An Editorial Commentary on 2-Methyl-3-Pentanone: Science, Safety, and Society
Looking Back: The Historical Thread
Chemists first explored the world of aliphatic ketones in the early years of the twentieth century. 2-Methyl-3-pentanone stands out as an early example of how simple changes to a carbon skeleton led to different chemical and physical behaviors. The compound’s path runs parallel to broader developments in synthetic organic chemistry, as new distillation and purification techniques opened up more routes to produce such molecules. Plant-derived alcohols gave way to industrial feedstock, and as manufacturing scaled up, this ketone found its way from research laboratories into industrial application, even if it never starred as a blockbuster commodity. Nobody carved a monument to it, but its modest contributions reveal bigger ideas about versatility in chemistry.
What 2-Methyl-3-Pentanone Really Is
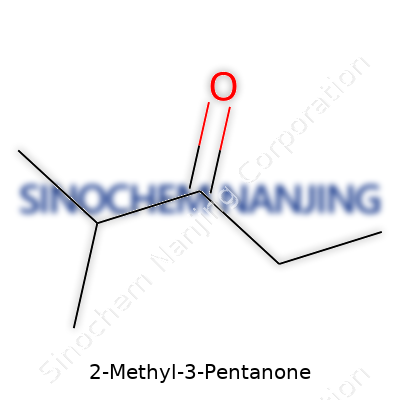
Ask a chemist to describe 2-Methyl-3-pentanone and you’ll get a straight answer: a six-carbon ketone where a methyl group branches from the third carbon. The molecular formula spells out C6H12O, and the IUPAC name frames its structure clearly. Compared to bulkier ketones, this one delivers a moderate volatility, noticeable odor, and a liquid state over a wide temperature range. Its odor won’t win awards—it lands somewhere between sweet and pungent. In the bottle, the colorless clarity makes it easy to mistake for other solvents, but it’s the branching that gives rise to special reactivity. Anyone who’s handled it in the lab knows that chemical properties flow from that arrangement of carbons as much as from the carbonyl group.
Diving into Physical and Chemical Nature
Physical data sheets put the boiling point of 2-Methyl-3-pentanone around 115 degrees Celsius. The density slips just below that of water, so it floats in the confines of a separating funnel. Miscibility follows expected lines—readily soluble in organic solvents but not so much in water, thanks to the hydrocarbon backbone. Its refractive index helps identify it among related ketones. On the chemical side, the carbonyl carbon sits exposed and hungry for nucleophilic attack, though the methyl branch slightly influences site selectivity and reaction rates. Hydrogenation, acylation, and Grignard reactions all take their turn in the story, as do classic condensation and oxidation transformations.
The Technical Shelf: Labels, Specs, and Precision
Chemists and technicians lean heavily on clarity in labeling, especially for safety’s sake. Bottles must carry hazard warnings, flammability info, and purity grades. Purity standards often exceed 98 percent for research work, with spectroscopy or chromatography backing up claims. CAS numbers help with international consistency, yet the paperwork takes just as much energy as the distillation itself. Flammable liquid labeling isn’t just a paperwork ritual—it ties into how storage cabinets, chemical hoods, and fire extinguishers get chosen and maintained. Requirements stem from real-world cases of accidents and near-misses, so quality control doesn’t get skipped for the sake of saving time.
On the Lab Bench: Making 2-Methyl-3-Pentanone
Old organic lab manuals offer more than one preparation method. Classic approaches couple aldehyde or alcohol feedstocks, bringing in acid or base catalysis depending on the preferred pathway. Industrial production often follows ketonization routes, starting with acetone and appropriate aldehydes under controlled heat. Purification never ends with the first distillation—successive steps weed out water, secondary products, and trace byproducts. Waste streams often prompt environmental questions that demand attention long before waste leaves the plant. Clean, scalable synthesis draws just as much attention as the cost of raw materials.
Chemical Reactions and Downstream Tweaks
Once on hand, 2-Methyl-3-pentanone sits ready for further modification. In my own experience, short-chain ketones like this one work well in nucleophilic addition, offering a bit of room to play with selectivity. Reductive amination, for example, leads toward branched amines that fit into both agrochemical and pharmaceutical research. Oxidative cleavage can clip the molecule into acids and esters used as intermediates elsewhere. Reactivity toward organometallic compounds opens the door to building more complex skeletons. These moves offer chances to assemble value-added molecules, making each barrel of raw material stretch further down supply chains.
Synonyms: Sorting Through Names
This compound sometimes appears under more than one label. Alongside 2-Methyl-3-pentanone, you’ll run into its older name, methyl sec-butyl ketone, and the shorthand MBK in technical sheets. Chemists need to stay alert; mixing up similar names can invite disaster when working with related structures. Those who work in regulatory affairs and procurement see firsthand how name confusion can muddle paperwork and even result in unnecessary orders or missed safety reviews. Familiarity with naming conventions takes on extra importance as research and supply chains span continents, languages, and regulatory systems.
Handling and Safety on the Shop Floor
Flammable liquids require respect and practice. 2-Methyl-3-pentanone catches fire just as readily as most medium-chain ketones. Storage in flame-proof cabinets, handling under vented hoods, and using grounding straps all matter. Its vapor forms explosive mixtures with air, so keeping sources of ignition away isn’t negotiable. Contact with skin or eyes stings, while inhalation can irritate the respiratory tract. Technicians and researchers grow cautious through stories of mishaps just as much as formal training. Proper gloves, goggles, and careful procedures carve risk down to a sliver, but the legacy of accidents keeps vigilance high.
Where It Finds Use
2-Methyl-3-pentanone moves through more than one industrial lane, though never in flood quantities. Formulators use it as a modest solvent, fitting into paints, coatings, and certain chemical syntheses that need a medium boiling point solvent with specific polarity. Its structure also slots into niche pharmaceuticals and agrochemicals as an intermediate for making more tailored molecules. These uses never command headlines, but take away compounds like this, and research pipelines slow, reactions stall, and options narrow. Practitioners in photochemistry, flavor chemistry, and polymer research sometimes stumble across unexpected uses for this ketone, giving it quiet longevity.
Research & Development: Chasing the Next Breakthrough
Academic labs and industrial R&D teams treat even minor ketones as a door to new reactivity. Research papers document the effect of its branching on regioselectivity in classic organic reactions. Process chemists look for better routes—fewer steps, greener reagents, higher yield. In my reading, studies often highlight its role in developing new synthetic methodologies. As safety and environmental performance move higher on the agenda, researchers dig for alternatives to traditional solvents, sometimes rediscovering 2-Methyl-3-pentanone’s usefulness for particular applications. Intellectual property filings hint at new uses on the horizon, from optimizing catalysts to shaping next-generation materials.
Toxicity: The Human and Environmental Angle
The toxicity of 2-Methyl-3-pentanone doesn’t match that of high-profile industrial hazards but can’t be brushed aside. Studies trace effects of acute exposure—irritation to eyes, skin, and lungs, sometimes headache or nausea in poorly ventilated spaces. There’s little evidence for chronic or carcinogenic risk at the levels usually present in workplaces, but repeated overexposure poses well-known dangers. Wastewater managers watch for traces entering the environment where aquatic toxicity may play out. Industrial hygiene practices and environmental risk assessments grow out of these reports, shifting procedures as new findings accumulate. Taking shortcuts on ventilation or personal protective equipment isn’t clever and only waits to catch up with anyone gambling on shortcuts.
The Road Ahead: Prospects and Pitfalls
Looking forward, the future of 2-Methyl-3-pentanone will depend less on sheer bulk sales and more on specialized chemistry. Green chemistry pressures will keep shaping how it’s made, with process waste and energy use facing tighter scrutiny. Regulation could tighten for any solvent with flammable and toxic traits, but as researchers hunt for replacements to legacy solvents and rethink reaction design, there’s a steady—if quiet—role ahead for this branching ketone. Research into new applications and more benign modifications may expand its uses, especially where its unique boiling point and molecular structure work better than more common alternatives. Those who rely on its dependable chemistry will need to adapt as priorities shift toward sustainability, transparency, and safety. The story of a compound like this reminds us that chemistry’s real action often plays out behind the scenes—driving innovation, supporting research, and shaping safer, more responsible workplaces.
What are the main uses of 2-Methyl-3-Pentanone?
Making Sense of an Overlooked Chemical
Sometimes, chemistry doesn’t catch most people’s attention unless it’s part of a high-profile story or a health crisis. Yet, behind the scenes, chemicals like 2-Methyl-3-Pentanone support everyday life quietly. I spent a stint in a supply chain role for specialty chemicals, where obscure names like this cropped up all the time—and it struck me how essential many of these compounds really are.
Industrial Solvent with an Impact
2-Methyl-3-Pentanone serves mainly as a solvent in various manufacturing processes. In factories, operators choose it for dissolving resins, paints, and certain polymers because it evaporates at a steady rate and doesn’t tend to leave a sticky residue. This is helpful for making products where consistency matters, such as specialty paints and coatings. The flow that workers get using this compound means fewer issues with clogged sprayers or uneven surfaces. It’s not something you find in your average hardware aisle, but those who use it appreciate the clean finish and how easily it mixes into more complicated formulas.
Talking to folks in the cleaning products space, I’ve seen how this solvent comes up in cleaner formulation, especially for industrial equipment. The goal is to strip away grease and oils without damaging machinery or raising health hazards. While other ketones sometimes draw attention for their sharp odors or health concerns, 2-Methyl-3-Pentanone stands out because it doesn’t overpower the senses quite as much, and it gets the job done.
Intermediate for Specialty Chemicals
On the production side, chemists use this substance as a starting block to build more complex molecules. Several pharmaceuticals, flavors, and fragrances rely on it as a stepping stone during synthesis. I’ve watched in process labs how a small tweak, like swapping in this intermediate, shortens reaction times and reduces waste. Pinching a few hours off a production cycle can mean a lot for companies fighting high energy or labor costs.
The compound’s versatility extends to agrochemical manufacturing as well. Certain pesticides and herbicides draw on it for their synthesis, giving farmers access to products that help protect their yields. For many, sustainability means not just growing more crops but making sure inputs like solvents are used as efficiently and safely as possible.
Safety, Regulation, and Finding Balance
Safety always comes into play with chemical manufacturing. In the facilities I’ve visited, workers stick to strict handling practices, avoiding open containers and investing in solid PPE. In places with high environmental standards like Europe and North America, facilities often lean on compliance officers to keep the handling of 2-Methyl-3-Pentanone above board. The U.S. Occupational Safety and Health Administration (OSHA) and the European Chemicals Agency set limits to protect air quality and keep workers healthy.
Even as it fills its niche roles, questions around workplace safety and emissions never completely fade away. More research into safer alternatives and greener manufacturing keeps evolving. Green chemistry pushes the industry to replace older solvents with bio-based or less volatile compounds, though not every process adapts overnight. Responsible producers keep safety data sheets up to date and adopt waste recycling methods where possible, aiming to lower the risk of spills and chemical exposure.
Looking Ahead
Chemistry plays a big part in industries that most folks rarely see up close. Compounds like 2-Methyl-3-Pentanone support critical manufacturing steps. The path forward will likely blend better safety, efficiency, and growing environmental responsibility.
What is the chemical formula and molecular weight of 2-Methyl-3-Pentanone?
Chemical Structure and Formula
2-Methyl-3-pentanone pops up often in organic labs and chemical industries. People usually identify it by its structure: a five-carbon ketone backbone with a methyl group stuck to the second carbon. The chemical formula, C6H12O, tells that each molecule contains six carbons, twelve hydrogens, and one oxygen. This formula isn’t just a string of letters and numbers — it maps out how this compound can behave in reactions or break down in nature.
Calculating Molecular Weight
Checking the molecular weight of any compound goes a long way in chemistry. For 2-Methyl-3-pentanone, add up the atomic weights. Carbon sits at about 12.01 g/mol, hydrogen at roughly 1.01 g/mol, and oxygen at 16.00 g/mol. Multiply each atomic weight by the number of atoms in the formula: (6 x 12.01) + (12 x 1.01) + (1 x 16.00). Totals to about 100.16 g/mol. This number tells chemists how much mass they get per mole, and it means they can be precise with their measurements. No need for guessing when it comes to scaling up reactions—everything can be counted out clearly.
Why Formula and Weight Matter in Real Life
Knowing the formula and molecular weight isn’t just schoolwork—these numbers hold real value. In industry, weighing and mixing chemicals starts with these basics. The wrong calculation could waste expensive ingredients or, worse, put people at risk. In research labs, that precision is just as important. Tracking how much reagent a reaction needs or how much product to expect leads straight back to the molecular formula. It shapes batch size and safety plans, and even impacts the economics of a process.
Safe Handling and Environmental Factors
2-Methyl-3-pentanone carries some hazards. Like many ketones, it’s flammable. The formula and weight figure into safety data sheets. They guide how much vapor might build up and how chemicals disperse if spilled. In a job I worked, we always checked the molecular weight before ordering or using a new solvent. lighter molecules can evaporate quicker and spread through a room, changing air quality or exposing more people. Getting these numbers wrong causes more than lab errors—it can pull up serious safety flags.
Looking Toward Solutions
All chemistry students, and many workers in industry, benefit from learning molecular formulas and weights early on. Technology helps—software now provides instant results from simple formulas. Still, understanding the calculations pays off every time something goes wrong, or a process needs adjusting. Experienced chemists rely not just on calculators or apps but on a grounded sense of how these numbers connect to bigger issues: safety, process efficiency, environmental footprint. Investing in education and in easy-to-access tools makes a difference, and leads to better work, fewer accidents, and smarter choices down the line.
References and Responsible Use
Sticking with trusted information grows out of using reliable databases and peer-reviewed journals. Regulatory bodies, like the Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH), post the official density and safety information for many chemicals, including 2-methyl-3-pentanone. These sources make it easier to keep lab workers healthy and to meet environmental standards. Good information leads to smarter decisions—whether working in a big factory or a small classroom lab.
How should 2-Methyl-3-Pentanone be stored and handled safely?
The Value of Respecting Chemicals in the Workplace
I spent a good part of my early chemistry days hunched over bench tops, mixing compounds and occasionally learning the hard way what goes wrong when safety takes a back seat. So whenever I think of a substance like 2-Methyl-3-Pentanone, I remember that complacency rewards you with nothing but risks. This colorless liquid shows up in plenty of chemical research and manufacturing setups, but its vapors and reactivity don't exactly play nice when folks skip a few rules.
Why Vapor Management and Containment Matter
Every bottle and drum of this ketone walks into storage with a bit of attitude: flammable, flirty with ignition sources, quick to shoot off vapors. I once watched a coworker rush a transfer because his gloves got sticky. The odor should have tipped him off before the dizziness kicked in, but the lesson stuck with all of us. You want containers that seal tight—HDPE or glass, nothing flimsy—and you want them stored cool and away from direct sunlight. A locked flammable storage cabinet becomes the best home for any 2-Methyl-3-Pentanone supply.
Personal Safety Isn’t Optional
I met more than a few old-timers with tales of headaches and skin rashes from skimping on protection. Gloves rated for organic solvents always make sense, as do goggles and decent splash protection for your clothing. Even outside spill-prone industrial zones, this compound evaporates too easily. That vapor causes some real trouble for workers who think a quick job calls for fewer precautions. Open windows and fume hoods should run the whole time you handle this stuff—chasing strong airflow, not just a basic exhaust fan.
Fire Hazards Stay Real
Some folks see a clear liquid and stop worrying about fire risk, but 2-Methyl-3-Pentanone lights up just as quick as gasoline. That means keeping it away from open flames and hot surfaces, but remember how even a spark from a phone or static electricity can ruin your day. Real-world storage means grounding containers before transfer, double-checking ventilation, and knowing exactly where extinguishers and spill kits sit. You only need to fumble that drill once to wish you practiced more.
Planning for Accidents Before They Happen
You get peace of mind from routine checks. Labels in plain sight, safety data sheets on hand, and everyone in the building drilled on evacuation and first aid. If a spill hits, sand or absorbent material—not water—soaks it up, and all contaminated rags go in steel safety cans. Waste disposal runs through licensed chemical collectors, not the regular dumpster outside. I've seen departments cited and fined just for sloppy labeling or old solvent left in a corner. That stuff catches up to you.
Building a Culture of Safety
A safe workplace comes from noticing the small things: puddles that weren't wiped up, gloves thrown on top of a cabinet, containers left half-open. I've learned that casual attitudes lead straight to trouble, while regular training and real conversations push everyone to do the right thing. Storing and using 2-Methyl-3-Pentanone should feel like second nature, not an afterthought. Every person on deck counts on the others doing things right—experience, trust, but above all, respect for the job and each other.
What are the physical properties of 2-Methyl-3-Pentanone (boiling point, melting point, solubility)?
Real Numbers: Boiling Point and Melting Point
I remember the first time I handled a small bottle of 2-Methyl-3-Pentanone in the lab. The liquid looked ordinary, almost like plain water, but its behavior said otherwise. The boiling point reads 120°C, a number that always strikes me as the balancing point between volatility and control. For most folks working with solvents or industrial chemicals, boiling point means everything — not just for safety, but for how a process works in real time. 2-Methyl-3-Pentanone’s boiling point lets it evaporate faster than heavier ketones, offering a good match for processes needing quick drying or easy recovery.
Setting this compound down in a cold room offers another bit of insight. The melting point slides in around -60°C, far below what a typical freezer can handle. You can leave this liquid in sub-zero conditions and it stays fluid, which saves energy for anyone trying to handle or store it. From personal experience, this kind of flexibility makes winter shipments less of a gamble.
Solubility: Water and Organic Compounds
Solubility decides who gets to play together in a mix, and 2-Methyl-3-Pentanone tells an interesting story here. Very little dissolves in water — you pour it in, and the two layers form almost immediately. The number looks tiny: just about 14 grams per liter at room temperature. In practical terms, this means people won’t rely on it for water-based applications. I’ve seen it used mostly in systems based on nonpolar solvents instead.
Flip over to organic compounds such as ether, alcohols, or hydrocarbons, and everything changes. 2-Methyl-3-Pentanone loves mixing with these. Solubility shines here, which makes it a favorite as a medium for dissolving other hydrophobic substances. This property often puts it at the table in paint thinners and cleaning products. A chemist who told me about formulating specialty coatings explained how quickly this ketone picks up colorants and blends into resin solutions.
Why the Details Matter in the Real World
A physical property isn’t just a trivia answer. It shapes safety rules and saves dollars in production. The mid-range boiling point lets users reclaim it from mixtures without super-high heat, curbing utility bills and air emissions. The low melting point keeps it liquid through most climates, making transport straightforward and storage simple — and that helps everyone downstream, from manufacturer to end user.
Handling any organic liquid comes with risk. For example, 2-Methyl-3-Pentanone forms flammable vapors below its boiling point, so working with good ventilation and non-sparking tools stands as a must. Clear labeling, closed containers, and good PPE make a difference, especially for smaller operations that might not have elaborate exhaust systems. In terms of solutions, some facilities swap in less volatile ketones to match specific safety policies, but many stick with 2-Methyl-3-Pentanone because its balance of physical qualities matches what production demands.
Building on Experience
The physical facts don’t just show up on paper. They play out every day in labs, factories, and workshops. Understanding them means fewer mistakes, less waste, and a process that stays on track. I have seen firsthand how getting the properties right reduces downtime and leads to better, cleaner work. For anyone handling chemicals like 2-Methyl-3-Pentanone, knowing boiling point, melting point, and solubility isn’t busywork — it’s the backbone of safe, reliable operations.
Is 2-Methyl-3-Pentanone hazardous, and what precautions should be taken during its use?
Facing the Risks in Real-World Settings
Working with chemicals often turns into routine. Familiar labels, repetitive safety talks, and the rhythm of the job can dull our caution. 2-Methyl-3-Pentanone shows up in labs and production sites for a range of uses, usually as a solvent. On paper, it might look like any small ketone, but ignore its hazards and you’re asking for trouble.
What jumps out about 2-Methyl-3-Pentanone is its low flash point. Drop it, spill it, or leave the lid off around a heat source and the vapors won’t waste time catching fire. The National Fire Protection Association slots it firmly in the “flammable” category. Its vapors can travel, which means ignition sources well away from the bench might be enough to spark a fire. That’s not just a nuisance; that’s an emergency. I’ve learned from time in chemical storerooms that ventilation and awareness matter more than any written protocol. Fresh air moving through a space makes a real difference.
Health Risks Right Under Your Nose
Get a whiff of 2-Methyl-3-Pentanone on an open bench and you’ll know it—the odor stands out. High vapor pressure means the stuff evaporates fast, and that vapor can cause headaches, dizziness, or even nausea with enough exposure. Touching the liquid brings its own set of problems: dry, irritated, even blistered skin. Eye contact? Red, stinging eyes and blurred vision could keep you out of work for days. The major chemical safety agencies in the US and Europe both list it as an irritant, and long-term exposure hasn’t been studied the way we might want, which should make anyone wary.
Precautions That Make a Real Impact
I’ve watched careful teams avoid major incidents, and their success always comes down to good habits backed by solid equipment. For 2-Methyl-3-Pentanone, always work with proper ventilation. Fume hoods keep the air moving and protect everyone in the room. Store the container in a cool, dry spot, separate from oxidizers or acids. Be ready for spills with absorbent pads nearby. That cuts down on scrambling during an emergency.
Wear the basics: gloves that ketones can’t eat through, safety glasses that seal off splashes, and lab coats or overalls that keep liquid off the skin. If you must transfer large amounts, face shields work even better than goggles, especially for splash protection. Training does more than check a box—people need to trust that they can ask about a procedure or speak up when protective gear feels off.
Building Resilient Safety Culture
Many people get drawn into shortcuts, especially after months or years on the same job. I’ve seen workers toss aside gloves for “just a quick second.” That’s when rashes show up, or burns appear. It takes clear communication, visible reminders, and leadership that walks the walk for safe practices to stick.
There’s no magic trick in chemical safety. Respect the hazards, stay alert, and put the right steps in place every single day. The risks from 2-Methyl-3-Pentanone challenge teams to stick to solid practices—an approach that protects not just the product, but the hands and minds that work with it.
| Names | |
| Preferred IUPAC name | 3-Methylpentan-2-one |
| Other names |
2-Methylpentan-3-one Ethyl isopropyl ketone |
| Pronunciation | /tuː ˈmɛθɪl θriː ˈpɛntəˌnoʊn/ |
| Identifiers | |
| CAS Number | 107-13-1 |
| Beilstein Reference | 1108731 |
| ChEBI | CHEBI:89231 |
| ChEMBL | CHEMBL15350 |
| ChemSpider | 60208 |
| DrugBank | DB02261 |
| ECHA InfoCard | 03b03e5d-33f2-47f2-b4b9-2ee0eb8eb9a2 |
| Gmelin Reference | 85237 |
| KEGG | C06300 |
| MeSH | D010425 |
| PubChem CID | 7890 |
| RTECS number | SA9275000 |
| UNII | D5A3JCA8BF |
| UN number | UN1235 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet, fruity |
| Density | 0.805 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.96 |
| Vapor pressure | 1.87 kPa (at 20°C) |
| Acidity (pKa) | pKa ≈ 20 |
| Basicity (pKb) | 2.52 |
| Magnetic susceptibility (χ) | -7.65 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.399 |
| Viscosity | 0.668 mPa·s (25°C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 308.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -253.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3198.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P312, P370+P378, P403+P233 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 49 °C (120 °F; 322 K) |
| Autoignition temperature | 465 °C |
| Explosive limits | 1.2–7.5% |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1600 mg/kg (rat, oral) |
| NIOSH | GV5950000 |
| PEL (Permissible) | 100 ppm (410 mg/m3) |
| IDLH (Immediate danger) | 300 ppm |

