2-Nitrobenzoyl Chloride: An In-Depth Commentary
Historical Development
Chemistry gained a new handle on selective synthesis after the early discovery of benzoyl chlorides. In the late nineteenth century, after advances in organic chemistry from Germany and England, chemists stumbled onto the synthesis of 2-nitrobenzoyl chloride as they looked for ways to functionalize aromatic rings. Professional curiosity often led people to experiment with various nitration and chlorination routes. Before electronic microscopes and chromatography hit their stride, researchers isolated and characterized this compound mostly by intensive manual work—recrystallizations, boiling points, melting points recorded with mercury thermometers. The development of this compound reflects the evolution from laborious, risky practices to modern, mechanized, and far safer chemical production.
Product Overview
2-Nitrobenzoyl chloride stands as a yellowish, pungent crystalline solid known best among chemists for its reactivity. Its demand rests in its niche position—too harsh for amateurs, vital for professionals in fine chemical synthesis. The smell alone signals trouble, and its corrosiveness can chew through gloves and glassware at the wrong moment. In my experience in organic labs, people set aside a dedicated hood just for acyl chlorides like this one. Its shelf life, even in sealed bottles, never inspires confidence. That’s why specialists treat it more like a reagent than a product for shelf display.
Physical & Chemical Properties
With a melting point often measured near 43-47°C and a boiling point a bit above 290°C (with decomposition), the substance turns liquid at room temperature in warm labs but will solidify in cooler conditions. Yellow crystals warn of its irritant character. In air, it hydrolyzes and gives off clouds of hydrogen chloride—an instant reminder to wear goggles and a respirator. The molecule’s structure, with a nitro group ortho to the carbonyl chloride, increases its reactivity and influences its interactions with nucleophiles. Even seasoned chemists respect its power: in a few minutes, an exposed surface shows acid burns.
Technical Specifications & Labeling
High-grade suppliers guarantee at least 98% purity, with impurities like 3- and 4-nitro isomers usually below 1%. Labels must highlight acute toxicity, environmental danger, and corrosivity using appropriate hazard pictograms. All relevant certificates—Certificate of Analysis, Material Safety Data Sheet, REACH pre-registration—should accompany shipments. Each shipment comes with lot numbers and tracked, tamper-proof containers. In industrial settings, guidelines require ventilation and continuous environmental monitoring to limit fugitive emissions.
Preparation Method
Preparation kicks off with starting material 2-nitrobenzoic acid, a product of selective nitration and carboxylation of benzene derivatives. An excess of thionyl chloride or phosphorus pentachloride then achieves the conversion to the corresponding acid chloride. Vapors released from these reagents prompt immediate evacuation, every time a flask gets heated. Modern setups employ reflux condensers, inert atmospheres, and automated venting for acid gases. Most researchers learned early that dry conditions and patience make or break the yield. Post-reaction workup involves slow distillation or crystallization and repeated purification—sometimes column chromatography, depending on the final purpose.
Chemical Reactions & Modifications
2-Nitrobenzoyl chloride’s most popular role comes as an acylating agent for nucleophiles—amines, alcohols, and occasionally thiols or active methylene compounds. Introduction of the 2-nitro group steers reactivity and stops certain side reactions. Chemists count on this selectivity when making pharmaceuticals, dyes, or functionalized polymers. Reduction or further substitution on the aromatic ring demands careful choice of catalysts and reaction conditions—catalytic hydrogenation or metal reductions pose risks of over-reduction. The acyl chloride function rarely survives basic or aqueous environments, and inadvertent exposure to even slight moisture ruins entire batches overnight. Safety controls and good practice remain the difference between a successful synthesis and an emergency.
Synonyms & Product Names
Other names include ortho-nitrobenzoyl chloride and 2-nitrobenzenecarbonyl chloride. In catalogues, you find it under CAS number 89-01-0. European and Asian suppliers offer it under similar names with international hazard codes. In patent literature, chemists abbreviate as ONBC, acknowledging its familiar status in research circles. The range of synonyms sometimes causes confusion, reinforcing the importance of clear labeling and communication between suppliers and end-users.
Safety & Operational Standards
Handling protocols for 2-nitrobenzoyl chloride draw from regrettable lab accidents and industrial incidents. Direct exposure to vapor or dust inflames lungs, eyes, and skin; gloves and goggles form only the first line of defense. Storage goes under strict ventilation, away from flammable or basic chemicals, and safety showers stand within arm’s reach. Training covers chemical spill response and specific neutralization agents such as sodium carbonate or ammonia solutions. In larger-scale operations, scrubbers and negative-pressure environments control accidental release. Documented cases show that complacency in chemical hygiene courts disaster—insurers and health agencies demand strict operational records. Emergency drills, backed by clear, practiced evacuation plans, remain part of every competent facility’s routine.
Application Area
Medicinal chemists turn to 2-nitrobenzoyl chloride as a building block in drug intermediate synthesis. The ortho-nitro functionality enables downstream transformation into amine groups—steps essential for some antibiotic and anti-inflammatory compounds. The dye and pigment industry uses the compound for color modification and permanent attachment onto aromatic scaffolds. Polymer chemists, exploring advanced functional materials, introduce this group to impart unique electronic or binding properties. I have seen analytical labs use it sparingly for derivatizing amines, boosting detection sensitivity in trace analysis. Its reach into agrochemicals, imaging agents, and specialty adhesives creates a persistent demand, despite strict regulation on transport and handling.
Research & Development
Recent research interests circle around less toxic preparation methods and more efficient reaction pathways. Scientists experiment with greener chlorinating agents, seeking ways to dodge wasteful and polluting byproducts common in classic protocols. Computational chemists try to model reaction mechanisms, hoping to guide experimentalists toward safer and more selective approaches. Teams in pharmaceutical development frequently publish case studies, sharing successes and near-disasters around handling sensitive acyl chlorides. In my circles, the chatter often involves swapping practical tips—minimizing exposure, securing supply lines, or troubleshooting contamination issues.
Toxicity Research
Researchers classify 2-nitrobenzoyl chloride as acutely toxic by inhalation, contact, or ingestion. Short-term exposure in animal studies confirms severe respiratory and dermal effects; repeated exposure stresses kidney and liver functions. Test results from regulatory filings show strong irritant potential and classified mutagenicity, putting workers on high alert. Authorities such as OSHA and EU-REACH regulate permissible exposure levels and require monitoring in occupational settings. Toxicological studies today try to balance necessary industrial use against environmental and worker health, spurring initiatives to find less hazardous substitutes or more contained production techniques.
Future Prospects
With tighter safety standards and greater public scrutiny on hazardous chemicals, the future of 2-nitrobenzoyl chloride lies partly in safer production and handling technologies. Automation, microreactor technology, and improved waste treatment may reduce exposure risks in both lab and industrial settings. Digital platforms now trace batch origins and movements to curb illegal diversion toward illicit synthesis. Researchers keep trying to tweak its chemistry to expand applications in biomedicine and materials science, with synthetic biologists eyeing modified analogs for target-specific drug release. Still, the core chemical risks won’t vanish—future growth depends on sustainable practices, continuous education, and robust engineering controls over mere cost savings.
What is 2-Nitrobenzoyl Chloride used for?
The Reach of 2-Nitrobenzoyl Chloride in the Lab
Some chemicals get swept under the radar, even though their fingerprints show up in all sorts of places. 2-Nitrobenzoyl chloride is one of those workhorses. This compound crops up in organic chemistry labs across the globe, thanks to its knack for starting reactions that lead to bigger, flashier molecules. Folks who spend their days coaxing new drugs and materials out of simple building blocks tend to keep this one within arm’s length, especially in pharmaceutical and materials science settings.
Building Blocks for Pharmaceuticals
A person who’s spent time at the bench knows the value of having the right “activator,” and 2-nitrobenzoyl chloride fits the bill beautifully. Researchers use it to tack nitrobenzoyl groups onto a variety of molecular “scaffolds.” This step matters because the nitro group is more than just a pretty functional group—it helps tune the properties of molecules. Sometimes, a drug candidate needs to slip past stubborn enzymes or hang around in the bloodstream longer. Tacking on a nitrobenzoyl group with the help of this compound can offer those tweaks.
Academics and drug developers put this chemistry to work for creating intermediates in the hunt for antibiotics, cardiovascular treatments, and anti-inflammatory agents. In my time working through research papers and helping students troubleshoot reactions, I’ve seen 2-nitrobenzoyl chloride show up again and again in protocols for drugs that block pain or infection. The basic chemistry stays the same, but the downstream effects make all the difference when working on something as life-changing as a new medication.
Stepping Stone in Dye and Pigment Production
This compound also helps out in making dyes and pigments. It serves as a starting material for synthesizing azobenzenes and other aromatic compounds, which show up in everything from fabric dye to diagnostic stains for lab tests. Coloring isn’t just about looks; dyes are tools for biologists who want to track disease or for engineers marking fibers in a manufacturing process. If someone ever wore a bright shirt or used a highlighter, there’s a nonzero chance some of the steps involved a molecule made using 2-nitrobenzoyl chloride.
Testing and Sensing Applications
Laboratories often put 2-nitrobenzoyl chloride to work for testing and analysis. For example, it can modify amino acids or peptides to prepare them for chromatographic analysis. Chemists often derivatize these molecules with 2-nitrobenzoyl chloride, making them easier to detect and measure reliably. As someone who has run more chromatography columns than I can count, I’ve come to appreciate reagents that produce sharp, clean signals—qualities this compound regularly delivers.
Concerns and Safer Handling
Every tool in the lab comes with its own set of risks. 2-Nitrobenzoyl chloride is no exception; its fumes irritate eyes and skin, and careless handling causes accidents. I learned early in my graduate work that fume hoods and gloves aren’t optional with this stuff. Waste management also matters, since unchecked release causes toxicity in wastewater or air. Most industry guidelines recommend closed systems and thorough employee training to keep everyone safe.
Looking Forward
As researchers call for cleaner reactions and chemicals with lower risk, the future pushes chemists to reimagine how these old standbys can be made safer or swapped out. Greener manufacturing routes and better personal safety practices strike me as the best ways forward. Until a perfect substitute appears, knowing and respecting chemicals like 2-nitrobenzoyl chloride remains part of responsible science.
What are the safety precautions for handling 2-Nitrobenzoyl Chloride?
Why 2-Nitrobenzoyl Chloride Deserves Respect
2-Nitrobenzoyl chloride doesn’t look outrageous among other white crystalline lab chemicals. Still, even small mistakes can punch a hole in your safety routine. This compound reacts quickly with moisture, producing hydrogen chloride gas—an inhalation hazard that irritates your eyes, throat, and lungs long before you spot the damage. It can blister your skin and eat through gloves if you grab whatever’s nearest instead of the right safety gear. There’s no glory in skipping the basics just to get things done quickly.
PPE: Your Everyday Armor
Many seasoned chemists swear by a well-stocked personal protective equipment drawer. Choose splash-proof goggles—nobody wins when a drop lands in your eye. A fitted lab coat keeps your clothes from soaking up spills or dust. Put on chemical-resistant nitrile gloves, not those thin plastic types. Closed shoes save your feet if glass breaks and powder scatters. Respirators aren’t just for formaldehyde. Use them when you measure out solids or open containers in a dry room. If you ever catch yourself wanting to skip one item, remember you might only have one face and two hands.
Respect the Fume Hood
Working with 2-nitrobenzoyl chloride out in the open doesn’t just risk your skin—it endangers everyone around you. I’ve watched newcomers think a tiny quantity in a big room is “safe enough.” Fume hoods aren’t just intimidating glass cupboards. They sweep up airborne particles and gases away from your nose. Don’t lean in to sniff or reach outside the sash. Set up all reagents inside, and check the airflow before you drop powder onto the scale.
Storage and Spills: Planning Matters
Uncapped bottles turn into headaches fast. 2-Nitrobenzoyl chloride should stay in tight, clearly-labeled bottles, dry and away from acids, bases, or water-reactive stuff. Try to squeeze ten bottles on a single shelf, and something will tumble. Segregate it in a cool, chemical-resistant cabinet with spill trays underneath. If your shelf is sticky, clean up right away. Wipe minor dust with damp towels and toss them into a hazardous waste bin. Don’t use your hands—grab forceps and use absorbent pads when the spill grows bigger. Practice drills so you know exactly who calls for help, grabs the spill kit, or safeguards the exits.
Training and Habit-Building
Any new chemist—intern to PhD—deserves more than a rushed safety briefing. Supervisors should make time for walk-throughs, pointing out both correct moves and common shortcuts. In tough moments, habits stick more than rules. I’ve learned to double-check container lids, label every batch, and count on others to do the same. One extra minute now can prevent weeks of injury or paperwork.
Waste Disposal: No Shortcuts
Disposal isn’t dumping powder down a sink and hoping for the best. 2-Nitrobenzoyl chloride waste goes straight into marked containers, separate from everything with water or strong bases. Neutralize only with explicit instructions; never just pour into water. Record all disposals, and don’t trust memory alone to keep labs and coworkers safe for the next shift.
Wrapping Up Safety Culture
2-Nitrobenzoyl chloride works well in the hands of people who treat safety as a shared project, not a box to check. Your coworkers depend on your attention as much as you depend on theirs. No deadline’s worth a scar or lost eyesight—inside a lab, teamwork and vigilance build real expertise.
What is the chemical formula and molecular weight of 2-Nitrobenzoyl Chloride?
Breaking Down the Basics
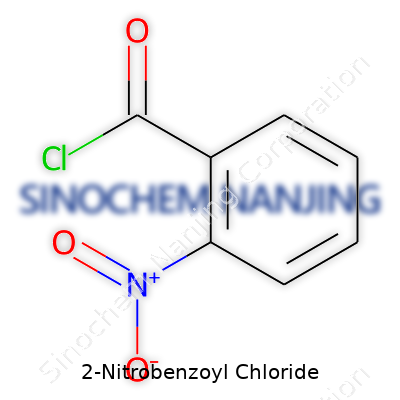
The story of 2-Nitrobenzoyl Chloride isn’t just wrapped up in its complicated name. In research labs, I’ve seen it used as a tool for organic synthesis and I remember the curiosity around any compound featuring both a nitro group and an acyl chloride. The chemical formula for 2-Nitrobenzoyl Chloride reads as C7H4ClNO3. This translates to a benzene ring carrying a nitro group at the second carbon, right next to a carbonyl chloride.
People sometimes underestimate the importance of molecular weight until the lab scales come out. With C7H4ClNO3, you add up each atom: carbon at 12.01, hydrogen at 1.008, chlorine at 35.45, nitrogen at 14.01, and oxygen at 16.00. Total it up, and the molecular weight lands at about 185.57 g/mol. This number matters every time someone draws up a reaction, because getting the wrong weight can wreck yields and waste expensive reagents.
Why Chemists Care About This Compound
Walk into any chemical synthesis class and the basics pop up quickly. Small molecules like 2-Nitrobenzoyl Chloride have shaped the dyes, pharmaceuticals, and agrochemicals industries over decades. Its acyl chloride group acts as a reliable partner in acylation reactions, attaching to amines and alcohols in a way that lets chemists build more complex structures. In this respect, it’s a workhorse, not just a theoretical formula tucked away in a textbook.
My own experience in graduate work revolved around building intermediates for bioactive compounds. Trying to source pure reagents became a regular struggle and knowing the exact formula cut that stress in half—you can’t risk contamination from compounds with similar names or close molecular weights. Some students in those crowded labs learned the hard way, mixing up similar-sounding benzoic acid derivatives and ending up with reactions that refused to cooperate. That’s not a minor inconvenience; it eats up time, funding, and sometimes reputation.
Handling and Hazards: Getting Safety Right
Chlorinated reagents bring their own baggage. 2-Nitrobenzoyl Chloride releases hydrochloric acid fumes when it meets water, and the nitro group signals potential toxicology issues. I’ve seen gloves eaten away, and labmates forced outside by eye-watering fumes. This underlines that safety data and the correct chemical identity must go hand in hand. Responsible storage, clear labeling, and appropriate handling gear serve as your shield.
Building Better Practices
Strong lab protocol doesn’t start with senior-level research, it has roots in freshman-year lessons. Writing clear labels—C7H4ClNO3, 185.57 g/mol—on containers may seem tedious but it saves headaches later. In teaching labs, I’ve urged students to double-check their calculations and never guess their way through reagent preparation.
In the chemical supply chain, accuracy matters just as much. Suppliers listing the exact chemical formula and molecular weight on their websites and datasheets help keep everyone informed and safe. Mistakes in labeling or documentation too easily cascade from supplier to bench to industrial scale, bringing regulatory investigations or even fines. This is why standardized documentation and digital tracking systems have become more widespread; they keep the details clear and mistakes traceable.
Practical Solutions for Lab Workers and Industry
Adopting digital inventory systems has made these concerns more manageable. These systems let you scan containers and pull up purity profiles, hazard ratings, and formula data. Automation takes away some of the guesswork, but doesn’t replace the need for vigilance—double-checking reagent identity before weighing or use remains a personal responsibility. My peers and I learned plenty from close calls that could have been caught by thirty extra seconds of cross-referencing a datasheet.
Small steps, such as mandatory safety refresher courses or on-the-spot calculations, form the backbone of reliable results and safer research. Compounds like 2-Nitrobenzoyl Chloride offer huge value, but that value depends absolutely on knowing—without doubt—what’s in your flask or bottle.
How should 2-Nitrobenzoyl Chloride be stored?
Understanding the Risks in Everyday Terms
2-Nitrobenzoyl chloride isn’t a compound most folks keep beneath the bathroom sink. Yet, in research labs and certain manufacturing facilities, it shows up more often than you might think. Having handled dozens of chemicals like it during long nights in the lab, I know hazards don’t always announce themselves with strobe lights and warning bells. Sometimes danger comes quietly, in an unassuming yellow jar.
I’ve watched colleagues scramble after a bottle cracked from a minor bump or—worse—after it corroded a leaky cap. Storing this compound poorly invites harm not only to the person handling it but to everyone nearby. Vapors burning the nose, splashes burning skin—these are more than inconveniences. Emergency rooms remember the names of people who misjudge such risks.
The Science Behind the Risks
Looking deeper, 2-nitrobenzoyl chloride stands out for its reactivity. Chloride groups react fast, especially in the presence of moisture. The result is hydrogen chloride gas—an acid that burns eyes, lungs, and any exposed skin. This isn’t framed as an abstract threat. According to government agencies like NIOSH and the CDC, acute exposure can send someone to the hospital for respiratory distress. Public reports confirm that just opening a rusty jar can fill a small room with acrid fumes.
Shelves and storage cabinets can hide past mistakes. I once saw a cap fused shut, eaten away by the very fumes meant to stay inside. Over time, even plastic seals can degrade, speeding up leaks and causing labels to slip into illegibility. In worst-case scenarios, forgotten jars become little chemical time bombs—waiting for an unsuspecting technician.
Practical Storage Solutions
Safe storage starts with location. Always keep 2-nitrobenzoyl chloride in a dry, cool chemical storeroom. Humidity invites disaster. Moist air seeps into ill-sealed bottles, and chemical breakdown begins before you even realize it. Store it away from windows, water sources, and anything organic—no neighboring acids or bases allowed. Combining it with other incompatible chemicals brings unnecessary risk.
Use containers designed for corrosive solids. Glass with tight-fitting, PTFE (Teflon) lined screw caps works well. Never trust basic polymer lids—these compounds will eat through soft plastics over time. Double-containment offers extra peace of mind. Place the original bottle inside a sealed, shatterproof secondary container. If the first one leaks or breaks, the backup keeps vapors from spreading. Good labeling matters—use chemical-resistant ink, including the date opened and the hazard class. Training staff to recognize and respect these signs makes a difference in keeping accidents at bay.
Relying on Experience and Facts
Keeping personal safety gear nearby should stay non-negotiable. Eye washes, chemical-resistant gloves, and fume hoods turn a risky encounter into manageable work. I’ve seen panic set in when someone touches an unknown powder only to read “Corrosive—May Cause Burns” belatedly. Fume hoods keep vapors away from eyes and lungs. Local policies should remind workers to check containers every week—for leaks, cracks, residue, or fading labels.
Lastly, always listen to those who came before you. Many senior chemists share stories filled with lessons not found in textbooks. Real-world injuries and near-misses can teach anyone more about chemical safety than lists of rules ever will. If a process or habit feels rushed or haphazard, press pause. One oversight can cost far more than a few minutes of careful preparation. Real safety comes from diligence, not luck.
What are the potential hazards and first aid measures for exposure to 2-Nitrobenzoyl Chloride?
Recognizing the Hazards
Even with years in a chemical lab, handling new substances like 2-Nitrobenzoyl Chloride keeps me alert. This compound releases a sharp, stinging odor. If a bottle gets knocked over or the powder escapes, eyes burn, throat itches, and skin starts to tingle. There’s something immediately personal about the risk — real harm waits behind careless moments.
The hazards don’t stop at irritation. 2-Nitrobenzoyl Chloride reacts strongly with water and moisture. Spill it on the floor or let a damp glove touch it, and you might see smoke or fumes. Those fumes can quickly lead to coughing, headache, or nausea. The lungs are vulnerable, and even short exposure may lead to long-term breathing problems. With enough dust or droplets in the air, some people experience chest pain or a feeling like asthma. On the skin, redness, blisters, or even burns develop in minutes if not washed away. Eyes exposed to a splash might suffer damage to the cornea, and permanent vision impairment is a real risk if action comes too late.
I’ve learned that the consequences stretch beyond immediate health. Any release in the workspace can disrupt hours of work, contaminate finished products, and trigger emergency protocols. Industry records and regulatory filings often cite serious violations over mishandling, so mistakes also invite audits and fines.
Personal Safety Measures
On any day working with 2-Nitrobenzoyl Chloride, full protection is non-negotiable. Safety goggles, face shields, and impervious gloves get checked for tears. The story of a colleague who trusted an old lab coat stays with me: one spill left permanent scars on the forearm. Double-layering — thick nitrile over latex — has spared a burn or two in my own experience. Chemical fume hoods matter, since air must stay clear of those noxious fumes. Respirators are never a luxury, especially for people prone to respiratory issues.
Immediate First Aid Steps
Speed is everything if exposure happens. Splash in the eye? I’ve had to rush someone to the eyewash station, holding their head down, holding their eyelids open, counting a slow fifteen minutes. No shortcuts — only after a good flush can you start to hope for a positive outcome. Water works best to dilute and carry away the chemical; don’t hunt for special solutions or try to neutralize, just use a steady, gentle stream.
Skin contact calls for fast action too. Remove any contaminated clothing and rinse the affected skin with copious amounts of water. No scrubbing. No harsh soaps. Keep rinsing for at least half an hour. I’ve seen minor rashes turn serious when people underestimated how much soaked through socks or under sleeves.
Inhaling fumes means immediate access to fresh air. Supervisors in my labs always check for signs of dizziness or shallow breathing. If someone has trouble, 911 gets dialed on the spot, oxygen support may be life-saving, and hospital care is the next stop. Swallowing 2-Nitrobenzoyl Chloride rarely happens, but if it did, emergency medical help is essential — never induce vomiting, and never give drinks unless told to by professionals.
Reducing the Odds of Accidents
Working with these risks, precautions aren’t a choice. Training sessions stick with people if they feature real stories, not just warnings off a slide. Safety data sheets belong right at hand and need regular review. Local exhaust ventilation upgrades, regular equipment checks, and reminder labels on bottles make a real difference in busy labs. Keeping a running log of near misses reveals repeat weak spots. Applauding caution, never speed or bravado, makes for a team that gets home in one piece. No short supply of gloves, eyewash water, or time for a double check has ever equaled the cost of one accident.
| Names | |
| Preferred IUPAC name | 2-nitrobenzenecarbonyl chloride |
| Other names |
2-Nitrobenzoyl chloride O-Nitrobenzoyl chloride Ortho-Nitrobenzoyl chloride α-Nitrobenzoyl chloride |
| Pronunciation | /ˌtuːˌnaɪtrəˈbɛn.zɔɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 88-19-7 |
| Beilstein Reference | 1366058 |
| ChEBI | CHEBI:27658 |
| ChEMBL | CHEMBL15919 |
| ChemSpider | 11667 |
| DrugBank | DB08635 |
| ECHA InfoCard | 03bfa7e2-6be1-4f59-b55f-790b174aa52a |
| EC Number | 211-034-9 |
| Gmelin Reference | 171157 |
| KEGG | C14327 |
| MeSH | D009647 |
| PubChem CID | 7285 |
| RTECS number | BY8575000 |
| UNII | E6DK63C4PW |
| UN number | UN1669 |
| Properties | |
| Chemical formula | C7H4ClNO3 |
| Molar mass | 169.55 g/mol |
| Appearance | Pale yellow crystalline powder |
| Odor | pungent |
| Density | 1.43 g/cm³ |
| Solubility in water | Reacts violently |
| log P | 1.04 |
| Vapor pressure | 0.04 mmHg (25°C) |
| Acidity (pKa) | 1.1 |
| Basicity (pKb) | Basicity (pKb) of 2-Nitrobenzoyl Chloride: "11.08 |
| Magnetic susceptibility (χ) | -49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 1.565 mPa·s (20°C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -110.0 kJ/mol |
| Pharmacology | |
| ATC code | ATC code not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H319, H332, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P309+P310 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 112 °C |
| Autoignition temperature | Autoignition temperature: 540 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1030 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 640 mg/kg |
| NIOSH | DH6825000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 20 mg/m3 |
| Related compounds | |
| Related compounds |
2-Nitrobenzoic acid 2-Nitrobenzamide 2-Nitrobenzaldehyde 2-Nitrobromobenzene 2-Nitroaniline 3-Nitrobenzoyl chloride 4-Nitrobenzoyl chloride |

