2-Nitrobenzenearsonic Acid: A Deep Dive
Historical Development
Scientists first encountered 2-nitrobenzenearsonic acid during the heyday of arsonic acid chemistry, roughly a century ago. Back then, interest revolved around developing specialized organoarsenic compounds for research, agriculture, and animal health. Chemists pushed boundaries, drawing from earlier discoveries such as atoxyl and arsonic acid derivatives. Over time, synthetic skills improved and researchers gained control over the introduction of nitro groups, giving rise to new compounds like 2-nitrobenzenearsonic acid. These milestones reflected both scientific curiosity and practical need, particularly for understanding how different substituents on an aromatic ring produced dramatic changes in a molecule’s biological reactivity and industrial usefulness.
Product Overview
2-Nitrobenzenearsonic acid stands out due to its distinct functional groups, featuring a nitro moiety and an arsonic acid group attached to the benzene ring. This setup influences its chemical behavior, solubility patterns, and reactivity. The balance between these groups determines how the compound performs in synthesis, its interactions with biological systems, and its stability during storage. Laboratories and manufacturers recognize these attributes, focusing on purity and consistent molecular structure to match research or industrial requirements.
Physical & Chemical Properties
The compound generally appears as a yellow to brownish crystalline solid. Its melting point hovers close to 200°C, and it demonstrates moderate solubility in water, with higher solubility in basic solutions. Chemically, both the nitro and arsonic acid groups contribute to acidity and electron-withdrawing effects, driving reactivity in both organic and inorganic transformations. These groups also impact how the molecule interacts with transition metals and biological molecules, making its chemistry rich for study. Researchers have established strong UV absorbance related to its aromatic and nitro components, and analytical chemists often rely on infrared and NMR techniques to verify structure.
Technical Specifications & Labeling
Producers usually specify content above 98% for research-grade material. Common practice includes detailed labeling with batch number, production date, recommended storage conditions, and warnings about toxicity and handling. Laboratories get product data sheets with spectra and analytical results for trace contaminants like heavy metals, residual solvents, or organic byproducts. Accurate labeling, safety codes, and compliance with international shipping standards remain essential for trustworthy sourcing, especially with regulatory scrutiny around arsenic-bearing chemicals.
Preparation Method
Crafting 2-nitrobenzenearsonic acid often starts with nitration of aniline derivatives, producing 2-nitroaniline, which then undergoes arsonation by reaction with arsenic acid or sodium arsenite under controlled conditions. This stepwise process requires meticulous temperature control and purification stages to remove unreacted materials and unwanted byproducts. Each phase of synthesis poses risks — exposure to arsenic reagents and strong acids — pushing experienced chemists to work under fume hoods, wear full PPE, and neutralize waste properly. Improvements in lab technique and analytical monitoring now minimize environmental release and worker exposure.
Chemical Reactions & Modifications
2-Nitrobenzenearsonic acid’s dual functional groups make it a starting point in synthesizing more complex organic molecules. Reducing the nitro group yields 2-aminobenzenearsonic acid, which opens up another world of reactivity, from diazotization to coupling reactions for dye manufacture or pharmaceutical research. The arsonic acid moiety can form metal salts or esters, some with unique biological activities. Modifying either group steers the molecule’s pharmacological profile, which matters in drug screening or agrochemical studies. Recent research points to selective transformations using mild catalysts, a nod to growing environmental limits on harsh chemicals.
Synonyms & Product Names
2-Nitrobenzenearsonic acid appears in catalogs under several names. Chemists might call it ortho-nitrobenzenearsonic acid or use trade names inspired by manufacturer history. Sometimes suppliers employ systematic names or CAS registry numbers for legal tracking. The wide array of labeling reflects both taxonomy debates between chemists and the evolution of chemical industries into global players, each wanting to brand their offerings competitively. This patchwork of names makes accurate specification in procurement documents and research literature critical to prevent costly mistakes.
Safety & Operational Standards
Handling 2-nitrobenzenearsonic acid brings substantial risk due to both arsenic and nitro content. Workers should avoid inhalation, skin contact, or ingestion. Labs and factories demand chemical-resistant gloves, full goggles, and properly vented workspaces. Safety data sheets point to toxicity concerns—chronic exposure risks organ damage, while acute exposure may trigger irritation or systemic symptoms. Any spill or solid waste requires strict disposal in line with hazardous waste protocols. Regular air and surface monitoring, ongoing staff training, and strict inventory control help safeguard both the workforce and the outside environment.
Application Area
The main uses for 2-nitrobenzenearsonic acid stem from its place as an intermediate. Animal nutrition research formerly relied on arsonic acid compounds to promote growth and control intestinal infections, though changing laws and public concern over residual arsenic in meat have pulled back usage. Synthetic organic chemists use the molecule to create dyes, sensors, or more advanced functionalized aromatics. Specialized uses surface in analytical chemistry, where its metal-binding ability aids in detecting certain ions. In some circles, the compound also figures in exploring the nuanced mechanisms of arsenic toxicity at the molecular scale.
Research & Development
Much current research now focuses on designing derivatives with lower toxicity or studying the environmental fate of arsonic acids. Some labs look for catalytic conversion processes to recover or destroy arsonic groups after industrial use. Others apply computational models to predict biological activity or environmental persistence. Funding agencies increasingly ask for green chemistry designs, meaning milder conditions, less waste, and alternative reagents that minimize ecological burden. Academic partnerships with regulatory bodies encourage greater transparency on risks and alternatives, setting higher bars for ongoing development.
Toxicity Research
Two main hazards shape the risk profile: arsonic acid’s link to systemic arsenic poisoning and the nitro group’s ability to induce oxidative stress in cells. Long-term exposure in animal models shows accumulation primarily in liver and kidney tissue, while environmental studies find slow breakdown and potential contamination concerns for soil and surface water. Regulators limit allowable residues sharply in food and feed. “No safe level” messaging drives precautionary handling and ever-stricter disposal regulations. Toxicologists continue mapping the subcellular impact of these compounds, striving for faster detection and effective antidotes.
Future Prospects
The spotlight now falls on safer alternatives, more selective synthesis, and thorough environmental risk assessment. Demand for arsonic acid derivatives in agriculture winds down in the face of consumer worry, while niche opportunities remain for tailored chemical syntheses or unique sensor technologies. New chemical modifications may unlock biocompatible versions for targeted research, though regulatory frameworks grow tighter each year. The big picture now involves balancing research curiosity, industrial utility, and public health, ensuring that knowledge about arsonic acids turns toward greener, lower-risk solutions for the laboratory bench and the broader world.
What are the main applications of 2-Nitrobenzenearsonic Acid?
How 2-Nitrobenzenearsonic Acid Made Its Mark in Animal Nutrition
For decades, chemists have pushed the boundaries of organic synthesis, but the world rarely hears about the sometimes hidden players like 2-nitrobenzenearsonic acid. Farmers once used this compound as an additive in the feed of livestock—especially chickens and pigs—to promote growth and fight off gut infections. Some called it by the trade name Roxarsone. The idea was that adding this material to an animal’s diet helped them convert feed into body weight more effectively. In my own experience growing up near poultry farms in the Midwest, I heard plenty about “feed supplements” that made birds grow faster. What people didn’t talk about as much was what happened to the byproducts.
Troubles Surrounding Its Use and Residues in the Environment
Use of 2-nitrobenzenearsonic acid sparked controversy. Big agencies like the U.S. Environmental Protection Agency (EPA) and the Food and Drug Administration (FDA) started to question whether arsenic-based additives were staying in the animals or finding their way elsewhere. Scientists tracked residues not just in chicken meat, but also in droppings. Arsenic from the compound could wind up in manure, and from there, into soil and nearby water. The evidence grew: arsenic doesn’t just vanish. It can linger, change form, and sometimes increase toxicity as it moves through the environment. Some chicken growers argued that they needed all the help they could get with disease control, but health researchers said the risks ultimately outweighed the benefits.
Shifting Toward Safer Feed Practices
By the early 2010s, mounting concern led to a full rethinking of this chemical’s place in farming. Big poultry producers like Tyson and Perdue dropped the use of arsenic-based additives and chose alternatives. Veterinary science shifted to using more probiotics and, in cases where necessary, carefully managed antibiotics. The move was slow and far from perfect, but it reflected a common-sense approach: if the potential harm to food and water is high, switch to something safer—especially when that food lands on so many kitchen tables. Growing up, I saw that local water supplies were always a big topic at community meetings, especially whenever new contaminants turned up.
Industrial, Research, and Forensic Applications
In the lab, some specialty researchers still work with 2-nitrobenzenearsonic acid. Certain scientists study its reactivity, trying to create materials for analytical tests or trace chemical changes during environmental monitoring. The compound has appeared in methods that detect heavy metals and in exploring how arsenic-containing molecules move through living systems. Research use comes with strict safety protocols because the hazards are well known.
What's Next and Possible Alternatives
Awareness leads to progress. Food safety groups and environmental chemists suggest sticking with non-arsenic-based growth enhancers and turning to better husbandry instead. Biological controls—think beneficial bacteria or yeast—now play a bigger role in animal health than such synthetic additives did years ago. Water monitoring and improved regulation keep pressure on manufacturers to find less harmful options.
People want to trust the food and water in their communities. Cutting out risky compounds like 2-nitrobenzenearsonic acid marks a step in that direction. Matter-of-fact alternatives and better oversight offer a safer path forward in both farming and environmental management.
What is the molecular formula and molar mass of 2-Nitrobenzenearsonic Acid?
Chemical Identity and Structure
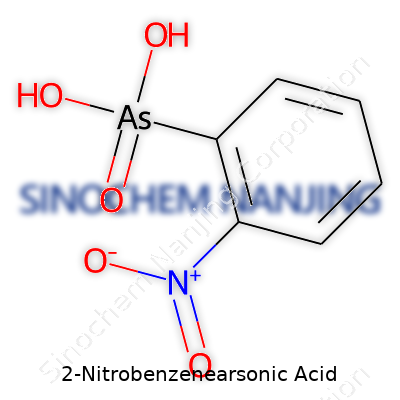
2-Nitrobenzenearsonic acid shows up in chemistry discussions, usually thanks to its mix of a benzene ring bolted together with two strong groups—arsonic acid and nitro. Its structure reveals a tale of carbon, hydrogen, nitrogen, oxygen, and arsenic holding hands. Chemists sketch its formula as C6H6AsNO5. Picture this: six carbons shape a ring, topped with six hydrogens. Slap on one arsenic, one nitrogen, and five oxygens. That’s chemistry’s way of combining the world of living things with the trickier elements.
Calculating Molar Mass: Digging into the Numbers
Back in graduate school, we spent hours with periodic tables, adding up every single atom to get the right molar mass. For anyone faced with a lab notebook and a need for precision, picking out the correct numbers is not only about passing a test; it determines quantities and reactions.
Let’s lay it out:
- Carbon (C): 12.01 g/mol × 6 = 72.06 g/mol
- Hydrogen (H): 1.008 g/mol × 6 = 6.048 g/mol
- Arsenic (As): 74.92 g/mol × 1 = 74.92 g/mol
- Nitrogen (N): 14.01 g/mol × 1 = 14.01 g/mol
- Oxygen (O): 16.00 g/mol × 5 = 80.00 g/mol
Totaling everything nets a molar mass of 247.04 g/mol. Good to remember that another decimal place now and then helps in a real lab, especially for researchers calibrating sensitive scales or engineers dosing up industrial batches.
Why Knowing the Formula and Mass Matters
Precision matters in the real world. Whether tweaking feed additives for animals or sorting out old environmental samples, one tiny mistake with formula or molar mass can ripple through an entire project. We saw contaminated fields, often the product of outdated chemical applications, where every piece of data needed double-checking. Getting the formula right meant saving time, money, and the environment. Bad math can lead to incomplete reactions, wasted materials, or even dangerous byproducts.
How This Information Fuels Solutions
Agriculture, toxicology labs, and chemical manufacturing all lean on rigorous, transparent record-keeping. The structure of 2-nitrobenzenearsonic acid provides the kind of data scientists depend on to build safer options, rework formulas, or develop alternatives where arsenic makes trouble. On paper, these numbers seem dry, but in practice, they inform everything from safety sheets to policies.
Clear labeling and accurate reference to the molecular formula, alongside precise molar mass, keep everyone on the same page—from factory floor to regulatory agencies. Years in quality control taught us that one digit off means rerunning batches or double-checking stocks. These details protect workers, ensure environmental compliance, and support the long trail of scientific research.
Paths Toward Better Practices
Going forward, industries and regulatory bodies need to promote open data and solid training for anyone handling complex compounds. Manufacturers should stress-cross-reference molecular information with batch records and demand periodic refresher training, so errors don’t snowball. Encouraging routine audits and real-talk with lab personnel can spot patterns before they lead to mistakes.
Innovation also helps. Digital tools, integrated with updated chemical databases, minimize the risk of mixing up similar-sounding formulas or masses. Sometimes, just automating calculations with up-to-date software keeps teams on track, especially during rushed production schedules or regulatory reviews. These steps don’t just stop errors—they push operations to a higher standard and make the science more reliable for every end user.
What are the storage conditions required for 2-Nitrobenzenearsonic Acid?
2-Nitrobenzenearsonic Acid doesn’t get much attention outside chemistry circles, but its storage makes a real difference both for safety and reliability in a lab. Many overlook the specifics, but small lapses can bring lingering headaches—from fire risks to inaccurate test results.
Understanding the Chemical
One look at the structure, and it’s clear this compound combines a potent arsonic acid group with a nitro ring. That recipe creates instability, particularly under certain conditions. People often assume shelf chemicals don’t warrant much thought, until a problem hits. Over time, even slight moisture or a rise in temperature can bring not just clumping but also decomposition. I’ve seen glass vials become fused shut from humidity exposure, turning a simple retrieval into a small crisis.
Climate Control Isn’t Optional
Room temperature doesn’t mean the same thing in every lab. In one of my earlier jobs, a few stubborn radiators kept the so-called ‘room temp zone’ at over 28°C some afternoons. A major chemical supplier, Sigma-Aldrich, warns that 2-Nitrobenzenearsonic Acid should always be stored cool—think below 25°C—and dry, well away from sunlight. That’s not micromanagement, that’s hazard prevention. Even a brief stint on a warm shelf can cause slow degradation, leading to unreliable assay results or worse, unwanted chemical reactions. If climate control feels out of reach, at least keep chemicals in insulated cabinets or near temperature monitors. Adding a cheap thermometer inside storage spaces made all the difference in labs I’ve managed.
The Trouble with Moisture
Hygroscopic isn’t just a fancy word—it means a chemical grabs onto moisture from thin air. 2-Nitrobenzenearsonic Acid absorbs water, and that opens the door to clumping, hydrolysis, and even unpredictable reactions when it’s used next. One careless lid left ajar in an ordinary storeroom is all it takes. Silica gel packs never go out of style for a reason. Tuck them into bottles, checkpoint them quarterly, and replace as soon as their colors shift. Even then, always reseal containers tightly between uses. Each ounce of caution saves hours lost during formula troubleshooting later.
Keep Incompatibles Apart
Some chemicals quietly sit together with no fuss. Others, like this one, don’t play nice near oxidizers, bases, or organic materials. I learned that lesson after a minor spill led to unexpected fumes from a shelf stacked too densely. Separate storage—clear labels, distinct shelving zones, and a quick reference chart on the door—curbs the chance of accidental cross-reaction. Local fire codes usually list this as a requirement, not a suggestion, for arsonic and nitro compounds. Even without a regulatory hammer, think ahead: a few minutes now prevents the sort of incident that ruins reputations.
Solutions that Last
Safe storage starts with training. Every new tech or student on my teams walked through chemical handling before so much as opening a bottle. Simple checklists posted near cabinets reinforce the message. Rotate stock, record every opening, and don’t skimp on expiry date checks. It pays to set reminders in digital calendars for regular audits. Large facilities, invest in temperature-warning alarms for critical storage rooms. Small spaces, batch chemicals by volatility and risk level to contain potential fallout. Good records mean fewer surprises and safer colleagues.
Every step here draws not just from textbooks, but scars and lessons learned in a working lab. One overlooked day or cracked seal may seem harmless, but for 2-Nitrobenzenearsonic Acid, that’s inviting preventable trouble. A little awareness and simple routines buy peace of mind and keep science moving forward, even on the busiest afternoons.
Is 2-Nitrobenzenearsonic Acid hazardous or toxic?
Why 2-Nitrobenzenearsonic Acid Attracts Concern
Anyone who’s spent a stretch of time learning about chemicals used in agriculture or industry watches for names like 2-nitrobenzenearsonic acid. This compound sometimes pops up in veterinary medicine, often in livestock feed to boost growth or stave off disease. The name itself signals a red flag—arsenic right there in the molecule. The association with arsenic means close attention is warranted, as many arsenic compounds built a history of toxicity that’s hard to ignore.
Hazards and Human Health Risks
Decades of scientific research tie arsenic exposure to a variety of human health problems. Inorganic arsenic can cause skin damage and create cancer risk. Higher exposure, even over short stretches, brings a risk of vomiting, abdominal pain, and even death. Organic forms such as those found in certain feed additives like 2-nitrobenzenearsonic acid have sometimes been called “less toxic.” That phrase can feel dangerously reassuring—yet evidence suggests these substances, under certain conditions, break down into more dangerous, inorganic forms. Once inside the body, arsenic doesn’t just disappear. It can accumulate in tissues, travel up the food chain, and show up in what people eat or drink.
Environmental Impact
Arsenical compounds introduced into animal feed don’t just affect the animals. Much of the arsenic gets excreted, and manure spreads widely in agriculture. Spreading manure from animals given 2-nitrobenzenearsonic acid means arsenic enters the soil, and potentially nearby water sources. Arsenic is persistent. Anyone who’s paid attention to groundwater contamination news knows how hard it is to remove once it gets into the system. Crops grown on contaminated soil pull up arsenic and transfer it through the food supply, setting off a cycle of exposure that doesn’t stop at the farm’s edge.
Community Experience and Regulation
People living near large-scale farms often carry the highest risk, with increased chances for their drinking water to contain arsenic. An Environmental Protection Agency (EPA) investigation found elevated arsenic levels in the vicinity of intensive poultry farms in multiple states. Concerns reached a point where regulatory agencies started to phase out or restrict certain arsenic-based feed additives. The U.S. Food and Drug Administration banned several such compounds, citing the health risk for both humans and animals, as well as the threat to water safety.
Looking Toward Better Practices
Industry can’t ignore public health in pursuit of profit. Transparent labeling and full disclosure help communities understand what chemicals make it into animal feed. Farms switching toward safer growth promoters and practicing responsible waste management take a big step in protecting both people and ecosystems. Responsible crop rotation and investment in water quality monitoring also matter.
For anyone buying food or making policy, paying attention to what goes into animal feed isn’t just about ethics—it’s also about keeping toxic legacy chemicals out of families’ water and dinner plates. The call for cleaner agriculture grows louder as more people see direct connections between chemical use on farms and wellbeing at home.
What is the purity and available packaging for 2-Nitrobenzenearsonic Acid?
Why Purity Level Matters
Purity isn’t a detail—it's the difference between successful research and wasted effort. In my years dealing with specialty chemicals, I’ve seen labs get tripped up by the smallest impurities, especially where reaction yields and regulatory benchmarks matter. With 2-Nitrobenzenearsonic Acid, most providers stamp each batch with numbers like 98% or 99%. These aren’t just badges: they’re promises for industries that can’t tolerate fuzziness, whether in synthesis or analytics.
Manufacturers achieve these levels with crystallization or high-performance chromatography. Labs need these methods since even tiny contaminants risk cross-reactions, or worse, misleading results. Chimera Research and Sigma-Aldrich, among others, regularly test batches using HPLC or elemental analysis. Certificates of Analysis (COA) back up every drum and bottle. For end users, selecting 2-Nitrobenzenearsonic Acid with a tested purity above 98% means one less variable to worry about.
Packaging Options Serve Real-World Demands
Researchers, process chemists, and plant managers know shipping and storing chemicals comes with more headaches than paperwork. Suppliers offer 2-Nitrobenzenearsonic Acid in sealed glass bottles, HDPE (high-density polyethylene) containers, and sometimes aluminum bags for bulk orders. Small-volume users—those running investigative syntheses or pilot studies—gravitate toward 5-gram, 10-gram, or 25-gram bottles. These sizes stay reasonable on budgets and keep leftover stock from sitting around absorbing moisture from the air (hygroscopicity is no joke).
For larger outfits, especially those scaling up promising drug candidates or setting up toxicological tests, larger packs become essential. You’ll see 100-gram, 500-gram, or even kilogram-scale drums packed for safe transit. Shipping regulations make sure nothing leaks, reacts, or poses extra hazard on route. I remember a colleague dealing with a leaking drum years ago—the paperwork and remediation cost more than the chemical inside. Today’s packaging standards reflect lessons learned the hard way.
Safety, Compliance, and the Traceability Factor
Every bottle and drum must trace back to a batch and COA. Oversight groups—think REACH, OSHA, China’s MEE—don’t cut any slack for companies offering ambiguous lots or vague purity details. Distributors who take shortcuts risk heavy fines, lawsuits, and harm to researchers. My own experience running quality checks taught me: check the COA before you sign the delivery sheet.
Transport remains tightly controlled. Each shipment of 2-Nitrobenzenearsonic Acid travels in packaging tested for pressure, impact, and chemical compatibility. Most reputable producers include tamper-evident seals, tracking barcodes, and hazard pictograms. It’s now rare to open a new bottle and get a surprise.
What Buyers Should Look For
People ordering this compound care about more than just the price tag. Ask for recent batch COAs that list inorganic and organic impurities below 2%. Request material safety data sheets (MSDS) and shipment tracking details—no serious supplier withholds those. If your project runs tight on specs, discuss packaging options with your source before approving purchase orders. That saves awkward surprises in the middle of a sensitive experiment.
In an ideal world, every researcher and industrial tech gets both high-purity 2-Nitrobenzenearsonic Acid and secure, no-hassle packaging. Good suppliers back up those goals with hard data and safety-conscious shipping—not wishful thinking.
| Names | |
| Preferred IUPAC name | 2-nitrobenzenearsonic acid |
| Other names |
Orsanilic acid 2-Nitrophenylarsonic acid ortho-Nitrophenylarsonic acid |
| Pronunciation | /tuː-ˌnaɪ.troʊ.bɛnˈziːn.ɑːrˈsɒn.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 98-88-4 |
| Beilstein Reference | 1368607 |
| ChEBI | CHEBI:34054 |
| ChEMBL | CHEMBL510492 |
| ChemSpider | 72894 |
| DrugBank | DB14005 |
| ECHA InfoCard | ECHA InfoCard: 100.013.798 |
| EC Number | 205-738-4 |
| Gmelin Reference | 127872 |
| KEGG | C18606 |
| MeSH | D017921 |
| PubChem CID | 69836 |
| RTECS number | CY8400000 |
| UNII | 6D8SQE43U9 |
| UN number | UN1660 |
| Properties | |
| Chemical formula | C6H6AsNO5 |
| Molar mass | 247.05 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.83 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -0.86 |
| Vapor pressure | 3.35 x 10^-8 mm Hg (25 °C) |
| Acidity (pKa) | 2.31 |
| Basicity (pKb) | 8.08 |
| Magnetic susceptibility (χ) | -72.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.661 |
| Dipole moment | 6.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -138.1 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1079.3 kJ/mol |
| Pharmacology | |
| ATC code | P53AG06 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P308+P311, P501 |
| NFPA 704 (fire diamond) | 2-2-0-ox |
| Flash point | 317.7 °C |
| Explosive limits | Lower: 0.22%, Upper: 1.8% |
| Lethal dose or concentration | LD50 oral rat 930 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 955 mg/kg |
| NIOSH | SN8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Arsanilic acid p-Aminophenylarsonic acid Roxarsone Nitrobenzene |

