2-Nitro-4-Cresol: History, Properties, and Possibilities
Historical Development
Few chemicals have walked such an interesting path through industrial history as 2-Nitro-4-Cresol. During the industrial revolution, aromatic nitro compounds gained attention for their dye-forming capacity and as precursors for pharmaceuticals and agrochemicals. Chemists in Europe began producing 2-nitro derivatives of cresol by the late nineteenth century, guided by an interest in synthetic intermediates. Applications grew from textiles into photography and electronics manufacturing, bolstered by rapid improvements in nitration technology. By the mid-1900s, global demand surged, prompting finer approaches to synthesis and purification. The continued development reflects both industrial needs and the ongoing drive to control environmental and health impacts.
Product Overview
2-Nitro-4-Cresol, sometimes called para-nitro-meta-cresol, forms a pale yellow solid, easily distinguished from its isomers in both appearance and reactivity. Its use as an intermediate extends into fields such as dyes, pigments, pharmaceuticals, and even as a stabilizer in specialty polymers. Suppliers offer grades designed for general industry or high purity applications, and labeling practices now highlight trace impurity content along with packaging and shelf-life details, which proves helpful for handling in both large and small-scale labs.
Physical & Chemical Properties
The compound carries the molecular formula C7H7NO3 and tips the scales just over 153 g/mol. Its melting point typically falls between 93 and 97 Celsius. The substance dissolves sparingly in water, although it finds better solubility in ethanol and other organic solvents. A nitro group in the ortho position and a methyl group meta to the hydroxyl ring location give it distinct chemical personality: it acts as both a weak acid and an electrophile, opening the door to nuanced organic reactivity. The nitro group modifies its color and absorption, making it detectable in spectroscopic analyses common in synthesis labs.
Technical Specifications & Labeling
Producers test each lot for purity, usually reporting levels above 98% for laboratory supply and bulk shipments that serve industry. Trace metals and related aromatic compounds find mention in certificate of analysis documents. Packaging typically uses sealed, inert-atmosphere containers, marked clearly with hazard warnings, batch identifiers, and best-before dates to discourage degradation from moisture or air. Material safety datasheets and REACH compliance paperwork arrive together, reflecting a shift towards transparency prompted by tightening global chemical safety expectations.
Preparation Method
Lab-scale synthesis of 2-Nitro-4-Cresol starts from 4-cresol and relies on direct nitration with mixed acid—usually concentrated nitric with sulfuric acids—under carefully chilled conditions. After allowing completion of the reaction, the crude product requires washing, neutralization, and recrystallization. Manufacturers running commercial operations usually standardize on continuous-flow technology and acid recovery systems, which cut down both waste volumes and risks of runaway reactions. I once worked alongside a process chemist who said the key to safety during nitration isn't just precise temperature control but also the patience to verify purification at each step, as small impurities can magnify risks in downstream production.
Chemical Reactions & Modifications
2-Nitro-4-cresol serves as a flexible platform for synthetic chemists. The nitro group invites reduction to an amine, leading to intermediates for azo dyes or pharmaceutical compounds. The methyl group stands ready for halogenation or oxidation, while the phenolic hydroxyl can support etherification or coupling reactions. Electrophilic aromatic substitution proceeds more selectively on this core ring than on simpler cresols, supporting the manufacture of high-value specialty products. The ability to customize modification paths continues to drive interest, particularly for applications where structural clarity and purity matter, such as in regulated drugs or advanced materials.
Synonyms & Product Names
The name 2-Nitro-4-Cresol remains the most direct, though product catalogs and regulatory databases include several synonyms. These include 4-Hydroxy-3-Methyl-2-Nitrobenzene, para-Nitro-meta-cresol, and sometimes abbreviated forms like 2N4C. European chemical inventories track it as EINECS 208-727-6, while importers in the US refer to it by its CAS number 1575-77-5. Clarity in nomenclature helps users trace the origin, safety data, and historical literature, which matters as supply chains stretch across countries and regulatory bodies.
Safety & Operational Standards
Handling 2-Nitro-4-Cresol brings genuine risks, as inhalation or skin contact can cause irritation or more severe systemic symptoms. Manufacturers emphasize the need for appropriate ventilation, splash protection, and gloves in all handling. Storage away from acids, bases, and oxidizers reduces accident potential, and fire suppression equipment caters to its combustibility. I remember a training seminar where an experienced handler walked newcomers through incident simulations, underlining how routine discipline—not just alarms or paperwork—prevents serious cases. Standard operating procedures now incorporate real-time sensor monitoring and automated shutoffs for large reactors, aligning with updated workplace safety laws seen in both Europe and North America.
Application Area
Industrial dye manufacturing pulls most of the product, especially for yellow and orange pigments. Beyond that, 2-Nitro-4-Cresol steps into pharmaceuticals, agrochemicals, photographics, and even as a process standard in some analytical settings. The electronics industry applies it as a soldering flux component and in microfabrication chemistry. Academic curiosity still fuels additional application areas—novel catalysts, biodegradable agrochemicals, specialty resins—often supported by minor tweaks to the parent molecule. Its role grows as demand for traceable, consistent intermediates rises in highly-regulated markets.
Research & Development
Ongoing research covers greener nitration methods that use less acid and produce less byproduct, echoing industry trends towards environmental responsibility. Analytical chemists push for better, faster tests to gauge impurity profiles or detect illegal dumping. Universities trace reaction pathways by isotope labeling experiments, teaching students that even “simple” compounds bring surprises and lessons about molecular design. My own dive into the literature turned up work on encapsulated forms for controlled industrial release, signaling where process safety technology may head. Cooperative R&D among chemical suppliers, users, and regulators has shortened the lag between lab discovery and safe commercialization.
Toxicity Research
Animal and cell studies chart both acute and chronic exposure ratings, typically aligning 2-Nitro-4-Cresol with moderate toxicity. Most findings connect inhalation or dermal contact to short-term irritation and sometimes longer-term liver or kidney impacts. Increased focus falls on environmental pathways, such as breakdown rates in soil or uptake in aquatic organisms. Regulatory limits keep trending tighter as more is learned about persistence and bioaccumulation. Efforts from academic groups and in-house toxicologists now peer into subtle, long-term effects with new biomarker tools, not just the older observable symptoms.
Future Prospects
As the push for green chemistry gathers steam, 2-Nitro-4-Cresol will likely face further scrutiny in both its manufacturing methods and its end-of-life breakdown in the environment. Progress towards bio-based phenolic inputs, recyclable catalyst systems, and effluent-free production holds promise. Demand in dyes, semiconductors, and controlled pharmaceutical syntheses continues to grow, especially where traceability and certification matter. Calls for new derivatives in digital printing and functional polymers keep laboratories busy on the development front. Every advance in safety, sustainability, and product design finds its roots in well-understood classics like 2-Nitro-4-Cresol, and new generations of researchers seem poised to stretch its boundaries further.
What are the primary uses of 2-Nitro-4-Cresol?
Across the World of Dyes
2-Nitro-4-cresol holds a well-worn seat in the dye industry. Factories turn to this compound when a bright, consistent color is the goal. Whether it’s for textiles or inks, this chemical plays a reliable part in achieving crisp, yellow to orange shades found on shirts, banners, and paper napkins. Chemists rely on 2-nitro-4-cresol as a building block for various azo dyes. Synthetic colors last longer and resist washing out, saving both the planet’s water supply from excessive dye runoff and people from frustration over faded laundry. Markets for dyed leather and fur also make use of this substance to ensure the finished product turns out with the expected hue.
Pharmaceutical Applications
Pharma laboratories count on chemicals like 2-nitro-4-cresol not as drugs themselves but as intermediates. Medicines require complex molecules, made step by step. 2-Nitro-4-cresol often sits early in that chain, offering a functional base for antihistamines and antiseptics. Anyone reaching for allergy tablets or certain skin creams might be holding the result of a process that started with this raw material, long before it touched a blister pack or tube.
Pesticide Production
Farming communities face real struggles with insects attacking crops. The pesticide market has a long history of using building blocks like 2-nitro-4-cresol to produce agents that protect grains and vegetables. This chemical contributes to the synthesis of some insecticides and herbicides. It acts as a bridge in chemical transformations, helping chemists produce substances that defend fields and greenhouses. Farmers depend on these solutions to avoid financial loss, while scientists keep working to minimize negative impacts on groundwater and beneficial insects.
Laboratory Reagent in Synthesis and Research
University and industry researchers use 2-nitro-4-cresol as a flagpole in organic chemistry labs. It reacts with other compounds, making it a staple for small-scale experiments or teaching laboratories. Students, graduate researchers, and project leaders look for substances with well-understood behaviors, and this compound fits the bill. When learning about kinetics or tackling synthesis of new molecules, a reliable reagent like this saves time and keeps results straightforward.
Safety and Environmental Perspective
People working with chemicals like 2-nitro-4-cresol can’t afford to take short-cuts on safety. Irritation to the skin, respiratory tract, or eyes can occur, so gloves and goggles are not optional. Manufacturing plants install scrubbers and capture systems to cut down on pollution. Waste handling matters—a spill affects not just workers, but the local soil and waterways. Regulations from agencies like OSHA and the EPA exist because safe practices are often hard won lessons. On the consumer end, most folks never see this compound directly, but what happens upstream can trickle into daily life. Progress in greener chemistry could swap out some of these tougher substances for safer options, if researchers keep at it.
Finding a Middle Ground
Society reaps clear benefits from chemicals like 2-nitro-4-cresol, from well-dyed shirts to secure harvests and reliable medicines. Warnings against careless handling have kept communities safer compared to previous decades. Investing in better disposal systems, improved personal protective equipment, and ongoing worker training can go a long way. Funding new chemistry is expensive, but consumer demand for safer products, along with tighter environmental rules, keep the pressure on. Everyone from the factory floor to the research bench has a stake in how chemicals get put to use now and in the future.
Is 2-Nitro-4-Cresol hazardous to health?
What Is 2-Nitro-4-Cresol?
2-Nitro-4-cresol pops up in industrial settings, especially where dyes, pigments, and certain chemical processes get attention. Looking at the yellowish solid, you probably wouldn’t expect much fuss, but history has taught me that a chemical’s looks tell us nothing about its true personality.
Exposure and Health Effects
Skin contact with this compound can lead to irritation or even burns if exposure drags on without proper protection. Over the years, working near similar aromatic nitro compounds, I realized how easy it is to underestimate even tiny particles—especially once dust rises. Knowing many folks skip gloves or masks on hot days drives home just how crucial work habits become with chemicals like this one. Reports from workers show redness, itching, or—if someone gets unlucky—severe allergic responses. Eyes sting, too, if dust or liquid splashes catch you off guard. Inhaling the stuff causes coughing, wheezing, or headaches, especially in tight indoor spaces lacking good airflow.
Going beyond the surface, the real worry turns toward long-term effects from nitro compounds as a group. Some studies tie extended inhalation or frequent skin contact to more serious health risks, like organ damage. Animal tests suggest the risk isn’t imaginary. Evidence shows that compounds similar to 2-nitro-4-cresol, when entering the bloodstream over months or years, stress out the liver and kidneys.
Cancer Risks
Cancer risks always draw attention. For 2-nitro-4-cresol, no smoking gun exists suggesting it causes cancer directly in people. Research on close relatives in the nitro group paints a mixed picture, though. Some have caused tumors in lab animals, especially with high doses and long exposure. This opens the door for caution. Without big human studies yet, I’d never feel comfortable brushing aside concerns.
Guidelines and Worker Safety
Regulators like OSHA, NIOSH, and the European Chemicals Agency review compounds such as this with two questions: Are people getting sick? Are current guidelines enough? Some authorities recommend strict exposure limits even for those working short shifts. For example, the European Union lists 2-nitro-4-cresol as a substance of concern and highlights the value of gloves, goggles, and industrial ventilation.
Personal experience says these protections work—if people wear them consistently and use them right. Too many times, I’ve seen workplaces cut corners, letting fumes gather or skipping gear to save money or time. Such decisions stack risks for staff and the wider community. Even if company rules sit on the books, real safety only happens if leaders make health a daily priority, not a slogan above the door.
Looking Ahead: Safer Alternatives and Solutions
Most companies benefit from reviewing their chemical inventory every now and then. Some industries have swapped out nitro-based additives and dyes for safer alternatives, often with no drop in product quality. Bigger firms have even shifted their entire colorant lines to plant-based compounds or less-reactive synthetics. For small businesses, basic steps like modern fume hoods, regular air monitoring, and up-to-date training cut down exposure risks dramatically. Open communication between managers and staff—plus listening to workers who handle chemicals every day—pushes real-world solutions forward faster than any safety poster.
Staying alert, asking tough questions, and following up with action beats regret every time. Chemicals like 2-nitro-4-cresol don’t get headlines often. That’s no reason to treat them lightly once you take a closer look at what the science and shared experience have to say.
What is the proper storage method for 2-Nitro-4-Cresol?
Understanding the Risks Behind The Bottle
2-Nitro-4-Cresol stands out in my mind as one of those lab chemicals that teaches respect for attention to detail. A tiny mistake—in storage, in handling—brings headaches you never want. I remember years ago working late in a facility with an aging chemical storage room, and someone neglected to check the cap was tight on a similar aromatic compound. Odor spread, safety flags popped up, and everyone’s evening was spent in evacuation drills. Small missteps easily snowball with compounds like this.
Why Dry, Cool, and Sealed Storage Counts
2-Nitro-4-Cresol shows sensitivity to several environmental factors. Exposure to moisture, heat, or sunlight opens the door to degradation. Left unchecked, it produces noxious vapors or—much worse—combines with other chemicals to create a toxic mix. I’ve seen colleagues assume moderate room conditions were safe, not factoring in how rapidly temperature spikes in unventilated storage spaces over a long weekend. Any rise in temperature increases volatility, and any hint of humidity triggers slow decomposition. Reliable storage starts with a tightly sealed container made of glass or high-grade plastic, never metal, and the label must stay visible, undamaged, and clear. I cannot overstate the ease with which a faded label creates confusion in any busy lab.
Ventilation: Not an Afterthought
Fresh air makes all the difference. A storage cabinet set aside for hazardous chemicals, built with ventilation or exhaust fans, blocks harmful vapors from accumulating and creating risk for fellow researchers or maintenance staff. Too many stories circulate of forgotten storage closets with a strange smell and discolored shelves. Fumes don’t have to be strong to cause problems. Repeated, low-level exposure to aromatic nitro compounds causes long-term health effects—skin irritation, headaches, and potential liver impact, as public health reports have documented. Ensuring proper airflow, installing chemical fume hoods, and checking cabinet integrity reduce the odds of a hidden threat building up over months.
No Surprises: Fireproofing and Logbooks
I’ve learned over the years that even seasoned chemists miss small signs of motor overheating, a dropped rag, or a poorly managed electrical outlet. With flammable or potentially combustible substances, fireproof cabinets are essential. Storing 2-Nitro-4-Cresol next to sources of ignition stands out as one of the more careless mistakes. Separation keeps small accidents from spiraling into major incidents. A written inventory, checked frequently, catches missing containers and tracks amounts better than memory ever will. If one bottle goes missing, it’s not just a math problem—someone’s health or even life could hang in the balance.
Training, Habit, and Accountability
Everyone who handles this material ought to get regular training, not just a quick show-and-tell. Dry runs—fake incidents, timed evacuations, quizzes—ensure the team treats storage and handling seriously. I’ve watched new interns spot errors veterans missed, simply because they approached the task with fresh eyes. Keeping open lines between supervisors, chemists, and safety officers fosters an atmosphere where everyone feels responsible for keeping dangerous chemicals locked down. Peer checks, posted storage instructions, and shared accountability reinforce good habits.
Proper storage of 2-Nitro-4-Cresol isn’t about following a checklist or looking good during a lab audit. Every step, from shutting the cap to maintaining the right environment, centers on protecting people—not just meeting a regulation. In my experience, it’s the routines and vigilance of everyday practice that prove their worth when something unexpected happens.
How should 2-Nitro-4-Cresol be handled and disposed of safely?
Why Proper Care Matters
2-Nitro-4-cresol turns up in the world of chemical manufacturing and research labs. Anyone who’s spent time around organic labs remembers the sharp, searching scent it gives off, the distinctive yellow color, and the way gloves seem absolutely necessary even before looking at the safety data sheet. This chemical isn’t just another bottle on the shelf. Skin exposure burns. Inhalation irritates the lungs. Spill some, and the cleanup stays fresh in your mind. Safety isn’t only for show; it’s about walking out at the end of the day with your health in one piece.
Everyday Precautions in the Workplace
Walk into a lab using 2-nitro-4-cresol and you’ll notice a few things. Respirators sit on hooks. Nitrile gloves never run low. Fume hoods hum along, pulling air away from faces. Nobody takes shortcuts. A few times, I’ve seen what happens when simple steps get skipped—eye-watering fumes, stains on the countertop that don’t wash clean, even a few trips to the health nurse. Direct skin contact causes burns, and dust sneaks into the air without warning. Wearing a well-fitted respirator and working behind glass shields limits exposure. Lab coats come off before anyone enters a break room, and contaminated gloves land in a marked bin.
Storage: More Than Just Shelving
2-nitro-4-cresol belongs in a cool, dry place, away from direct sunlight, far from sources of heat or open flame. I’ve seen old storerooms with leaky bottles, labels smeared by who-knows-what, and the mess becomes a risk in itself. Store this chemical in tightly sealed containers, away from oxidizers and strong acids. Metal shelves with spill trays serve well, giving a reminder that leaks do happen—and floors soak up more than water. A storage list on the door keeps everyone honest. If something looks off, a supervisor steps in immediately.
Disposal: Looking Beyond the Bin
Disposing of 2-nitro-4-cresol takes more than tossing it in the trash. This substance qualifies as hazardous waste under most local laws. Down the drain isn’t an option. Improper disposal leads to groundwater contamination and environmental harm. Having seen cleanup crews dig up old waste sites, it sticks with me that every shortcut leaves a scar in the soil or water. Labs set up dedicated waste containers for this chemical, clearly marked and securely closed. No mixing with incompatible materials, and regular pickups by licensed hazardous waste handlers keep things on track.
Practical Steps for Everyone Handling It
Training happens before anyone opens a bottle. Workers know what symptoms to watch for—skin irritation, breathing complaints, a spatter in the eye. Emergency showers sit near workspaces, never blocked by carts or boxes. Clean-up kits don’t collect dust; they get checked each month. If a spill happens, response gears up fast, using absorbent pads and chemical neutralizers, all with proper protective equipment in place. The focus always stays on doing things right the first time.
The Responsibility of Safe Practice
Handling and disposal of 2-nitro-4-cresol brings responsibility. It affects people today and the environment tomorrow. Upholding best practices isn’t about avoiding fines—it’s about keeping colleagues safe and leaving fewer problems for the next generation. Every workplace has a voice in shaping a safer, cleaner future. All it takes is a willingness to follow sound procedures, read every label, and treat each step as if it matters—because it truly does.
What is the chemical formula and structure of 2-Nitro-4-Cresol?
Chemical Formula and Structure of 2-Nitro-4-Cresol
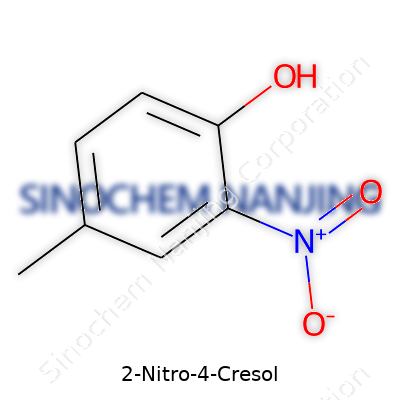
2-Nitro-4-cresol carries the formula C7H7NO3. That means seven carbon atoms, seven hydrogens, one nitrogen, and three oxygens work together in this molecule. I remember seeing its structure during lab prep—once you spot the nitro and methyl groups attached to the benzene ring, it stands out.
The name tells a story. The “cresol” part means you’re dealing with a methylphenol skeleton. “2-nitro” signals a nitro group sitting on the second carbon. Systematic naming puts the methyl at the para (4th) position relative to the hydroxy group. Picture a benzene ring: at the “1” point sits the hydroxy (OH), at “2” is the nitro (NO2), and at “4” is the methyl (CH3).
Drawing it out, it's not hard to spot the influence each group brings. The nitro group pulls electron density, makes things more acidic, and shifts the molecule’s reactivity. The methyl group can make reactions a little more selective. This is what my professor meant by “functional groups controlling the game.”
Why Structure Matters in Real Work
Plenty of folks overlook the nitty-gritty of a chemical’s shape and build, but structure shapes everything—a lesson that stuck with me pulling odd hours in a research lab. For 2-Nitro-4-cresol, its unique arrangement influences how it behaves in industrial and environmental settings. In dyes and pigments, this molecule offers vibrant color yet raises eyebrows due to its toxicity profile. Workers handling it in factories need solid safety protocols.
Those nitro and methyl arrangements aren't just trivia. The nitro group increases solubility, which means run-off isn't just a small issue. Streams and groundwater can see traces if disposal happens carelessly. Once, during a site audit, I saw how lax waste practices put entire water sources at risk. Change only came after fines piled up.
Looking at human health, 2-Nitro-4-cresol lands on several hazardous substance lists. Its ability to cross biological membranes leads to harmful effects, especially if inhaled or if skin comes in contact. Regulatory bodies—such as OSHA and the EPA—demand strict handling and labeling for a reason. Supporting research shows repeated exposure can cause irritation of skin, eyes, and respiratory tract, as well as potential systemic effects.
Paths Forward With Safer and Smarter Practices
The chemistry world stays on its toes, adapting protocols and inventing new ways to study and modify molecules like this one. Start with strict monitoring at manufacturing sites, build accountability for those involved, and use personal protective gear. Education helps, too—regular safety workshops, posted signage, and real-world case studies make abstract warnings concrete for staff.
On the environmental front, I’ve seen some firms switch to closed-loop processes that recycle solvent streams. Wet scrubbing systems capture airborne particles, leaving cleaner air behind. Thinking upstream, some innovators examine greener alternatives—reformulating dyes that reduce, or eliminate, harmful byproducts like 2-Nitro-4-cresol.
No chemical exists in isolation. Every structural tweak carries ripple effects in labs, factories, neighborhoods, and wild spaces. The chemical formula and arrangement might seem like just numbers and lines, but for many, they define daily routines and long-term health. Responsible use hinges on knowing—and respecting—exactly what sits inside every barrel or beaker.
| Names | |
| Preferred IUPAC name | 2-methyl-5-nitrophenol |
| Other names |
2-Nitro-4-Cresol 2-Nitro-p-cresol 4-Hydroxy-2-nitrotoluene 4-Methyl-2-nitrophenol 2-Nitro-4-methylphenol |
| Pronunciation | /tuː-ˈnaɪtroʊ-fɔːr-ˈkriːsoʊl/ |
| Identifiers | |
| CAS Number | 1575-58-8 |
| Beilstein Reference | 883132 |
| ChEBI | CHEBI:16470 |
| ChEMBL | CHEMBL165696 |
| ChemSpider | 12396 |
| DrugBank | DB14095 |
| ECHA InfoCard | 03e6e5b3-d997-44d9-b836-1a4e1c9c5dad |
| EC Number | 202-204-7 |
| Gmelin Reference | 7877 |
| KEGG | C01582 |
| MeSH | D009625 |
| PubChem CID | 7419 |
| RTECS number | GO8575000 |
| UNII | 5Y8A2UJ6VI |
| UN number | UN1323 |
| Properties | |
| Chemical formula | C7H7NO3 |
| Molar mass | 153.14 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.31 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 1.1E-4 mmHg at 25°C |
| Acidity (pKa) | 8.1 |
| Basicity (pKb) | 11.63 |
| Magnetic susceptibility (χ) | -70.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.629 |
| Dipole moment | 3.2061 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 142.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -71.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3084.5 kJ/mol |
| Pharmacology | |
| ATC code | D08AX06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H318, H411 |
| Precautionary statements | Precautionary statements of product 2-Nitro-4-Cresol are: "P280, P305+P351+P338, P302+P352, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-0-🌟 |
| Flash point | 113°C |
| Autoignition temperature | Autoignition temperature: 540°C (1004°F) |
| Lethal dose or concentration | LD50 oral rat 283 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 283 mg/kg |
| NIOSH | SN3850000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m3 |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
4-Cresol 2-Nitrophenol 4-Nitrophenol |

