2-Nitroanisole: Insight, Industry, and Impact
Historical Development
The story of 2-Nitroanisole starts over a century back, when synthetic dyes and fine chemicals began shaping entire sectors. In the early 20th century, researchers explored aromatic compounds to drive industrial chemistry forward. Chemists first produced 2-Nitroanisole by nitrating anisole, paving the way for its use in dye manufacture. It sparked curiosity for its nitrated aromatic ring, and as chemical manufacturing ramped up worldwide, this compound earned a seat in dyes, pigments, and beyond. The interest didn’t end with those early days; chemists and engineers kept refining production processes to achieve higher purity and better safety, mirroring parallel advances in the global chemical industry.
Product Overview
2-Nitroanisole stands recognizable by its pale yellow crystalline look and mild aromatic odor. This compound occupies a spot in the toolbox for companies focused on chemical synthesis, intermediates, and research labs. Being a prime building block, it flows into production lines for dyes, pigments, and certain pharmaceuticals. With its aromatic nitro group and methoxy substituent, 2-Nitroanisole bridges a gap between primary organic synthesis and advanced specialty chemicals, often showing up in formulations that need stability or effective electron-withdrawing power. Along its journey, it’s picked up several names, yet one thing stays constant—its value in the heart of process chemistry.
Physical & Chemical Properties
This compound presents as a faintly yellow solid when pure, and it tends to melt somewhere between 32 and 35°C. Its boiling point rises above 240°C, and the substance holds a low vapor pressure at ambient conditions. It’s only sparingly soluble in water, but combines well with many organic solvents like ether, chloroform, and benzene. The nitro group sits at the ortho position on the benzene ring, right next to the methoxy group. This arrangement matters for electronic effects in chemical reactions. Its molecular structure, C7H7NO3, produces a molecular weight of 153.14 g/mol. Stability remains good under normal storage conditions, but it should stay away from strong oxidizers, acids, and open flames.
Technical Specifications & Labeling
Producers and distributors typically offer this compound at purities ranging above 98%, fitting the strict standards that specialty chemicals demand. Batches often display yellow crystalline granules with minimal impurity content. Packaging tends to involve sealed glass or high-density polyethylene containers, complete with clear hazard labeling for flammability and toxicity. Safety data sheets warn of risks such as eye and skin irritation, respiratory sensitization, and environmental impact. Responsible labeling adheres to international chemical management protocols, including GHS and REACH requirements. Diligent traceability along the supply chain stands as a minimum expectation today, and record-keeping ensures compliance, efficiency, and transparency.
Preparation Method
The go-to synthetic route involves nitration of anisole with a mix of concentrated nitric and sulfuric acids. Under controlled temperatures, this direct reaction introduces the nitro group preferentially to the ortho position, courtesy of the electron-donating effects of the methoxy group. Chemists found the balance between temperature, acid concentration, and reaction time to maximize yield and minimize by-products. Newer developments include continuous flow methods and greener nitration strategies, such as solid acid catalysts that reduce waste and improve selectivity. On a larger scale, keeping stringent controls over raw material quality, reaction temperature, and acid recovery helps both profit margins and environmental safety.
Chemical Reactions & Modifications
Both the nitro group and the methoxy group open doors for further chemistry. Reducing the nitro group transforms 2-Nitroanisole into o-anisidine, a valuable precursor in dye production. The methoxy group’s electron-donating quality allows selective aromatic substitutions, crucial in complex molecule synthesis. Halogenation, sulfonation, and further nitration all find a role in derivative manufacture. Chemists still use this platform to synthesize compounds for agrochemicals, pharmaceuticals, and advanced materials. Its reactivity, paired with relative controllability, makes it a mainstay in scale-up studies looking to optimize downstream chemical products.
Synonyms & Product Names
Over time, 2-Nitroanisole gained several labels. Trade catalogs list it as o-Nitroanisole, 2-Methoxynitrobenzene, and ortho-Nitroanisole. Product codes may differ based on supplier or region, but the core compound remains unchanged. In chemical literature, these names refer to the same substance, ensuring that researchers, manufacturers, and procurement professionals can identify it regardless of source or market.
Safety & Operational Standards
Work with 2-Nitroanisole always demands attention to best-practice lab safety. Proper personal protective gear—gloves, goggles, and lab coats—cuts down exposure risk. Local exhaust ventilation or fume hoods keep fumes under control, and emergency showers or eyewash stations anchor responsible lab planning. Spills demand swift cleanup using absorbent materials and safe disposal as hazardous waste. Training remains vital, since untrained staff represent the biggest safety x-factor. Most facilities restrict handling access to trained personnel. Regulatory compliance requires secondary containment, regular inventory checks, and prompt reporting of incidents. Unsafe habits or neglected protocols continue fueling incidents, reinforcing the necessity for both operational and cultural diligence.
Application Area
Most of the world’s 2-Nitroanisole finds a home in the dye and pigment industry, feeding into synthesis of azo dyes that color textiles and printing inks. Some of it finds additional value as a starting point in specialty pharmaceuticals, where downstream transformations help shape advanced intermediates or molecular building blocks. Environmental fate studies use 2-Nitroanisole as a model compound to track nitroaromatic breakdown. In analytical chemistry, it sometimes serves as a standard for chromatography techniques, and research labs keep it in stock for a wide range of organic reactions and educational demonstrations. Global demand usually follows trends in dye, pigment, and specialty chemical production.
Research & Development
Recent years have brought renewed attention to cleaner manufacturing and improved product stewardship for aromatic nitro compounds like 2-Nitroanisole. Chemists now research solvent-free or low-waste nitration, improved catalyst systems, and advanced recovery of process by-products. Analytical chemists continue refining detection methods—using high-performance liquid chromatography and mass spectrometry—to track trace impurities or breakdown products, both in manufacturing and in the environment. Research in toxicology and risk assessment guides safer workplace standards and consumer protections, connecting upstream innovation with real-world impact. Thanks to collaborations across industry and academia, safer, greener, and more efficient routes keep appearing on the horizon.
Toxicity Research
2-Nitroanisole draws consistent attention for potential human and environmental risks. Animal studies point to toxic effects from both acute and chronic exposure, with risks concentrating around liver and renal function following high-dose or long-term contact. The compound’s nitroaromatic nature gives reason for caution—some nitro compounds have links to carcinogenic or mutagenic effects, though no evidence puts 2-Nitroanisole in the highest hazard categories. Guidelines push for limiting workplace exposure, tightly controlling waste discharges, and monitoring contaminated sites. Environmental studies show it can resist quick biodegradation, so responsible disposal and remediation planning help limit its footprint. Ongoing studies process hazard data for regulatory agencies, so both local and international buyers can set safe handling parameters for workers and communities.
Future Prospects
The horizon for 2-Nitroanisole keeps shifting with technology and sustainability demands. Cleaner synthesis methods, exposure reduction, and smarter risk management shape the market for raw materials across the dye and specialty chemical sectors. Digital monitoring tools help keep track of both inventories and emissions, while automation and process improvements cut down on waste and accidents. Alternative, more benign chemistries for dyes and advanced materials spark new competition, but 2-Nitroanisole’s reactivity and utility keep it relevant for now. The push for non-toxic, biodegradable dyes could influence future demand and regulatory oversight. Developers, environmentalists, and production engineers share the same goal: new materials and technologies that don’t sacrifice safety or environmental health. Each improvement builds on decades of experience and research, keeping this compound at the center of industrial progress—responsibly managed and carefully monitored at every step.
What is 2-Nitroanisole used for?
What's Behind the Name?
Every once in a while, a chemical comes up in conversation that sounds pretty niche. 2-Nitroanisole doesn’t exactly roll off the tongue, but it helps shape a few things we run into more often than we think. This compound belongs to the nitroanisole family, marked by a nitro group hooked to a methoxybenzene ring. It isn’t just another obscure item on a lab shelf. Production workers, environmental scientists, and regulators all deal with its presence and impacts.
How Industry Puts It to Work
The story of 2-Nitroanisole revolves mostly around the dye and pigment industry. Factories use it to manufacture dyes that tint everything from synthetic fibers to plastics. Without certain intermediates like this one, those vivid colors that fill store shelves just wouldn’t pop the same way. In my own experience hanging around textile plants, the shelves always seem packed with these sorts of chemicals. Production lines lean on this molecule to streamline syntheses, making whole batches faster and sometimes with less waste.
There’s a knock-on effect. Chemical companies also integrate 2-Nitroanisole into the production of other compounds. It acts as a starting block for making antioxidants, which help to maintain the quality and longevity of everyday products, like lubricants and some coatings. You turn up the volume on chemical flexibility with building blocks like this — a chemist can branch off in several directions from one starting point.
Is It Safe Around People?
Most folks never realize when they cross paths with anything related to 2-Nitroanisole, but its presence matters for worker safety and community health. The International Agency for Research on Cancer (IARC) classifies it as possibly carcinogenic. Studies involving lab animals found it could spur tumor development in certain tissues. Now, that doesn’t mean just walking by a textile plant puts someone at risk, but factory safety officers and environmental engineers keep tight controls on how it’s stored, transported, and disposed of.
Workplace exposure limits help set guardrails. Protective gear, ventilated workspaces, closed production systems — these are all proven ways to keep chemical exposure down. Regulators encourage companies to track air quality and leaks, keeping the risks firmly in check.
Environmental Impact Takes the Spotlight
Every chemical has to go somewhere when a factory’s done with it. Some nitro compounds hang around in the environment for a while, resisting breakdown by sunlight or microbes. That persistence means local authorities and watchdog groups test water and soil near major users regularly. If 2-Nitroanisole escapes into rivers or fields, it doesn’t just vanish, so responsible operators invest in proper waste treatment and containment.
I’ve seen some pilot programs add advanced filtration and track waste streams with sensors. It’s not just about following rules — many in the industry realize that cleaner operations protect both their bottom line and local communities. Cleaner water, better air, fewer liabilities.
Better Practices, Safer Outcomes
While the chemistry gets technical quickly, companies can adopt safer substitutes for many applications. Green chemistry initiatives encourage researchers to find dyes and antioxidants that keep risk low without giving up quality. Engineers continue to build smarter factories that use less, waste less, and protect more. A focus on safety, innovation, and transparency helps chemicals like 2-Nitroanisole stay in line with modern expectations.
People may never see or handle 2-Nitroanisole directly, but the choices industry makes with it echo beyond the lab bench. Active monitoring, honest labeling, and good waste management stand out as vital. Through dialogue between companies, health experts, and regulators, safe use can keep pace with industry’s needs and the planet’s limits.
Is 2-Nitroanisole toxic or hazardous?
Getting to Know 2-Nitroanisole
2-Nitroanisole shows up as a pale-yellow liquid with a mild smell, and you won’t find it in everyday household products. Factories use it for making dyes, especially for coloring in textiles, inks, and sometimes pharmaceuticals. It’s a compound built by combining nitro and methoxy groups onto a benzene ring, which looks harmless to a casual observer. But its chemical structure makes it less than friendly to humans or the environment.
Health Risks People Should Recognize
Personal experience working in industrial labs reminds me that even chemicals with neutral appearances often carry hidden risks. Scientific studies tie 2-nitroanisole to cell-level damage. The National Toxicology Program lists it as “reasonably anticipated to be a human carcinogen,” based on animal tests where rats and mice developed cancers after exposure. Breathing in 2-nitroanisole dust or vapor in work settings can irritate skin and eyes. If swallowed or absorbed through the skin, it can stress out the liver and kidneys. Many factory workers use it unknowingly since its yellow tint and weak odor don’t stand out like a warning sign.
The Environmental Protection Agency (EPA) notes that nothing good comes from lax handling of this substance. The Occupational Safety and Health Administration (OSHA) has not set a strict exposure limit, though Europe and several Asian countries put tighter controls in place after several workplace incidents.
Environmental Impact
Factories that handle 2-nitroanisole run the risk of spills and releases. The compound breaks down slowly. Unlike some chemicals that quickly degrade, 2-nitroanisole can stick around in soil and seep into groundwater. Wildlife exposed to contaminated water may face hormone disruption or organ damage, much like people do. Pollution from dye industries doesn’t just fade; particles leach for months or even years, becoming a source of chronic harm for nearby communities.
Worker Safety and Prevention Measures
Weekly jobsite safety meetings sometimes brushed over lesser-known chemicals like 2-nitroanisole, but real-world experience shows this is a mistake. Workers need gloves and chemical splash goggles even for short tasks. Good air flow and reliable exhaust fans matter more than fancy signage when avoiding chemical vapors. Sites using 2-nitroanisole should invest in tight storage containers and spill response kits. Emergency showers and eyewash stations earn their keep in dye factories. Basic workplace education makes a difference: people deserve to know that inhaling fumes or skin contact isn’t just uncomfortable — it increases cancer risk with each exposure.
Reducing Hazards and Protecting Communities
Switching to safer dye precursors should be a priority for manufacturers. Many labs now use greener alternatives that break down quickly and don’t harm living cells the way 2-nitroanisole does. Ongoing health screenings can catch signs of trouble early for workers in at-risk industries. Routine ground and water monitoring keeps toxins from slipping into food supplies or public water. Governments can push for clearer labeling and transparency from companies importing or using hazardous dyes.
Factories serve a purpose, but people’s health and clean water matter more. It takes effort to swap old chemicals for safer ones, but community voices and worker experience have driven change before, and they can again.
What is the chemical structure of 2-Nitroanisole?
The Shape of 2-Nitroanisole
2-Nitroanisole stands out as a simple yet significant molecule in organic chemistry. It consists of a benzene ring, a methoxy group (-OCH3), and a nitro group (-NO2). The methoxy and nitro groups attach themselves to the benzene ring, specifically at the 1 and 2 positions. Chemists refer to this arrangement as the “ortho” position. The structure especially matters for anyone working with aromatic compounds or involved in synthesizing dyes, pharmaceuticals, or pesticides.
Why Structure Matters in Life and the Lab
At first glance, it’s easy to think molecules like this remain tucked away in the world of textbooks or tucked in chemical storerooms. Realistically, their structure impacts much more. A small shift in position, even a single group on a benzene ring, can shape how a substance behaves. In the case of 2-nitroanisole, the relative positioning of the methoxy and nitro groups changes its reactivity and its health risks compared to its other isomers. The way I’ve seen colleagues worry about handling it says a lot.
As someone who spent years around chemistry labs, that ortho nitro group always raises eyebrows. You start to watch out for things like how easy it is to nitrate something and whether you’ll accidentally end up with the ortho over the para or meta forms. These small details affect everything — downstream synthesis, toxicity, and how to dispose of it after use.
Putting Safety and Science First
Facts share a less friendly side of this molecule. 2-Nitroanisole has a chemical formula of C7H7NO3. While it looks innocent on paper, the presence of the nitro group flags it as something to watch for health and environmental impact. Several studies show repeated exposure can damage DNA in living cells. The U.S. National Toxicology Program classifies it as “reasonably anticipated to be a human carcinogen.” I still recall a professor warning the class: “Treat every nitroaromatic as hazardous until proven otherwise.” That wisdom rings true, not just for students, but also for anyone handling this compound in industry.
What Happens in Industry?
Industrial producers rely on 2-nitroanisole mostly for making dyes. Large-scale production means there’s a bigger risk for spills or downstream waste finding its way into streams or soils. Wastewater can carry residues that don’t break down easily in the environment. There’s still a long way to go in ensuring factories put protective measures first. Closed systems, regular monitoring, and investment in better catalytic processes cut down on waste and worker exposure. It takes company leadership willing to invest in safety, rather than just following the rules on paper.
Solutions Are Within Reach
Every lab, every factory, and every worker plays a part in lessening risk. I once saw a team overhaul its waste disposal process, not because of a mandate, but because someone cared enough to look closer. Doubling down on training, using technologies that trap nitroaromatics before they escape into water or air, and adopting green chemistry principles can move the needle forward. Simple chemical structures like 2-nitroanisole remind us that every detail—where a group sits, how it reacts, whether we take shortcuts—ends up mattering in the big picture.
How should 2-Nitroanisole be stored and handled?
Working with 2-Nitroanisole: More Than a Simple Label
2-Nitroanisole has its place in dyes, organic synthesis, and research labs, but its risks get overlooked. I’ve seen far too many bottles pushed into the back of cabinets or left on open benches because handling guidelines sounded straightforward. A look at accident reports makes the dangers clear: lapses in storage practices sit behind every spill, fire, or poisoning. It only takes one moment of oversight.
Storage: Out of Sight, Out of Mind Causes Trouble
A compound like 2-Nitroanisole brings a host of chemical dangers. Flammable, potentially toxic, and sensitive to heat and air, it does not belong in an ordinary cabinet or next to snacks in the breakroom fridge. I remember walking into a small lab some years back and spotting a faded bottle of 2-Nitroanisole parked next to acetone and peroxides. I flagged it, but the team there didn't recognize the hazard of vapor buildup and how easily a little static electricity can trigger a fire. Safety means thinking ahead: use a well-ventilated, dedicated flammable storage cabinet. Keep it away from strong acids, oxidizers, and anything combustible—metal shelves or acid lockers won’t cut it.
Logbooks help track just how much enters and leaves storage. Most workplace incidents start with someone forgetting what sits in back or what was partially used months ago. Chemical hygiene programs that include monthly checks and a real culture of speaking out about storage issues cut these problems way down.
Handling Means Respecting What You Work With
Open a bottle of 2-Nitroanisole, and even if the odor seems faint, microdroplets can drift off and linger. Direct skin contact or inhalation brings headaches and nausea; chronic exposure runs even deeper risks, with organ and blood system damage possible. Gloves and goggles serve as first-line barriers, but good practice asks for more: wear a long-sleeve lab coat, use a certified fume hood, and make sure gloves actually hold up against organics.
Some labs toss caution aside after collecting a few safe years, swapping full PPE for thin nitrile gloves. I once watched a seasoned chemist wash a spill off bare hands under a tap—she shrugged it off, but six months later, a routine checkup caught blood chemistry changes linked to chronic absorption. Rushing through cleanups tempts fate. Every spill needs an absorbent material approved for organics, not just a towel. Sealed waste containers end up in hazardous chemical streams, not the general trash, and regular reminders about correct disposal keep teamwork strong.
Training sessions grow stale when they only show slides. I’ve seen real progress when everyone practices spill drills and glove changes. Think muscle memory, not just knowledge.
Managing Risks Takes Persistent Effort
Regulatory bodies like OSHA spell out clear minimums, but real safety goes beyond rules. Peer reminders and walk-throughs, not just warning posters, make people pause before popping open a bottle. Labs keep records handy, update safety data sheets, and avoid storing even a single extra gram more than necessary. Old chemicals and debris add up, and periodic inventory control not only reduces clutter but also keeps everyone aware of what's on the shelf.
The bottom line: mishandling 2-Nitroanisole isn’t only about breaking rules. It shortens careers and sometimes lives. Every person who works in a lab can tighten up storage, improve personal habits, and ask better questions. A strong safety culture relies on vigilance, not shortcuts.
What are the safety precautions when working with 2-Nitroanisole?
Why 2-Nitroanisole Deserves Respect in the Lab
2-Nitroanisole isn’t the kind of chemical you just splash around. It’s used for specialty dyes and some research, but its health hazards land it high on the list of substances to handle with care. I’ve found a clean lab and strong habits go further than any warning label. Breathing in its vapors or letting the liquid touch your skin can end poorly—some folks have reported everything from allergic reactions to symptoms tied to its link with cancer in animal studies. NIOSH and OSHA both flag it as a substance that can’t be treated casually.
Personal Protection Counts for More than Looks
Before anything else, grab the right gear. I always reach for a lab coat, gloves made from nitrile, and goggles that fit. 2-Nitroanisole soaks through latex and vinyl a little too easily, so nitrile or neoprene offers better defense. When you know the chemical might splash, face shields aren’t overkill. I’ve seen people skimp on this and regret it later—skin exposure is no joke with organics like this.
No Fumes Left to Chance
Ventilation matters more than folks realize. I never work with 2-Nitroanisole outside a fume hood. It doesn’t take much to send vapors rising, and once you smell it, you’re already being exposed. If there’s no fume hood, it’s time to rethink whether the work can go ahead safely. Some rely on bench-top fans, but that just spreads the vapors around. Good air flow pulls fumes away, keeps breathing space safer, and cuts risks for everyone in the room.
Cleanliness Isn’t Just About Tidiness
Spilling 2-Nitroanisole—or leaving drops around on a bench—always creates extra work and tension. I make it a point to have a spill kit ready: absorbent pads, neutralizer, and bags tough enough to hold toxic waste. It’s easy to think a paper towel can do the trick, but that just moves danger from one place to another. Once, after seeing someone try that, I watched a custodian get exposed during trash pickup—a situation nobody wants to repeat. Always label every container with clear, big letters, and double-check seals before leaving the lab for the day.
Waste Doesn’t Belong Down the Drain
Chemicals like this have no business heading into sewage or regular garbage. I collect every drop used or spilled, pour it into marked waste containers, and deliver it to hazardous waste disposal with a strict log. If left in ordinary trash, its toxic effects can spread far beyond the lab—risking water supplies and, yes, wildlife and people in a wider area. I learned early on that local regulations dictate how and when to hand over such waste, but the golden rule stands: treat every bit as hazardous from start to end.
Looking Out for the People in the Room
I never work alone with 2-Nitroanisole. There’s peace of mind in knowing someone nearby can call for help or grab an eye wash station in an emergency. Building a culture of safety—regular training, honest talk about close calls, and quick action on spills—keeps everyone sharper. Where I’ve seen best practices, labs run smoother, accidents drop, and new folks pick up good habits fast.
Knowledge Goes Further Than Labels
Digging into the chemical’s safety data sheet (SDS) and keeping it handy lets me work with confidence. The facts are right there, from fire risks—since 2-Nitroanisole burns hot and fuels strong flames—to proven first aid steps. Experience tells me reading isn’t enough; practicing those steps preps everyone to react if something ever goes wrong. Storing the chemical away from oxidizers and heat turns a risky job into manageable business.
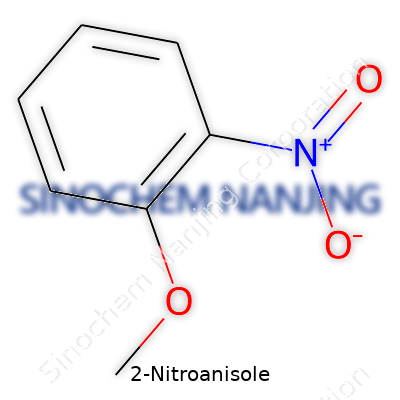
| Names | |
| Preferred IUPAC name | 1-Methoxy-2-nitrobenzene |
| Other names |
o-Nitroanisole 2-Methoxynitrobenzene Ortho-Nitroanisole |
| Pronunciation | /tuːˌnaɪ.trəʊˈæn.ɪ.soʊl/ |
| Identifiers | |
| CAS Number | 91-23-6 |
| Beilstein Reference | 1362960 |
| ChEBI | CHEBI:34582 |
| ChEMBL | CHEMBL32114 |
| ChemSpider | 211063 |
| DrugBank | DB08311 |
| ECHA InfoCard | 03b090e9-d358-43c7-833c-022f3afd4fc8 |
| EC Number | 4.1.1.33 |
| Gmelin Reference | 8640 |
| KEGG | C01795 |
| MeSH | D009602 |
| PubChem CID | 7418 |
| RTECS number | RN3675000 |
| UNII | 8A2J3KZ1K7 |
| UN number | 1669 |
| Properties | |
| Chemical formula | C7H7NO3 |
| Molar mass | 153.15 g/mol |
| Appearance | Pale yellow liquid |
| Odor | aromatic |
| Density | 1.206 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.028 mmHg (25°C) |
| Acidity (pKa) | 15.3 |
| Basicity (pKb) | 9.85 |
| Magnetic susceptibility (χ) | -70.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.573 |
| Viscosity | 2.021 mPa·s (25 °C) |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 68.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3474 kJ/mol |
| Hazards | |
| Main hazards | Suspected human carcinogen, toxic if inhaled, swallowed, or in contact with skin, causes skin and eye irritation, harmful to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H351, H335 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 Health:2 Flammability:3 Instability:0 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 670°F (354°C) |
| Explosive limits | Explosive limits: 1.3–8.5% |
| Lethal dose or concentration | LD50 (oral, rat): 670 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 760 mg/kg |
| NIOSH | SN45500 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Nitroanisole: "No OSHA PEL established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
Anisole Nitrobenzene 2-Nitrophenol 2-Nitrotoluene 4-Nitroanisole |

