2-Nitro-4-Toluidine: An In-Depth Look
Historical Development
Chemistry as a field keeps opening new doors, and looking through its history shows how certain compounds left a big mark. 2-Nitro-4-toluidine has quite a backstory, tracing roots to late 19th-century organic synthesis. Back then, dyestuff manufacturers searched for better colorfast dyes. Chemists found that tweaking aromatic amines gave vibrant hues, leading to compounds like 2-Nitro-4-toluidine. Synthetic dyes based on nitroaromatics like this one flooded industries, allowing mass-market textiles, inks, and pigments. Over time, production methods toughened up, focus shifted onto both yield and worker safety—long before today's regulations were in play. My own time studying aromatic nitro compounds in the lab introduced me to the mix of creativity and caution needed in technical chemistry, especially after seeing legacy procedures that risked both people and product because the science hadn’t quite caught up with safer ways of doing things.
Product Overview
Sitting alongside scores of nitroaniline derivatives, 2-Nitro-4-toluidine stands out for a unique mix of stability and reactivity. The toluene ring, spiced up with a methyl group, creates a gentle but persistent aroma, a reminder of its aromatic ancestry. Pale yellow to orange crystalline granules signal high purity. Companies value this compound not just for color, but for how predictably it fits into further reaction schemes. As a solid, it handles easily under lab conditions—no puff of dust, no overwhelming scent wafting through. In my experience, that's a relief compared to some volatile or sticky intermediates you might meet in organic synthesis.
Physical & Chemical Properties
The melting point of 2-Nitro-4-toluidine comes in between 70 and 74 degrees Celsius, perfect for controlled recrystallization. The compound dissolves well in organic solvents like ethanol and ether, but water doesn’t do the trick. That difference means the compound stays put in water-based processes, letting chemists isolate it without much trouble. The nitro and amine groups, lodged on opposite ends of the ring, make room for all kinds of targeted reactions—reduction, diazotization, acetylation, and more. Its density and molecular weight let formulators predict reaction yields with a reasonable margin. Lab techs respect the deep yellow color as an early sign of purity and the absence of decomposition. If you’ve ever handled impure aromatic amines, you know the tarry mess that can gum up glassware—2-Nitro-4-toluidine, handled right, dodges that headache.
Technical Specifications & Labeling
Products come sealed in drums or stout plastic bags, protected against moisture and sunlight—the usual enemies. Labels read out the chemical’s name, formula C7H8N2O2, batch number, date of manufacture, expiration if stability data suggests it. Regulations in places like REACH and the US EPA lay out hazard warnings, signal words, precautionary statements, and GHS pictograms. Accurate labeling spares buyers and staff confusion, tracking each shipment from plant to end-user. During my internship in a chemical plant, watching the diligence surrounding cargo tags helped me appreciate how details at the loading dock keep people and processes safe all the way down the chain.
Preparation Method
Synthesis usually starts with p-toluidine as a key raw material. Treatment with a nitrating mixture—sulfuric and nitric acid at cold temperatures—brings the nitro group precisely to the ortho position. This step demands sound temperature control and careful venting since runaway reactions don’t give much warning. After acid work-up, the crude product falls out, then recrystallization from alcohol or a mixed solvent bath strips out unwanted isomers. The result gives an efficient, scalable preparation route that doesn’t burn through specialty reagents or equipment. Leaning on a method that's both time-tested and robust strikes a balance between throughput and purity.
Chemical Reactions & Modifications
2-Nitro-4-toluidine opens doors to further synthetic work. Chemists reduce the nitro group to an amine, tack on sulfonic acid residues, or set up diazonium salts for coupling reactions. These modifications fuel color chemistry, agrochemical intermediates, and specialty materials. In practical terms, the reactivity of the nitro and amine groups makes the compound a “two-lane highway,” with each end offering a chance at a new product. Research groups around the world leverage these features to bridge gaps in medicinal chemistry or material science. Years working alongside synthetic chemists showed me how a well-placed methyl group or shifted nitro moiety, like those found here, can mean the difference between a dead end and a breakthrough.
Synonyms & Product Names
Suppliers catalog this compound under several trade names: 2-Nitro-p-toluidine, 4-methyl-2-nitroaniline, or sometimes simply as NT intermediate. Customers sometimes ask for “para-toluidine nitro derivative,” relying on the methyl placement to separate it from similar items. Naming consistency isn’t just about convenience; it prevents costly mistakes down the production line. If you’ve ever read a safety sheet, scratched your head at ambiguous nouns, and double-checked a sample because of it, you appreciate the role of good, precise labeling in high-stakes manufacturing.
Safety & Operational Standards
Workplace safety rules focus hard on 2-Nitro-4-toluidine. Contract labs and plants store it separately from strong acids and oxidizers, in cool, dry rooms. Workers use gloves, goggles, and sometimes respirators if handling powders for extended periods. Spills mean instant cleanup with absorbent material, then careful disposal according to national hazardous waste rules. European and U.S. regulations call for monitoring of airborne particles, training for first aid related to aromatic amine exposure, and strict incident reporting. Direct skin or inhalation exposure risks allergic reactions or more severe toxicity, so carefully written rules—and common sense—matter. Industry partners audit facilities to spot gaps, driving continuous improvement. From my own time in safety training, one thing stands out: people trust processes when they see hazards taken seriously.
Application Area
Out in the field, 2-Nitro-4-toluidine hides inside dyes and pigments that color our fabrics, plastics, and occasionally paper goods. Textile factories lean on its stability and reactivity to create reds, oranges, and specialty shades that resist fading even after long use. Beyond colorants, modified forms serve as intermediates to build ingredients for crop protection and specialty medications. In my lab days, residues from these syntheses showed up everywhere—testifying to both the ubiquity and resilience of these small but mighty molecules. Not just a background player, this compound shapes whole supply chains connecting manufacturers and end-users in a tight loop.
Research & Development
Innovation never stands still. Research teams keep digging up new uses for aromatic nitro compounds, including 2-Nitro-4-toluidine. Efforts target more selective syntheses, improved environmental friendliness, and better worker protection. Analytical chemists refine ways to detect trace impurities that could foul up downstream products, while materials science groups study new derivatives as sensors or responsive polymers. Collaboration between academic chemists and industry players pushes the envelope—remaking an “old” compound for modern needs. Having worked in research myself, I saw firsthand how a question posed by a twenty-year veteran could steer an entire project onto new ground, especially when the stakes included efficiency or ecological impact.
Toxicity Research
Toxicity studies for nitroaromatic compounds go back many decades. Direct exposure at even moderate levels risks skin sensitization, respiratory irritation, methemoglobinemia, and, for some related amines, possible carcinogenic effects. Animal studies with 2-Nitro-4-toluidine show concern for chronic exposure rather than acute incidents. Regulatory agencies flag it for careful review, keeping daily exposure levels very low in industrial and consumer settings. Labs use blood and urine tests to check for occupational exposure, offering an early warning system that can prevent long-term harm. Drawing on incidents I’ve seen, an educated workforce—prompted to use personal protective equipment, wash promptly, and respect chemical limits—dodges most risks effectively.
Future Prospects
Looking ahead, change rolls in on several fronts. Greener chemistry principles push the field to replace harsh reagents and minimize runoff, both to stay legal and to attract eco-conscious buyers. Companies invest in closed-loop manufacturing, energy-saving technologies, and less toxic analogs to keep pace with public expectations. Academic labs, drawn to a familiar molecule with room for improvement, keep testing new synthetic strategies. Market demand for bold colors and high-purity intermediates remains robust, though under the watchful eye of stricter safety standards. Judging by industry movements in the last decade, the compound may never escape close regulatory attention, but with careful stewardship and creative organic chemistry, it looks set to evolve as both a legacy staple and a subject of fresh innovation.
What is 2-Nitro-4-Toluidine used for?
What This Chemical Touches in Everyday Life
Most people rarely think about the small ingredients behind the colorful products they use. 2-Nitro-4-Toluidine belongs to a class of compounds that help bring vibrant colors into the world, especially in the textile and dye industry. Factories use this chemical as an intermediate step when making azo dyes, the types often responsible for bright reds and yellows in clothing, plastics, and inks. Without such chemical building blocks, manufacturers would struggle to bring out rich shades that catch the eye.
The Role in Dye Production
During work visits to textile production plants over the years, I watched workers measure out powders like 2-Nitro-4-Toluidine for dye synthesis. Their work transforms base chemicals into dyes that color everyday items, including school uniforms and curtains. Chemists rely on 2-Nitro-4-Toluidine because it reacts well with other components, giving predictable results in dye quality. In one factory, managers mentioned that the right intermediate can improve efficiency during scaling, saving both time and resources.
Beyond Clothing: More Than Meets the Eye
You’ll find traces of this substance in more than just textiles. Some companies use 2-Nitro-4-Toluidine when producing colored inks for commercial printing and certain plastic parts that require colorfast pigments. On the shop floor, engineers have told me how color consistency makes a difference in brand recognition—an example is toys or consumer packaging. Without effective intermediates, those vivid hues can fade quickly or become patchy.
Concerns and Potential Dangers
Working with aromatic amines like 2-Nitro-4-Toluidine can put factory workers at risk. The International Agency for Research on Cancer classifies related substances as possible carcinogens, linking long-term exposure to increased health dangers. During safety training sessions, there’s always a strong focus on protective gear—masks, gloves, and ventilation systems become mandatory, not optional. If facilities ignore these steps, local communities sometimes pay the price in the form of contamination or rising cancer rates, as studies from parts of Asia and textile clusters in Europe have shown.
Environmental Realities
The waste produced from dye manufacturing doesn’t vanish without effort. In several river valleys worldwide, untreated wastewater carrying remnants of 2-Nitro-4-Toluidine and related compounds has stained water and damaged aquatic life. I once interviewed local farmers downstream from a dye plant in northern India—many blamed declining fish catches and odd health symptoms on what they called “factory water.” This problem rarely fixes itself. Companies and governments need strict monitoring and investment in water treatment to avoid turning streams into chemical dumping grounds.
Building a Better Path Forward
Stronger regulation and clear labeling help keep industries honest. Some consumer-facing brands now ask for chemical safety reports from their suppliers, trying to filter out substances linked to health or environmental harm. On the scientific side, teams keep researching ways to switch from hazardous intermediates to greener alternatives. Supporting those efforts, both as an informed worker and a consumer, makes a real difference. Clear standards, regular checks, and collective diligence create safer workspaces and healthier communities. Each link in the chain takes responsibility for the chemicals that color our lives.
What are the safety precautions when handling 2-Nitro-4-Toluidine?
What’s the Deal with 2-Nitro-4-Toluidine?
2-Nitro-4-toluidine looks like a yellowish powder, useful for dye and pigment production, but it hides some serious risks behind its practical uses. The experience of working around chemicals like this one teaches fast: every shortcut invites trouble. Its vapors and dust have no mercy—getting careless means putting yourself and coworkers in danger.
Don’t Skimp on Personal Protective Equipment (PPE)
Many shops try to pinch pennies on gear, but that decision often leads to regret. Nitrile gloves and a lab coat form the frontline for skin. Add splash goggles and a sturdy face shield since this stuff burns eyes fast. Respirators keep your lungs guarded because even a small whiff can mean headache, nausea, or worse. Good gear costs less than a hospital bill or lifelong discomfort from chemical exposure.
Work in Well-Ventilated Spaces
Stuffy rooms make chemical risks worse. An open window can help, but real safety comes from ventilated fume hoods. I’ve watched folks try to wing it only to get headaches from lingering fumes. A fume hood draws vapors away and gives peace of mind. Skipping this step means gambling with your health.
Label and Store with Caution
Mixing up containers has ended labs and ruined careers. Clear, bold labels in waterproof ink matter more than slick appearances. Keep 2-nitro-4-toluidine sealed tight in dedicated chemical cabinets, far from food, water, and materials that react easily, such as strong acids and oxidizers. Lock cabinets at the end of the shift—not as a formality, but because someone always forgets or gets too curious.
Master Your Spill Plan
Lab veterans know that spills happen, even with good training. If powder spills, scooping gently with a small plastic dustpan works, and using wet towels helps prevent dust from spreading. Never sweep—it just floats dangerous dust around. For liquid messes, keep up with inert absorbents and have a proper disposal drum on hand. Notify your supervisor and record every incident, no matter how minor it seems. Hiding mistakes just keeps risks alive for the next person.
Wash Up Before You Eat—Always
This sounds trivial but deserves repeating: wash your hands before grabbing lunch. Toxins linger on skin and under fingernails, especially after a long session at the workbench. Rubbing your eyes or biting your nails exposes your body in ways gloves never fully prevent. Soap and water after handling chemicals become simple habits that protect families, too, since chemical traces like to hitch rides home.
Companies Need to Prioritize Training
I’ve seen training treated as a checkbox when it should be the foundation. The best labs schedule refreshers every few months, introduce case studies from real accidents, and encourage questions. Practical drills prove more valuable than any PowerPoint. People remember simulations long after they forget technical jargon. Encouraging curiosity leads to a culture where spotting near-misses and reporting them keeps everyone sharp.
Why Precautions Matter
2-Nitro-4-toluidine can inflame skin, cause respiratory issues, and if mismanaged, threaten lives. Nobody wants a visit from regulators after a preventable accident, or worse, having to call an ambulance for a friend. With the right steps, most chemical hazards shrink. Good habits, honest reporting, and real respect for the risks make a safer workplace—nothing too fancy, just common sense backed by experience.
What is the chemical structure and formula of 2-Nitro-4-Toluidine?
What 2-Nitro-4-Toluidine Looks Like Chemically
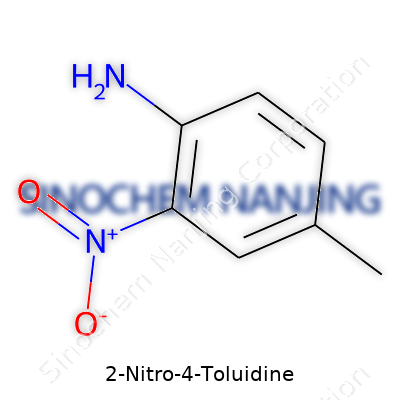
2-Nitro-4-Toluidine’s name reveals a lot about its chemical makeup. Its backbone comes from toluene, which you can think of as a benzene ring with a single methyl group attached. Add to that, at the fourth position on the ring, an amino group clings on, while at the second position, a nitro group takes its spot. This placement changes the way this molecule interacts compared to other isomers.
The structural formula spells this out: begin with a six-carbon benzene ring. Number the carbon where the methyl group (CH3) sits as carbon 1. At carbon 2, attach the nitro group (NO2). At carbon 4, an amino group (NH2) hangs on. All together, the molecular formula becomes C7H8N2O2. That means it has:
- Seven carbon atoms
- Eight hydrogen atoms
- Two nitrogen atoms
- Two oxygen atoms
This formula, despite its simplicity, packs a punch in terms of chemical properties and uses.
Why the Structure Matters
Living through a couple of chemistry lab explosions in college teaches you to appreciate the tiny differences groups like nitro and amino make. A slight shift in position can turn something docile into something reactive. Here, the nitro group at the ortho position (right next door to the methyl group) changes how electrons move across the ring and how the compound interacts with other chemicals.
This matters in industrial applications. Textile dyes and pigments rely on the way molecules like 2-Nitro-4-Toluidine behave when exposed to heat, light, and acids. Its unique structure leads to the specific colors and stability people look for. I’ve seen firsthand how a slight misplacement in the group leads to unwanted hues or poor fastness in the final fabric.
Real-World Concerns: Toxicity and Handling
The presence of a nitro group raises red flags from a safety perspective. Too many years around labs and shop floors, and you see what improper handling can do. Hard data from industrial hygiene studies backs this up—aromatic nitro and amino compounds often carry health risks. 2-Nitro-4-Toluidine is no stranger here; studies have flagged it for possible links to cancer and environmental persistence.
Policy and process changes make a difference. Employers who provide proper fume hoods, personal protective equipment, and closed systems keep workers safer. Regular air monitoring and disposal controls help protect local water tables. Education beats ignorance every time. Early in my career, I saw a peer hospitalized from exposure to aniline derivatives. Workplace training stopped those accidents cold.
Better Chemistry for the Future
As more markets seek greener textiles and less pollution, swapping out risky chemicals earns genuine business rewards. Chemists continue to build new molecules that mimic the best features of aromatic amines without the baggage. Techniques like flow chemistry and catalytic hydrogenation reduce waste. Research keeps pushing the field further, reminding us daily that one atom’s position shapes our world in unexpected ways.
How should 2-Nitro-4-Toluidine be stored?
Why This Chemical Calls for Respect
Chemistry classes only scratch the surface. Reading warning labels on certain chemicals years later pushed me to think differently about storage. 2-Nitro-4-Toluidine shows up in dye and pigment manufacturing, but you might not realize how dangerous a yellowish crystalline powder can turn if handled carelessly. It isn’t just about keeping a container on a back shelf. There are real health risks. Anyone who works with raw aromatic amines has seen burns and breathing trouble. Skin or eye contact causes serious reactions, and inhaling dust leads to headaches and nausea. I once saw a lab tech hospitalized after a splash. So, proper storage has real consequences.
Choosing the Right Place
Not every storage room works. This compound reacts to sunlight and heat, sometimes decomposing to release dangerous fumes. It should never go near radiators, steam pipes, or anywhere summer sun could heat the storage space. The air temperature matters; cool and dry means between 2°C and 8°C. Humidity brings headaches—literally. Moisture can trigger breakdown and unpredictable reactions. Low humidity cuts risk. In practice, a well-ventilated chemical storeroom with reliable climate control offers far fewer surprises. If local weather bounces all over, investing in an insulated cabinet makes sense.
Packing Matters Just as Much
Original, tightly-sealed containers always work best, but never trust a loose lid. Vapors seep out, and those sneaky fumes turn headaches into something worse. Glass or high-grade plastic stands up against corrosion and accidental splashes. Keep containers upright, clearly labeled, and free of crusty buildup. Mixing up labels leads to accidents. I’ve seen busy techs grab a white powder from a faded jar, thinking one aromatic amine was another. That slip could send someone to the ER.
Keeping Incompatibles Apart
Tossing every jar on one shelf invites trouble. Strong acids, oxidizers, and bases don’t belong anywhere near 2-Nitro-4-Toluidine. An accidental spill could make a fire or toxic cloud. Experience says: keep each chemical family on their own shelves. If space is tight, use rated chemical storage bins for physical barriers. Manufacturers and training guides always make the same point: keep the incompatible stuff separate. Real-life mistakes prove them right every time.
Handling Spills and Emergencies
PPE is non-negotiable. Gloves, eye protection, and lab coats stay on, even for quick tasks. Any spill demands prompt clean-up with absorbent material—not rags or paper towels that can smolder. Properly marked waste containers mean nobody drops used towels in an ordinary trash bin, risking exposure outside the lab. Adequate ventilation helps, but a chemical fume hood handles powders best.
Documentation and Education
Records of who uses what, when it was received, and where it is stored, reduce confusion. The most recent Safety Data Sheet should sit close by. Regular staff training prevents forgetfulness. Supervisors who skip safety audits don’t just break rules, they ignore the common sense that keeps teams healthy. From small academic labs to big industrial warehouses, a shared respect for danger pairs with practical storage to keep everyone safe and healthy.
What are the potential health hazards of 2-Nitro-4-Toluidine exposure?
What Makes 2-Nitro-4-Toluidine a Concern?
2-Nitro-4-toluidine brings color to dyes, pigments, and plastics. It pops up in textile factories, toner production lines, and some laboratories. Working with this chemical feels routine to many, but even small spills or careless habits can take a toll on health. Stories from production floors show that folks often underestimate these dangers until symptoms creep into daily life. A few bad episodes—persistent cough, skin rashes, tougher headaches—often push people to dig deeper into what they’re around, and facts about this chemical surface quickly.
Inhalation: Breathing in Trouble
Dust and fumes seem minor at first. You don’t notice trouble from a quick whiff, but workers breathing 2-nitro-4-toluidine dust over days or weeks may end up with sore throats, irritated lungs, or, over longer periods, more severe breathing problems. Occupational health research connects regular inhalation to increased asthma-like symptoms. Longer exposures set off chronic cough. The lungs lay down the first protest against careless handling here, sending a message that should not be ignored.
Skin Contact: More Than an Itch
Splash this substance on your hand and burned, itchy skin tells the story. My own stint in a dye plant taught me that personal protective equipment is not just for show. A sloppy moment led to bright red inflammation along my wrist, persisting for days. Others have reported rashes, cracks, or even sores after direct exposure. Toxicology data back up these experiences, reporting allergic reactions and delayed sensitivity. Over time, repeat contact may prime your immune system to overreact, making even tiny exposures a problem down the line.
Beyond Immediate Effects: Cancer Risk Lurks
Longer-term health matters most. Studies on chemicals similar to 2-nitro-4-toluidine—aromatic amines—raise flags for cancer links. Laboratory animals, after repeated, high doses, develop tumors in organs like the bladder and liver. Agencies like the International Agency for Research on Cancer keep a close eye on these substances because the molecular structure—the nitro and amino groups—look like other chemicals that damage DNA. Industry health records from the mid-20th century reveal that cancer clusters showed up in dye factory workers long before modern regulations joined the scene.
Protection Means Action
Safer handling comes down to informed respect. Gloves, masks, and goggles form the basic armor on the job. Ventilation keeps airborne dust down. In my experience, training new staff by showing photobooks of injuries makes rules hit home faster than lecturing. Regular health checks also turn up problems before they spiral. Companies that enforce clean practices—frequent hand-washing, safe storage, fast cleanup of spills—report fewer incidents.
The Road Forward
Stronger oversight still matters. Switching to safer chemical alternatives, where possible, helps health and cuts down on long-term risks. Workers deserve plain talk about symptoms: odd skin changes or stubborn coughs should trigger a doctor’s visit, not just a shrug. Scientists keep studying these compounds, and industry watchdogs use new data to update rules. Real progress comes from honesty about risks—shared from old hands to newcomers—allowing all of us to keep health in our own two hands every shift.
| Names | |
| Preferred IUPAC name | 4-Methyl-2-nitroaniline |
| Other names |
2-Nitro-4-methylaniline 4-Methyl-2-nitroaniline 2-Nitro-p-toluidine p-Toluidine, 2-nitro- 4-Methyl-o-nitroaniline |
| Pronunciation | /tuː-ˈnaɪtroʊ-fɔːr-təˈluːɪdiːn/ |
| Identifiers | |
| CAS Number | 99-55-8 |
| Beilstein Reference | **1458733** |
| ChEBI | CHEBI:34656 |
| ChEMBL | CHEMBL3331029 |
| ChemSpider | 13253 |
| DrugBank | DB11438 |
| ECHA InfoCard | 100.007.726 |
| EC Number | 612-028-00-4 |
| Gmelin Reference | 72469 |
| KEGG | C14351 |
| MeSH | D014068 |
| PubChem CID | 7415 |
| RTECS number | XZ9625000 |
| UNII | 4BO9Z9D6F7 |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C7H8N2O2 |
| Molar mass | 137.14 g/mol |
| Appearance | Orange to brown crystalline solid |
| Odor | aromatic |
| Density | 1.22 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.85 |
| Vapor pressure | 3.2E-4 mmHg at 25°C |
| Acidity (pKa) | 4.16 |
| Basicity (pKb) | 10.10 |
| Magnetic susceptibility (χ) | -79.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.607 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 159.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -26.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3478 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction, suspected of causing genetic defects, may cause cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H301, H315, H319, H317, H334, H341, H351, H410 |
| Precautionary statements | P260, P264, P270, P273, P280, P301+P312, P302+P352, P304+P340, P308+P313, P311, P312, P321, P330, P332+P313, P337+P313, P362, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-** |
| Flash point | 120°C |
| Autoignition temperature | 540°C |
| Explosive limits | Explosive limits: 2.0–6.0% |
| Lethal dose or concentration | LD₅₀ oral rat 1190 mg/kg |
| LD50 (median dose) | LD50 (median dose): 600 mg/kg (oral, rat) |
| NIOSH | TT0700000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 0.5 mg/m3 |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
4-Nitroaniline 2-Nitroaniline 2,4-Dinitrotoluene 2-Methyl-4-nitrophenol 2-Nitrotoluene |

