2-Nitrobenzohydrazide: Deep Dive into a Key Chemical
Historical Development
2-Nitrobenzohydrazide holds a unique spot in the annals of organic chemistry, especially through its involvement in the synthesis of heterocyclic compounds. Decades ago, organic syntheses pointed towards nitroaromatic building blocks as some of the most reliable intermediates for pharmaceuticals and dyes. Chemists searching for new ways to tap into the versatility of benzoic acid derivatives found promise here. Research articles from the late 20th century, especially during the innovation bursts in the 1970s and 1980s, began emphasizing hydrazide functional groups and their reactivity with nitro-substituted aromatic rings. The pathway to 2-nitrobenzohydrazide tracks with the progress of hydrazinolysis protocols and echoes the growing sophistication of aromatic substitution reactions. Modern chemistry textbooks treat this molecule as more than just an academic curiosity—it sits right in the engine room of research for medicinal, dye, and material science applications.
Product Overview
2-Nitrobenzohydrazide appears as a light yellow crystalline powder. Commercial suppliers offer material for research labs, typically in fine or granular form, and with purity dissected down to decimals. This compound turns up across synthesis projects looking for a nitro group ready for reduction or a hydrazide tail prepared to open doors to diverse derivatives. Bench chemists reach for it because it can act as a linchpin for building larger, more complex molecules, especially those chasing selective biological action or color stability.
Physical & Chemical Properties
A closer look at its properties shows a melting point sitting tight between 180°C and 190°C under normal lab conditions, marking it as appropriately robust for heating reactions. Solubility in most organic solvents stays modest—solvents like dimethyl sulfoxide (DMSO) or dimethylformamide (DMF) work best for getting this solid into solution. The bright yellow tint signals the nitro group’s presence. Reactivity targets the hydrazide and nitro functionality: this combo creates both nucleophilic and electrophilic hot spots. The compound gives a sharp IR absorption from the nitro stretch near 1500 cm-1 and a broad stretch from the hydrazide NH groups. In well-kept dry storage, the material holds up admirably, resisting light oxidation and hydrolysis.
Technical Specifications & Labeling
Standard chemical suppliers classify 2-nitrobenzohydrazide with a CAS number 619-48-1. Material safety data sheets dictate labeling requirements: you’ll see hazard statements, precautionary codes, and information on emergency procedures for skin or eye contact. Laboratories aiming for top-tier purity generally look for assays above 98%, with water and residual solvent limits posted clearly on certificates of analysis. Product containers come with batch numbers for traceability, control lot numbers, and sometimes QR code tracking for digital record-keeping. The packaging aims to limit light and moisture ingress, using dark amber or opaque bottles, foam padding, and sealed liners.
Preparation Method
Large-scale and academic syntheses lean on a straightforward approach that begins with 2-nitrobenzoic acid. A stepwise conversion converts the acid chloride with thionyl chloride or oxalyl chloride, followed by treatment with excess hydrazine hydrate. The process generates a crystalline precipitate, filtered and washed repeatedly to shed reaction byproducts and residual acids. Careful temperature control prevents unwanted substitution on the aromatic ring or overreaction yielding polyhydrazide byproducts. A high-vacuum drying stage further curtails the risk of hydrolysis or decomposition during storage. Most practitioners avoid column chromatography at scale for this product, focusing instead on recrystallization from ethanol or isopropanol to obtain a sharp melting solid suitable for downstream chemistry.
Chemical Reactions & Modifications
2-Nitrobenzohydrazide does a heavy share of work in substitution and condensation chemistry. Under reducing conditions, the nitro group smoothly converts to an amine, opening new paths to anthranilic acid derivatives. With carbonyl-containing partners, the hydrazide tail forms hydrazones and a variety of heterocycles like phthalazines or benzotriazoles, both of which get prime billing in drug discovery. Under basic or acidic catalysis, acylation of the hydrazide nitrogen gives rise to new amide and urea chemistries. This molecule acts as a springboard to further tailor activity and physicochemical properties for advanced targets—pharmaceutical, pigment, and analytical reagent industries eagerly follow these leads for new innovations.
Synonyms & Product Names
Researchers and suppliers sometimes write 2-nitrobenzohydrazide as o-nitrobenzohydrazide, 2-nitrobenzhydrazide, or 2-nitrohydrazinobenzoic acid. Laboratory catalogues might list synonyms based on differing hydrazide naming conventions, but each points to the same molecule: a derivative of benzoic acid with a nitro group at the ortho-position and a hydrazide at the carboxy end. Some product sheets include the IUPAC name—2-nitrobenzohydrazide—or rely on English descriptors such as ortho-nitrobenzoic acid hydrazide, ensuring clarity and reducing confusion during ordering and chemical handling.
Safety & Operational Standards
Handling 2-nitrobenzohydrazide demands solid chemical hygiene and a level head for personal protection. Even for experienced hands, using gloves, goggles, and lab coats stands as standard practice due to the risk of skin sensitization or mild toxicity, especially during weighing or dissolution. Powder handling under fume hoods guards against inhalation—hydrazide dust, even in trace levels, warrants caution. Storage stays firmly at room temperature in dry, cool conditions, buffered from direct sunlight. Following local chemical hygiene protocols keeps bench areas and personnel safe, while spill kits for nitroaromatic materials should sit close at hand. Safety data, incident logs, and staff training get updated annually, especially for regulated or public-facing labs. Waste disposal runs through hazardous chemical streams, avoiding ordinary landfill or sink disposal because both the nitro and hydrazide groups mark this as persistent and potentially bioactive.
Application Area
Research labs keep 2-nitrobenzohydrazide near the top of the order list for heterocyclic synthesis, specifically when looking at antimicrobial screens or anti-tumor lead compounds. Dye manufacturers have used derivatives to tweak color fastness and stability in textile dyeing. Analytical chemists run it out in spot tests for aldehydes and carbonyl compounds—its hydrazide group forms robust colored hydrazones, useful in titrations and detection kits. Medicinal chemistry teams pick over the molecule for its ability to provide both nitro and hydrazide functionalities in a single scaffold, ideal for rapid exploration of chemical space.
Research & Development
Ongoing innovation circles around improved routes for synthesizing 2-nitrobenzohydrazide and derivatives. Green chemistry trends steer new prep methods away from hazardous chlorinating agents, probing catalytic systems and alternative dehydrating agents. Structure-activity relationships push teams to modify both the aromatic and hydrazide parts, mapping out bioactivity landscapes for the next round of antibiotics, anticancer drugs, and novel pigments. Analytical scientists refine detection methods, turning to LC-MS/MS and NMR for fast, sensitive quantification of this compound in complex mixtures. Patents on newer derivatives roll out steadily, underlining the competitive race to discover the next application.
Toxicity Research
Safety data sheds light on moderate acute toxicity: rodent studies at high doses point to irritation effects and mild organ impact, mainly at exposures far beyond typical laboratory use. The nitro group triggers caution—some aromatic nitro compounds cause methemoglobinemia or mutagenic effects, which prompts routine in vitro screens and regulatory oversight. Published work reports no evidence of carcinogenicity, though chronic exposure studies remain limited. Lab management prefers strict exposure minimization, air monitoring, and tidy documentation to stay compliant with occupational safety guidelines. Waste streams get bottled and sent for specialized chemical destruction, never left to evaporate or drain into municipal systems, underscoring a defensive approach toward environmental stewardship.
Future Prospects
Progress in synthetic methodology will shape the next wave of 2-nitrobenzohydrazide applications. More sustainable synthesis using eco-friendly reagents and solvent recycling jumps near the top of the R&D wish list. Bioactive derivatives call for new rounds of lab trials and patents, especially as pharmaceutical companies scan for next-generation antimicrobial scaffolds. Digital chemistry—AI driven screening, reaction optimization, and predictive modeling—may accelerate the pace and precision of designing new hydrazide derivatives. The demand for safer, greener, and more versatile building blocks means 2-nitrobenzohydrazide will not fade into the background of chemical catalogs any time soon. Researchers, safety officers, and manufacturers alike watch closely for every advancement, as each step forward promises both scientific and commercial rewards on multiple fronts.
What is 2-Nitrobenzohydrazide used for?
The Workhorse in Synthesis
In any decent chemistry lab, you find compounds like 2-nitrobenzohydrazide quietly doing the heavy lifting behind the scenes. This chemical isn’t something most folks encounter on a walk through the park, yet it’s a supporting player in the creation of plenty of things we actually use. Lab work involving 2-nitrobenzohydrazide tends to focus on making more complicated molecules, especially those found in medicines and agrochemicals. The structure gives chemists a handy tool for building up new rings or helping shape specific features in a molecule, which is a big deal in drug discovery and agricultural science.
Medicinal Chemistry and Drug Discovery
Pharmaceutical chemists use 2-nitrobenzohydrazide when searching for new drugs. The molecule acts as a starting point for assembling other, more complex compounds known as heterocycles. These ring-shaped chemicals show up often in medicines, including certain cancer drugs, antivirals, and antibiotics. Real progress against disease sometimes comes down to finding the right scaffold, and 2-nitrobenzohydrazide provides just that kind of base. Published studies, like those listed in journals such as the European Journal of Medicinal Chemistry, underline how scientists use this molecule to help create targeted treatments that fight disease right where it starts — at the cellular level.
Supporting Crop Protection and Food Security
Chemistry isn't just about pills and potions — it has an impact on farm fields and gardens, too. Researchers in crop science use 2-nitrobenzohydrazide to build pesticide candidates. Actual tests show some of these derivatives work well against fungi or beetles that threaten harvests. Food security matters more than ever as the world’s population grows, so substances like this pick up a lot of importance, even if nobody ever hears their odd names on the evening news.
Lab Techniques and Analysis
Certain scientists turn to 2-nitrobenzohydrazide for chemical analysis and detection tricks. This molecule reacts with aldehydes and ketones, letting researchers spot these groups in complicated mixtures, which can serve as a fast check of quality and purity in products and ingredients. Analytical chemistry often needs specialized, trustworthy reactants, and this one fits the bill for those particular reactions.
Safe Handling and Environmental Impact
Just because a substance is useful doesn’t make it harmless. I’ve seen enough lab safety posters and accident reports to know handling nitro-compounds requires care. Manufacturers and universities sign off on careful storage and disposal rules, and regulators expect companies to track waste and air emissions from production sites. Balancing utility and responsibility stays essential throughout the chemical industry, whether that means training staff or investing in cleaner processes.
Support for Future Breakthroughs
Breakthroughs in science rarely arrive from nowhere — they build on years of steady progress. 2-nitrobenzohydrazide acts as one of those building blocks. Chemists need access to versatile, reliable materials if they want to solve the new generation of problems, from antibiotic resistance to food shortages. I’ve seen promising research projects drop out of contention simply because key reagents weren’t available on time, so supporting production and distribution pipelines for chemicals like this deserves real attention.
What are the safety precautions when handling 2-Nitrobenzohydrazide?
Why Care About Safety?
Working with chemicals always brings back memories of my early days in the lab: lot of curiosity, but also a few rookie mistakes. Whether you’re in school, research, or industry, safety changes the outcome—often in ways you only realize after things go wrong. 2-Nitrobenzohydrazide, a hydrazide derivative, carries extra risk. The nitro group sets up a possibility for strong reactions, and the hydrazide structure brings toxicity and instability to the table. Not just a lab myth; I once saw a bottle degrade because someone left it near heat, leading to an expensive cleanup that could have been much worse.
Risks to Watch Closely
2-Nitrobenzohydrazide can cause skin rashes, eye irritation, or breathing problems. It might sound routine, but these symptoms can land folks in the doctor’s office. The compound’s dust lingers in the air if spilled, so proper air filtration matters as much as good gloves. There’s also a serious fire risk—especially if the compound reacts with reducing agents, metals, or strong bases. Keeping it locked away from sunlight and moisture protects both the handler and the building.
Making Good Habits Stick
People like to talk about safety goggles and gloves, but grabbing the right set is more than a box-ticking exercise. I stick with nitrile gloves and goggles that won’t fog up, because missing a spill in your field of view happens when you take shortcuts. A lab apron and solid footwear give protection from the chest down. Once, I watched someone get a compound splash on canvas sneakers; they limped for a week. Don’t skip the fume hood—even quick measurements send vapor into the room.
Best Storage Practices
Nobody wants to open a cabinet and smell something odd. Store 2-nitrobenzohydrazide in a tightly sealed container, in a cool and dry space that only trained folks can access. The right hazard labels on every bottle keep confusion low. Storing hydrazides away from acids, bases, and oxidizers cuts reaction risks down. Inventory logs save the day if a shipment arrives damaged—you know who managed each transfer, and when.
What Works in Case of Accidents
If the chemical touches your skin, rinse it with running water for at least fifteen minutes, and swap out contaminated clothing. Splashing in eyes calls for immediate eyewash. Every lab I work in keeps eyewash bottles at the door. Breathing difficulty after exposure can be serious: clear the area fast, move the person to fresh air, and call emergency medical help. Fire blankets, absorbent pads, and chemical spill kits should stay within reach. Posting emergency numbers in sight of every workspace keeps panic low if something unexpected happens.
Training Builds Confidence
Lab safety drills shouldn’t just be for show. Practicing cleanup and spill containment, from mock table-top simulations to real spill drills, means everyone feels confident if the real thing ever arrives. Staying curious about updates in chemical safety guides pays off. I’ve learned more from swapping stories with old pros than from the thickest manuals. Sharing direct experience keeps the next person from repeating costly mistakes.
Doing It Right, Every Time
Chemistry rewards the steady hand. You don't need a white coat and a PhD to care about safety. 2-Nitrobenzohydrazide can be a helper or a hazard, depending on how smartly it’s treated. Using personal protective gear, storing chemicals right, knowing what to do in an emergency, and keeping everybody trained build a safer environment for all. Careful habits don’t happen by accident—they show up through practice, respect for the risks, and learning from others’ hard-earned lessons.
What is the chemical structure of 2-Nitrobenzohydrazide?
Looking at the Skeleton
In chemistry class, drawing molecules can feel routine, but tracing out the structure of something like 2-nitrobenzohydrazide still gives a rush. The name itself throws out a challenge: you get a benzene ring, a nitro group, and a hydrazide. Breaking it down, that benzene ring holds onto a nitro group in the second spot and links up with a hydrazide. Seeing these three parts together, you know you’ve got a molecule ready to jump into interesting chemistry.
Structure Stitched Together
2-Nitrobenzohydrazide has a benzene ring—the backbone. That nitro group (NO2) sits on the second carbon. Opposite the nitro, the structure features a hydrazide moiety, which is basically a CONHNH2 chunk. In plain sight, this setup gives the molecule places to bond, react, and even attach hinges for building other compounds. It isn’t just a random arrangement. The nitro group pulls electrons, creating spots on the benzene ring that become more reactive. That’s how chemists know where to poke and prod—one reason these positioning details matter so much for anyone hoping to modify or use this in the lab.
Why Position and Groups Matter
It’s easy to gloss over chemical location. Stick a nitro in the wrong place, and you lose the exact qualities the molecule promises. At the second position, this group makes the whole structure act differently—polarizing certain bonds, opening up new doors for reactivity. In the hands of organic chemists, that feature means the difference between a useless compound and a stepping stone for new drugs or dyes.
Hydrazides themselves open doors for making hydrazones. The structure’s unique blend of groups often turns up in medicinal chemistry circles, where researchers spam the benzene ring with tests for biological activity. Some teams are looking at hydrazide-based scaffolds as lead compounds in pharmaceutical development. With the right tweaks, the molecule might show antibacterial or antifungal traits. And that starts right with the way each atom nests around the ring.
Lab Synthesis Concerns
Anybody who’s run a reaction with nitrobenzenes knows they don’t always play nice. Nitro groups can make things tricky; they’re not just placeholders but working components. That means safety goggles, good fume hoods, and some practical respect for the possibility of unwanted side reactions. More than once, I’ve watched colleagues triple-check a reaction scheme so everything lands in the correct spot—on the right carbon, in proper orientation. The chemical structure dictates these safety and handling needs, not some arbitrary guideline.
Applications and Next Steps
Lab teams have their eyes on compounds like 2-nitrobenzohydrazide for more than academic reasons. Designers want easy starting points for forming links (like hydrazones), which help create sensors, pharmaceuticals, and even specialty dyes. The unique pattern of groups means this molecule fits with others like a puzzle piece, letting researchers assemble test drugs with a known, tunable backbone.
Creating safer methods for handling and synthesizing hydrazides, as well as mapping out better routes for derivatization, could cut down on lab hazards and open up faster innovation. By keeping the focus on the molecule’s shape and functionality, research can inch toward building smarter compounds for health, agriculture, or industry—one carefully placed atom at a time.
How should 2-Nitrobenzohydrazide be stored?
Understanding What’s at Stake
Safety in a lab should never become a box-checking exercise. Chemicals like 2-Nitrobenzohydrazide deserve special handling because a mistake here can mean more than just paperwork. This compound, part of the benzohydrazide family, carries both nitro and hydrazide groups. That mix often points to sensitivity toward light, temperature, and air. For many chemists—myself included—the storage setup isn’t just a technicality. Strong habits keep incidents away, and the right storage builds that sort of trust in your workspace.
Core Storage Principles That Work
Solid experience says nobody should trust a warm shelf in a sunlit room for chemical storage. For 2-Nitrobenzohydrazide, the smart move is always a tightly sealed glass bottle kept far from heat and moisture. Most flammable or reactive chemicals tell part of the same story, but 2-Nitrobenzohydrazide asks for even more care because hydrazides don’t age well in damp or bright conditions.
Direct light kicks off slow decomposition, and moisture leaks in through loose lids or even thin plastic. Even before reading any technical data, I’ve lost count of the times I spotted ruined chemicals just from careless cap habits or overcrowded cabinets. You buy a fresh bottle, skip the storage protocol, and soon enough, there’s a smell no one wants to describe.
Storing for Stability
Best practice in most labs involves putting light-sensitive solids inside amber glass bottles, then sliding those into a cool, dry chemical cabinet. At home, my go-to setup included hygrometers and desiccant packs. Modern labs step it up with desiccator cabinets—especially where humidity spikes are common. The right container, the right seal, and the right shelf go together every single time. This isn’t just for insurance; poor storage often leads to impurities or even pressure build-up in the container.
Lab Culture and Training
People make systems strong or weak. New hires get sloppy if they never see examples set or receive reminders. As someone who trained younger researchers, I always walked them to the cabinet and showed what failed storage looks like. Once you see a yellowed, clumpy mass inside a clear jar, it makes the lesson stick. Older bottles, poorly cleaned lids, or labels stained by leaks all point to trouble. Supervisors can’t spot every slip, but regular checks and straightforward guidelines pay off. Lab manuals alone don’t do much; real safety culture comes from habits everyone actually follows.
Risk Reduction and Emergency Prep
Lowering risks comes down to vigilance. Fireproof cabinets help, but pairing that with proper temperature controls (usually between 2–8°C) and total separation from acids, oxidizers, or bases is needed. I’ve seen labs store incompatible chemicals side by side; that sort of shortcut courts disaster. Safety data sheets (SDS) exist for reasons—skipping them out of routine is like driving with your eyes closed. Knowing spill cleanup and basic response protocols saves time and, potentially, lives.
Solutions for Modern Labs
Automation helps but doesn’t solve everything. Tracking chemicals electronically helps managers spot risky patterns or expired bottles. Regular inventory clears away forgotten hazards. Working with 2-Nitrobenzohydrazide means accepting that careful storage isn’t just technical—it has moral weight too. Colleagues trust that what’s in one cabinet won’t put everyone at risk. That trust grows only with repeated, careful actions every day.
What is the molecular weight of 2-Nitrobenzohydrazide?
Numbers Behind the Name
The name “2-Nitrobenzohydrazide” can look intimidating at first, but there’s value in learning how these molecules break down in terms of their weight. For anyone who works with chemicals, molecular size gives clues about reactivity, toxicity, and even how a substance handles in the lab. Let’s peel it apart: the formula for 2-Nitrobenzohydrazide is C7H7N3O3.
Each atom pulls in its own weight. Carbon clocks in at about 12, hydrogen sits around 1, nitrogen is roughly 14, and oxygen is 16. Multiply those out: 7 carbons (84), 7 hydrogens (7), 3 nitrogens (42), and 3 oxygens (48). All together, the total molecular mass ends up at 181 grams per mole. In the lab, that small shift of a digit can mean the difference between a successful process and a glitch, especially if a chemist needs to scale reactions or predict yields. The number isn’t just academic trivia—it’s groundwork for actual work on the bench.
Why Weight Guides Research
I learned early on to triple-check these numbers. During a stint analyzing organic compounds at a small research company, I messed up a calculation through a typo—off by a single hydrogen. The mix wouldn’t react the way the literature predicted. That pound of caution I picked up from getting it wrong helped later on. In pharmaceuticals and crop science, these small molecules become the building blocks for much bigger plans. Incorrect weights create risk, from wasted time to failed syntheses and costly recalls.
On a practical level, knowing the mass of a compound like 2-Nitrobenzohydrazide matters for everything from ordering the right amount of material to setting dosages in an animal trial. No one wants to find out mid-project that the math doesn’t line up. For university students, accurate weights can be the difference between passing an exam or starting over in a lab practical. In manufacturing, tracking mass keeps batch-to-batch quality in check and sets the table for safe chemical handling. It’s a foundation, but one that supports everything stacked above.
Transparency and Trust: Why Numbers Count
Accuracy in reporting chemical weights forms the backbone of trust—between researchers, journals, and the wider public. Laboratories depend on reference databases like PubChem or Sigma-Aldrich to provide reliable numbers. Publishing the right molecular mass lets others repeat findings and verify claims. As an advocate for open science, I see honest reporting as a key part of any successful research project. A lab notebook with fuzzy or rounded numbers just invites problems later, especially under audit or review. That accuracy and transparency save headaches and reinforce the credibility of scientific work.
Making the Most of the Data
For anyone newly working with 2-Nitrobenzohydrazide, take the time to double-check and cross-reference sources. Include the precise molecular mass in datasheets and protocols. If suppliers or reference books report slightly different values, don’t be afraid to ask questions—quality control demands it. Sometimes manufacturers round numbers for packaging or inventory, but the science doesn’t allow shortcuts.
One smart habit is to use computer-based calculators or reputable chemical databases. Those digital tools pull from vetted information and do fast math without the risk of finger slips. Scientists, students, and industry partners all benefit when the number 181 lines up across every experiment, order, and report. Keeping these figures front and center builds confidence and helps everyone get on with the actual work of making and discovering new things.
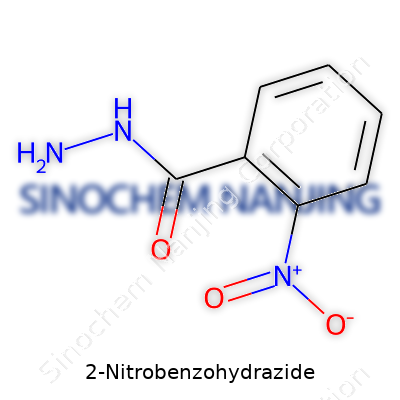
| Names | |
| Preferred IUPAC name | 2-nitrobenzenecarbohydrazide |
| Other names |
2-Nitrobenzhydrazide o-Nitrobenzhydrazide 2-Nitrobenzohydrazide o-Nitrobenzohydrazide 2-nitrobenzohydrazide |
| Pronunciation | /tuː-ˌnaɪ.troʊ.bɛnˈzoʊ.haɪ.drə.zaɪd/ |
| Identifiers | |
| CAS Number | 5751-55-3 |
| Beilstein Reference | 82164 |
| ChEBI | CHEBI:94877 |
| ChEMBL | CHEMBL1692807 |
| ChemSpider | 167357 |
| DrugBank | DB07757 |
| ECHA InfoCard | 03d1a01c-3999-4d57-8096-425d21536c7a |
| EC Number | 2.4.1." |
| Gmelin Reference | 211644 |
| KEGG | C06501 |
| MeSH | D009805 |
| PubChem CID | 107186 |
| RTECS number | SN6650000 |
| UNII | 6TRN2U8A7M |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C7H7N3O3 |
| Molar mass | 181.15 g/mol |
| Appearance | White to yellow crystalline powder |
| Odor | Odorless |
| Density | 1.46 g/cm3 |
| Solubility in water | soluble |
| log P | 0.01 |
| Vapor pressure | 1.41E-7 mmHg at 25°C |
| Acidity (pKa) | 11.17 |
| Basicity (pKb) | Basicity (pKb): 11.85 |
| Magnetic susceptibility (χ) | -53.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.687 |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 191.8 J mol⁻¹ K⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes skin irritation. Causes serious eye irritation. May cause an allergic skin reaction. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P501 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | 151°C |
| Lethal dose or concentration | LD₅₀ (oral, rat) > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 2-Nitrobenzohydrazide: "LD50 (oral, rat): >2000 mg/kg |
| NIOSH | NA |
| PEL (Permissible) | No PEL established |
| REL (Recommended) | 0.02 mg/m3 |
| Related compounds | |
| Related compounds |
2-Nitrobenzoic acid 2-Nitrobenzoyl chloride 2-Nitroaniline 2-Aminobenzohydrazide Benzohydrazide |

