2-Methylallyl Alcohol: Perspective on Its Path and Place in Chemistry
Historical Development
Looking back, 2-methylallyl alcohol first drew attention among organic chemists decades ago, threading itself through the chapters of synthetic organic chemistry as both a building block and as a challenge. Laboratories in the mid-20th century sought small alkene-based alcohols to explore new routes for pharmaceuticals and specialty additives, and 2-methylallyl alcohol soon stood out. The compound offered double-bond reactivity with a structural twist due to the methyl group. This led to deeper investigation during the rise of physical organic chemistry, where people wanted to know how substituents could direct or hinder transformation. Those early experiments gave substance to the blueprints written by past chemists, who simply saw a three-carbon chain, an alcohol, and a branching point. Libraries from the late 1900s echo the incremental knowledge gained as researchers coaxed new reactions from this simple structure, and even now, universities use it as an example in introductory classes for understanding alcohol chemistry beyond ethanol or propanol.
Product Overview
2-Methylallyl alcohol, sometimes called methallyl alcohol, occupies a peculiar niche. Too reactive to ignore, but niche enough that it never gained household familiarity. It comes as a clear liquid, often slightly pungent. The industry sources it primarily for organic synthesis where having a reactive alcohol and a double bond together brings options for further modifications. A little goes a long way in many reactions, and its accessibility as a relatively small molecule means chemists can experiment without sinking heavy costs into specialized reagents. People outside academic labs rarely encounter the material on its own, yet the changes it can help introduce make it central to the family of functionalized alkenes and alcohols.
Physical & Chemical Properties
2-Methylallyl alcohol carries the chemical formula C4H8O and has a boiling point close to 113°C. The structural formula places a methyl group at the 2-position of an allyl alcohol, an arrangement that creates both opportunities and headaches for organic chemists. The molecule is moderately soluble in water and dissolves with ease in many organic solvents—a trait that eases its handling in multi-step syntheses. The presence of the double bond right next to the alcohol group means it's tuned for both nucleophilic and electrophilic routes, letting manufacturers choose reactions that suit their specific downstream product. The pungency is unmistakable, acting as a warning for those in the lab; the vapor shouldn’t be inhaled directly, judging by my own quick recoil during my first hands-on experience.
Technical Specifications & Labeling
Regulators classify 2-methylallyl alcohol under several chemical inventory lists, and bottles must feature proper hazard warnings. Labels on lab-grade material highlight its flammable nature and the risks linked to skin and eye contact. Purity grades often run above 97%, but researchers working in fine synthesis look for even tighter controls on contaminant levels. Storage specifications demand tightly sealed glass containers to prevent evaporation and contamination. Refrigeration contributes to longer shelf-life, as spontaneous polymerization or slow oxidation tend to degrade open samples. Those tasked with handling this alcohol learn quickly to respect its volatility and to keep flammable sources or hot surfaces well away.
Preparation Method
Factories rely on classic organic reactions to produce 2-methylallyl alcohol. Most routes take isobutene or closely related precursors and subject them to hydroformylation followed by careful reduction, or they draw it from methylallyl chloride via hydrolysis. These methods produce the desired molecule in sizable yield, although the specifics require operators to balance throughput with waste concerns and side-product formation. Some research teams have tested biocatalytic approaches, but cost and scalability remain limitations. Lab syntheses sometimes turn to Grignard-type additions for small batches, but the scale jumps to industry often favor catalytic processes using metals like palladium or rhodium, chosen for their selectivity and reliability.
Chemical Reactions & Modifications
In hands-on synthesis, 2-methylallyl alcohol shines because its double bond and alcohol group enable a web of chemical possibilities. Oxidation transforms the alcohol into aldehydes or acids, with the methyl branch influencing reaction rates and conditions. It accepts addition reactions across the alkene, making it attractive for growing larger, more complex molecules—common in agrochemicals and pharmaceutical intermediates. The molecule reacts efficiently with acylating agents, which opens a path to esters valued for both research and industrial product streams. In my bench work, the balance sits in controlling overreaction, as the electron-rich alkene can sometimes draw in reagents meant just for the alcohol part, making selectivity as much about craft as science.
Synonyms & Product Names
Chemists prefer alternate names such as methallyl alcohol or 2-methyl-2-propen-1-ol. Literature from different corners of the world references it under a handful of identifiers, which sometimes prompts confusion in multi-jurisdictional shipments. CAS Registry lists it as 513-42-8. People in laboratory supply simply call it "methallyl alcohol," usually enough to keep orders and paperwork unambiguous. Over the years, I've seen trade catalogs pile up synonyms, but the chemistry, in the end, doesn’t change—a compact piece of reactivity, by any name.
Safety & Operational Standards
Guidelines for 2-methylallyl alcohol parallel those for other volatile, flammable liquids. While working with even a moderate quantity, good ventilation is non-negotiable, and the routine calls for eye protection, gloves, and temperate handling to avoid vapor buildup. The compound irritates skin and mucous membranes. Lab teams train to contain and neutralize small spills, keeping sand or absorbent material within arm’s reach. Fire extinguishers must be ready at hand since neglecting the flammability hazard courts real risk. I've seen supervisors drill new students on vapor density and flashpoints, and the lessons tend to stick: one carelessly opened container can set off alarms faster than any safety demo could warn.
Application Area
Manufacturers and researchers work 2-methylallyl alcohol into a surprising number of downstream products. The molecule often serves as an intermediate in the synthesis of perfumes, agrochemical ingredients, and pharmaceutical intermediates. People developing specialty polymers or cross-linking agents turn to its alkene-alcohol framework to introduce unique branching or reactivity into their products. In fine chemical synthesis, this alcohol finds favor because its structure lets chemists tuck reactive handles into larger, more complicated molecules—a luxury that expands both method development and production pipelines. Those processes require careful control at every step, as the double bond sometimes invites unwanted side reactions.
Research & Development
Over the past decade, academic groups and specialty chemical firms have published thousands of reactions involving 2-methylallyl alcohol, each exploring new ways to expand its value. Topics range from its use as a precursor for complex ring systems, to tweaking the alcohol and alkene groups for pharmaceutical candidates. The alcohol often serves as a model compound for studying regioselectivity, particularly in cases where adjacent functional groups battle for priority. I’ve seen graduate students struggle and succeed by using this molecule to hone their understanding of organic synthesis, especially in asymmetric transformations and transition-metal catalyzed couplings. These experiences reveal just how much subtlety hides in a structure that seems simple at a glance.
Toxicity Research
Toxicological studies on 2-methylallyl alcohol paint a clear picture of a material that demands respect, if not outright caution. Direct contact irritates skin, eyes, and respiratory tracts. Inhalation of high concentrations triggers dizziness, headaches, or more severe respiratory symptoms; even low-level, repeated exposure should be avoided. Agencies in North America and Europe catalog it as an irritant, and workplace standards regulate air concentrations to minimize long-term risk. Chronic toxicity data remains less abundant, and animal studies suggest possible hepatic and renal impact at sustained high doses. As always, minimizing exposure, careful handling, and immediate cleanup of spills stand as clear rules in any lab or pilot plant, echoing lessons learned over generations.
Future Prospects
Current directions for 2-methylallyl alcohol point toward more selective synthetic transformations and greener, less wasteful manufacturing. As chemical industries chase reduced emissions and circular economy goals, interest grows in finding routes to produce this and similar molecules from renewable feedstocks. Process engineers search for catalysts that cut energy use and raise selectivity, helping shrink the environmental footprint tied to specialty alcohol production. Research teams also explore tailored modifications of 2-methylallyl alcohol to open new paths in drug synthesis or materials science, particularly in the hunt for alternatives to less environmentally friendly building blocks. The molecule’s track record as a flexible, reliable intermediate means it isn’t set to disappear soon, even as chemists try to squeeze more performance, safety, and sustainability from every step.
What is 2-Methylallyl Alcohol used for?
What Sets 2-Methylallyl Alcohol Apart
2-Methylallyl alcohol shows up as a small, colorless liquid with a sharp, slightly sweet odor. The name alone might sound like it belongs in some obscure research paper, but this chemical finds its way into more corners of modern life than most people realize. The unique structure—a three-carbon chain with an alcohol group and a methyl branch—gives it a reactive edge that sparks the interest of researchers, manufacturers, and tinkerers alike.
From Laboratory Benches to Production Floors
Its most well-known use sprouts in the lab. Organic chemists value 2-methylallyl alcohol as a building block—something to snap together with other pieces on the way to larger, more complex molecules. Due to its reactivity, synthesizing specialized compounds such as pharmaceuticals and flavoring agents gets a lot smoother. I’ve seen it pop up in reaction schemes during pharmaceutical projects, especially when creating chemical scaffolds—those backbones new medicines get built around.
Production of certain pesticides and agricultural chemicals relies on 2-methylallyl alcohol too. Its reactive backbone can anchor chemical changes, letting chemists tweak molecules to become more effective for crop protection. In my experience talking with agriculture researchers, these modifications often lead to safer, more targeted solutions for farmers and growers.
Flavor and Fragrance: Inspiration from Simplicity
Fragrance and flavor experts also value 2-methylallyl alcohol. Starting with this alcohol, synthesis teams can branch off into esters—compounds that infuse soaps, lotions, and even processed foods with scent and taste. Many of the subtle, bright notes in scented products depend on well-designed building blocks from the lab. The small size and manageable behavior of 2-methylallyl alcohol make it particularly suited for these controlled, creative transformations.
Safety, Regulation, and Handling Concerns
People working around 2-methylallyl alcohol quickly learn that it carries health and safety risks. Vapors irritate eyes, skin, and lungs, so regulation keeps tight reins on its sale, storage, and use. The European Chemicals Agency and the U.S. Environmental Protection Agency require transparent reporting for production, exposure, and transport data. Good laboratory practices—wearing gloves and goggles, using fume hoods—come standard. I remember my own first experience handling this chemical, and the lessons in ventilation and containment measures have stuck with me since.
Seeking Better Alternatives and Cleaner Synthesis
Some folks in the green chemistry field aim for alternatives that cut down on toxicity while offering similar chemical reactivity. The focus here isn't just making something new, but reducing environmental impact during the process. Exploring plant-based feedstocks or milder reaction conditions can shrink the overall environmental cost, and the push for bio-based ingredients continues to pick up speed in industry circles.
Why 2-Methylallyl Alcohol Matters
A single molecule like 2-methylallyl alcohol doesn’t dominate headlines, but its presence in pharmaceuticals, agriculture, and fragrance production keeps modern society moving. For chemists and product designers, it’s a tool to solve problems and invent new possibilities. Balancing its usefulness against health and environmental risks demands ongoing work, collaboration, and responsible innovation. The path forward keeps evolving alongside chemistry’s reach, and responsible handling makes sure that both workers and the wider world stay protected.
What are the safety precautions when handling 2-Methylallyl Alcohol?
Understanding 2-Methylallyl Alcohol
2-Methylallyl alcohol comes up in some chemical labs and factories. You’ll find it under names like 3-buten-2-ol or methallyl alcohol. Labs use it for organic syntheses, and it has a sweetish, sharp smell. The real story here: 2-methylallyl alcohol isn’t something to splash around without thought. Health concerns demand respect, both for your sake and anyone nearby.
Risks That Matter
If you’ve worked in any halfway decent shop or lab, they make a habit of prepping folks for nasty splashes and breathing trouble. This alcohol irritates skin and eyes, and the fumes tug at your lungs. It can enter your body through contact or air. I learned quickly not to trust my nose because not every risky vapor announces itself. Reports and data from the National Institute for Occupational Safety and Health show that repeated exposure burns the skin, inflames eyes, and can damage the nervous system. Long-term effects kick in after careless handling, and that isn’t a lesson anyone wants to learn by accident.
Proper Gear Is Critical
Goggles and nitrile gloves sit on my workbench at all times for a reason. Chemical splash goggles keep droplets away from your eyes, while thin, cheap gloves don’t make the cut. In hot labs, vapors want to creep around, so chemical fume hoods stay in use through every step—spraying, mixing, pouring. If the hood doesn’t draw, nobody stays in the room. It helps to use lab coats with cuffs to avoid droplets running down and soaking wrists, plus closed shoes—no sandals, ever.
Storing It Tight
Leaving 2-methylallyl alcohol in a flimsy container or somewhere warm spells disaster. I keep all volatile alcohols inside dedicated flammable storage cabinets. The right polyethylene containers with tight-fitting caps cut down spill risks and block fumes. Room temperature must stay cool, no direct sunlight. Experience taught me to make a habit of checking labels and lids—especially before and after use—because one mistake can ruin more than just a lab coat.
What To Do If Something Goes Wrong
Quick response saves pain and sometimes careers. If a spill touches skin, rinse with water for a good fifteen minutes. Eyes exposed need an eyewash station straight away, not just a splash from a cup. Inhaling fumes sometimes leaves a bad cough or dizziness; get fresh air fast, and if symptoms linger, don’t tough it out—see a doctor. I always keep spill kits handy, with absorbent pads and plastic tubs ready. Disposal happens with approved hazardous waste bins, not tossed out with common trash. This keeps the people who haul trash safe, too.
Building a Culture of Safety
Nobody works in a vacuum. Keeping others in the loop about which chemicals you use makes a difference. In my best work groups, we go over safety data sheets before anyone brings out new bottles. Training routines keep old habits sharp, whether newbies or veterans. Sharing lessons and swapping tips is key. I remember a colleague who blew a fume hood breaker, leaving vapors to escape—everyone learned from that scare, and now, we check electrical circuits as part of our routine.
Real Solutions for Everyday Work
Rules shouldn’t sit unused in dusty binders. Clear labeling, updated data sheets, and regular safety checks turn safety into habit. An eyewash and shower within dashing distance give real peace of mind. Smart companies reward teams for keeping a clean safety record and keep training real with hands-on practice.
Treating 2-methylallyl alcohol with care isn’t about following orders—it’s about coming home healthy. Each step protects you and those who work beside you. With simple preparation and the right mindset, risks drop, and confidence grows.
What is the chemical structure and formula of 2-Methylallyl Alcohol?
A Closer Look at the Chemistry
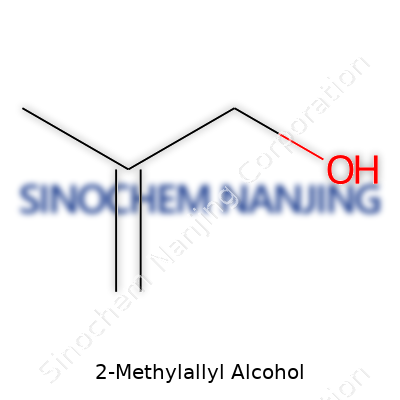
2-Methylallyl alcohol, also called 3-buten-2-ol, offers a simple yet intriguing structure. The formula is C4H8O. Breaking it down, this molecule has four carbon atoms, eight hydrogens, and one oxygen. The name hints at its design—a methyl group attached next to an allylic alcohol. In straightforward terms, this creates a parent carbon chain with a double bond and a hydroxyl (–OH) group.
To picture 2-methylallyl alcohol, focus on its backbone: CH2=C(CH3)CH2OH. Here’s how it lines up: the double bond links the first and second carbons, a methyl group branches off the second carbon, and the alcohol group sits at the end. Organic chemists recognize this as an unsaturated alcohol, with both an alkene and an alcohol group on show.
What Sets This Molecule Apart
The dual presence of both unsaturation (that double bond) and the alcohol group gives this molecule flexibility. It reacts easily in various organic transformations, making it useful for researchers and manufacturers alike. Such a structure invites hydrophilic and hydrophobic interactions, setting the stage for reactivity that can create new building blocks. These features help companies create everything from flavorings to pharmaceutical agents.
Practical Significance
Working in the lab, I noticed that 2-methylallyl alcohol’s structure makes it suitable for reactions such as esterification and epoxidation. Its double bond attracts chemists hunting for starting points in the synthesis of complex molecules. For those producing specialty chemicals, its versatility offers freedom: turn the alcohol into esters, or engage the double bond in addition reactions. This adaptability encourages innovation in labs focusing on materials science and fine chemicals.
This kind of alcohol lands between simple alcohols like ethanol and more complex branched structures. Instead of becoming a bench staple, it helps carve out specialty pathways. Medical researchers rely on intermediate compounds like this for synthesizing active pharmaceutical ingredients. Flavors or fragrances developers appreciate its ability to serve as a foundation for new aroma compounds. Its combination of double bond and an alcohol group doesn’t show up in every molecule, so it deserves attention from those working on unique projects.
Addressing Safety and Handling
The balance of alcohol and unsaturation brings rewards and risks. 2-Methylallyl alcohol can irritate the skin and eyes on contact, requiring the right gear in every workplace. It evaporates fast, so labs need good ventilation. Even though the molecule looks plain on paper, it serves as a reminder to stay alert. As regulations tighten around chemical safety, users have to keep safety training updated and storage areas organized.
Paths Toward Responsible Use
Sustainable chemistry asks us to reconsider how molecules are made and handled. Green synthesis techniques push towards safer, less wasteful production. In my experience, researchers build better systems by switching to less toxic reagents and improving reaction yields. This molecule’s reactivity also brings challenges for waste disposal. Strict procedures for neutralizing or recycling excess chemicals cut costs and environmental impact, while ensuring everyone in the lab stays safe.
2-Methylallyl alcohol doesn’t always headline major discoveries, but its clear structure and flexible chemistry put it in the toolkit of innovators across many fields. By respecting its properties and using smart lab practices, researchers and industry experts can keep unlocking new opportunities with this small but valuable molecule.
How should 2-Methylallyl Alcohol be stored?
Why Handling 2-Methylallyl Alcohol Demands Respect
2-Methylallyl alcohol isn’t your average household chemical. It has a pungent, sweet odor and flammable properties that put it in the same category as other high-risk organic solvents. In labs and factories, it’s often used for making pharmaceuticals, perfumes, and specialty chemicals. This substance can irritate eyes, skin, and lungs, and doesn’t forgive mistakes. Every slip-up has the potential to create a real mess—sometimes one that leads to serious health hazards or fires.
The Right Environment for Storage
Years of storing aggressive solvents taught me that most chemical accidents start with carelessness. 2-Methylallyl alcohol reacts badly to heat, sparks, cigarettes, and open flames. A fire-resistant storage room, tucked away from busy workspaces, makes a solid starting point. I always check storage temperature stays below 25°C, well out of reach of any heating vents or sunlight. The substance belongs in tightly sealed, labeled steel or glass containers. Plastic might leach or crack over time, so I avoid it unless the manufacturer says otherwise.
Moisture and Air: Two Hidden Enemies
Even if flammability draws the biggest warning symbols, humidity and oxygen quietly tear down container integrity from within. I’ve seen containers swell, leak, and even burst when air found its way in. Keeping containers tightly closed and vented only in chemical fume hoods solves much of this trouble. Silica gel desiccants tossed into storage cabinets pull away sneaky moisture. Nitrogen atmosphere storage helps too, since it chokes out reactive oxygen.
The Real Dangers Lurking on the Shelf
Spilled alcohol vaporizes fast, filling a room or storage cabinet before anyone knows it. Vapor builds up and just one static spark sets off a fireball. That’s why I keep grounding straps on metal containers. No rags, cardboard, or combustible trash ever share the same cabinet. Company policy bans food and drinks in chemical storage too—just seeing a spilled sandwich near these chemicals makes me cringe.
Training Makes All the Difference
Labeling seems like a small step, but I remember times labels wore off and caused confusion no one saw coming. Clear hazard signs, storage dates, and simple instructions make a huge difference, especially when new staff rotate in. Regular inspections catch rusty caps or leaky stoppers before they make trouble. Emergency showers, eye-wash stations, and spill kits sit close by. Practice drills covering all storage chemicals smooth over panic in real emergencies.
Don’t Ignore the Rules
Laws don’t exist just to complicate life. The fire marshal showed me how even short-term shortcutting the rules can create domino effects—violations can lead to closed labs or big fines, even injuries. OSHA guidelines recommend explosion-proof refrigerators for temperature-sensitive storage. Separate acids, oxidizers, and flammable liquids. Good records give you a fighting chance when safety audits or surprise inspections roll around.
Solutions for Safer Storage
Simple habits and regular checks serve everyone better than over-complicated systems. I recommend only storing the amount needed soon, returning unused portions immediately, and logging every in-and-out container. That means less clutter and fresher stocks on the shelf. Automatic chemical inventory systems help reduce human error. If a container looks off-color or bulging, treat it as an emergency and alert the right team. These steps may slow things down but protect people, property, and peace of mind.
What are the potential health hazards of 2-Methylallyl Alcohol exposure?
What Happens During Exposure
2-Methylallyl Alcohol, sometimes called prenol, often pops up in labs and factories where synthetic flavors, fragrances, and resins get produced. Many workers don’t know much about it, and most folks never talk about the hazards unless something goes wrong. I’ve spent time walking plant floors and talking to safety officers. From those conversations, one thing stands out: Chemical safety rarely gets the same attention as physical hazards like moving machinery. The effects of breathing in or getting this stuff on your skin can sneak up slowly or hit fast, depending on how exposed you are.
How the Body Responds
Short contact with 2-Methylallyl Alcohol can bring burning eyes, sore throats, coughing, and chest pain. It doesn’t take much to get a headache or feel dizzy. If the liquid hits your skin, redness and itching start up quickly. The real trouble kicks in with repeated or high exposure. Over time, the liver and kidneys can feel the brunt of the damage, and lungs might start working less efficiently. Cases show people who handle large doses without the right protection can lose coordination and develop more serious nervous system problems.
What Hard Data Shows
Reports from the National Institute for Occupational Safety and Health (NIOSH) caution that only a small amount—just a few parts per million in the air—can trigger symptoms. Animal testing reveals just how potent this alcohol is in damaging organ tissues and messing with normal breathing. There’s also worry about cancer risk, since any chemical that stresses cells in the liver for long stretches raises suspicion. The EPA considers the risk high enough that they require strict tracking of workplace levels.
Experience From the Field
I’ve seen factories where safety glasses and gloves sit unused even as fumes float around. Many workers blame headaches or rashes on stress or dry air instead of the chemicals they handle every shift. It’s not always a matter of negligence; sometimes the labels don’t spell out enough warning, and safety information arrives piecemeal. I’ve watched safety teams scramble during inspections or after a co-worker landed in urgent care with breathing problems. These moments lead to better habits for a while, but without steady reminders, old routines come back. Long-term health checks still seem rare, even in industries where chemical splashes and vapor clouds happen daily.
How to Cut Down the Risks
Basic steps help a lot. Good ventilation, accurate air monitors, and regular training make a real difference. It helps to keep the material stored in well-sealed containers. Training doesn’t mean another boring PowerPoint session; hands-on demonstrations stick better. If bosses set up health screenings and encourage honest reporting of symptoms, problems get caught early. I've seen big improvements in companies that offer quick access to wash stations and hang clear warning signs at every point where the chemical gets used.
The Importance of Awareness
Moving forward, knowledge needs to keep pace with production. Awareness campaigns and well-placed information go a long way. Everyone—from line workers to supervisors—benefits when people know what they’re dealing with and how to protect themselves. Real improvements come from listening to workers on the ground, not just ticking boxes on a compliance sheet.
| Names | |
| Preferred IUPAC name | 3-Methylbut-2-en-1-ol |
| Other names |
Methallyl alcohol 3-Buten-2-ol Isocrotyl alcohol 2-Methyl-2-propen-1-ol |
| Pronunciation | /tuːˈmɛθɪlˌælaɪl ˈæl.kə.hɒl/ |
| Identifiers | |
| CAS Number | 115-18-4 |
| Beilstein Reference | 0418737 |
| ChEBI | CHEBI:52970 |
| ChEMBL | CHEMBL15700 |
| ChemSpider | 16284 |
| DrugBank | DB04537 |
| ECHA InfoCard | 100.018.898 |
| EC Number | 200-922-6 |
| Gmelin Reference | 186491 |
| KEGG | C06166 |
| MeSH | D004528 |
| PubChem CID | 6566 |
| RTECS number | RR0350000 |
| UNII | W420LQ60A9 |
| UN number | UN2054 |
| Properties | |
| Chemical formula | C4H8O |
| Molar mass | 86.13 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Pungent |
| Density | 0.857 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.49 |
| Vapor pressure | 3.5 mmHg (20°C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 15.20 |
| Magnetic susceptibility (χ) | -6.53 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.429 |
| Viscosity | 1.77 mPa·s (20 °C) |
| Dipole moment | 1.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 318.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -48.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2222 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H312, H315, H319, H332 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0-ALC |
| Flash point | 46 °C |
| Autoignition temperature | 220 °C |
| Explosive limits | 1.9–11.2% |
| Lethal dose or concentration | LD50 oral rat 620 mg/kg |
| LD50 (median dose) | LD50 (median dose): 790 mg/kg (rat, oral) |
| NIOSH | NIOSH = "AS0425000 |
| PEL (Permissible) | PEL = "No OSHA PEL assigned |
| REL (Recommended) | 2 ppm |
| IDLH (Immediate danger) | 60 ppm |
| Related compounds | |
| Related compounds |
3-Buten-2-ol Crotyl alcohol Allyl alcohol Methallyl chloride |

