Looking at 2-Methyl-4,6-Dinitrophenol: Insights From Laboratory to the World
Historical Development
The story of 2-Methyl-4,6-Dinitrophenol unfolds from an age when organic chemistry began finding its feet in both industry and medicine. My own research pulled me into archives describing early synthetic efforts. Driven by curiosity about ring nitration and phenolic chemistry, chemists of the nineteenth and twentieth centuries shaped the landscape that gave rise to compounds like this. Academic papers trace its synthesis back to the era when scientists explored the possibilities of simple aromatic nitro derivatives. As industry grew, so did the urge to create dye intermediates and potential pharmaceuticals. Interest picked up through its resemblance to dinitrophenol, known then for use in manufacturing and regrettably in misguided weight loss efforts. That history reminds me just how quickly science leaps from laboratory discovery to real-world impact, sometimes skipping safety on the way. What stands out is the way chemists learned by trial and error, binding experience to each new structural variant they investigated.
Product Overview
2-Methyl-4,6-Dinitrophenol remains a specialty chemical with a distinct yellow hue. Its structure builds on a phenol ring carrying a methyl group and two nitro groups, giving it unique reactivity. You don’t run across it on a grocery shelf; it’s more the kind of compound handling teams find in research labs, or in industrial settings focused on specialized synthesis. The nitrophenol backbone has attracted both opportunity and caution, drawing attention for use in screening, staining, or as a chemical intermediate—and on rare occasions, in the more niche world of explosives research and analytical testing. Its legacy as a tool in organic synthesis continues, as experimental chemists keep returning to its reliable behavior as a building block.
Physical & Chemical Properties
If you’ve ever worked with nitrophenols, you know the powdery, often crystalline appearance and the bold colors these compounds bring. 2-Methyl-4,6-Dinitrophenol stands out with that deep yellow, a property used in early dye work. On the bench I’ve noticed how nitro-aromatic compounds carry a distinct, almost earthy odor. Melting points are telling: these chemicals demand clean technique since small impurities can shift those numbers quickly. The molecule dissolves in organic solvents like ethanol and ether without much fuss, but in water, that nitro-heavy backbone slows things down. The placement of methyl and nitro groups can lead to underappreciated shifts in reactivity: oxidative reactions go differently than with its better-known cousin, 2,4-Dinitrophenol. Human error often comes from underestimating the volatility and sensitivity those extra groups introduce. Handling demands a healthy respect and a steady hand.
Technical Specifications & Labeling
Labeling for compounds of this kind often brings out the importance of clarity—mix-ups can have serious consequences. In my own lab, I’ve seen how vital it is to carry accurate labels specifying concentration, hazards, and distinct chemical names. Even a minor spill of such compounds triggers immediate attention due to their nitro content and potential volatility. Storage guidance usually demands cool, dry places and airtight bottles, away from acids or reducing agents. Always, the bright yellow stands as its own warning sign—visual cues save time and lives far more often than they get credit for, making strict technical labeling a front-line defense in chemical safety.
Preparation Method
Coming up with 2-Methyl-4,6-Dinitrophenol means starting from a methylphenol and subjecting it to directed nitration. Most synthetic chemists learn early that putting nitro groups on an aromatic ring isn’t just about pouring acid; it takes finesse. Control of temperature, acid ratios, and reaction time can mean the difference between a clean product and a brown, unusable tar. I once observed a graduate student ruin a whole batch through a rushed procedure—nitration chemistry punishes carelessness. Most labs mix nitric and sulfuric acids, often under chilling conditions, because runaway exotherms spell disaster. Once the desired substitution happens, careful crystallization helps isolate the product. I learned the hard way that taking shortcuts with purification leaves stubborn byproducts.
Chemical Reactions & Modifications
2-Methyl-4,6-Dinitrophenol opens up a toolkit of possible reactions, most of them driven by the activating and deactivating effects of the methyl and nitro groups. Its phenolic hydrogen takes part in esterification or etherification, but the nitro functions steal the show in reduction or nucleophilic substitution. Catalytic hydrogenation yields aminophenols, which serve a completely different set of uses. Attempts to further modify this molecule require strict controls—some transformations can trigger dangerous decomposition. My early work in this area showed that slight procedural variations produce vastly different results, underlining why reproducibility matters in research.
Synonyms & Product Names
Like many phenolic derivatives, 2-Methyl-4,6-Dinitrophenol shows up under several names. Researchers switching between trade, IUPAC, and common names easily lose track. In scientific papers and chemical catalogs, I’ve found synonyms such as 2-methyl-dinitrocresol or cresyldinitrophenol. Such variety causes confusion unless supported by rigorous labeling in storage and documentation. Nobody in a busy lab wants to discover too late that their stack of naming conventions led to a costly or dangerous mix-up—precision in names remains as important as precision on the balance.
Safety & Operational Standards
Among organic nitro compounds, few deserve more respect than 2-Methyl-4,6-Dinitrophenol. Its systemic toxicity is no secret among seasoned researchers. I remember my first safety training, where the instructor recounted cases of phenol burns and inhalation risks. Standard operating procedures call for gloves and full goggles, and even the experienced chemists double-check ventilation. Many incidents in chemistry stem from a failure to appreciate cumulative exposure—handling nitro groups without constant vigilance rarely ends well. Chemical hygiene practices make the difference: periodic air monitoring and careful waste segregation reduce risk to both people and the environment. Emergencies remind us there’s no substitute for preparedness.
Application Area
By reputation, 2-Methyl-4,6-Dinitrophenol finds few applications compared with other similar molecules, but its impact in those areas counts. Most notably, industrial labs rely on it as an intermediate for synthesizing more complex molecules—especially certain dyes and pesticides. Its role as a tool for analytical chemistry and as a substrate in research settings does not slip under the radar. Over the years, I saw several institutions employ it as a test chemical for enzymatic studies or as a marker in chemical kinetics. Environmental and pharmaceutical research continues to apply such molecules to unravel biological transformation pathways or assess degradation under field conditions. My colleagues’ work in pollutant tracing highlights the lasting relevance of such compounds despite limited direct consumer use.
Research & Development
Research involving 2-Methyl-4,6-Dinitrophenol has shifted over time. Early projects stressed new synthetic pathways, while modern efforts dig into selective reduction and environmental fate. Colleagues in environmental chemistry have looked at its role as a breakdown product or surrogate in modeling studies of more hazardous nitrophenols. Methods development has improved, with greener chemistry getting a bigger push lately as labs weigh process yields against health and planetary concerns. Researchers continue developing sensors and detection techniques due to the low thresholds at which these compounds impact health and safety. Regulatory shifts, alongside academic curiosity, drive deeper investigation into long-term stability, interactions with soil and water, and implications for remediation technologies. Lab budgets rarely stretch far, so partnerships and interdisciplinary projects keep this field moving forward.
Toxicity Research
Toxicity profiles for nitrophenic compounds are not kind. 2-Methyl-4,6-Dinitrophenol, in particular, brings recognized dangers—its resemblance to notorious dinitrophenol gave me pause the first time I encountered it. Ingestion, inhalation, or skin contact can all lead to acute health risks. The mechanisms of toxicity relate to its interference with mitochondrial function, a trait common to this family. I have seen toxicologists document incidents where improper protective measures led to hospital visits, usually marked by skin burns and respiratory distress. Chronic exposure studies uncover potential for organ damage, and accidental environmental releases lead to persistent soil and water contamination. These findings fuel calls for better risk assessment, mandatory training, and the kind of transparency that allows responding to incidents in real time.
Future Prospects
The future for 2-Methyl-4,6-Dinitrophenol depends on new approaches to chemical risk and sustainable production. Ongoing research leans into greener reagents and less hazardous substitutes, but classic nitro-aromatics won’t vanish overnight. Cleaning up legacy contamination and narrowing the scope for accidental misuse stand as immediate challenges. I hope to see more advances in in-situ remediation and alternative uses for these compounds that are safer and cleaner without losing functional value. Real progress will come from combining robust safety regulations, transparent reporting, and cooperative scientific research to close knowledge gaps about long-term impacts on health and the environment. My experience points toward the need for stronger links between industrial practice and academic discovery, making room for safer technology while respecting the lessons those bright yellow crystals have already taught the field.
What are the primary uses of 2-Methyl-4,6-Dinitrophenol?
Understanding the Chemical
2-Methyl-4,6-dinitrophenol isn’t something most people encounter in daily life, but it plays a part in industries that affect people all over the world. This yellowish compound shows up in small vials in labs and factories. From the formula alone, you can sense its punch: nitro groups, plus a methyl group, all hooked to a phenol ring. That's the structure that gives this compound its strong effects.
Industrial Applications
Across the chemical manufacturing field, 2-Methyl-4,6-dinitrophenol is used to make dyes and pigments. That yellow on synthetic fabrics, the bright tints in plastics, or some printing inks—they often owe their color intensity to compounds like this one. Textile production, in particular, relies on these nitrophenol derivatives. Their stable colorfastness gives clothing and upholstery bright, long-lasting hues.
Pharmaceutical synthesis, though not a household-word application, often requires unique starting compounds to build more complicated molecules. Chemists turn to 2-Methyl-4,6-dinitrophenol as an intermediate. By attaching or removing small chemical groups, they create medication components or crop protection agents.
Pesticide and Explosive Roles
If there’s trouble with bugs or fungi in crops, some older pesticide recipes contain dinitrophenol compounds because they disrupt metabolic processes in pests. Before regulations tightened, farms favored these chemicals for quick results. Risks from exposure, both for farmworkers and the environment, led to a sharp drop in use. Some countries still report traces, especially where local restrictions do not fully cover these older substances.
In the field of explosives, the nitro groups make this compound energetic. Manufacturers mix or modify these chemicals into detonators or propellants. Safety around nitrated organics always demands attention. Explosives based on dinitrophenol structures offer reliable ignition for mining, demolition, or even specialized military operations. Handling, transport, and storage get regulated through national security laws and environmental protection agencies to limit accidental release or misuse.
Environmental and Health Considerations
Workers exposed to dinitrophenol chemicals must handle them with care. Inhaling dust or direct skin contact can set off headaches, skin irritation, or in serious cases, toxic reactions in the liver or nervous system. I recall reading safety data in a plant: gloves, masks, and full ventilation were essential in all handling and packaging steps. Regulators insist on limit thresholds, and regular monitoring reduces long-term harm.
In the wider environment, runoff or leaks from factories introduce these chemicals into water and soil. Aquatic life shows sensitivity at low doses, and bioaccumulation threatens predators further up the food chain. Technologies now focus on containment—closed systems, strict waste processing, and routine site audits help keep accidental releases in check.
Improving Responsibility
Greater transparency around chemical sourcing, traceability, and safe end-of-life disposal gives people more faith in how these industries operate. Consumers and watchdog groups call for newer dye molecules with less toxicity, encouraging research into plant-based or biodegradable alternatives. In the meantime, constant review of protocols and risk assessments helps factories keep exposure to a minimum.
Teaching workers how to spot symptoms and respond to spills, while updating gear and techniques, brings health and safety right to the shop floor. As these practices spread, the industries that depend on strong colorants or energetic materials can keep pushing forward without costing people or ecosystems too much.
Is 2-Methyl-4,6-Dinitrophenol hazardous to human health?
Everyday Exposure Isn't Harmless
Running into complicated names like 2-Methyl-4,6-Dinitrophenol tends to make most folks’ eyes glaze over. Still, this stuff matters. Used in lab research, explosives, and dyes, 2-Methyl-4,6-Dinitrophenol carries a heavy health risk if not handled with care. It’s not your average cleaning agent or food additive. Its toxic nature is well-established—just a small amount can spell real trouble.
What Happens Inside the Body?
Chemicals like this one disrupt the way cells create and use energy. It interferes with the body’s own “battery,” called mitochondria. The result isn’t pretty. Overheating, rapid breathing, confusion—symptoms spiral quickly for people who come in direct contact or inhale dust. Just look at dinitrophenol’s history: its close relatives have led to deaths in weight-loss crazes and industrial misuses.
I’ve seen seasoned lab workers double-check their protective gear when they even hear mention of phenol compounds like this one. And for good reason. The body has very little defense against accidental absorption or inhalation. Sweating, sudden fever, even organ failure—risk ramps up, not down, the longer it lingers in your system.
Science Backs Up the Concern
Research doesn’t sugarcoat the threat. 2-Methyl-4,6-Dinitrophenol has shown severe toxicity in animal studies, with rapid onset of poisoning and low thresholds for harmful effects. Regulators classify it as an environmental hazard partly because it breaks down slowly; once released, it sticks around and keeps causing harm—not just to people, but to aquatic life, soil microbes, and any ecosystem downwind.
No Room for Carelessness at Work or Home
Leaving dangerous chemicals accessible, even in a garage or storage room, isn’t just careless—it can be deadly. A quick spill or a careless dust-up means risk for pets, kids, or even neighbors. Stories of accidental poisoning don’t just come from factories or labs—they happen in homes, too, especially where chemicals are stored near food or water supplies.
How Policy Can Step Up
To address risks, honest-to-goodness policy changes matter. Access should stay tight. Only trained pros need a reason to handle or store 2-Methyl-4,6-Dinitrophenol. Lawmakers did right by clamping down on similar substances after reports of misuse and poisonings. Consumer access should get the same hard look, with better labeling requirements and mandatory education for anyone buying or using these substances.
Available evidence underscores why protective equipment isn't just a suggestion, but a requirement. Ventilation, gloves, and respirators belong in every workspace where this chemical could show up. Also, quick-response plans need real practice, not just paperwork. Workers benefit from knowing what action steps to take if accidental exposure occurs.
Protecting Health and the Environment
Chemicals with a reputation for harming people and the environment deserve real respect—never casual handling. Cleaning up spills, keeping detailed records, and testing local water or air make a big difference, especially near schools or residential areas. Clear public information goes further than technical jargon. Families expect transparency about pollution or potential exposure from factories or waste facilities nearby.
Protecting health starts with straightforward action and clear communication. The threat from 2-Methyl-4,6-Dinitrophenol isn’t just a theory—it’s proven, and no one can afford to look the other way.
How should 2-Methyl-4,6-Dinitrophenol be safely stored and handled?
Understanding the Risks
Experience in the lab has taught me that some chemicals need more respect than others. 2-Methyl-4,6-Dinitrophenol (often called 2,4,6-DNP or Dinitrocresol) is one of those substances you don’t forget after the first encounter. This yellow crystalline solid is highly toxic, both through inhalation and skin contact. It doesn’t just pose short-term acute symptoms – repeated exposure can hit the nervous system, liver, and kidneys. In my years working in research, stories circulate about careless practices leading to disaster. Every bottle, every gram, expects vigilance.
Choosing the Right Storage
Throwing 2,4,6-DNP on any shelf in a storeroom increases the odds of an accident. Its sensitivity to heat, friction, and shock means temperature control isn’t a suggestion—it’s a demand. I’ve worked in labs where specialized flame-proof cabinets make all the difference. Storing DNP means using containers that seal tightly, keeping out moisture and minimizing volatility. Labels with clear hazard icons help new staff avoid mistakes. There’s no cutting corners—each jar or drum demands its own secondary containment tray, because even a minor spill can turn routine cleanup into a medical emergency.
Staying on Top of Ventilation
Poor air circulation stacks risk. Small spills release fumes that linger for days in stale air, even long after a cleanup. I once helped design a storage room with a dedicated exhaust and negative pressure system just for chemicals like DNP. Opening a container only inside a fume hood, never on an open countertop, adds a layer of safety that matters—a lesson often learned the hard way.
Personal Protection: No Excuses
Goggles, gloves, lab coats—this is the unglamorous uniform needed around DNP. Some colleagues get tempted to swap nitrile for latex gloves, thinking it’s just another chemical, yet latex breaks down faster when handling harsh organics. Nitrile offers better resistance, and double-gloving adds peace of mind. A splash or accidental dusting on bare skin can be extremely dangerous, not just for you, but for anyone who crosses your path afterward. Closed shoes, long sleeves, and strict “no eating or drinking” policies inside work zones become second nature fast, often enforced by the memory of old accidents.
Dealing with Spills and Waste
Spills never seem to happen at a convenient time. Absorbent pads, neutralizing agents, and specialized disposal bins need to be at arm’s reach. Training regularly on quick clean-up means less panic in the real event. Any contaminated item—say, a paper towel or even a chunk of bench liner—turns into hazardous waste. Each piece waits in a clearly labeled drum for a professional hazardous waste pickup. Unlike some compounds, pouring anything with DNP down the drain or placing it in communal trash is simply reckless, inviting fines and endangering lives.
Training Should Never Stop
Most accidents with DNP start with a breakdown in communication or a forgotten detail. Refresher courses on handling, signage, and emergency drills wake up complacency. Sharing real-case stories, not just dry rules, drives home why safe handling can't rely on memory alone. Veteran staff mentoring new hires helps build good habits early. No one wants to be the reason for a headline accident.
Looking for Safer Alternatives
In some settings, DNP proves tough to replace, but whenever possible, finding a less hazardous chemical saves effort and risk. Decision-makers should weigh the payoff of safer substitutes against the real costs—financial, legal, and most importantly, human—of a single mishap.
What precautions are necessary when disposing of 2-Methyl-4,6-Dinitrophenol?
Why Getting Rid of This Chemical Matters
Anyone who’s spent time in a lab knows chemicals like 2-Methyl-4,6-Dinitrophenol don’t belong anywhere near a trash can or the drain. This isn’t just some rule made up by cautious managers. Years back, I saw firsthand what mishandling a nasty compound did to a workbench and almost to a colleague’s hand. That burned-in memory drives home why it’s not just about ticking boxes, but protecting yourself, your coworkers, and the environment.
What’s So Bad About 2-Methyl-4,6-Dinitrophenol?
People sometimes underestimate this yellow powder. On paper, it looks like just another aromatic nitro compound. At the bench, its toxicity jumps out. Studies show it cranks up metabolic rates and damages internal organs with just small exposures. Its dust irritates eyes, skin, lungs, and even tiny spills bring dangerous risk. Over time, even low-level exposure builds up—think cumulative damage, not just accidents.
Research out of industrial safety publications backs this up. Folks exposed to similar compounds have ended up with nervous system issues and organ failure. Stories like those convince me to treat this with the same respect as corrosives and strong oxidizers.
Proper Disposal: Steps That Keep People Safe
Sending 2-Methyl-4,6-Dinitrophenol down the drain or mixing it with general waste sets everyone up for trouble. Instead, I always start by reading the Safety Data Sheet. That means gloves, goggles, and a real lab coat every time. Keeping it in sealed, labeled bottles made from glass or compatible plastics matters—nitro compounds sometimes chew through the wrong materials.
After containerizing the waste, I bring in the waste management team. These professionals, equipped with respirators and protective gear, specialize in removing material that can’t go through regular municipal routes. The chemical heads to a licensed hazardous waste incinerator facility, where controlled, high-heat burning breaks it down without sending toxins or particles into the air. Reports from environmental audits show these sites have multiple scrubbers and filters to capture anything released.
Some university labs store their spent chemicals in secured rooms under constant supervision until pickup. I remember a time our inventory process flagged a missing bottle. That prompted a lockdown, review, and extra staff training. Incidents like that always reinforce clear tracking and strong communication.
How Regulations Play In
Government agencies, from the EPA in the US to the European Chemicals Agency, set firm ground rules on disposal. They issue fines when folks cut corners. These rules didn’t pop up overnight—they’re responses to things going wrong. Personally, I’d rather deal with some extra paperwork than environmental contamination or a medical emergency.
Community transparency also matters. Some facilities post online logs of hazardous chemical handling. Local residents have a right to know what happens near them—we owe that to them as part of ethical practice.
Better Habits Moving Forward
Minimizing use in daily lab work goes a long way. In research groups I’ve worked with, we always tracked how much hazardous material we ordered and aimed to keep surplus low. Keeping up with new disposal methods helps too; the field keeps developing less risky destruction or neutralization routes.
Teaching every new lab assistant about chemical risks, not as scary stories but as real hazards, makes the culture safer for everyone. No amount of paperwork or legal requirement teaches as well as stories paired with clear instructions and follow-up conversations.
What are the key physical and chemical properties of 2-Methyl-4,6-Dinitrophenol?
Looking Closer at Its Structure
2-Methyl-4,6-dinitrophenol, known to some by its shorthand Dinitro-o-cresol or DNOC, brings a punchy chemical profile. This yellow, powdery solid owes its intensity to two nitro groups locked onto a benzene ring, plus a lone methyl group hanging off the edge. Diagrams may look simple, but its molecular setup gives it an edge in reactivity and stability — two traits people in chemistry or manufacturing rarely take for granted.
Handling the Physical Side
From a lab bench perspective, this substance stands out. The yellow-orange crystals are unmistakable. It melts at around 87–89°C, which means regular room temperatures leave it as a solid. Slightly higher heat quickly brings it to a melt, something I’ve found sometimes complicates storage in older facilities with poor temperature control. Moisture doesn’t claim it easily, but it does dissolve modestly in hot water and ethanol. Even a faint whiff can tingle the nose, showing it doesn’t hide in plain sight.
In my experience, working with it without gloves is a risky move. Its dust moves in the air and can settle on skin, staining yellow. Folks in older industrial shops sometimes still talk about the “DNOC dust” on hands at the end of a shift. It sticks because the compound binds with natural oils. I learned to double-check for traces before heading home.
Chemical Reactivity and Safety Concerns
Chemically, DNOC lives up to its potential as an oxidizer, especially because of those nitro groups. Any source of ignition pushes it into dangerous territory. While mixing it with other organics or reducing agents, one slip can provoke a violent reaction. There’s a reason shops invest so heavily in proper containment and ventilation wherever this compound piles up.
Exposure isn’t just a matter of physical contact. Inhalation and ingestion form real risks, leading to headaches, nausea, even more severe symptoms from chronic poisoning. Cases from the 20th century documented health incidents from agricultural and pest control use before better training and protective rules cut down on accidents. DNOC blocks cellular respiration, which disrupts energy production inside cells. Anyone dealing with it keeps emergency wash stations within reach.
Why These Properties Matter
DNOC’s physical and chemical behavior set it apart from less-reactive phenols. It has powered everything from pesticides to industrial dyes. Anyone working with it can’t ignore its potential for harm, or its environmental impact. It hangs around in soil and water. Some research has linked it to nervous system damage and other chronic issues, especially in places where regulation or oversight still lags.
Mitigating these risks takes both real-world knowledge and attention to detail: sealed storage, consistent monitoring, proper personal protective equipment, and robust disposal methods. If regulatory agencies flag concerns, it’s for good reason. I’ve seen facilities invest heavily in upgraded storage and handling equipment because the penalties for ignoring safety can last a lifetime.
Moving Forward
Controlling exposure at every stage is key. Up-to-date labeling, better staff training, spill response planning, and clear guidelines reduce mishaps. Some groups push for biodegradable alternatives or less toxic options wherever possible, especially in farming. Staying current on regulations and hazards keeps people and environments safe. These aren’t abstract concerns — they come straight from shared experience on the lab floor and in the warehouse.
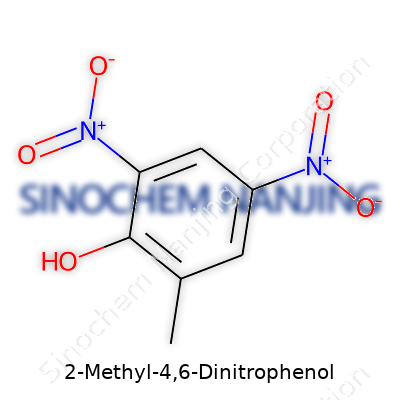
| Names | |
| Preferred IUPAC name | 2-methyl-4,6-dinitrophenol |
| Other names |
2-Methyl-4,6-dinitrophenol 2-Methyl DNOC DNOC-methyl 4,6-Dinitro-o-cresol DNOC Dinitro-o-cresol |
| Pronunciation | /tuː ˈmɛθɪl ˈdaɪnaɪtrəʊˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 88-85-7 |
| Beilstein Reference | '1742977' |
| ChEBI | CHEBI:34481 |
| ChEMBL | CHEMBL1277 |
| ChemSpider | 10372 |
| DrugBank | DB08210 |
| ECHA InfoCard | 100.007.679 |
| EC Number | 209-193-3 |
| Gmelin Reference | 89304 |
| KEGG | C02573 |
| MeSH | D008945 |
| PubChem CID | 14373 |
| RTECS number | SJ9625000 |
| UNII | 62X7618A6S |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C7H6N2O5 |
| Molar mass | 228.13 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odor: odorless |
| Density | 1.57 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.67 |
| Vapor pressure | 1.71E-6 mm Hg at 25°C |
| Acidity (pKa) | 4.0 |
| Basicity (pKb) | 10.89 |
| Magnetic susceptibility (χ) | -30.9·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.639 |
| Viscosity | 6.8 cP (50°C) |
| Dipole moment | 4.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 249.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −210.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2487 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H318, H332 |
| Precautionary statements | H302 + H332: Harmful if swallowed or if inhaled. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| NFPA 704 (fire diamond) | 2-2-4- (Note: The specific values can vary, but commonly for 2-Methyl-4,6-Dinitrophenol: Health=2, Flammability=2, Instability=4, Special=blank) |
| Flash point | 99.0 °C |
| Lethal dose or concentration | LD50 oral rat 340 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (oral, rat) |
| NIOSH | SA9100000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Methyl-4,6-Dinitrophenol: 0.2 mg/m3 (Skin) |
| REL (Recommended) | REL: 1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 mg/m3 |
| Related compounds | |
| Related compounds |
2,4-Dinitrophenol 2-Methyl-4,6-dinitroaniline 2,6-Dinitro-4-methylphenol Picric acid 4,6-Dinitro-o-cresol |

