2-Nitro-1,3-Xylene: A Deep Dive
Historical Development
Chemistry’s roots run deep, and 2-Nitro-1,3-xylene’s story traces back to the days when nitration of aromatic hydrocarbons first shaped the direction of industrial chemical production. In the late nineteenth and early twentieth centuries, chemists experimented with xylene and its derivatives, motivated by the growing need for intermediates in the dye and explosives industries. As nitration technology improved and safety knowledge grew, workers began to recognize the significance of introducing nitro groups to xylenes. Over time, researchers established standard preparation methods, improved yields, reduced hazards, and created the foundation for what modern chemical manufacturing looks like. Today’s producers build on that line of work, balancing technological advances with lessons from the past.
Product Overview
2-Nitro-1,3-xylene comes from the xylene family, specifically as a nitro derivative. This compound stands out due to its dual methyl groups positioned on the benzene ring, with the nitro group occupying the “2” position. Laboratories and industrial sites look to this compound for its intermediate value, especially in the production of specialty chemicals, pharmaceuticals, and dyes. Its distinct attributes, notably reactivity and selectivity, set it apart among the various nitroaromatics. Bulk suppliers and small-scale labs alike keep this chemical in inventory due to its reliability and performance in synthesis pathways that depend on both electron-donating and electron-withdrawing properties.
Physical & Chemical Properties
2-Nitro-1,3-xylene presents as a yellowish solid at room temperature and offers a melting point that usually sits between 53 and 55°C. Solubility hinges on the polarity of available solvents; the compound dissolves better in organic liquids such as acetone, chloroform, or ethyl acetate, while showing very limited solubility in water. With a molecular formula of C8H9NO2 and a molar mass around 151.17 g/mol, it handles the chemical load expected of functional intermediates in organic synthesis. The electron-donating methyl groups drive certain substitution reactions, while the sizeable nitro group pushes reactivity toward reduction and nucleophilic steps, guiding researchers to clever chemical modifications.
Technical Specifications & Labeling
Industry expects precision. Technical data sheets describe necessary details: CAS number for identification, purity generally exceeding 98%, trace impurities below 1%, and moisture levels controlled under 0.5%. Suppliers include labeling for hazard classification—consistent with GHS guidelines—alongside UN numbers for regulated transport. The compound earns risk phrases calling out possible skin, eye, and respiratory irritation. Packaging for labs tends to use amber bottles with tamper-proof seals. On a larger scale, drums and bulk containers feature rigid materials to prevent leaks. Documentation includes handling recommendations, first-aid advice, and chemical inventory tagging. Proper labeling reduces risk and improves communication up and down the supply chain.
Preparation Method
Manufacturers derive 2-nitro-1,3-xylene by direct nitration of m-xylene. This process means stirring m-xylene into a cooled mixture of concentrated sulfuric and nitric acids, maintaining temperature controls to guide regioselectivity. Workers monitor acidity and reaction time to achieve the high yield of the 2-nitro product, with side reactions forming other isomers or leading to overnitration being common hurdles. Modern operations employ continuous stirring reactors and in-line quality monitoring. Once the reaction completes, neutralization with aqueous base, solvent extraction, and recrystallization purify the compound. Lab technicians in research settings might adjust process conditions for custom quantities or isomeric purity, reflecting customer demand for tight specifications in specialty chemical markets.
Chemical Reactions & Modifications
The aromatic backbone of 2-nitro-1,3-xylene invites both reduction and substitution. Many industrial chemists exploit the nitro group, transforming it into an amino group through catalytic hydrogenation. This step unlocks possibilities for further derivatization—think sulfonation, halogenation, or even cross-coupling with selected substrates. The activation from methyl group positioning also shapes the type and speed of reactions with electrophiles. For dyestuff synthesis, the compound serves as a building block for constructing more complex aromatic amines or for introducing functional groups at defined ring locations. In some protocols, oxidation of the methyl groups yields corresponding acid or aldehyde derivatives, often under controlled temperature and specific reagent choices to limit by-product formation.
Synonyms & Product Names
Chemical complexity often comes with a long list of alternate names. People in research, logistics, or different geographies call 2-nitro-1,3-xylene by various labels: 2-nitro-m-xylene, 2-methyl-1,3-dimethyl-4-nitrobenzene, or o-nitro-m-xylene. Some catalogs reference its registry numbers, while safety offices rely on international names for compliance documentation. This multitude of synonyms can confuse procurement or regulatory reviews, but it highlights the diversity of uses and historical naming conventions. Accurate naming plays a big part in preventing mix-ups, especially in multistep synthesis campaigns where even small errors risk entire batch losses.
Safety & Operational Standards
Safety in working with 2-nitro-1,3-xylene depends on established training and alert systems in production environments. Inhalation or prolonged skin contact can irritate, so protective gear matters: gloves, safety goggles, and use of local exhaust systems reduce exposure risk. Plant operators use chemical sensors to keep track of airborne levels, especially during transfer or open handling. This compound’s combustibility requires storing it away from heat and strong oxidizers. Chemical hygiene plans encompass storage in well-ventilated spaces, routine leak checks, and regular safety drills. Workplace monitoring results guide updates for personal protective measures and emergency response steps. Safe waste handling, including neutralization and certified disposal, protects both workers and the environment.
Application Area
Industry workers turn to 2-nitro-1,3-xylene in several fields. It stands as an intermediate for producing azo dyes, a class known for intense colors and reliability in textile applications. It anchors many steps in agrochemical development, such as routes to active herbicide molecules. Pharmaceutical teams value it for coupling in certain therapeutic ingredient pathways, using its versatile reactivity to link molecular structures that might otherwise remain elusive. Student chemists use it in teaching labs when demonstrating principles of aromatic substitution, reduction, or building complex ring systems. Its technical performance and well-characterized nature mean that, across large and small operations, it holds a valued spot.
Research & Development
Research teams continue to probe new uses for 2-nitro-1,3-xylene as catalysts, selective ligands, or molecular building blocks. Some projects examine green synthesis approaches, targeting nitration methods that generate less hazardous waste or make use of recyclable solvents. Analytical chemists use the compound as a model substrate when developing new monitoring techniques—think HPLC, GC-MS, or new sensor technology—with an eye toward improving detection limits and speed. Computational chemists map out its electronic environment, hunting for optimized reaction recipes or looking for ways to harness its unique features for future organic synthesis innovations. Collaborative projects between universities and industrial labs often lean on this compound to bridge theory and bench-scale implementation.
Toxicity Research
Toxicologists and environmental scientists have a long-running interest in nitroaromatic compounds, and 2-nitro-1,3-xylene stands front and center in toxicity screens, environmental fate studies, and risk assessments. Inhalation studies, skin exposure models, and chronic ingestion assessments point to organ-specific effects when safety guidelines aren’t followed. Research groups have found that long-term exposure may affect the liver and kidneys, and breakdown products can persist in some environmental settings. Regulatory agencies such as the EPA and ECHA set workplace exposure limits and require reporting for spills or environmental release. Cross-disciplinary teams often run bioaccumulation testing to forecast the impact if releases move beyond controlled zones. These data help companies and regulators update safety protocols over time.
Future Prospects
Green chemistry has shifted the focus in chemical manufacturing toward less waste and safer products. New research tries to streamline the nitration step, using alternative catalysts, flow reactors, or enzymatic approaches to boost both selectivity and environmental performance. Process intensification and digital monitoring now help operators detect off-spec materials before they reach production scale. Synthetic chemists keep looking for new downstream uses, like in advanced electronic materials, sensor devices, or pharmaceutical precursors. Tighter regulations on workplace exposures and waste streams push ongoing work to find biodegradable derivatives, offering similar functional flexibility while reducing ecological footprint. As innovation keeps moving, familiarity with the strengths and risks of 2-nitro-1,3-xylene equips chemists and manufacturers to adapt, discover, and use this compound safely in a world with growing technical and environmental expectations.
What is 2-Nitro-1,3-Xylene used for?
A Patchwork Role in Modern Chemicals
2-Nitro-1,3-xylene pops up in surprising corners of the chemical industry. This isn’t some niche compound tucked away in a forgotten lab; it helps build the backbones for dyes, pigments, and pharmaceuticals. Shaped by two methyl groups and a nitro group, this xylene derivative pulls weight where the demand for specialty organic synthesis rises. Factories in Asia and Europe roll out barrels of this stuff to feed into larger supply chains.
Building Blocks for Color and Creativity
Anyone who worked in textile or paint development knows that most bright, lasting colors don’t just come from nature. Synthetic dyes and pigments often depend on molecules like 2-nitro-1,3-xylene. This compound links into larger molecules, shaping intense blues, reds, and violets that find a home on everything from blue jeans to car panels. Its chemical profile simplifies certain reactions, cutting down time, costs, and unwanted byproducts. In the world of color, a steady flow of 2-nitro-1,3-xylene means quicker rollouts for trendy shades.
Driving Innovation in Medicine
Pharmaceutical labs can’t ignore specialty aromatics. A compound like 2-nitro-1,3-xylene forms essential steps in crafting new medicines, especially antibiotics and anti-inflammatory drugs. Chemists count on its unique structure to add nitro and methyl groups at just the right spots on a growing molecule. In my own work years ago, I saw how this chemical enabled us to bypass certain tedious processes—saving months on projects and helping move drug discoveries forward. With drug resistance climbing and demand for new treatments rising, this time-saving edge becomes crucial.
Beyond the Lab: Safety, Supply, and the Environment
Any time an aromatic nitro compound shows up, safety experts pay attention. 2-nitro-1,3-xylene demands careful handling. It can irritate skin and lungs, and it doesn’t break down easily in water. Wastewater treatment plants working near chemical manufacturers may find traces in their intake. Researchers have flagged questions about how these substances might linger in groundwater and soil. In my local community, this led to calls for stricter storage and spill-response protocols—a move echoed in industry rulebooks worldwide. Factories have taken steps with double-walled tanks, enforced air filtering, and training for spill cleanups. These frontline changes help limit daily exposure for both workers and the environment.
Responsible Production and Fresh Solutions
Green chemistry offers new paths for producing and disposing of compounds like 2-nitro-1,3-xylene. Some firms turn to catalysts that run at lower temperatures and water-based solvents to cut down on hazardous waste. Efforts don’t stop at the plant gate—community partnerships invite third-party audits and neighborhood air sampling. These programs build trust and spot trouble before it spreads.
Directing resources toward safer chemistry has its share of hurdles, especially for smaller suppliers who keep the global industry moving. Regulatory agencies have offered grants and technical advice for greener upgrades—support that needs to continue if long-term change stands a chance. Back in the day, switching over to safer processes felt like a gamble on tighter margins, but new incentives now help companies weather those early costs. That creates more space for data sharing and innovation, both of which improve public health and turn industry knowledge into everyday protection.
What are the safety precautions when handling 2-Nitro-1,3-Xylene?
What’s in a Chemical: Everyday Reality with 2-Nitro-1,3-Xylene
Spend enough time in a chemical lab, and you quickly learn some materials earn extra respect. 2-Nitro-1,3-xylene — a yellowish crystalline powder — stands out. It serves well in synthetic dyes, making pigments, even some pharmaceutical work. But there’s no getting around it: this stuff brings health risks. I remember my first encounter in grad school. My supervisor didn’t rely on posters. He showed firsthand what can go wrong without proper steps. That lesson stuck harder than any warning label.
The Hazards: Not Just Numbers on a Chart
What makes 2-nitro-1,3-xylene worrisome? The data tells us it’s toxic if inhaled, ingested, or if it lands on skin. It can cause irritation in eyes, respiratory system, and skin. Prolonged or repeated exposure brings risk to your liver and kidneys, sometimes even hematopoietic effects. The fact that these dangers pop up in real lab accidents, not just technical papers, should change how anyone approaches this chemical.
Once, a colleague accidentally knocked over a beaker. No one felt immediate effects, but the strong odor stood out. Three hours later, two of us felt headaches and queasy. The cause was clear: ventilation in our older lab just couldn’t catch up. That single event convinced me to trust my nose and always double-check airflow.
Zero Room for Complacency: Everyday Prevention Steps
Good gloves matter. Not all nitrile gloves block aromatic nitro compounds effectively. Thicker gloves, and double gloving for longer procedures, add peace of mind. Don’t handle this stuff bare-handed.
Eye protection is a must. Safety glasses with side shields work, but a full face shield adds a layer of honesty—stuff splashes, and regular goggles sometimes miss the mark.
Lab coats and closed shoes. I stopped wearing open-toed shoes years ago after a minor chemical spill. And the less absorbent the clothing, the better. Those blue disposable coats seem light, but they give chemicals less to cling to.
Ventilation saves workdays. Open benches don’t cut it for 2-nitro-1,3-xylene work. A certified fume hood—regularly tested, sash down, nothing blocking the airflow—should be non-negotiable. I check airflow with smoke sticks even if the indicator light glows green.
Label and store right. In my lab, clear secondary containers and labels (with dates) stop confusion. This isn’t a chemical for the bottom shelf or crowded storage; dedicated bins limit mixing accidents.
Emergency Prep: No Time to Guess
Every workplace should know the protocol. For skin or eye contact, reach for the safety shower or eye wash, not just a wet paper towel. Speed matters. Have the emergency number for local poison control and occupational health in plain sight. I urge new lab members to run a drill, not just read the instructions.
Spill kits need to handle organic compounds—no high school solutions like sweeping it up dry. Absorbent pads, gloves, and dedicated disposal bags come out at the first sign of trouble.
Real Safety Culture Grows from Experience
I’ve heard people call comprehensive safety routines “overkill.” The folks who say that haven’t been through a HAZMAT cleanup. Every short-cut skipped comes back twice as hard. Safety with 2-nitro-1,3-xylene isn’t about paranoia or ticking boxes; it’s about respect for a material that doesn’t care if you’re new, rushed, or “just being careful.”
A strong safety culture starts with hands-on learning and honest talk about mistakes. No shame in asking for a second set of eyes on your procedure. In the world of 2-nitro-1,3-xylene, the person who double-checks often winds up the healthiest.
It only takes one exposure to change how you work, but it shouldn’t take an accident for people to get it right.
What is the chemical structure and formula of 2-Nitro-1,3-Xylene?
Understanding the Basics of 2-Nitro-1,3-Xylene
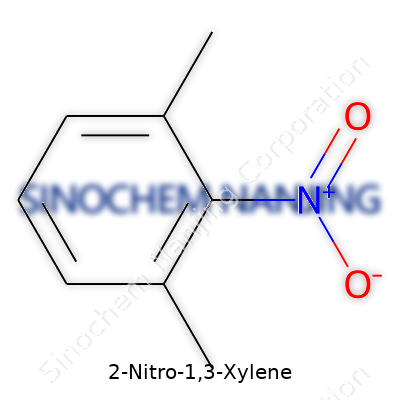
2-Nitro-1,3-xylene comes from the family of xylene derivatives. Its backbone rests on a benzene ring, with two methyl groups and a nitro group attached. The name tells you exactly where everything sits: methyl groups at positions 1 and 3, a nitro group locking into position 2. In chemical terms, its formula is C8H9NO2.
These numbers matter in labs and factories. Lining up the groups on the ring isn’t just a formula game. Placement changes how this compound behaves. Nothing in chemistry seems random. The positions set up the electronic cloud on that benzene ring, which in turn shapes reactivity and toxicity.
How the Structure Matters in Real Life
I remember the first time I handled a sample of substituted xylene. Safety training hammered home how even a tiny change on an aromatic ring transforms its impact on humans and the environment. Methyl and nitro groups are powerful actors. The nitro group at the 2-position bumps reactivity, especially under conditions used in organic synthesis. It also throws up red flags for toxicity screening.
Take the chemical structure: benzene looks simple, but adding two methyl groups (CH3) at the 1 and 3 spots keeps the molecule pretty electron-rich. The nitro group (NO2) at position 2 drains electrons, pulling the ring’s charge in new directions. In a molecular drawing, think of the ring as a clock—with the methyls at noon and 4, the nitro at 2. Chemists read this shorthand and picture exactly where reactions might start or clog up.
Why Structure Drives Importance
Chemicals like 2-nitro-1,3-xylene don’t exist in a vacuum. Out in the real world, they become blending agents, intermediates for dyes, and even contributors to industrial pollution. Factories often choose specific xylene derivatives to push reactions the right way. This compound’s structure makes it easier to introduce the nitro group for further reactions, like reduction or coupling. In hands-on work, this saves time and cuts steps—valuable when safety, cost, and product purity matter.
Unlike the more famous parent xylene (which has three isomers—ortho, meta, para), 2-nitro-1,3-xylene’s unique ring pattern lets scientists tailor synthetic routes. For dye manufacturing, this structure offers a strong launchpad for building more complex aromatic frameworks. This is the kind of information any chemist should have top of mind before scaling up or picking a substitute.
Looking Toward Safer and Smarter Chemistry
Safety with chemicals like 2-nitro-1,3-xylene can’t become an afterthought. Methylxylene derivatives with nitro groups raise concerns. They can cause respiratory irritation, and their environmental persistence demands careful handling. Studies published in Environmental Toxicology and Chemistry highlight the way substituted nitroxylenes don’t always break down quickly under standard wastewater conditions.
Better containment, active monitoring of storage areas, and improvements in personal protective equipment (PPE) are practical solutions. Researchers have started experimenting with enzymatic methods for breaking down nitro-aromatics. These approaches didn’t exist decades ago. Expanding green chemistry approaches, including catalyst recycling and benign synthesis routes, points to real progress. I’ve watched labs migrate from old cleanup solvents to safer alternatives. This shift usually starts after someone reads the MSDS and sees the long-term impact spelled out in plain language.
Key Details at a Glance
- Formula: C8H9NO2
- Structure: Benzene ring, methyls at 1 and 3, nitro at 2
- Main Uses: Dye intermediates, chemical synthesis
- Major Risks: Toxicity, environmental persistence
Knowing the arrangement and chemical formula of this compound gives scientists and manufacturers more control. Mistakes shrink when people understand what’s in the beaker. The right information can help prevent pollution, limit waste, and keep workers safe.
How should 2-Nitro-1,3-Xylene be stored?
Safety Lessons Learned the Hard Way
Every time I think about chemical storage, my mind goes straight to stories—some where things went wrong, others where the procedures caught problems before they turned serious. Chemicals like 2-Nitro-1,3-Xylene don’t care about your schedule or whether you’re in a rush. This stuff can ruin your day or your workplace fast if it leaks, overheats, or even mixes with the wrong container. So, keeping it in the right spot matters a lot more than it might seem at first glance.
Looking at the Science—Not Just the Labels
2-Nitro-1,3-Xylene has some traits that make it a risk in the wrong hands or poor conditions. It’s flammable, toxic, and it can turn nasty if it spills or vaporizes. Just storing it far from heat won’t cut it. I remember walking through a lab after someone ignored the warnings and put flammable chemicals next to a sunny window. The bottles were so warm that you could feel the heat radiate if you stood close. That’s why more than a shade or a locked door separates smart storage from a disaster.
Choosing the Right Container and Location
Keeping 2-Nitro-1,3-Xylene in a well-sealed, chemical-resistant container isn’t just regulation—it saves lives. Polyethylene or glass containers with tight-fitting lids keep vapors inside and moisture out. I’ve seen labels peel off cheap bottles in humid rooms, leaving mystery fluids behind. Real labeling and regular checks make sure nothing becomes a guessing game. A flammable safety cabinet that’s both grounded and ventilated stops sparks and static before they start trouble. I learned long ago to leave containers in the original packaging as much as possible. Once you start transferring chemicals, you invite mistakes.
Why Forgetting Ventilation Costs You Later
It’s tempting to think that shelving chemicals in a storeroom solves the issue. In reality, vapors build up. I once opened a storage closet and got hit with odors that made my eyes water. Mechanical ventilation pulls out vapors before they turn an enclosed space into a hazard. Good airflow can feel like a nuisance when it rattles or hums all day, but it keeps toxins out of your lungs and lowers the risk of a surprise fire. Don’t wait for the HVAC to break before asking if the chemical room even has a vent.
Keeping Records, Training People, and Acting Fast
Lax record-keeping turns storage into roulette. An up-to-date chemical inventory doesn’t just keep the boss happy; it helps in emergencies. Staff have to know what’s dangerous, where it lives, and what to do if things start spilling or burning. I’ve worked with teams that drilled emergency responses until they could do them with their eyes closed. In those places, accidents turned into stories about fast reactions, not messes in the local news.
Respecting the Rules Pays Off
The EPA, OSHA, and other agencies don’t write rules to make life harder; those standards come from real tragedy. Following those guidelines means using secondary containment trays, fire extinguishers within reach, and keeping incompatible chemicals far apart. A little attention to detail doesn’t just avoid fines—it saves lives and property. That’s not theory; it’s hard-won wisdom from people who learned the long way.
What are the potential health hazards of 2-Nitro-1,3-Xylene exposure?
The Hazard Behind a Familiar Name
Sometimes, the names you spot on a chemical drum sound like they belong in a distant lab, far removed from everyday life. 2-Nitro-1,3-Xylene is one of those, but its impact can cut much closer to home for those in chemical manufacturing or research. This yellowish-brown solid creeps into the workplace during the creation of dyes, pigments, or as a reference compound in laboratories. In my early days working alongside industrial hygiene teams, we handled compounds like this with care, but rarely did the dangers feel real until someone experienced the side effects firsthand.
What Breathing This Stuff Does
Inhaling even small amounts of 2-Nitro-1,3-Xylene starts an unpleasant chain of events. Fumes irritate the nose and throat right out of the gate. Workers often shrug off that burning, scratchy sensation, blaming it on a long shift or poor air flow. Ignoring it only brings on stronger headaches, dizziness, and sometimes a wave of nausea. Longer stretches around this chemical push it deeper into the body, where it can mess with the liver and kidneys. Blood tests sometimes show changes that hint at early organ stress, even before classic symptoms pop up.
Getting 2-Nitro-1,3-Xylene on Your Skin
Not every hazard comes from breathing. I once watched a coworker spill a splash of this stuff on his wrist. It did not look dramatic; a small, oily spot that he wiped off right away. Within hours, his skin flared up, turning red and itchy with a patchy rash. Many synthetic chemicals bite the skin, but this one sometimes leaves peeling or even blistered patches if not washed off quickly. Rubbing your eyes by accident spreads the problem—stinging and watering signals your mistake right away. Without fast rinsing, eye exposure means a trip to the doctor. Studies track slow-healing damage to eye tissue with prolonged or concentrated contact.
What Science Tells Us
The U.S. National Institute for Occupational Safety and Health (NIOSH) calls 2-Nitro-1,3-Xylene a hazardous substance for a reason. Animal studies show it triggers organ changes and sometimes sets off mutations in their cells. Long-term effects in people are hard to nail down, but repeated work exposure has lined up with higher risks of nervous system issues, memory loss, and chronic fatigue. People with chemical sensitivities or previous liver problems take an even bigger hit.
Facing the Threats: Smarter Work Practices
Gloves and goggles are not enough if the workplace does not take airborne exposures seriously. Engineers I know swear by local exhaust systems and closed transfer lines. I have seen the difference: in shops with good ventilation, headaches and complaints drop off fast. Emergency showers and eyewash stations need more than a "checklist" status. Real training and drills matter. Some businesses run regular air monitoring and rotate duties so no one spends hours at the hottest spots near open containers. A tight safety data sheet in every break room keeps everyone aware, not just those trained in chemical handling.
The Importance of Respecting the Risks
Many workers skip gloves or eye protection on busy days, trusting that a small mistake does no lasting harm. My experience says otherwise. A moment's shortcut often means weeks of follow-up. 2-Nitro-1,3-Xylene deserves every ounce of respect. Workplace safety is not just about following rules. It's about making sure everyone gets home healthy, ready to come back the next day.
| Names | |
| Preferred IUPAC name | 1,3-dimethyl-2-nitrobenzene |
| Other names |
2-Nitro-1,3-dimethylbenzene 1,3-Dimethyl-2-nitrobenzene 3,5-Dimethyl-2-nitrobenzene |
| Pronunciation | /ˈtuː ˈnɪtroʊ waɪl ˈθriː ˈzaɪliːn/ |
| Identifiers | |
| CAS Number | 603-72-1 |
| Beilstein Reference | 1208736 |
| ChEBI | CHEBI:34175 |
| ChEMBL | CHEMBL504431 |
| ChemSpider | 11776 |
| DrugBank | DB04011 |
| ECHA InfoCard | 03ba7ab9-6ddb-45ab-b7e0-30d481d27e6b |
| EC Number | 209-724-6 |
| Gmelin Reference | 7877 |
| KEGG | C08354 |
| MeSH | D015911 |
| PubChem CID | 72698 |
| RTECS number | XT8575000 |
| UNII | W2ZVT43U7D |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C8H9NO2 |
| Molar mass | *150.17 g/mol* |
| Appearance | Light yellow crystalline powder |
| Odor | odorless |
| Density | 1.114 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.004 mmHg (25°C) |
| Acidity (pKa) | 6.99 |
| Basicity (pKb) | 11.10 |
| Magnetic susceptibility (χ) | -69.7×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.595 |
| Viscosity | 1.175 cP (25°C) |
| Dipole moment | 3.31 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 226.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −26.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4077.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02,GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-OX |
| Flash point | 82 °C (180 °F; 355 K) |
| Autoignition temperature | 540°C |
| Explosive limits | Explosive limits: 1.4–7% |
| Lethal dose or concentration | LD50 oral rat 1070 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1590 mg/kg (rat, oral) |
| NIOSH | SN2100000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
1,3-Xylene 2-Nitrotoluene Nitromesitylene |

