2-Nitrobenzyl Chloride: A Deep Dive Into An Influential Chemical
Historical Development
Chemists got interested in nitrobenzyl derivatives over a hundred years ago, chasing after new intermediates for dyes and drugs. The roots of 2-Nitrobenzyl Chloride reach back to the late nineteenth century, when organic chemistry’s toolbox barely held much besides sulfuric acid and benzene. Researchers noticed that introducing a nitro group to benzyl chloride tweaked its reactivity in powerful ways. Over decades, European labs firmed up the basic synthesis and started mapping out its conversion pathways. The nitro group’s influence changed how the benzyl chloride interacted with nucleophiles, luring more attention from academia. By the 1960s, advances in photochemistry and peptide research pushed this molecule into the limelight. It caught the eye of organic chemists for its usefulness as a protecting group, particularly after discoveries in photocleavable systems. These origins shaped its role, and made 2-Nitrobenzyl Chloride a central figure in both bench chemistry and industrial labs.
Product Overview
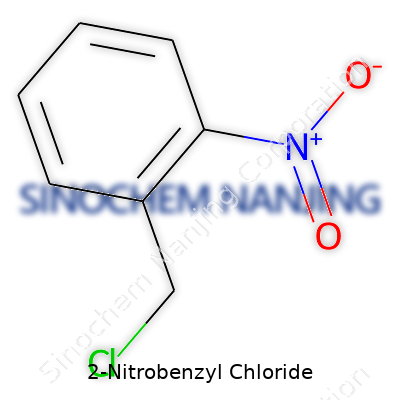
2-Nitrobenzyl Chloride comes off as a modest yellowish solid. Its structure, a benzene ring with a nitro and a chloromethyl group attached, makes it a valuable intermediate in many syntheses. Chemical suppliers stock it for research, and it often sits in the catalog next to its cousins, such as 2-Nitrobenzyl Alcohol and 4-Nitrobenzyl Chloride. It plays out its main role as a photoremovable protecting group, prized for how easily it sheds in the presence of UV light, but it also appears in the formation of pharmaceuticals, specialty polymers, and experimental bioconjugates. People in labs buy it in grams or kilograms, depending on the project, and those with bigger budgets appreciate the reasonable price-to-function ratio that stems from a fairly scalable production route.
Physical & Chemical Properties
This compound packs interesting physical and chemical features. Under the hood, 2-Nitrobenzyl Chloride holds a molecular formula of C7H6ClNO2. Its solid form melts above room temperature, stashing itself at 45–48°C, and if left long enough, its faint, pungent odor gives itself away in any unventilated chemistry department office. Its slight solubility in water barely makes a splash, but it dissolves far better in organic solvents—think dichloromethane, chloroform, and ether. The nitro and chloro substituents prime it for a spectrum of substitution and reduction reactions. Sitting inside a brown glass bottle, it demands to be kept away from bases and moisture, since both can kick off unwanted reactions. Exposure to UV causes breakdown, a feature that plays into its biggest utility.
Technical Specifications & Labeling
Producers label shipments with more than just a CAS number and purity. Common grades run from 95% up to 99%, tailing off with traces of related nitrobenzyl compounds or over-chlorinated side-products. The packaging tells you it’s moisture sensitive, and the hazard labels flag it under GHS symbols for corrosivity and acute toxicity. Modern suppliers stick QR codes on bottles for easy SDS access. Those working in regulated labs demand batch analysis sheets with each order, checking retention time and impurity profiles via gas chromatography or HPLC. Common synonyms written right on the label keep things clear for international buyers.
Preparation Method
Synthesis of 2-Nitrobenzyl Chloride starts with a run-of-the-mill nitration of toluene. Lab techs cool a pot, add a mix of concentrated nitric and sulfuric acids, and let the reaction go until the right isomer forms. Isolation takes some separation skills, since both ortho and para products drop out. The ortho-nitrotoluene collected then heads to a chlorination setup. Here, bubbling chlorine gas or using N-chlorosuccinimide in the presence of light snips off a hydrogen from the methyl group, installing that key benzylic chloride. Purification means distilling or recrystallizing under reduced pressure, catching the right melting point. Industrial plants push this synthesis at scale, securing better yields by tweaking solvent and temperature profiles.
Chemical Reactions & Modifications
It carries both reactive nitro and benzylic chloride handles, which unlock diverse chemistry. Nucleophiles, including alcohols, thiols, and amines, appreciate the benzylic position for SN2 attack—already more reactive thanks to the electron-withdrawing nitro group. Reduction of the nitro group leads to 2-aminobenzyl chloride, which is popular in agrochemical intermediates. Chemists hungry for photolabile groups tie 2-nitrobenzyl to carboxylic acids, phosphates, or phenols, creating caged compounds. UV light cleaves the bond, freeing whatever functional group was masked. This feature is the main draw for peptide and nucleotide chemistry, where controlled deprotection matters. Side reactions sometimes include hydrolysis to 2-nitrobenzyl alcohol, especially if moisture sneaks in during storage.
Synonyms & Product Names
In catalogs and literature, this chemical pops up as "o-Nitrobenzyl Chloride," "2-Chloromethyl Nitrobenzene," and sometimes plain old "NBCl." Overseas, buyers might call it "2-nitro-alpha-chlorotoluene," although rarely in English papers. The CAS number 89-98-5 links these names. A few catalogues use trademarks for their photo-cleavable derivatives, but the main chemical sticks to these standard monikers.
Safety & Operational Standards
2-Nitrobenzyl Chloride needs careful handling. Liquid and solid forms both sting skin and burn on contact. Fume hoods aren’t optional for measuring, weighing, or reactions—once you sniff its vapor, you remember to keep the sash down. Dust irritates eyes, sinuses, and lungs, so safety goggles and gloves are absolutely necessary. SDS documents warn of acute toxicity if inhaled or swallowed, citing animal studies. For spills, it pays not to improvise—absorb with inert material and double-bag for hazardous waste pickup. Anyone scaling up runs must check scrubber systems for vented HCl and verify that containment protocols are in place. Labs with a strict culture keep logs and inventory records, while periodic risk assessments confirm compliance with both local and GHS standards.
Application Area
Most users find their way to 2-Nitrobenzyl Chloride through the world of protecting groups in synthetic chemistry. It creates caged compounds that only become active or functional when zapped with a precisely timed blast of light. Researchers use this feature to control biochemical processes in everything from neuroscience, where they uncage neurotransmitters in live cells, to polymer science, where UV sensitivity directs crosslinking patterns. Medicinal chemists, too, like the orthogonality of the group—it can block sensitive sites on drugs or probes until the final synthesis step. Its use in photoresists, specialty polymers, perfume chemistry, and some dye intermediates rounds out the portfolio.
Research & Development
Research teams constantly push the boundaries with this molecule, working to extend the reach of caging and uncaging technologies. Photocleavable protecting groups didn’t become mainstream overnight—it took years of trials to boost reaction yields, reduce byproducts, and suppress unwanted side reactions. Standard groups like FMOC and BOC shielded amines, but 2-Nitrobenzyl showed new pathways with its visible-light response. Lately, labs in photopharmacology and bioconjugation use it to control biomolecule activation with deep spatial precision. Some startups and academic groups seek derivatives with even faster cleavage times or higher biocompatibility, blending new nitrobenzyl analogues into advanced assays.
Toxicity Research
The chemical community recognizes 2-Nitrobenzyl Chloride’s risks and keeps up with toxicity studies. Most focus on inhalation and ingestion pathways, since the compound is readily absorbed and causes tissue irritation. Animal studies flag acute toxicity, and chronic exposure at lower doses may trigger organ effects. Its classification as a hazardous chemical under EPA and REACH guidance isn’t casual—both agencies cite evidence of respiratory and central nervous system impacts. Research into bioaccumulation suggests low environmental persistence, but breakdown products deserve close watch. Anyone working with this material learns to treat it with respect, particularly in handling, waste disposal, and emergency response planning.
Future Prospects
Looking ahead, 2-Nitrobenzyl Chloride is positioned for expanded use in fields that depend on precise chemical control—photonics, optogenetics, and next-generation drug delivery all benefit from its established chemistry. Research into its derivatives looks promising, especially as scientists hunt for faster, less toxic photoremovable groups. Labs tweak substituents to shift activation wavelengths into safer ranges, supporting new experiments in living systems. Combining its old-school reactivity with new synthetic targets continues to ensure demand, while ongoing efforts in green chemistry may improve how it’s made and managed. Every innovation here relies on a deep understanding of its history, reactivity, and risks, ensuring this chemical keeps earning its place on the cutting edge of research and development.
What is the chemical formula of 2-Nitrobenzyl Chloride?
Getting to Know the Molecule
A lot of people in laboratories—students, researchers, and chemists alike—find themselves handling all sorts of complex chemicals. One that pops up from time to time in chemical literature is 2-Nitrobenzyl Chloride. If you’re keen on understanding its structure, here’s what’s at stake: a benzene ring with a nitro group and a chloromethyl group attached to it. In everyday lab work, being able to sketch or describe this molecule goes a long way in communicating with peers, running reactions, or even ordering the right compound.
Chemical Formula: C7H6ClNO2
The formula provides a glimpse into the elements and numbers of atoms making up each molecule: seven carbon atoms, six hydrogen atoms, one chlorine, one nitrogen, and two oxygen atoms. So, C7H6ClNO2 stands at the center of any discussion involving this compound. Once you see these numbers, it’s easier to calculate molar mass, figure out stoichiometry, and keep track of chemical inventories.
Practical Importance in Laboratories
From my own experience, maybe as a graduate researcher, these building blocks make a big difference in planning syntheses. For those unfamiliar, the nitro group at position two activates the benzene ring, allowing the chloromethyl group to act as a handle for further transformations. This kind of functionality proves essential in organic synthesis, particularly if you’re looking into photochemistry. The 2-nitrobenzyl group serves as a popular “protecting group” that can be removed with light, which is a neat trick for controlled release.
It’s not uncommon to see this molecule woven into complex pharmaceutical or materials science projects. Think back to a synthetic pathway that required hiding a reactive site; adding a 2-nitrobenzyl group often saved a lot of hassle. Being precise with formulas matters—not just for purity, but for safety. A small slip in calculation, or a mix-up with a similarly-named compound, can throw experiments off or even become dangerous.
Safety and Environmental Concerns
Working with benzyl chlorides demands careful handling. Most chemists know these compounds irritate skin, eyes, and mucous membranes. In poorly ventilated workspaces, vapors can cause real trouble. Chlorinated organics present disposal issues, too, as regulatory pressure grows to reduce hazardous waste. I’ve seen research groups revise protocols to reduce solvent use, substitute safer reagents, or choose greener disposal methods. Sustainability in the lab often begins by scrutinizing which chemicals we buy, use, and throw away.
Institutions and companies encourage training around safe handling, and monitoring usage. Being familiar with Material Safety Data Sheets and storing compounds in secure, labeled containers reduces risks. Solid record-keeping—logging lots, usage, and storage conditions—can save hours sorting out confusion after an accident.
Better Working Methods
For a researcher handling 2-Nitrobenzyl Chloride, the formula anchors all calculations. Tools like stoichiometric calculators, chemical inventory systems, and online safety platforms can help. By focusing attention on clear labeling and proper protocol, labs avoid confusion—even in high-throughput settings. Modern research depends on open sharing of information, and chemical nomenclature is the foundation.
Anyone diving into chemical synthesis, whether for class or advanced research, ought to have a sharp eye for detail. Chemistry advances on the backs of clear communication, accurate formulas, and respect for the hazards involved. That’s best practice, plain and simple.
What are the common applications of 2-Nitrobenzyl Chloride?
Exploring Its Role in Organic Synthesis
Everything in organic chemistry comes down to building blocks and tools. 2-Nitrobenzyl chloride fits that role as a reliable starting material for plenty of reactions. Chemists often reach for it when they need a “protecting group,” which means it temporarily guards sensitive bits of a molecule during multi-step syntheses. This blocking action allows reactions to run without unwanted side effects on certain spots of the molecule, saving both time and materials.
People in research labs, especially those working on pharmaceuticals, make use of this protection trick all the time. That’s because many drugs depend on precise, step-by-step assembly lines. With 2-nitrobenzyl chloride, a lab can “hide” functional groups, perform aggressive reactions elsewhere, and later “unmask” the protected group using light or chemical means.
Photoremovable Protecting Groups in Biochemistry
Interest in this chemical really took off with photochemistry, where light helps trigger reactions. Researchers use 2-nitrobenzyl groups to keep biological molecules—like peptides or nucleotides—inactive until they’re needed. Shine a specific wavelength of light on them, and the chemical bond breaks, releasing the active molecule right on cue.
One of the best real-world uses pops up in neuroscience and cell biology. For example, scientists want to see how a neurotransmitter works at a single nerve cell, so they cage it with a 2-nitrobenzyl group. A laser then “uncages” the neurotransmitter, and the cell responds at that moment. This unlocks experiments with razor-sharp timing, leading to discoveries about brain function and signal pathways.
Building Complex Molecules for Pharmaceuticals
Pharmaceutical chemists appreciate this molecule for more than just protection. The nitro and chloride groups set up opportunities for substitution, allowing the creation of compounds with new biological activities. In my experience, switching out these groups helps lead researchers to early-stage molecules with potential as cancer drugs or antibiotics.
An everyday example involves using 2-nitrobenzyl chloride to attach complex side chains onto existing molecules, changing how they act inside the body. Small tweaks at this stage can make huge differences in a drug’s effectiveness or safety. Several patented medicines owe part of their structure to reactions starting with this building block.
Industrial and Material Science Uses
Industries outside medicine find value here too. Specialty polymer companies have adopted 2-nitrobenzyl chloride for light-sensitive coatings and photographic chemicals. You walk into an electronics factory and see photoresists—light-activated materials used to etch circuit boards—where this compound plays a recurring part.
In the quest for better solar cells and sensor devices, engineers test molecules that change properties after absorbing light. The nitrobenzyl group tends to deliver reliable results in this area. These advances wouldn’t move forward without stable, reproducible sources of tricky reagents.
Safety and Handling Concerns
It’s worth noting that 2-nitrobenzyl chloride should be treated with respect. Chlorinated and nitro compounds can trigger allergies, respiratory problems, or skin irritation. Working with it means using gloves, proper ventilation, and up-to-date safety data. Regulatory agencies keep lists of chemicals that need special attention in labs, and this one makes frequent appearances. The path to safer innovation involves not just finding new uses but also keeping a close eye on how these chemicals interact with people and the environment.
Moving Toward Greener Chemistry
There’s no shortage of demand for substances that offer strong performance without causing downstream problems. As a chemist who has tried to reduce hazardous waste in my own lab, I see potential in developing alternatives to nitrobenzyl compounds or in recycling methods that lower the impact. Teams around the world are working on substitutes that use light-activated chemistry but break down more easily or pose less risk to workers. Until those options fully mature, precise protocols and responsible sourcing remain key for anyone using 2-nitrobenzyl chloride.
What safety precautions should be taken when handling 2-Nitrobenzyl Chloride?
Respecting Chemistry in Practice
Every chemist remembers that sharp, odd smell from the hood during an organic synthesis. 2-Nitrobenzyl chloride is one of those chemicals that makes you double-check if you’re doing things right. With a strong irritant reputation and a history of causing chemical burns, it makes safety feel real, not hypothetical.
Personal Experience: Gloves, Gowns, and Immediate Cleanup
Working with 2-nitrobenzyl chloride once left a faint burn on my glove. That moment stuck with me. No glove is too thick, no splash goggles too large. Skin does not forgive accidental exposure. Latex or nitrile gloves add a basic layer; double-gloving feels snug, but it works. A face shield with safety goggles stopped a fine mist from getting near my eyes once. Proper lab coats—long-sleeved and buttoned—save clothes and, more importantly, skin. Closed shoes aren’t optional; spilled drops find weak spots on sandals and old sneakers.
Using Fume Hoods and Avoiding Vapors
Once you smell the acrid odor, you realize ventilation matters. The fume hood should be down, sash pulled as low as comfortable for the hands. It’s not about storage; it’s about working directly under the hood vacuum. Even a small open bottle can give off vapors that irritate the throat and eyes. If you ever coughed while pipetting, you know how essential this step is. No experiment is worth lung damage.
Storage Wisdom Based on Reactivity
2-Nitrobenzyl chloride reacts with water and many organics, so storing it properly avoids ruined supplies or, worse, accidental fires. Clear labeling, tight caps, and keeping it on a separate shelf—preferably in a ventilated, locked cabinet—keeps curious hands and accidents at bay. Never store above eye level—reaching up for a hazardous chemical has gone wrong for many, and it burns into memory.
Dealing with Spills and Skin Contact
Spills don’t give second chances. If it hits a bench, grab absorbent pads doused in neutralizer. Work as if that chemical is always hot, even when it isn’t. Skin contact means running water, at minimum, for 15 minutes. No shortcuts. I remember an incident when a peer didn't want to be "trouble" for the safety shower after a splash. The wound took weeks to heal. Safety showers look intimidating, but they save tissue.
Waste: No Shortcuts with Disposal
Waste collection jars exist for a reason. Dumping leftovers down the sink spreads the problem, hurting drainage, lab workers, and the environment. Segregate waste into halogenated organic bins, seal tightly, and log entries. Not all labs have perfect waste systems—speak up if you spot clogs or leaks.
Health Monitoring and Training Matter
Chemistry professors do not forget to train students on hostile compounds. Refresher courses, warning signs posted at doors, and strict buddy systems mean more eyes on the problem. Rashes, odd coughing, uncoordinated spills—these are red flags. Having emergency numbers posted close by and practicing quick response drills isn’t just bureaucracy; it’s a habit that prevents injury and panic.
Solutions: Fostering a Culture of Responsibility
Trust in protocols comes with experience, but learning from each near miss makes the biggest difference. Encouraging transparent reporting of accidents, investing in good PPE, and keeping equipment up-to-date help protect everyone. Handling dangerous reagents like 2-nitrobenzyl chloride starts as a checklist but becomes second nature as you respect the risks and value the people around you.
What are the storage conditions for 2-Nitrobenzyl Chloride?
Safety Takes Center Stage
Dealing with 2-Nitrobenzyl Chloride isn’t something to take lightly. I’ve handled plenty of chemicals in my work—some seem harmless, but this one deserves respect. This compound reacts with water and produces corrosive gases. Even a hint of humidity in your storage area could lead to trouble. Keep it bone dry. Shelves near sink areas or under leaky pipes won’t cut it. Use tightly sealed glass or HDPE containers and double check for cracks.
Temperature and Light Matter
Hot spots in the storage room create problems. Excessive heat, even from a window in summer, will break down this chemical. You want a cool spot, well away from direct sunlight or radiators. Store it below 25°C—around room temperature works. Don’t let the temperature drop too far below freezing since condensation forms on containers as things warm up, inviting moisture where it doesn’t belong.
Ventilation Prevents Problems
If a container breaks, fumes can spread quickly. A good storage space has clear airflow—ideally, a chemical fume hood or a locked, well-ventilated cabinet set apart from general-use shelving. From my own lab experience, good air movement makes everything less stressful if a spill happens, and it helps with detection of leaks before they build up.
Away From Incompatibles
Don’t stash 2-Nitrobenzyl Chloride near strong bases, acids, or reducing agents. Mixing chemicals by accident is a rookie mistake. Every time I have organized a chemical stockroom, proper separation prevented the kind of disasters that insurance companies write long reports about. Your best bet is a dedicated shelf or locked box away from common reagents and flammable solvents.
Labeling, Inventory, and Routine Checks
I’ve seen so many jars outlive their labels or lose them altogether. For this compound, every container needs a clear, durable chemical label with the full name, hazard warnings, and date of storage. Record every accession and withdrawal. Open containers or those with questionable seals should get replaced, not ignored. Regularly check for signs of brownish or yellowing residues around lids or stoppers—these point to slow leaks or contamination.
PPE and Emergency Gear
Storing dangerous chemicals means you need the right protective gear on hand: nitrile gloves, splash goggles, and a lab coat at minimum. Spill kits should always sit within easy reach. Too many labs overlook routine practice drills. Even the best-labeled shelf won’t help if you freeze up in an emergency or can't find the eyewash station.
Complying With Laws and Guidelines
Local, national, and international regulations aren’t just red tape—they safeguard your lab, your people, and the community. I rely on the Globally Harmonized System (GHS) and local chemical safety data sheets for specifics. Regular training and updated protocols keep everyone alert. Neglecting legal storage requirements opens the door to fines, lab shutdowns, or worse.
Responsible Storage Means Fewer Accidents
Proper storage isn’t about following a checklist. It’s built from experience, a little patience, and learning from those who got it wrong. If you give 2-Nitrobenzyl Chloride the respect it demands, your workspace will run safer, smoother, and with plenty of peace of mind.
Is 2-Nitrobenzyl Chloride available in different purity grades?
Why Purity Matters with Chemicals Like 2-Nitrobenzyl Chloride
Anyone who’s spent time in a lab knows the frustration that comes with inconsistent results. More often than not, purity levels in reagents sit at the center of the issue. That’s especially true with compounds like 2-Nitrobenzyl Chloride. A slight variation in composition can throw off entire processes, whether it’s in pharmaceuticals, materials research, or synthesis work.
What Purity Levels Mean On the Bench
You won’t always find a one-size-fits-all purity grade for 2-Nitrobenzyl Chloride. Manufacturers turn out different grades to suit different needs. Many sources offer what’s labeled as “reagent grade,” with purity above 98%, targeting research and development settings. Analytical work often goes a step further, sourcing higher-purity lots to keep interference at bay. Lab teams working on small-molecule synthesis typically reach for higher grades to reduce the risk of side-products, especially when traces can sabotage downstream reactions or analytical processes.
Industrial buyers sometimes choose lower purity grades. This isn’t about cost-cutting so much as practicality: some processes can tolerate small amounts of impurity. Bulk synthesis may not demand the same clean reaction profile as drug discovery work. An old colleague, who spent years scaling up reactions for a specialty chemical producer, said it bluntly: “Chasing that last percent of purity is fine if you’re making an API. But a little contaminant hardly matters in coatings precursors if it stays within spec.”
Risks Linked to Lower Purity
Every bit of added impurity carries baggage. Residual solvents, water, or unreacted starting material can all skew results. Even equipment performance can suffer—impurities deposit in valves or lines, leading to maintenance issues. Contaminants sometimes mask themselves, producing subtle changes in yield or color but eluding detection until late in the process.
Labs under regulatory oversight—like anything regulated by the FDA—face tight scrutiny, so purity documentation becomes part of the daily grind. Hiding behind incomplete certificates of analysis (CoA) can spark real trouble, both legally and scientifically.
Transparency and Verification
Suppliers still play a key role. Good manufacturers provide a reliable CoA, with comprehensive breakdowns of impurities and testing methods. I’ve learned the hard way not to settle for vague documentation. Authentic CoAs and open communication help maintain trust and reduce costly mistakes. Smart buyers verify the actual certificate against the lot to catch any slip-ups.
Third-party testing adds another layer of confidence. External labs help spot red flags, especially for lots imported from new vendors or regions where standards vary. Spot checks on purity, using NMR or chromatography for example, add shield against unexpected failures.
Improving Safety and Quality in the Supply Chain
Open demands for full traceability start upstream. Buyers should press for details on manufacturing processes, solvents involved, and storage practices. Simple storage missteps can lead to degradation, especially given how 2-Nitrobenzyl Chloride reacts with moisture. Keeping this compound dry and sealed prevents surprise degradation products from turning up.
Anyone working with these chemicals benefits from collaboration. Sharing experiences—good or bad—between labs helps close knowledge gaps and pressure suppliers to do better. Community effort smooths out surprises and ensures safer, more consistent work for everyone involved.
Solutions for Getting the Right Purity Level
Start by knowing the specs that matter for your project, then source from suppliers who back up their claims with solid data. Building relationships with reputable vendors goes a long way. In-house testing should stay routine, especially with new purchases or critical experiments. If a project can tolerate less-than-perfect material, document it and flag any downstream risks.
Raising expectations and refusing to cut corners turns purity from a “nice to have” into a project-saving necessity. No one wants to redo work because of hidden contaminants or poor documentation. Investing in quality cuts stress and keeps research moving forward.
| Names | |
| Preferred IUPAC name | 1-chloro-2-(nitromethyl)benzene |
| Other names |
2-Chloromethyl-1-nitrobenzene o-Nitrobenzyl chloride 1-Nitro-2-(chloromethyl)benzene α-Chlor-o-nitrotoluene |
| Pronunciation | /tuː ˌnɪtroʊˈbɛn.zɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 612-23-7 |
| Beilstein Reference | 867175 |
| ChEBI | CHEBI:76260 |
| ChEMBL | CHEMBL410250 |
| ChemSpider | 10799 |
| DrugBank | DB08358 |
| ECHA InfoCard | 03b757e0-1bce-41e7-9d88-47c0e5f6e8f7 |
| EC Number | 202-204-0 |
| Gmelin Reference | 83378 |
| KEGG | C02569 |
| MeSH | D015894 |
| PubChem CID | 6924 |
| RTECS number | CU5950000 |
| UNII | L966C1S47J |
| UN number | 1668 |
| CompTox Dashboard (EPA) | `DTXSID3048415` |
| Properties | |
| Chemical formula | C7H6ClNO2 |
| Molar mass | 169.57 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Aromatic |
| Density | 1.34 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.90 |
| Vapor pressure | 0.02 mmHg (25°C) |
| Acidity (pKa) | 14.8 |
| Basicity (pKb) | 13.21 |
| Magnetic susceptibility (χ) | -59.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.595 |
| Viscosity | 1.623 mPa.s (20°C) |
| Dipole moment | 3.47 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.3 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P271, P301+P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-X |
| Flash point | 108°C |
| Autoignition temperature | Autoignition temperature: 590°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 2-Nitrobenzyl Chloride: "670 mg/kg (oral, rat) |
| NIOSH | CY8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | IDLH: 20 ppm |
| Related compounds | |
| Related compounds |
2-Nitrobenzyl bromide 2-Nitrobenzyl alcohol o-Nitrobenzyl acetate 2-Nitrobenzylamine 4-Nitrobenzyl chloride |

