2-Methylvaleraldehyde: Context, Science, and Future
Historical Development
Chemists first started paying attention to aldehydes in the 19th century, recognizing their value in both synthesis and scent. Years later, as labs explored the possibilities within branched-chain molecules, 2-methylvaleraldehyde emerged as a compound that didn’t just check the books—it sparked new ways of thinking about structure-activity relationships. My own early academic exposure to aldehydes drove home the point that even small changes in a molecule’s structure, like adding a methyl group to valeraldehyde, could turn a stable substance into something with a sharper aroma or a different reactivity profile. That willingness to experiment, to take the foundation of earlier discoveries and push them, led to the isolation and characterization of 2-methylvaleraldehyde. It became clear that this molecule had uses well beyond the lab bench, fitting into both industrial and niche scientific pursuits.
Product Overview
2-Methylvaleraldehyde draws attention for its simple structure—a branched aldehyde with a short carbon chain. Unlike many chemicals, its role isn't limited to just being a base for further synthesis. Industries value it for perfumery, flavorings, and specialty chemicals. Having handled it in a lab setting, I can say it’s not merely a chemical name to memorize. The distinct, pungent odor proves hard to forget, hinting at a wide reach in scent formulation. Whether used as a building block for amines, acids, or larger aldehydes, this molecule sits firmly in the lineup of basic chemical feedstocks.
Physical & Chemical Properties
The substance usually arrives as a clear, colorless to slightly yellow liquid, carrying a strong, sharp scent. Its molecular formula—C6H12O—signals a moderate weight and a volatility that matters for storage and handling. It evaporates at room temperature, making ventilation a priority. Its density and boiling point put it in the range of many common organic liquids, not especially dense but quick to respond to heat. That volatility can catch users off guard; my first encounter with a bottle left a lasting sensory impression. Its chemical backbone features the characteristic aldehyde group, making it a ready partner for reactions involving nucleophiles or oxidants.
Technical Specifications & Labeling
Labels typically highlight the need for care: flammability takes center stage, and warnings about skin and respiratory exposure remind users that this isn’t a chemical to treat casually. In regulated labs, each container demands clear hazard pictograms. Spec sheets provided by suppliers outline minimum purity requirements and shelf-life estimates—handy for anyone running reactions where impurities could skew results or threaten safety. In my experience, labs with good labeling habits protect not only the current team but anyone visiting or working in the same space down the line.
Preparation Method
Production of 2-methylvaleraldehyde has roots in classic organic synthesis, often using aldol condensation or controlled oxidation of related alcohols. Commercial operations usually take advantage of higher yields from catalytic processes, making the economics work on a larger scale. Recollections from my time in academic chemistry remind me that side reactions and byproducts kept us on our toes. Purification by distillation wasn’t just a chore—it was a test of patience and technique, since the product’s volatility called for careful temperature control. Factories chasing purity deploy similar tactics but marry them with modern continuous separation to keep up with demand and waste regulations.
Chemical Reactions & Modifications
Reactivity is where this aldehyde really shines. That exposed carbonyl group eagerly participates in further transformations. Reduction yields the corresponding alcohol, a step often seen in fine chemical synthesis. Oxidation pushes the substance toward acid derivatives, opening routes to specialty solvents or intermediates for pharmaceuticals. In lab work, Grignard or Wittig reactions harness the aldehyde’s affinity for nucleophiles, letting chemists stitch together tailored molecules. Experience shows that most mishaps happen not because the chemistry is difficult, but due to underestimating the speed or vigor of these reactions—reminders that familiar molecules can still pack a punch.
Synonyms & Product Names
Depending on source or application, users might bump into names like 2-methylpentanal or methyl isoamyl aldehyde. This confusion sometimes leads to mix-ups in shipment or documentation. Any practitioner who has managed a chemical inventory knows the frustration of cross-referencing synonyms. The consistency in nomenclature serves more than just paperwork: it prevents accidents and missed opportunities, connecting dots between research studies or industrial best practices.
Safety & Operational Standards
Dealing with volatile aldehydes means prioritizing safety. 2-Methylvaleraldehyde doesn’t play by everyday rules; it requires tightly closed containers, flameproof environments, and proper ventilation. I’ve watched seasoned staff wear out before their time, thanks to overlooked solvent fumes or chronic skin irritation. Training and compliance audits help, but what truly counts is the culture—teams supporting one another to suit up properly, monitor exposure, and reinforce routine checks. Regulatory frameworks, such as OSHA and REACH in Europe, don’t just enforce paperwork. These set the standard for handling, disposal, and permissible exposure.
Application Area
Perfumery often leads the roster, as 2-methylvaleraldehyde’s unique odor becomes a key note in blends aiming for fruitiness or mown grass. Food flavor chemists add minute amounts for certain savory or fatty profiles. Beyond the senses, chemical manufacturers incorporate it into syntheses for agrochemicals, plasticizers, or pharmaceutical intermediates. Lab work with this aldehyde regularly functions as a training ground for students new to carbonyl chemistry. My experience in academic and industrial projects shows that its versatility earns repeat attention, especially for projects balancing cost, accessibility, and performance.
Research & Development
Curiosity keeps this molecule relevant. Ongoing R&D looks at catalysis that saves energy, green chemistry options that reduce side-waste, and safer packaging—each effort born out of lessons learned from years of trial and error. Recent years saw scientists push for more efficient asymmetric syntheses and methods using renewable feedstocks. From my own reading, collaborations between universities and industry often push the field further, replacing fossil-derived sources with plant-based precursors or leveraging enzymes for improved selectivity. Publications and patents continue to expand around bio-based routes and downstream modifications, hinting at a future where the carbon footprint shrinks as performance rises.
Toxicity Research
Toxicity sits high on the list of priorities. Exposure through inhalation or skin links to irritation, and chronic studies investigate more subtle effects. Regulatory agencies run tests on animals for acute and chronic exposure levels. Data suggest caution, though not panic, for proper lab and factory conditions. Having talked with occupational health specialists, I learned the gap between what’s known and what’s practiced means incidents often come not from ignorance, but from overconfidence or short cuts. The push for lower-threshold exposure guidelines reflects not just compliance but care for long-term worker well-being.
Future Prospects
As the push for sustainability grows, interest in greener synthesis methods climbs. There’s momentum for harnessing renewable resources, not just fossil fuels, to generate chemical feedstocks like 2-methylvaleraldehyde. Advances in catalysis and process engineering offer pathways that promise lower waste, better energy use, and improved safety. Researchers explore ways to tune the molecule’s reactivity for ever more complex final products, signaling roles in pharmaceuticals or specialty materials yet untapped. Field conversations with professionals hint that, though well-known, the molecule could see a resurgence as more sustainable supply chains and tailored applications emerge. The link between science and real-world impact pushes both industry and academia to refine, rethink, and expand possibilities.
What is 2-Methylvaleraldehyde used for?
Aldehydes Aren’t Just for Labs
Step into a perfume lab, and the aroma tells you a story built on chemistry. In that tale, 2-methylvaleraldehyde, a molecule that doesn’t catch headlines, plays an important role. This little aldehyde—with its short, sharp scent—sits quietly behind the scenes in making flavors and fragrances that people encounter every day.
What’s the Appeal in Scents?
Perfume and flavor companies have leaned into aldehydes for decades. The reason sits in their ability to lift other notes, serving up those fresh, noticeable top notes you get in soaps, cosmetics, and even some foods. 2-Methylvaleraldehyde pops up as a building block, giving a punchy, green, crisp quality to both fine fragrances and more utilitarian cleaners. Having worked with olfactory raw materials myself, nose fatigue hits fast without these bright additives to cut through the clutter of ingredients. It isn’t just about the aroma — it’s about what the scent triggers in the brain. A small tweak here, a short burst of 2-methylvaleraldehyde there, and a soap suddenly smells cleaner, more vibrant, more like a fresh start.
Where Else Does It Show Up?
Beyond prettifying products, this aldehyde finds a second life in pure chemistry and industrial synthesis. Chemical plants roll out batches of 2-methylvaleraldehyde as a starting material to make other chemicals—mainly through changes that add new groups or build larger molecules. For instance, turning the aldehyde into an acid or an alcohol forms links in a bigger chain that could end up in plastics, solvents, or even pharmaceuticals. Many of us don’t consider how apple flavors appear in candies or drinks. Often, that comes down to tweaks on molecules like 2-methylvaleraldehyde, which can be converted to fruity esters widely used in the industry.
Health and Safety: Facts Over Fear
Experience with lab chemicals always brings up safety. Working hands-on with volatile aldehydes, I learned to respect their reactivity. 2-Methylvaleraldehyde’s sharpness in smell signals that careful handling matters. Even at low levels, exposure to vapors can irritate eyes and skin. Strict handling protocols, good ventilation, and personal protective equipment aren’t negotiable. For end consumers, though, the quantities in finished products sit far below levels that could cause harm. Fragrance trade groups follow tight rules on how much of these materials can go in perfumes, providing published studies that show where safe thresholds lie. Transparency helps build trust, and companies post safety data sheets that reveal how risk drops as products get diluted and controlled through the manufacturing process.
Looking Ahead: Safer and Cleaner Chemistry
The industry faces ongoing challenges. Environmental sustainability sits squarely on the table in any R&D department meeting these days. Making flavor and fragrance molecules from renewable sources gets lots of attention. Today, some companies start with plant-based feedstocks or engineer microbes to craft aldehydes like 2-methylvaleraldehyde, lessening reliance on fossil fuels. Safer processes also mean better monitoring and equipment to limit worker exposure and environmental release. In my view, this brings a better reputation for chemistry and a much cleaner workplace than just a generation ago.
Solutions with Everyday Impact
It’s easy to overlook simple chemicals hiding in household products. The story of 2-methylvaleraldehyde shows how specialized chemistry meets everyday comfort, from soaps that smell inviting to flavors that brighten the snack aisle. Responsible production, clear communication, and sustainable innovation let us enjoy these tiny molecules without worry. The next time a bar of soap lifts your mood, remember—hidden chemistry deserves a little gratitude.
What are the safety precautions when handling 2-Methylvaleraldehyde?
What 2-Methylvaleraldehyde Brings to the Bench
2-Methylvaleraldehyde has its place in chemical synthesis and fragrance creation. It’s got an unmistakable scent and a knack for bringing special value to chemical reactions, where its reactive carbonyl group gets plenty of action. But that sharp, fruity smell isn’t just a sign of character. It signals that careful handling counts. Anyone who has opened a bottle of this aldehyde feels that strong vapor—so it’s smart not to trust your sense of smell for safety. It doesn’t matter if you’re in a research lab or a flavor facility: working with this chemical means keeping your guard up from the minute you start pouring.
Why Personal Protection Always Matters
Gloves aren’t optional here; nitrile or neoprene outshines thin latex when dealing with aldehydes. If some leaks onto bare skin, you’ll get irritation and redness fast. Goggles come out before the bottle gets opened. Even an accidental splash can sting your eyes, so keep those safety glasses close—nothing ruins your day like rinsing with an eyewash station while your vision clouds up. And a classic white lab coat does more than keep your clothes from stains. It becomes a shield against splashes, which do happen no matter how tidy you like to work.
Ventilation: The Unsung Hero
Every chemist knows the fume hood becomes your best friend here. 2-Methylvaleraldehyde evaporates quick, and that vapor can sting your throat or set off a nasty cough if you get close. Step out of the hood and even a little spillage leaves a scent cloud hanging in the air. If a hood isn’t handy, fans and open windows help, but nothing matches a true laboratory vent system. Don’t settle for a cracked window—let the proper airflow do the heavy lifting.
No Room for Sloppy Handling
Every surface that gets a drop of this stuff should be wiped with proper cleaning solvents after use. Paper towels and water won’t cut it—organic solvents like ethanol pick up the residue without leaving the place sticky. Never eat near your work let alone touch your face until you wash your hands. I once watched a colleague grab a snack after cleaning up an aldehyde spill; his throat burned for an hour and lunch got cut short.
Spill Response Demands Focus
Even with steady hands, spills happen. The first instinct shouldn’t be panic; grab absorbent pads that don’t dissolve or crumble. Once soaked up, throw the pads in a fume hood or seal them inside a hazardous waste bag. For bigger spills, sand or a universal absorbent powder controls the mess, then a chemical-resistant mop takes the rest. Label waste containers right away—nobody wants to deal with a mystery jug of old aldehyde sludge in the storeroom weeks later. The cleanup crew will thank you.
Storage: Planning Ahead Saves Trouble Later
Store 2-Methylvaleraldehyde in tightly capped bottles away from light and heat. This chemical reacts with oxygen over time, so checking the bottle caps for tightness and dated labels means less guesswork months down the line. Never rely on eyesight alone; if you see crusts around the rim or catch an odd smell near the storage area, investigate. Flammable storage cabinets beat shelves—organic solvents and aldehydes shouldn’t sit next to acids or oxidizers.
Training Makes a Real Difference
No one really learns safe handling from reading one data sheet. I learned my habits from skilled supervisors who repeated good practice at every step, and that kind of hands-on instruction always sticks better than safety posters. Training sessions, real walk-throughs, and honest mistakes (as long as no one gets hurt) make up the groundwork for lasting habits. That experience feeds the trust needed when you’ve got a bottle of something potent in hand.
What is the chemical formula of 2-Methylvaleraldehyde?
Real Insight into a Simple Compound
Sometimes, questions in chemistry look deceptively simple—what’s the formula for 2-Methylvaleraldehyde? In real life, practical chemistry rarely stops at the answer. Most folks remember college labs: glassware, odd smells, a twinge of dread over mixing something wrong. I do, anyway. But curiosity could drive anyone to look closer at the pattern of atoms behind a name like 2-Methylvaleraldehyde.
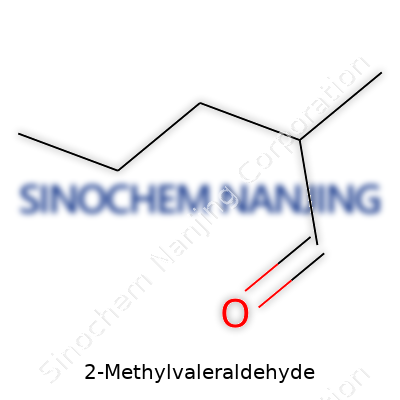
The heart of this question lies in recognizing the language chemists use. “Valeraldehyde” rings a bell for anyone who’s ever sniffed out flavor compounds in the food and fragrance industries. Its formal name, pentanal, shows five carbons lined up with an aldehyde group lounging off the end. Stick a methyl group on the second carbon, you get 2-Methylvaleraldehyde. The simple math for its chemical formula: C6H12O.
Why Should Anyone Care About a Formula?
Working in manufacturing and food research, it quickly becomes clear that recognizing the right structure saves everyone headaches. If you miss a methyl group on a production scale, your batch won’t perform as expected. The way molecules line up determines how they smell, taste, or fit into bigger chemical chains. For 2-Methylvaleraldehyde, the methyl branch changes reactivity, and that small switch gives new flavors and scents. Think of the difference a pinch of salt makes—same principle, but at the atomic level.
For factories dealing in aromas or specialty chemicals, knowing precise formulas is more than textbook learning. Inconsistent raw materials don’t just ruin one order—they undermine trust with clients who demand every batch matches the last. Many food safety recalls trace back to small irregularities in compounds like aldehydes. Reputations and sales swing on accurate chemistry, not just marketing or nice labels. The European Food Safety Authority (EFSA) and FDA require clean, detailed records before such compounds hit market shelves. So a deep understanding isn’t just for scientists or university types; it’s for anyone who eats, sniffs a candle, or sprays perfume in the morning.
Issues with Mislabeling and Solutions
Errors with chemical names or formulas seem harmless until they scale up. Mislabeling can leave workers exposed to unpredictable hazards or send tainted goods across borders. In my early lab days, a simple swap between isomers caused delays, rewrites, and safety reviews. The solution? Foster a no-shortcuts culture in chemical training and reporting. Double-checking formulas against IUPAC naming guidelines—or using software that verifies chemical structures—stops most issues before they reach shipping docks.
Education can’t stop at basics, either. Blending technical training with practical, real-life scenarios cements why accuracy matters. Most companies who succeed at this mix old-fashioned mentorship with digital checks. Government enforcement helps, but self-driven accuracy across the supply chain works faster. 2-Methylvaleraldehyde’s formula, C6H12O, carries more weight once people see the stakes tied to every digit and letter.
Looking Forward: Chemistry in the Real World
Mistakes in chemistry stories rarely start from ignorance. They start from assuming details aren’t worth double-checking. Getting the chemical formula right isn’t just for passing an exam—it’s a building block for safety, trust, and quality in modern business. Precision with even the simplest molecules often means the difference between success and setback. In the end, there’s no shortcut around getting the basics right.
How should 2-Methylvaleraldehyde be stored?
Getting the Basics Right
Storing chemicals like 2-Methylvaleraldehyde isn’t just about keeping bottles on shelves and hoping for the best. Anyone who’s worked in a lab or a plant knows odor and reactivity can turn a small oversight into a real safety problem. I remember a time at a facility where a misplaced aldehyde bottle ended up corroding nearby plastic supplies because we overlooked the power of vapor. These details stick with me every time I’m around volatile chemicals like this one.
Keeping Vapors Contained
This compound has a pungent smell for a reason—it evaporates easily. Vapors not only stink up workspaces, they can irritate eyes and skin. A tightly sealed glass bottle with a good chemical-resistant cap always beats cheap plastics, which degrade over time, especially againt aggressive compounds. Don't stash it near HVAC intakes or open shelves. Give it a home in a proper chemical storage cabinet designed for organic solvents. Keep that cabinet cool, dry, and ventilated, and don’t overcrowd it so you can spot leaks early.
Heat and Sunlight: Enemies of Stability
Direct sunlight speeds up breakdown, and even mild warmth nudges up vapor pressure. Store 2-Methylvaleraldehyde somewhere away from windows and hot machinery. Room temperature makes sense, provided there’s no sunlight. Refrigerated chemical storage helps if heat can’t be controlled—just keep things clearly labeled to avoid confusion with food or less hazardous chemicals. I’ve seen interns use a shared breakroom fridge for samples, and that’s a headache waiting to happen.
Keeping Air and Water Away
This stuff reacts with moisture and oxygen, which can lead to dangerous byproducts. Using a dry atmosphere—like with desiccant packs or nitrogen flushing—stops unwanted reactions. Don’t keep containers open longer than necessary. Label and date every bottle, and use chemicals on a “first in, first out” system. Open a fresh bottle only if an old one is empty or spoiled—waste adds up quick, and expired aldehydes can be more hazardous than you think.
Avoiding Potential Disasters
Storing this aldehyde near oxidizers, acids, and strong bases is a recipe for trouble. Since mixing even invisible traces of reactive chemicals can trigger pressure buildup or fire, I always double-check separation. Personal experience taught me that spills often come from overcrowded shelves and forgotten old stock, not from complex accidents. Regular inspections and inventory help keep things honest—seeing cracked caps or yellowed labels should send a signal to act fast.
Training and Emergency Readiness
Labels, safety data sheets, and training go further than any rulebook. Everyone who works near 2-Methylvaleraldehyde should know what it smells like, know the signs of leaks, and understand cleanup steps. Spills can ruin more than inventory; they can cost you your health. Simple PPE like gloves, goggles, and lab coats mean a lot. If you’re not sure what to do, reach out to experienced staff or occupational safety officers before improvising.
Wrapping Up Good Practices
Safe storage starts with respecting what chemicals can do when they escape the bottle. Focus on airtight, chemically compatible containers; keep them out of sunlight and away from heat, air, and incompatible substances. Regular checks and clear labeling make it hard to overlook problems until they're big. Good chemical stewardship builds trust, keeps people healthy, and protects valuable work.
Is 2-Methylvaleraldehyde hazardous to health or the environment?
What 2-Methylvaleraldehyde Brings to the Table
Almost every chemical has a story. 2-Methylvaleraldehyde isn’t on every household shelf, but it shows up across several industries. It helps synthesize flavors, fragrances, and finds a role in producing certain specialty chemicals. Just like any industrial chemical, seeing it pop up warrants a clear look at what it does to people and the places we live.
Health Hazards: Risks Deserve Respect
Take a whiff of 2-Methylvaleraldehyde in the lab and you’ll notice a strong, sharp smell. Inhaling vapors can lead to throat and eye irritation, and even headaches or nausea. A few studies from chemical safety data sheets point out that skin contact brings irritation. That gets worse with repeated exposure.
The Centers for Disease Control (CDC) and the Globally Harmonized System (GHS) have highlighted risks with this compound. Breathing in high concentrations may affect your nervous system. Some workers in manufacturing settings have reported dizziness or even sluggishness after exposure. Scientists haven’t linked it directly to cancer, but the lasting impact of repeated low-level contact remains largely unstudied in people. That gap means caution makes a lot of sense.
Environmental Fate: No Guarantee of Safety
Once 2-Methylvaleraldehyde leaves a factory, some of it lands in the air, water, or soil. Significant risks show up in aquatic environments. Fish and other water life react badly to aldehydes in general. Tests in lab conditions point out that 2-Methylvaleraldehyde can knock back aquatic organisms at relatively low concentrations. Its presence in waterways—no matter how small—leads to questions, especially if spills or leaks go unnoticed.
In the atmosphere, this aldehyde reacts and breaks down within a few hours to days. That seems like good news, but the products of its breakdown, such as smaller organic acids and ozone, carry environmental baggage. Ozone in the lower atmosphere doesn’t help the planet. It can damage crops and aggravate asthma in people.
We don’t see mountains of research focused specifically on soil and plant health with this chemical. Given how aldehydes act, a level of care looks warranted until deeper investigation comes along.
Managing the Risks: Steps for Safer Handling
Many industrial users stick to safety basics—ventilation, gloves, and face protection—when dealing with 2-Methylvaleraldehyde. Where I’ve seen it in use, local exhaust systems and careful bottle labeling stop most incidents before they start. Spill kits and emergency procedures matter more than some workers expect. Training plays a bigger role than any fancy device: new staff should know what the compound does and how to respond if things go sideways.
For waste handling, sending it off as hazardous material—never down the drain—makes a difference. The Environmental Protection Agency (EPA) tracks chemicals like this one under the Toxic Substances Control Act, so companies face penalties for careless disposal.
Better substitution sometimes beats better safety gear. Some businesses now opt for less volatile aldehydes or tweak their operations to avoid using 2-Methylvaleraldehyde altogether. Green chemistry has offered up alternatives for flavor and fragrance production. Each change needs real-world proof, but these switches reduce workplace risks and keep extra pollutants out of the environment.
The Road Ahead: Staying Watchful
Real protection comes from understanding, not ignoring. Calling for wider independent studies can help fill gaps about long-term exposure and subtle environmental effects. Workers and neighbors of factories alike benefit most when companies and regulators make transparency a standard practice.
Personal experience has shown me that small investments in safety—not just personal gear but in open conversations and practical protocols—pay off in the most important ways. 2-Methylvaleraldehyde proves one thing: it’s better to handle tough questions now than to face bigger problems down the road.
| Names | |
| Preferred IUPAC name | 3-Methylpentanal |
| Other names |
2-Methylpentanal α-Ethylbutyraldehyde |
| Pronunciation | /tuːˌmɛθɪl.vəˈlɛr.əl.daɪd/ |
| Identifiers | |
| CAS Number | 123-15-9 |
| Beilstein Reference | 83252 |
| ChEBI | CHEBI:89357 |
| ChEMBL | CHEMBL142205 |
| ChemSpider | 64251 |
| DrugBank | DB11338 |
| ECHA InfoCard | 100.007.848 |
| EC Number | 202-590-6 |
| Gmelin Reference | 7876 |
| KEGG | C02352 |
| MeSH | D008756 |
| PubChem CID | 12241 |
| RTECS number | OI8575000 |
| UNII | YF44F2P0GW |
| UN number | UN2378 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | strong, pungent, fruity |
| Density | 0.809 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 1.68 |
| Vapor pressure | 1.94 kPa (20 °C) |
| Acidity (pKa) | 14.68 |
| Basicity (pKb) | 12.74 |
| Magnetic susceptibility (χ) | -6.46 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.397 |
| Viscosity | 1.315 mPa·s (20 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -234.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3356.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-1-0 |
| Flash point | 45 °C (113 °F; 318 K) |
| Autoignition temperature | 207 °C |
| Explosive limits | Explosive limits: 1.3% - 6.9% |
| Lethal dose or concentration | LD50 (oral, rat): 740 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1600 mg/kg |
| NIOSH | WA9425000 |
| PEL (Permissible) | PEL = "100 ppm (410 mg/m3) |
| REL (Recommended) | REL (Recommended): 5 ppm |
| IDLH (Immediate danger) | IDLH: 300 ppm |
| Related compounds | |
| Related compounds |
Isovaleraldehyde 2-Methylbutanal Valeraldehyde 3-Methylbutanal |

