Digging into 2-(N-Acetylcarbamoyl)-4-(3,4-Dimethylbenzenesulfonyl)Benzenediazonium Hydrogen Sulfate: Science, Safety, and What Lies Ahead
Looking Back: Where This Molecule Comes From
Chemistry pulls us along a winding path, sometimes just to stretch the boundaries of what's possible. Long before 2-(N-Acetylcarbamoyl)-4-(3,4-Dimethylbenzenesulfonyl)Benzenediazonium Hydrogen Sulfate ever found its stride, the story began with organic dyes and coupling reactions. Synthetic chemists in the last century tinkered with diazonium salts, hunting for new colors, improved stability, specialized materials, and tools for molecular design. Sitting among these came the targeted engineering of sulfonylated diazonium compounds. Adding the N-acetylcarbamoyl group did more than dress it up—it carved out a new place in laboratories working with complex molecular scaffolds. This path, built by years of gradual synthesis and accident, shapes how modern research teams look at molecules like this one: not just as an endpoint but as a platform that keeps evolving.
What's Special About It: Breaking Down the Structure
2-(N-Acetylcarbamoyl)-4-(3,4-Dimethylbenzenesulfonyl)Benzenediazonium Hydrogen Sulfate deserves a closer look, right down to its bones. Sporting a diazonium group on a benzene ring, paired with a bulky 3,4-dimethylbenzenesulfonyl moiety, it has a knack for stability compared to the old-school diazonium salts that chemists know too well for being jumpy and poorly behaved. The N-acetylcarbamoyl component doesn't just ride along—it tunes reactivity and opens a path to different pigment and pharmaceutical intermediates. When used in the lab, the sulfate counterion gives it a boost in water solubility while shaping its shelf-life. Holding this molecule in your hand, you’re working with something that bridges functional groups rarely seen together outside specialty synthesis.
Physical and Chemical Details
Handling this diazonium salt, a pale to orange solid, means smelling a whiff of acrid chemical sharpness and respecting the granulated form. While it's heavier than it looks, the powder disperses in water and select organic solvents. The melting point lands lower than similar compounds—a point to remember, since heat breaks diazonium salts into simpler fragments, some of which can pack a punch. Anyone in the lab knows storage means cool, dry, and dim conditions: too much light or a stray spark, and you’re courting decomposition. Chemists who’ve worked with it remember seeing its fading color—the sign of breakdown in the presence of air. The pH and solubility provide an edge in aqueous media, and the reactivity won’t give you much time once you pop the cap.
Digging Into the Details: Specification and Labeling
Getting this chemical off the shelf, the user expects upfront, honest labeling, since mistakes have a price in fine chemical work. Purity matters. Most labs target at least 98% for reliable results. Impurities hiding in the bag can ruin entire syntheses, triggering unintended byproducts that confuse analysts and slow down research. Storage temperatures tend to get nailed onto labels with red or bold print, and the shelf life, if listed, is rarely more than six months out. Those handling instructions—use gloves, protective eyewear, run a vent hood—aren’t just legal fluff. The stakes for mishandling are high, and awareness saves lab coats and hospital visits.
Rolling Up Sleeves in Synthesis: How It's Made
The route to this molecule demands precision, patience, and an in-depth understanding of diazotization chemistry. You don’t dash off a batch in any old glassware. The backbone starts with nitration and sulfonation of a precursor benzenoid, followed by the careful attachment of N-acetylcarbamoyl and 3,4-dimethylbenzenesulfonyl groups—organic transformations that require dry reagents and carefully controlled temperatures. Converting the amine function to a diazonium salt needs cool sulfuric acid, sodium nitrite, a well-timed addition, and constant monitoring. One slip-up with the acid or temperature, and the reaction either stalls or explodes—something organic chemists remember, sometimes too well. The final product, after precipitation with hydrogen sulfate, dries under vacuum to a powder that moves directly into cold storage.
Reactivity and “Tinkering” with Structure
What makes this compound so appealing in modern organic synthesis lies in its reactivity. Diazoniation gives you a launchpad for introducing new groups in aromatic substitution. Crafty chemists have used it to build complex molecules on demand, from pharmaceuticals to embedded pigments in polymers. Modifications don't stop with the parent structure—subtle tweaks lead to whole new branches of chemical families. Want a different solubility? Swap a methyl for an ethyl. Need to change stability or reactivity? Adjust the acetyl piece or shuffle partners on the sulfonyl ring. The possibilities mushroom, limited mainly by the imagination and nerve of the researcher at the bench.
Synonyms in the Trade and Research Circles
This molecule doesn’t stick to just one name. In the world of patents, scientific articles, or supply catalogs, chemists might see jargon like “N-acetylaminocarbonyl-dimethylbenzenesulfonyl diazonium sulfate” or shorthand that captures its key motifs. Keeping track of synonyms is more than a paperwork issue. Mishaps happen when researchers get caught between naming conventions, and there’s no forgiving a mix-up at that scale. Each name frames it for a different audience—for synthetic chemists, for patent lawyers, or high-throughput screening labs.
Prioritizing Safety Without Shortcutting Discovery
Working safely means never underestimating the risk of diazonium salts, especially those with hidden reactivity. Stories circulate in the community—an unmarked vial left in a warm corner, a dust mask not worn, or a forgotten acid bath. These tales end with at best a mess to clean, at worst an emergency call. Safety data flags respiratory, skin, and eye irritation, as well as acute toxicity with careless contact. Training is more than a sign-off sheet in research environments, where hands-on knowledge and repetition build respect for these chemicals. Waste disposal follows tight protocols, since running them down the drain isn’t just illegal, it pollutes. Every bottle handled is a lesson in caution and respect, not just compliance.
Seeing Its Reach: Uses in Today’s Lab and Beyond
Labs across the globe leverage this compound for precise coupling reactions—making azo dyes, functionalizing aromatic rings, or forming key intermediates for agrochemical and pharmaceutical pipelines. Sometimes it pops up in the design of light-fast pigments for plastics and fibers, with the diazonium group offering a handy handle. Researchers working on electronic materials have turned to these diazonium derivatives to build up complicated layers on electrodes and sensors. Their specialty value comes from tailored modification, allowing targeted reactions that few other groups can pull off with such control or efficiency. Consequences ripple out beyond the bench, since innovations here trickle into finished goods, sustainability projects, and new classes of biomedical tools.
Research, Risk, and the Push for Safer Chemistry
Research takes center stage on two fronts: unlocking what the molecule can do, and defining its limits. Labs continue investigating novel coupling reactions, expanding the catalog of what’s possible. But another camp works on the flip side, probing toxicity and environmental persistence. Diazonium salts sometimes break down into aromatic amines and other byproducts—some with known toxicity issues. Animal studies, cell line screenings, and computational modeling search for answers: how dangerous are these fragments, what concentrations persist, and how quickly does the body clear them? Balancing breakthrough with responsibility means tackling these questions head-on, exploring greener solvents, streamlined purification steps, and biodegradable alternatives wherever feasible.
Looking Forward: Where This Chemistry Could Land Next
Future prospects depend on innovation, sharper regulation, and industry pressure for greener, safer molecules. There's a clear drive to design diazonium compounds with lower toxicity and improved stability, so that breakthroughs in the lab don’t lead to headaches in waste disposal or safety training. Advances in computer-aided design bring new variants to the table, as researchers map out reactivity and hazards before the flask ever gets warm. Industries hungry for precision—next-generation dyes, high-performance materials, and pharmaceutical intermediates—stand to gain from tweaks to structure and function. New diagnostic tools, smart sensors, and custom coatings all tie back to what began in traditional dye chemistry and branched far afield. Developers working in this space know the reputation of diazonium chemistry: tricky but rewarding, risky but packed with potential. The challenge is clear—keep safety standards sky high, push discovery forward, and never lose sight of the responsibility that comes with every tube and bottle.
What is the primary use of 2-(N-Acetylcarbamoyl)-4-(3,4-Dimethylbenzenesulfonyl)Benzenediazonium Hydrogen Sulfate?
Understanding Its Place in Chemical Manufacturing
Every time I visit a research lab, I spot shelves packed with compounds hardly mentioned in everyday conversations. One of those is 2-(N-Acetylcarbamoyl)-4-(3,4-Dimethylbenzenesulfonyl)Benzenediazonium Hydrogen Sulfate, usually shortened for sanity’s sake. This mouthful of a molecule carries serious weight in organic synthesis, and chemists respect its powerful diazonium group, especially in dye and pigment creation.
Pigment Production and Beyond
With so much color in our lives, it’s easy to forget that most synthetic dyes borrow their vibrance from compounds like this one. The diazonium component acts as a bridge for coupling reactions with phenols and aromatic amines, which become the basis for azo dyes. I’ve seen technicians adjust shade or stability by tweaking the substituent groups, and this molecule’s sturdy framework allows targeted color development. The entire textile and printing sector owes much of its palette to clever chemistry like this, supporting billions in economic activity every year.
Why Purity and Structure Matter
Friends in analytical chemistry talk about structure with reverence. The presence of the N-acetylcarbamoyl and dimethylbenzenesulfonyl groups increases water solubility and helps control the reactivity. It also reduces the odds of unwanted side reactions, which can ruin a dye’s consistency. Academic studies point out that impurities, even at low concentrations, lead to unexpected results in the final product—think about faded fabrics or unstable inks. Labs rely on this compound’s predictable behavior, and standardization saves a lot of troubleshooting later in the production pipeline.
Environmental Impact and Safety
Concerns about sustainability grow louder each year. Azo dyes made from diazonium salts have received criticism for their environmental persistence. Some regulatory agencies now demand strict monitoring, especially for wastewater. Factory managers must follow disposal protocols and invest in pre-treatment to neutralize byproducts before they hit the municipal system. Investing in cleaner synthesis routes and lifecycle tracking makes a difference—not only for compliance, but for the river ecosystems that sit downstream from plants.
Improving the Process and Reducing Waste
Years ago, a colleague worked on a pilot project that re-engineered diazonium reactions to use less hazardous solvents. The results looked promising: fewer emissions, reduced worker exposure, and lower costs. Breakthroughs like flow chemistry now allow more precise control over conditions, which means fewer side reactions, leading to better yields and less waste. As the sector pushes further down this road, everyone from raw material suppliers to end-product manufacturers should see benefits, both economic and societal.
Supporting Research and Innovation
Universities dedicate real resources to finding alternatives to classic diazonium salts where possible. At the same time, they study ways to enhance the efficiency and specificity of those still needed. A strong research pipeline, funded and guided by clear regulatory frameworks, can accelerate the transition toward safer, greener dye production methods. For students, this is a chance to blend academic inquiry with industry partnerships, gaining hands-on experience with both synthesis and real-world applications.
Looking Forward in Specialty Chemicals
This compound’s role in color chemistry demonstrates how a single molecule can punch above its weight. By investing in process upgrades and greener disposal, companies serve markets without neglecting their environmental responsibilities. Success here comes from a balance between proven chemistry and fresh solutions—a lesson every sector can take to heart.
What are the storage requirements for this chemical?
Looking Closer at What Storage Involves
Storing chemicals isn’t just about putting barrels or bottles on a shelf. My years in industrial labs taught me that even a misplaced container can create a headache or put people at risk. Every chemical comes with its own story—corrosives eat through weak spots, volatiles sneak off as fumes, oxidizers react with the wrong neighbors. Safe storage starts by understanding the unique behavior of each substance, since one-size-fits-all storage could turn a chemical storeroom into disaster central.
Why Location Makes the Difference
In an old paint plant job, I watched a newcomer set down solvent cans close to acids, thinking it saved a few steps on busy days. One spill could turn that shelf into a fire hazard. Strong acids and bases demand separate, leak-proof cabinets—polyethylene or enamel often works best. For flammable materials, local regulations call for metal cabinets with self-closing doors, vented to prevent fumes from collecting. Keeping oxidizers away from anything that burns turns every fire code from theory into real protection for people and property.
Temperature and Moisture: More Than Just Details
I’ve seen cheap fixes backfire when humidity creeps into the storage room. Desiccants or sealed containers keep moisture away from water-reactive chemicals. Many products want a temperature range—some offer more safety if refrigerated, which means a spark-free lab fridge, not the same one holding someone’s lunch. Temperature swings can cause expansion, leaks or even explosions. Facts support this: the U.S. Chemical Safety Board has counted dozens of incidents that started with overheated chemicals.
Labeling and Inventory: More Than a Checklist
Safe storage leans heavily on labeling. A simple, bold label can stop confusion when seconds matter. During a training stint, I learned to double-check every incoming container against its Safety Data Sheet, not only for the right name, but expiration date, hazards, and handling tips. Out-of-date or mystery bottles belong nowhere except the hazardous waste bin. Good recordkeeping makes inventory easy—a worn label or missing entry usually hints at bigger problems down the line.
Fume Control and Air Flow
Chemical odors tell you something is leaking or exposed. I remember a storeroom where improper ventilation led to headaches and complaints until we improved airflow and added exhaust fans. Some chemicals, even in closed bottles, let out vapors that build up without fresh air. Strong fume hoods or local exhaust keep air inside safe for anyone who walks in, and help meet occupational safety standards.
Personal Protective Equipment and Training
Even the best storage plan fails if people take shortcuts. I’ve learned to never overlook goggles, gloves, and lab coats—one careless moment, and acid can splash or vapors sting eyes. Training staff on chemical hazards and emergency steps creates a culture where safety isn’t just a manual, it’s muscle memory. OSHA rules draw a line, but a strong safety mindset builds trust among coworkers.
Finding Better Solutions
Modern storage systems save lives—spill trays catch drips, alarms warn of leaks, and digital inventory controls cut out guesswork. Still, nothing replaces daily vigilance. Whether in a big facility or a small workshop, storing chemicals with care protects not only the people nearby but the community outside the door. Solid storage practices save money, reputation, and sometimes, much more than that.
What safety precautions should be taken when handling this product?
Pay Attention to Labels and Warnings
Most accidents at home or work happen because warnings get ignored. I’ve seen cases where people trusted their instincts and skipped reading the fine print, or used a cleaner in a small bathroom with the window shut. A label isn’t just there for show—it tells you where the real dangers lie. Corrosive, flammable, toxic—these words spell out what could go wrong. Pick up the package, read every bold word, and don’t rush through the steps. People in the medical field or chemical plants never skip the instructions, because mistakes hurt.
Personal Protective Equipment Matters
I remember the stinging smell from a batch of ammonia I once helped mix for a friend’s backyard cleanup. My gloves bought at a discount store melted in minutes. Quality goggles, sturdy gloves, masks, and even aprons matter. Research shows about 80,000 chemical exposures reported in U.S. workplaces each year, many caused by poor gear or no gear at all. Nitrile gloves work for solvents and oils, but cheap latex ones won’t stop harsh detergents. Cotton t-shirts don’t block splashes; a long-sleeved old shirt helps, but dedicated protective wear stops burns and rashes. The proper gear might not feel stylish, but it saves trouble later.
Good Ventilation Saves Lives
Opening windows and doors isn’t always top of mind, especially when weather takes a turn. Toxic fumes stay trapped indoors and they build up fast. One story that sticks is a family hospitalized after using a pesticide spray in their kitchen. They kept the air sealed in to keep the smell away from neighbors, but those invisible gases went straight to their lungs. A fan in the window, or even a small extractor, can keep air moving and prevent headaches, nausea, and longer-term damage. If that’s not enough, taking work outside beats risking your health indoors.
Storage: Out of Reach and Out of Trouble
Some chemicals react if mixed together, like bleach and ammonia. I still remember a garage where someone kept half-used paint thinner beside a tray of fertilizer. A small leak could set off a fire or toxic cloud. Safety experts advise putting flammable and corrosive things in locked cabinets, away from kids and pets. Pharmacy bottles have child-resistant caps, but many garage and cleaning products do not. A summary from poison control centers lists storage issues as a leading cause of accidental poisoning in kids. Secure shelving and separate containers protect more than just your floors.
Disposal: Don’t Cut Corners
A lot of towns set up hazardous waste drop-off days. It’s tempting to pour old chemicals down the drain or toss them with regular trash, but they travel into water supplies or the soil. My neighbor nearly ruined his backyard by dumping leftover paint thinner beside his compost bin. City waste management and recycling programs have resources and staff trained to keep everyone safe. Bringing products to them takes longer, but keeps the entire community healthier.
Common Sense Wins Every Time
Nothing replaces being careful. Double-check labels, don’t mix unknown substances, wear real protection, and let others know what you’re handling. If something feels off—a strange smell or a burning sensation—stop right away and get fresh air. Familiar products can still surprise you, so a bit of respect and planning goes a long way.
Is this compound stable under normal laboratory conditions?
It Matters More Than Most Think
People often ask if a compound is stable under normal laboratory conditions. Nobody wants to waste time, risk safety, or blow grant money on something that won’t last. Having worked in a few busy research labs, I know the smallest oversight causes problems. Think glassware filled with smoke, repeated orders for the same reagent, or burnt eyebrows. Stability is a practical issue, not just a box to check in the safety sheet.
Why Stability Decides More Than Safety
Stability really means you can store a substance without worrying it will lose effectiveness, react with air, or cause trouble. Sodium chloride, for example, stays the same for months sitting on a shelf, no fancy storage required. On the other hand, reagents like sodium hydride react violently with moisture. One careless moment with the lid can ruin your experiment or hurt someone nearby.
Fact is, the US Occupational Safety and Health Administration (OSHA) tracks incidents rooted in improper handling of reactive or unstable chemicals. In 2021, unstable chemicals accounted for dozens of lab accidents. Each case led to schedule delays and untold costs.
Learning From Others’ Errors
It’s easy to forget that basic details like light sensitivity or humidity influence outcomes. I once kept a sensitive dye on my bench, thinking the cap would keep it safe for a few hours. By afternoon, every sample turned brown—the price of skipping a dark drawer and dry atmosphere. Turns out, even a few hours in ambient light can destroy some compounds’ structure, especially those with lots of electronic double bonds or unstable ring systems.
A quick glance at published research shows thermally unstable compounds trip up both beginners and seasoned chemists. The journal Nature discussed a case where polymer researchers misjudged the stability of a new material, leading to failed batches and a recall of data sets. People who’ve spent years in industry will nod knowingly—failure to respect shelf life has shut down more than one promising project.
Logic and Fixes
Clear, readable labels help—date received, hazard notes, recommended storage. Digital inventory tools now provide automated reminders, reducing brain strain for over-worked grad students and staff. Ask old timers, they’ll tell you: don’t trust memory alone.
Simpler fixes work, too. Keep checklists for regular inspections. If that jar of peroxide looks cloudy, dump it. Train every new team member to check MSDS sheets before moving anything. Discard expired materials, no second guessing.
Production-scale labs often use dedicated climate-controlled rooms for sensitive stocks. It’s not just because someone said so; it prevents fires or lost batches when someone accidentally stores ammonia nitrate next to organic solvents.
The Stakes
Stable compounds mean reliable experiments, safe workspaces, and less waste. Long after I left academic research, every new project still starts with one question: can I trust this stuff to sit on my bench for a week without turning toxic, useless, or dangerous? The answer decides everything from cost to safety to scientific credibility.
Chemistry works best with a little humility and a lot of preparation. Everyone thinks they know how stable their compound is until they don’t. Use the data, respect the warnings, and learn from those who’ve made mistakes before.
Does it require any special disposal procedures?
Why Safe Disposal Matters
Most folks don’t often think about what happens to things after tossing them in the trash. Out of sight, out of mind seems to rule the day. Still, the world doesn’t run on magic. Garbage doesn’t just disappear. Items that seem harmless, like batteries or old lightbulbs, have ingredients inside that can seep into the soil or water if dumped without care. That’s not just bad for critters and plants; it comes back around to us, too.
Hazardous Waste: More Common Than You Think
Household cleaners, paint cans, leftover medications, and electronics all count as hazardous waste. These items carry chemicals that, if handled carelessly, can build up in groundwater and hurt local wildlife for years. I remember a neighbor pouring paint thinner down the storm drain after a garage cleanout. A few days later, that classic chemical smell wafted through our street—somebody’s good intentions, but not such a great move for our creek that runs past the playground.
The EPA estimates Americans toss out over three million tons of electronics every year. Only about a quarter ends up recycled. Televisions, computers, and phones often contain lead, mercury, and other heavy metals. Old medicine flushed down the toilet shows up later in trace amounts in rivers and, eventually, tap water. It’s not paranoia; studies in dozens of cities have found this link.
Finding Better Paths Forward
Clear laws lay out what counts as hazardous waste. Paint, pesticides, and large batteries must go to approved collection sites or local events. Call your city’s public works or visit their website to find out where to drop things off. Many hardware stores take back used batteries and bulbs. Police stations often run drug take-back drives, making it easy to keep pills off the street and out of the water supply.
People sometimes hold onto old chemicals or electronics for years, worried they’ll get in trouble or pay a hefty fee for safe disposal. In reality, most community programs make it painless. Some locations even offer pickup services for special items. Cost rarely stands in the way; most places waive fees on collection days.
Everyday Habits Make a Difference
It helps to know what’s in household items. Look for labels that warn of corrosive, poisonous, or flammable contents. Before tossing, check if something could leach toxins where you live. Keeping these items out of regular trash protects garbage collectors and landfill workers, too.
At home, my family keeps a plastic bin in the garage for questionable items. Old batteries, spent vaping cartridges, and defunct smoke alarms go there until we haul them to the local hazardous drop-off. This small habit keeps our conscience clear and ensures the wrong stuff doesn’t wind up in the landfill just because it fit in a garbage can.
Solutions: Start with Local Resources
No one expects you to memorize the EPA’s chemical registry. Yet, looking up waste rules takes only minutes and prevents years of downstream harm. Reach out to local officials or visit local government websites. Some apps even list disposal options by zip code. As more neighbors follow these steps, entire communities see less pollution and healthier water. That trade-off makes the hassle of proper disposal a lot easier to accept.
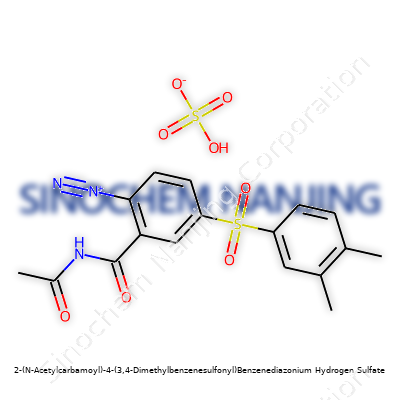
| Names | |
| Preferred IUPAC name | 2-[Acetyl(aminocarbonyl)amino]-4-[(3,4-dimethylphenyl)sulfonyl]benzenediazonium hydrogen sulfate |
| Other names |
Fast Blue B Salt C.I. 37235 Benzenediazonium, 2-(acetylamino)-4-[(3,4-dimethylphenyl)sulfonyl]-, hydrogen sulfate 2-(Acetylamino)-4-(3,4-dimethylbenzenesulfonyl)benzenediazonium hydrogen sulfate Fast Blue B BN Salt |
| Pronunciation | /too ɛn ə-ˈsiː-təlˌkɑːr-bə-ˌmɔɪl fɔːr θriː, fɔːr ˈdaɪˌmɛθ-ɪlˌbɛn-ˈziːnˌsʌl-ˌfə-nɪl ˈbɛn-ziːn-ˈdaɪ-ə-zoʊ-ni-əm ˈhaɪ-drə-dʒən ˈsʌl-feɪt/ |
| Identifiers | |
| CAS Number | 941678-36-6 |
| Beilstein Reference | 3956764 |
| ChEBI | CHEBI:139476 |
| ChEMBL | CHEMBL4643112 |
| ChemSpider | 30853819 |
| DrugBank | DB08747 |
| ECHA InfoCard | 03f3c7ff-cc6e-49db-8d9b-60f49e9bede7 |
| EC Number | NA |
| Gmelin Reference | Gmelin Reference: "Gmelin 100086 |
| KEGG | C17435 |
| MeSH | D014332 |
| PubChem CID | 187470223 |
| RTECS number | GV0700000 |
| UNII | I6W8TZG07E |
| UN number | NA1993 |
| CompTox Dashboard (EPA) | DTXSID90192469 |
| Properties | |
| Chemical formula | C17H18N3O6S2 |
| Molar mass | 528.57 g/mol |
| Appearance | Light yellow solid |
| Odor | Odorless |
| Density | 1.54 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.14 |
| Acidity (pKa) | -2 |
| Basicity (pKb) | 4.5 |
| Magnetic susceptibility (χ) | -73.76 × 10⁻⁶ cm³/mol |
| Dipole moment | 6.98 D |
| Pharmacology | |
| ATC code | N02BE01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS07,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H331 + H315 + H319 + H317 + H334 + H335 + H410 |
| Precautionary statements | P264, P280, P301+P312, P302+P352, P305+P351+P338, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 1-1-0-☢ |
| Flash point | 137 °C |
| LD50 (median dose) | LD50 oral rat >2000 mg/kg |
| REL (Recommended) | 0.02 mg/m³ |
| Related compounds | |
| Related compounds |
Benzenediazonium tetrafluoroborate p-Toluenesulfonyl hydrazide Diazomethane 3,4-Dimethylaniline Acetylcarbamic acid |

