauthorized folks have access to storage spaces, consider locking cabinets. Mistakes at home can become disasters in labs or classrooms.
Label Everything
Clear labeling sounds simple, but plenty of people skip details. I’ve seen jars marked with a single green dot. That's a great way to start a guessing game with something toxic. For 2-Nitrobiphenyl, use labels that include the full name, hazard symbols, date of receipt, and storage temperature guidelines. Emergency crews look for legible information during accidents; don’t make their jobs harder. GHS labels, OSHA color coding, and simple language keep everyone on the same page.
Temperature and Humidity Control
Heat speeds up deterioration. Humidity causes powders to clump together or, worse, react by accident. I keep a thermometer and humidity meter taped to the cabinet door at home. For 2-Nitrobiphenyl, 20–25°C is a good bet, closer to the lower end if you can manage. Never store next to strong oxidizers or acids, or next to cleaning supplies for that matter. A spill on nearby bleach could trigger chemical reactions in seconds.
Disposal and Emergency Plans Keep You Ready
Storage doesn’t end with placement on the shelf. At every site I’ve worked, staff reviewed chemical spill drills once a quarter—where to find the shower, who calls the emergency number, where to grab gloves and goggles. Waste builds up, and old 2-Nitrobiphenyl poses more risks than fresh stock. Follow local hazardous waste rules. If anything spills, evacuate fast and let a trained team handle clean-up.
Respect for Protocol Keeps Everyone Safe
Plenty of errors trace back to overconfidence or cutting corners. Routine can set in, but chemical safety asks for constant respect. Up-to-date material safety data sheets, organized inventory, and honest talk about risks keep accidents rare. Looking after hazardous chemicals depends on habits, vigilance, and remembering the consequences that come from looking the other way.
Is 2-Nitrobiphenyl hazardous to health or the environment?
The Substance at a Glance
2-Nitrobiphenyl is an industrial chemical, mainly getting used in research and, on occasion, in dye production. Out in public, most people would never run into it, and for good reason. It never comes up in household products or food. The main set of folks who face direct exposure are workers in chemical labs or at plants that manufacture specialty chemicals.
The Health Risks of 2-Nitrobiphenyl
Handling 2-Nitrobiphenyl isn’t a game. Scientists flagged it as a possible cancer-causing chemical. The International Agency for Research on Cancer (IARC) classified it as a Group 2B carcinogen, which means enough animal studies raised suspicion to treat the substance with a heavy dose of caution. Studies in rats and mice show long-term exposure leads to tumors in the bladder and liver. There’s not enough solid data on what happens to humans after accidental or repeated exposure, but with cancer worries in animals, nobody jumps at the chance to take that risk.
There are also short-term effects. Breathing dust or touching 2-Nitrobiphenyl might cause skin irritation, coughing, or sore throats. The chemical sneaks into the body easily through the skin, lungs, or digestive system, so glove and mask use turn into essentials, not just recommendations.
Environmental Threats
Chemicals don’t just stay where they land. If 2-Nitrobiphenyl makes its way into wastewater or spills out during production, the risk grows well beyond the factory gate. The molecule breaks down slowly in soil and water, and that means it can hang around for years, moving up the food chain or leaching into drinking supplies. Fish and aquatic plants take up these compounds and pass them along to larger animals—including, eventually, people.
Personal Experience and Lessons Learned
While working as an environmental reporter, I’ve seen similar compounds cause harm when chemical oversight slips. A single leak somewhere upstream, and suddenly there’s an uptick in strange wildlife deaths and stories from families about rashes or odd smells in tap water. These chemical traces aren’t the main event for most people, but ask anyone who lived near a chemical spill—these events can haunt a community for decades. Even with strict rules, unplanned releases happen, so calling 2-Nitrobiphenyl low-risk misses the real story.
Better Practices and Safer Workplaces
Workers in these environments shouldn’t have to gamble with their health. Employers can invest in closed systems for chemical processing, top-grade protective equipment, and frequent air and water monitoring around the site. I know people who have gone from old-style, open-bench work to modern glovebox systems, and exposure rates drop significantly with the right setup. Real-world health data always lags behind, so prevention becomes the only safe bet.
Agencies like OSHA and EPA insist on tough limits and regular checks. It pays off, because once this kind of chemical escapes into the environment, cleanup takes years and enormous resources. Many countries already require companies to track every ounce of 2-Nitrobiphenyl, treating small spills like a serious emergency.
Final Thoughts
Everyone wins with extra caution—workers stay healthy, nearby communities dodge long-term threats, and expensive disasters get headed off before they start. Scrutinizing every hazard may feel tedious, but for chemicals as notorious as 2-Nitrobiphenyl, the stakes are just too high to cut corners.
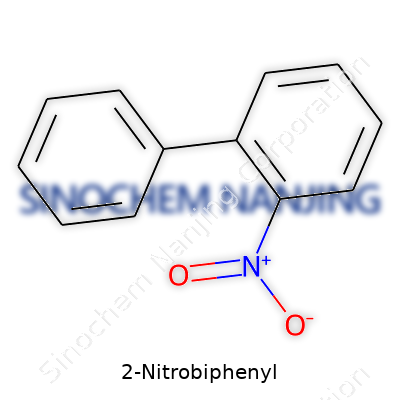
| Names | |
| Preferred IUPAC name | 2-nitro-1,1'-biphenyl |
| Other names |
2-Nitrobiphenyl o-Nitrobiphenyl 2-Phenyl-nitrobenzene o-Phenyl nitrobenzene |
| Pronunciation | /tuː ˌnaɪtroʊ baɪˈfiːnɪl/ |
| Identifiers | |
| CAS Number | 86-19-5 |
| Beilstein Reference | 2040007 |
| ChEBI | CHEBI:34343 |
| ChEMBL | CHEMBL16222 |
| ChemSpider | 21820055 |
| DrugBank | DB07714 |
| ECHA InfoCard | 100.017.423 |
| EC Number | 202-204-7 |
| Gmelin Reference | 65360 |
| KEGG | C14151 |
| MeSH | D009617 |
| PubChem CID | 6947 |
| RTECS number | DJ3325000 |
| UNII | C2D7B4144E |
| UN number | 1660 |
| Properties | |
| Chemical formula | C12H9NO2 |
| Molar mass | 245.24 g/mol |
| Appearance | Pale yellow crystalline powder |
| Odor | Odorless |
| Density | 1.233 g/cm3 |
| Solubility in water | insoluble |
| log P | 3.8 |
| Vapor pressure | 0.000224 mmHg at 25°C |
| Acidity (pKa) | 15.17 |
| Basicity (pKb) | 6.40 |
| Magnetic susceptibility (χ) | -64.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.662 |
| Viscosity | 23 mPa·s (160°C) |
| Dipole moment | 2.86 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 321.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 113 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5313 kJ/mol |
| Hazards | |
| Main hazards | Suspected of causing cancer. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H351 |
| Precautionary statements | Precautionary statements for 2-Nitrobiphenyl: "P261, P264, P270, P271, P301+P312, P304+P340, P308+P311, P405, P501 |
| NFPA 704 (fire diamond) | 2-Nitrobiphenyl: "2-2-0 |
| Flash point | Flash point: 162°C |
| Autoignition temperature | Autoignition temperature: 550°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 470 mg/kg |
| LD50 (median dose) | LD50 (median dose): 340 mg/kg (oral, rat) |
| NIOSH | NIOSH = "T3662000 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | 500 mg/L |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Biphenyl 2-Aminobiphenyl 3-Nitrobiphenyl 4-Nitrobiphenyl |

