2-Nitrobenzenesulfonyl Chloride: Perspectives from Science to Safety
Historical Development
Back in the mid-20th century, as organic synthesis kept moving forward, chemists needed something that could offer reliable sulfonylation without a lot of side reactions. 2-Nitrobenzenesulfonyl chloride emerged from research into nitroaromatic compounds, with early papers in the 1950s and 1960s exploring its strong electron-withdrawing capacity thanks to the nitro group. This little tweak meant scientists could not only protect amines but could also fine-tune how they exposed molecules to selective reactivity. The sulfonyl chlorides scene already had its big names—tosyl and nosyl—but the 2-nitrophenyl flavor found favor where heightened reactivity or tuneable lability made the difference. Chemical suppliers caught on, so by the 1980s, bottles of this yellow powder started cropping up on university shelves and in industrial research catalogs across continents.
Product Overview
2-Nitrobenzenesulfonyl chloride crops up in organic synthesis, playing a big part in amine protection and sulfonamide formation. The presence of the nitro group right on the aromatic ring brings unique attributes not found in similar compounds, giving chemists more control over reactivity. Vendors usually pack it in dark glass with tight seals to stop moisture from creeping in. Chemists buy it in everything from gram vials for bench research to kilogram drums for bulk operations, depending on the scale of their projects.
Physical & Chemical Properties
This compound appears as a pale to canary yellow crystalline powder, often with a sharp, biting odor common among sulfonyl chlorides. Its melting point hovers around 75-78 °C. It stays stable under dry conditions, but water kicks up a fuss—hydrolyzing to yield the corresponding sulfonic acid along with hydrochloric acid fumes. Its solubility tilts toward the organic end, with decent dissolution in acetone, DMF, DCM, and chloroform, while it shows little interest in water. With a molecular formula of C6H4NO5SCl and a molar mass that fits cleanly into the hands of any chemical calculator, 2-nitrobenzenesulfonyl chloride lines up as a practical reagent for many synthetic protocols. The nitro group really tweaks the electron density, steering nucleophilic attacks in a more predictable direction than other sulfonyl chlorides.
Technical Specifications & Labeling
Manufacturers label shipments with hazard pictograms covering corrosiveness, environmental risk, and acute toxicity, as expected under GHS regulations. A good supplier lists not only the CAS and molecular structure, but also batch-specific purity (usually not less than 99%), melting point, and loss-on-drying data. Batches often undergo HPLC to confirm organic purity, while heavy metal and residual solvent limits follow international pharma and fine chemical standards. Labels carry clear warnings about handling under dry conditions, as hydrolysis can be exothermic and produce toxic fumes. Storage advice points toward tightly sealed, moisture-free containers in well-ventilated chemical storage lockers.
Preparation Method
Chemists prepare 2-nitrobenzenesulfonyl chloride by sulfonation of nitrobenzene with chlorosulfonic acid, often under chilled conditions, followed by chlorination. The process demands careful control—temperature swings or contamination with water leads to nasty side-products and harder purification. Industrial processes optimize flow and agitation while minimizing waste, keeping the yield high and impurity count low. On a bench scale, scientists often generate smaller batches to avoid losses from hydrolysis. After sulfonation and chlorination, solvents like ethyl acetate help extract the product, while repeated recrystallization removes lingering impurities. This method has stood the test of time, reflecting both industrial efficiency and lab practicality.
Chemical Reactions & Modifications
In organic synthesis, 2-nitrobenzenesulfonyl chloride swings into action during sulfonamide and sulfonate ester formation. With amines and hydroxyls, it reacts briskly, often forming crystalline solids that are easy to purify. Its heightened reactivity—thanks to the nitro group—means milder conditions work, which is handy when dealing with sensitive substrates. The sulfonamide derivatives show useful properties not just as protecting groups but also as stepping stones in more elaborate syntheses. Reduction of the nitro group to an amine, followed by further substitutions, opens a window to a whole suite of chemical libraries, frequently used in medicinal chemistry. Furthermore, the highly electron-deficient aromatic ring takes part in cross-coupling reactions, such as Suzuki or Stille, under carefully tweaked conditions.
Synonyms & Product Names
Chemists coming across this compound in the literature might spot it listed under a crowd of names—2-nitrophenylsulfonyl chloride, o-nitrobenzenesulfonyl chloride, NSCl, or even just “nosyl chloride” in shorthand. Some catalogs have used older names like o-nitrobenzenesulfonic acid chloride, so anyone browsing chemical vendors or research papers benefits from knowing these tags to avoid confusion or potential mix-ups at the bench.
Safety & Operational Standards
This compound demands real attention to protective gear and ventilation. Skin or eye contact causes burns, and inhalation brings severe danger to the respiratory system. Hydrolysis in air or with water generates hydrogen chloride—no friend to mucous membranes. Every experienced chemist knows to keep work strictly inside functioning fume hoods, with nitrile gloves, goggles, and sometimes full-face shields, making accidental release less likely. Spill protocols require neutralizing acid vapors and removing contaminated materials in sealed disposal bags. Waste goes straight to designated hazardous chemical bins, keeping the chemical drain-free. Emergency showers and eyewash stations should sit close to any bench where the reagent gets used.
Application Area
2-Nitrobenzenesulfonyl chloride finds heavy use where selective amine protection is a must—peptide synthesis, combinatorial chemistry, and building bioactive heterocycles, to name a few. In the hands of a skilled organic chemist, it helps create building blocks for pharmaceuticals, including enzyme inhibitors and neuroactive agents. The nitro group doesn’t just participate—it lets chemists “switch” reactivity, cleaving protective groups or launching further transformations on cue. In the materials world, its derivatives have roles in custom dye synthesis and specialty polymers, providing new properties to coatings or electronic materials. Even in life sciences, its controlled release in peptide manufacturing sets it apart from more pedestrian protecting groups.
Research & Development
Labs continually tweak both the reagent and its applications. Researchers focus on “green chemistry” approaches for its synthesis, hoping to move away from harsh chlorinating agents and generate less chemical waste per batch. In medicinal chemistry, nosyl-protected intermediates stay popular, as the group’s stability helps during complicated multi-step syntheses. Disposable plastics, environmental catalysts, and advanced dyes built on the 2-nitrobenzenesulfonyl skeleton find their way into research papers each year. Chemical education also relies on small-scale use, as nosyl chemistry illustrates key concepts in selectivity and protection in organic labs around the world.
Toxicity Research
Toxicology studies highlight risks posed by inhalation, ingestion, or skin absorption, with animal studies reporting acute and chronic effects. The compound irritates the lungs and triggers coughing and nausea even at low exposures. Degradation products, particularly nitroaromatic intermediates, present mutagenic and ecological hazards. Modern toxicity assessments push for comprehensive evaluation, using both animal and in vitro studies to build guidelines. Research partners with regulatory agencies, providing data that shapes workplace exposure limits and waste disposal standards. Workers, including those in downstream industries using nosyl-protected intermediates, benefit from better education and stricter monitoring, lessening the old risks faced in the chemical industry’s early years.
Future Prospects
Looking ahead, green chemistry stands as a beacon. Developing milder, less polluting ways to manufacture and use 2-nitrobenzenesulfonyl chloride will keep it relevant in both industry and academia. Synthetic biologists and medicinal chemists clamor for more toolkits—compounds that allow precise building, cutting, and recombining of molecular frameworks. New analogs based on the nitrobenzenesulfonyl group may deliver even greater control or shed persistent toxicity concerns. Digital modeling, high-throughput screening, and automation give a boost to research, letting chemists sift through dozens of potential derivatives without pouring as many liters of solvent. In the long run, safety and sustainability will walk hand in hand with innovation, proving that even a chemical from the last century can evolve and find fresh purpose in the decades to come.
What is 2-Nitrobenzenesulfonyl Chloride used for?
Behind the Scenes in the Lab: The Real Utility
2-Nitrobenzenesulfonyl chloride, often called o-NsCl by people who spend time in synthetic labs, doesn’t show up on talk shows. You don’t find it on supermarket shelves or in the news most days. But for chemists and researchers, it has a lived importance in making molecules that eventually work their way into drugs, advanced materials, or once simply served to prove a theory out in some dusty PhD thesis. I’ve watched folks pull out this powdery stuff with careful hands, letting a little bit transform a clear flask into something colored, something new. That’s the excitement of organic chemistry, where everything depends on the small, the obscure, the reactive.
A Selective Tool for Tough Jobs
This compound plays a starring role as a reagent for protecting amines. In synthetic chemistry, protecting groups keep certain parts of a molecule from reacting while chemists tinker with other sites. If you’re trying to build a drug molecule, often you want to mask an amine until the last steps. 2-Nitrobenzenesulfonyl chloride makes a tough, reliable shield. It works quickly and doesn’t get peeled away too soon. Later, with the right conditions, chemists can remove it without damaging the rest of the molecule.
The significance stretches further. Research teams in pharmaceutical discovery use this chemical for complex peptide synthesis—stringing together chains of amino acids that form part of medicines, biologics, or imaging tools. The nitro group also acts as a visible marker in some procedures. If the color in your reaction flask changes, you know something happened.
Making Science Happen Safely
Sulfonyl chlorides like o-NsCl don’t earn safety awards. They’ve got a sharp odor and can irritate skin, eyes, or lungs. Good lab technique isn’t optional when using these chemicals. I remember old mentors insisting on fresh gloves, well-sealed goggles, and patience during weighing. Proper ventilation and clear labeling make a difference, especially for students or new lab staff who might not recognize the risks. It’s the kind of practical knowledge that sets apart a well-run lab from a dangerous one.
Why Use o-NsCl and Not Something Simpler?
There are other sulfonyl chlorides and protecting groups. What sets this one apart is the combination of high selectivity and easy removal when the synthetic sequence demands it. In tricky syntheses, especially those targeting new antibiotics or cancer drug candidates, shortcuts cause delays. Using a proven tool cuts down on wasted time and failed batches. Publications and patents often reference o-NsCl as an industry standard, and the fact that graduate students worldwide keep using it means it fills a real need.
Challenges and Safer Directions
The downsides deserve attention. Handling generates hazardous waste that must be neutralized and disposed of properly. Modern organic chemistry stresses greener approaches. There’s a push to design alternative protecting groups that avoid churning out toxic byproducts. New research explores enzyme-based or recyclable protecting agents, drawing inspiration from nature’s efficiency. As these ideas get refined, hopefully, future chemists will handle fewer dangerous powders without sacrificing the precision required for advanced synthesis.
In a World of Small Details
2-Nitrobenzenesulfonyl chloride carries a quiet but essential weight in labs committed to building ever more complex molecules. Its value comes from decades spent sorting out when and how to use it, balancing performance with health and environmental awareness. Tools like this don’t stand out to most people, but the work they power leads to discoveries that shape medicine, technology, and science more broadly.
What is the chemical formula of 2-Nitrobenzenesulfonyl Chloride?
Understanding the Compound In Practice
Step into any organic chemistry lab, and odds are you’ll find bottles labeled with long names and cryptic formulas. Somewhere on one of those shelves, 2-nitrobenzenesulfonyl chloride might show up, almost as a rite of passage for anyone who has spent much time manipulating sulfonylation reactions. This compound’s formula — C6H4(NO2)SO2Cl — doesn’t only represent the atoms tied together; it points toward a specialty reagent that often finds its way into peptide chemistry or as a tool for protecting amines.
Digging Into the Structure
What stands out is how the molecule combines three functional groups onto a single benzene ring: a nitro group (NO2) anchored at the 2-position, a sulfonyl chloride group (SO2Cl) attached directly to the ring, and the signature aromatic foundation. This construction gives it power and selectivity in reactions. In the lab, you can almost smell the unique edge chlorosulfonic reagents bring — if you’re experienced, you also respect their bite and reactivity, especially in moist air.
Practical Impact in Chemistry Labs
Sizing up its uses shows why it matters. It’s not just a bit player in theory lectures or a curiosity for textbook charts. Researchers rely on this molecule to protect nitrogen atoms in drugs and intermediates. This protection simplifies reaction pathways, prevents unwanted side reactions, and lets chemists build complexity molecule by molecule. Experienced chemists often turn to 2-nitrobenzenesulfonyl chloride when they want precise protection and deprotection strategies—especially when making substances like antibiotics, enzyme inhibitors, or even in early-stage diagnostics.
Health and Environmental Considerations
Mixing powerful chemistry with safety brings its own challenges. 2-nitrobenzenesulfonyl chloride, armed with both a chlorinated end and a nitro group, demands respect in the fume hood. Even seasoned professionals know to keep it behind glass, with gloves tight on their hands, and eyes wary of how fumes or even particles can sneak into lungs or eyes. Environmental impact shouldn’t be ignored, either. Sulfonyl chlorides carry risks during disposal, and chemists who take stewardship seriously look for greener approaches and tighter waste management.
Pushing Toward Safer, Cleaner Reactions
Solutions don’t just fall from the sky, but there’s steady progress. Green chemistry aims for alternatives that sidestep hazardous byproducts, often swapping simpler protection groups or turning to enzyme-catalyzed reactions that create less waste. Some chemists design new routes that cut down on the number of steps, trimming out stages that require harsh reagents like sulfonyl chlorides altogether. Others push for capture technologies to trap and neutralize toxic off-gassing before it can drift out of the lab and into the world.
The Formula Speaks Volumes
It’s easy to see C6H4(NO2)SO2Cl as a random soup of elements, but the real world sees in it more than just a formula. Each atom links to practical realities: lab workflow, safety, professional responsibility, and environmental care. Experience teaches that every decision, from how reagents are chosen to how waste exits the lab, shapes both science and the world around it.
How should 2-Nitrobenzenesulfonyl Chloride be stored?
Practical Safety Starts With the Basics
I’ve worked in a few different labs – from small university set-ups full of beakers and big dreams to busier industrial spaces where chemical drums crowd every aisle. No matter the setting, most people learn pretty quickly: cut corners while storing reactive chemicals and trouble shows up sooner or later. 2-Nitrobenzenesulfonyl chloride isn’t just another bottle that gets tucked away. Its reactivity can make a lab tech’s day considerably more eventful—often in the worst possible way—if it’s left to its own devices.
The first thing that stands out once you’ve handled this chemical for a while is its sensitivity. The compound reacts with water, so letting it face humid air will lead to decomposition. Forget about pouring it back in quickly after weighing—it starts breaking down as soon as a container sits open, and anyone who’s walked into a storage closet to find mysterious white crust on lids knows the headache that follows.
Real-World Storage Solutions
Glass bottles with airtight seals help a lot. Polyethylene containers might look sturdy, but many degrade over time or fail to provide reliable barriers. Sticking with well-made glass matters, especially since this chemical also gives off fumes that slowly eat at rubber or some plastics. Once, I saw a screw cap fail and drip – just a tiny bit of vapor ruined a cardboard shelf overnight.
The storage spot itself asks for care. 2-Nitrobenzenesulfonyl chloride stays more stable at lower temperatures. Putting it in a fridge at 2-8°C lengthens shelf life while calming down any slow decomposition. Anything colder, like a deep freeze, usually isn’t necessary, but I wouldn’t suggest leaving this chemical out at room temperature in the middle of a muggy summer. Every lab has stories of unexpected heat spikes pushing chemicals over their line.
Keeping Moisture and Light Out
Desiccant packs make a difference. Tossing a packet or two inside the storage box soaks up stray moisture. In big stockrooms, dry boxes with humidity indicators let staff know before water vapor creeps in. I can remember walking the aisles with a notebook, watching little circles turn pink—an easy fix that kept things running smoothly.
Light throws another wrench. Sunbeams streaming through a window warm up shelf space fast and can cause certain chemicals to degrade. For this reason, all our sensitive compounds stayed in amber bottles or behind solid cabinet doors where light couldn’t sneak in.
Label Carefully, Lock It Up
A legible label with the full chemical name, hazard indicators, and the date received reduces confusion. I once spent half a morning sorting unmarked jars after a coworker tried abbreviating everything in Sharpie. Simple, clear labeling keeps everyone safe and makes audits or emergency response faster.
Hazardous chemicals deserve a locked, ventilated cabinet, ideally with a chemical spill tray beneath. In my experience, theft isn’t the only risk: curious colleagues and clumsy hands cause more damage. Having quick access to spill kits, goggles, and gloves nearby means nobody needs to run across the lab for emergency gear.
Training That Sticks
Handling lessons help everyone remember the basics: never return unused material to stock, always clean up spills right away, and treat every open container as a fresh risk. I’ve seen routines break down when people chase efficiency—reminders posted near cabinets kept us honest. Regular refreshers and honest talk about close calls can prevent bigger mistakes.
What are the safety precautions when handling 2-Nitrobenzenesulfonyl Chloride?
Why 2-Nitrobenzenesulfonyl Chloride Demands Respect in the Lab
Anyone who has spent time working with complex organic chemicals knows how easily an overlooked precaution can send a smooth day sideways. 2-Nitrobenzenesulfonyl chloride isn’t as notorious as fuming acids or pyrophoric powders, but it’s not something to take lightly. Ineffective handling can invite real trouble—health risks from inhalation and skin contact, property damage, or environmental headaches if spills escape containment.
Keeping Health Risks Under Control
I learned pretty early not to underestimate sulfonyl chlorides. Vapors bite at the eyes and nose, and the powder itself has no business on bare skin. That harsh odor isn’t just annoying; it’s a warning. Treat this compound as a respiratory and skin hazard from start to finish. A pair of safety goggles stops splashes before they touch your eyes. Adding a face shield keeps stray droplets away during more active transfers. Chemical-resistant gloves (nitrile or neoprene) actually make a difference, since latex doesn’t hold up against this stuff. A standard lab coat blocks accidental drips, and buttoning it up stops the chemical from getting trapped close to the skin.
Air Quality: Keeping Toxic Clouds at Bay
Fresh air means everything. Good chemical fume hoods trap vapors and make sure the room doesn’t fill up with invisible nastiness during weighing or mixing. A regular old exhaust fan doesn’t cut it. Folks without access to a certified fume hood really ought to reconsider the work, or move it somewhere better equipped.
Smart Storage—Not Just Shelf Space
2-Nitrobenzenesulfonyl chloride needs dry, cool, and tightly sealed quarters. Humidity and heat degrade the chemical, and that weakens your reactions while potentially releasing toxic fumes. Polyethylene or glass bottles work well—anything metal is a bad idea, given sulfonyl chlorides’ habit of corroding some alloys. Each bottle gets dated, labeled in bold, and placed away from bases, water, and strong oxidizers. Good storage practice means never stacking heavy stuff on top or crowding the shelf. If you’ve ever tried to dig an important reagent out from behind a pile, you know the risk that comes with a rushed grab.
Dealing With Spills and Waste
Spills demand immediate cleanup with absorbent pads meant for acids or reactive chemicals. Make sure the workspace has a spill response kit (not just a broom and dustpan). Scraps and rinse water go straight into dedicated waste containers—don’t dump anything down the drain. Most local regulations treat these wastes with strict oversight, and for good reason. Chlorinated waste finds its way downstream easily if not managed.
Training and Emergency Prep
Every researcher, intern, or technician must know emergency procedures. It’s not enough to just point to an eyewash station or humor a quick demo on spill control. Practicing emergency drills and walking through the steps in real time makes the difference during a real incident. I’ve seen nerves freeze good scientists who never rehearsed what to do with an accidental splash or leak.
Constant Respect Pays Off
Safe handling can sound routine until something skips a step. 2-Nitrobenzenesulfonyl chloride commands caution. Shortcuts—gloveless hands, open benches, ignored ventilation—almost always bring regret. A well-designed lab setup, good training, the right gear, and respect for waste disposal protect everyone involved. Following these simple habits keeps days productive and sends everyone home healthy.
What is the appearance and physical properties of 2-Nitrobenzenesulfonyl Chloride?
An Insider’s Look at a Key Reagent
Working in a lab, you get a hands-on feel for chemical substances, and 2-nitrobenzenesulfonyl chloride has its share of quirks. You’ll notice right away that this compound carries a yellow tint, and it appears as fine crystals or sometimes as a more clumpy powder. Forget anything transparent or subtle — even a quick glance in the weighing dish tells you this stuff doesn’t belong among the bland, white crowd of typical sulfonyl chlorides. That yellow color gives away its aromatic nitro group right off the bat, and it stands apart from similar compounds.
Physical Properties That Stand Out
If you’ve handled reagents that react to air and moisture, you know the drill. This one is no exception. The slightest interaction with the room’s humidity creates a hint of pungency in the air. That sharp, almost acidic smell reminds you to reach for gloves and a good fume hood. Its melting point hovers near 70°C, a detail that comes up during purification. Priming your hotplate to navigate the narrow gap between solid and liquid means that spills can quickly turn messy — so folks tend to respect the yellow stuff. Once melted, it doesn’t hang around as a liquid for long, and exposure to heat can make it decompose, releasing sulfur dioxide and hydrochloric acid gases.
Solubility often determines how a reagent can be manipulated. Here, water only causes trouble. Even a splash triggers hydrolysis, which means the compound readily breaks down, producing sulfuric acid derivatives and plenty of heat. Tossing some into dichloromethane or chloroform gets better results, since these solvents keep the chemical in solution without wrecking its structure. Trying to clean glassware touched by this chemical leaves behind a sour memory because residues cling tight — and anyone with years behind the bench learns to check with indicator paper just in case.
Safety Matters
No getting around it: 2-nitrobenzenesulfonyl chloride can hurt if it meets bare skin or eyes. Irritation isn’t a maybe, it’s a fact you remember long after the burning stops. That’s not just hearsay; safety data backs this up. Gloves, eye protection, and a hood aren’t “suggested” — they’re as standard as a fire extinguisher in a kitchen. Those gases, sulfur dioxide and hydrochloric acid, do not make a pleasant perfume, and anyone ignoring basic ventilation is in for an ugly surprise. Your colleagues will remember who poisoned the lab air, and it’s best not to earn that reputation.
A Role in Chemical Synthesis
This compound finds its place when building more complicated molecules, thanks to the sulfonyl chloride group. It acts as an activating or protecting agent, most commonly for amines or alcohols in organic synthesis. Its reactivity comes as both a blessing and a curse. On one hand, you can get efficient transformations. On the other, mishandling means wasted time, ruined experiments, and safety headaches.
Years of experience underscore the importance of both skill and respect in chemical work. Reliable data from trusted sources, like the Merck Index and peer-reviewed journals, reveal the same facts: yellow crystals, strong odor, eager to react, and unforgiving if handled carelessly. Over time, you learn the limits, the shortcuts, and, more importantly, you learn the reasons for every rule in the book when dealing with 2-nitrobenzenesulfonyl chloride.
Looking Ahead
Moving toward safer handling starts with solid training and reliable infrastructure. Clear labeling, diligent storage in sealed containers, and handling waste responsibly all make a difference. Labs benefit by fostering a culture where every scientist slows down and pays attention — not just to succeed in their synthesis, but to keep every set of eyes and lungs in the building protected. The reality remains: mastery isn't only about knowing the chemical formula. True know-how comes from recognizing the warnings your senses and your colleagues send your way.
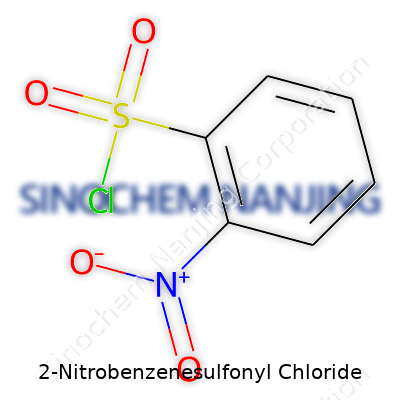
| Names | |
| Preferred IUPAC name | 2-nitrobenzenesulfonyl chloride |
| Other names |
o-Nitrobenzenesulfonyl chloride 2-Nitrobenzenzenesulfonyl chloride o-Nosyl chloride 2-Nosyl chloride 2-Nitrobenzenesulfonic acid chloride |
| Pronunciation | /tuː ˌnaɪtroʊˌbɛnˈziːnˌsʌlˈfəʊnɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 612-89-7 |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:51768 |
| ChEMBL | CHEMBL15406 |
| ChemSpider | 12744 |
| DrugBank | DB14115 |
| ECHA InfoCard | 100.011.202 |
| EC Number | 221-002-6 |
| Gmelin Reference | 83857 |
| KEGG | C14321 |
| MeSH | D017967 |
| PubChem CID | 74822 |
| RTECS number | DB6125000 |
| UNII | TXF86B86D2 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DJZWSCHWQSLFPO-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | C6H4ClNO4S |
| Molar mass | 205.63 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.56 g/cm³ |
| Solubility in water | Decomposes in water |
| log P | 0.35 |
| Vapor pressure | 0.00111 mmHg (25°C) |
| Acidity (pKa) | -6.5 |
| Basicity (pKb) | pKb = 11.92 |
| Magnetic susceptibility (χ) | -55.0e-6 cm³/mol |
| Refractive index (nD) | 1.624 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 357.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -223.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -634 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | Precautionary statements: "P261-P280-P305+P351+P338-P304+P340-P312 |
| NFPA 704 (fire diamond) | 2-3-0-W |
| Flash point | 158°C |
| Lethal dose or concentration | LD50 oral rat 508 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 1750 mg/kg |
| NIOSH | SN8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
2-Nitrobenzenesulfonamide o-Nitrobenzenesulfonic acid 2-Nitrobenzenesulfonic anhydride 4-Nitrobenzenesulfonyl chloride Benzene-1,2-disulfonyl chloride |

