2-Methyl-3-Pentanol: Understanding Its Role and Relevance
Tracing the Story of 2-Methyl-3-Pentanol
Curiosity about odd-sounding chemical names probably packs less punch at most dinner tables than the latest streaming show, but 2-Methyl-3-Pentanol tells a story much older than YouTube. Through the 20th century, chemists hunted for ways to understand and work with alcohols not just as everyday beverages, but as building blocks for flavors, fragrances, solvents, and novel drugs. This compound started popping up in industrial catalogs as modern labs gained tighter control over synthetic organic chemistry, and it stuck around because it fit the needs of more than one industry. Its six-carbon skeleton and secondary alcohol group offered the kind of flexibility that gets chemists excited. Early literature shows that people first made it through classic Grignard and reduction approaches, and found that its branched nature opened up some unique reaction paths. Once manufacturing scaled up, folks realized they could depend on its predictable properties and stability. It’s not famous, but it speaks to a time when chemical innovation felt wild and full of risk and reward.
The Practical Face of 2-Methyl-3-Pentanol
Walking into a lab and seeing a bottle labeled with this chemical, you wouldn’t guess it’s part of countless industrial routines. This colorless liquid carries a faint smell and a moderate boiling point. It slips easily into mixtures, which makes it favored as a special-purpose solvent. Technically, it isn’t hydrophobic enough to ignore water, but not fully friendly with it either. Physical data from the CRC Handbook and confirmed by Sigma-Aldrich puts its boiling point at just over 146 °C, with a density sitting right around 0.81 g/cm³ at room temperature. Unlike more volatile alcohols, it handles gentle heating without alarming vapor buildup. That gives it an edge in manufacturing settings where control beats chaos. In practice, its refractive index and solubility profile lend it versatility, though it never shows up in consumer goods where stricter regulations clamp down on specialized alcohols.
Preparation and Hands-on Chemistry
In labs, experienced chemists often reach for old favorites when preparing this alcohol—building out from pentanone precursors through straightforward reduction, or using Grignard reagents for a more textbook synthesis. The steps aren’t glamorous, but reliability counts in chemical work. You start with 2-methyl-3-pentanone or similar ketones, add a reducing agent—sodium borohydride keeps side reactions at bay—and, in well-controlled conditions, out comes the desired secondary alcohol. Improvements and tweaks follow the same cautious approach, minimizing waste and focusing on yield. Scale-up means careful distillation and drying, since purity affects every downstream application, especially in specialty chemical supply. Over the years, process improvements simplified the route—greener reducing systems, safer workups—but at heart, it’s always about making sure the final liquid matches the demanded specs and functions robustly wherever it lands.
Names Meaning the Same Thing
Chemists seem to love variety in naming. 2-Methyl-3-pentanol also answers to names like 2-methylpentan-3-ol, 3-hydroxy-2-methylpentane, and less formally, sometimes just “sec-hexanol” or “secondary methylpentanol.” Catalogs might organize it by Cas No. 565-60-6, tying science’s penchant for precision to practical storage and handling. Each of these names circles back to the same six-carbon frame, sliced and rearranged depending on which naming tradition you prefer. For those navigating chemical supply, precision in labeling prevents mistakes, especially since isomers with just one shifted methyl group can behave very differently in real reactions.
Getting Down to Safety and Operational Realities
In every lab I’ve worked in, handling medium-weight alcohols like this one demands respect but not panic. It’s not known for wildly toxic fumes, yet gloves and decent ventilation always matter. Standard practice means keeping it away from open flames, since even moderate heat can send vapors climbing to dangerous levels. SDS documents stress typical alcohol precautions—avoid skin and eye contact, don’t breathe in concentrated fumes, and keep containers sealed. Regulations often group it under “flam-liquids, n.o.s.” or general secondary alcohol classes, so seasoned techs spot the hazards fast. For disposal, environmental protocols ask for containment and controlled incineration, since pouring this kind of solvent down the drain means risking groundwater trouble and regulatory fines. Every responsible operation trains on spill kits, and well-run facilities check for residual vapors during clean-ups.
Where 2-Methyl-3-Pentanol Finds a Use
The real world calls this alcohol to the front for niche jobs rather than retail shelves. In my own time in product development, its role as a specialty solvent for high-precision coatings and resins showed up time and again. Some pharmaceutical intermediates rely on its branched skeleton for chiral control—meaning its subtle shape shifts can help guide reactions that favor one mirror-image form of a molecule, which becomes critical when building drugs. Paint chemists blend it in for fast, predictable flow, while some fragrance and flavor formulators chase its subtle notes for intermediate synthesis, not final products. Pilot-scale operations tinker with it for making finer alcohol derivatives, since its chemical backbone cracks open unusual reactivity not found in simpler liquids like ethanol or propanol.
Digging Into Chemical Behavior
Drop this alcohol into a reaction setup and it’s ready to play ball with all the classic transformations. Oxidation with chromium or permanganate pushes it to the corresponding ketone, 2-methyl-3-pentanone. Tried-and-true dehydration with acids veers off into branched alkenes, and you can find epoxide or ether formation under the right base conditions. Chemists value its secondary nature—neither too eager nor too sluggish—when sizing up routes for further modification. Its steric bulk means crowding can slow down nucleophilic substitution, so you see selectivity that’s tough to engineer with straight-chain alcohols. If your workflow demands forming reactive esters, 2-Methyl-3-Pentanol can deliver those with a bit of acid or anhydride, then snap back to the bare alcohol with controlled hydrolysis. This reliability means it slots into agile discovery chemistry, a hidden player driving bigger breakthroughs.
On the Trail of Recent Research
Studies keep crawling forward. Academic and industrial chemists run tests to understand how this alcohol behaves in advanced material synthesis. Green chemistry approaches investigate ways to recycle or bio-produce branched alcohols, seeking routes that trim hazardous waste and lower energy costs. Some research uses 2-Methyl-3-Pentanol as a starting point for creating novel surfactants or solvent blends, focusing on performance in applications where volatility and miscibility play a part in efficiency. Analytical chemists track trace levels after use in sealed environments, measuring environmental fate and breakdown during waste processing. Publications in the last decade show expanded use in preparing reference standards or controlling stereochemistry in new pharmaceuticals, making use of its moderate reactivity and chiral center.
Understanding Toxicity
The science on toxicity paints a nuanced picture. Animal studies point out that this secondary alcohol, like others in its family, can create central nervous system depression at high doses, and lethal doses stack up at levels far beyond casual exposure in normal lab work. Health officers warn that repeated, careless handling risks skin dryness or irritation, and large spills mean risk to aquatic environments before breakdown occurs. No study attaches the acute danger label seen with halogenated solvents or the most volatile ethers, but safety analysts treat it with steady caution. Chronic data lags behind more common alcohols, so risk assessment professionals look for structural similarities with better-studied compounds and err on the side of conservative exposure limits. As research continues, public data sets like PubChem and ToxNet collect results, giving regulators, labs, and manufacturers a transparent window into emerging concerns and best practices.
Looking Ahead: Future Directions
Changes in industry and science keep the future of 2-Methyl-3-Pentanol open. If clever chemists crack routes to sustainable, biosourced alcohols with similar branching, that could shift the source away from oilfeed stocks and ease environmental arguments against synthetic production. Interest in fine chemical synthesis and greener solvent systems supports continued use, especially for research tools and specialty product lines that demand fine-tuned volatility and chemical reactivity. Researchers track regulatory shifts, weighing stricter controls on volatile organic compounds. Where green chemistry becomes a firmer requirement, modifications or analogs might take over, shaped by the same balance of safety, reactivity, and downstream utility. The story isn’t likely to land on the front page, but it keeps evolving at the edges of science and industry, where making, moving, and mastering modest molecules like 2-Methyl-3-Pentanol quietly shapes bigger trends in technology and sustainability.
What is 2-Methyl-3-Pentanol used for?
Understanding 2-Methyl-3-Pentanol
2-Methyl-3-pentanol doesn't make headlines like some chemicals, but its applications touch many products and processes. This clear, colorless alcohol, sometimes called isopropyl sec-butyl carbinol, turns up in places that rarely get a spotlight. It stands out as an important building block, mostly because chemists like its stable, easygoing nature.
Industrial Applications That Matter
Manufacturers use 2-Methyl-3-pentanol as a solvent. It’s especially handy when paint or varnish needs thinning. Some companies add it to special inks and coatings, because it helps dissolve ingredients that other ordinary solvents leave behind. This keeps surfaces looking smooth and finished. For adhesives, this alcohol plays a quiet but crucial role, encouraging all the sticky ingredients to mix together and stay mixed, so the final glue works as intended.
This alcohol pops up in labs, too. Sometimes chemists stir it into chemical reactions for pharmaceutical research. It helps them play with molecules by changing how chemicals interact, nudging them toward specific products. So a headache pill, for example, may depend on 2-Methyl-3-pentanol stepping in during its long journey from raw ingredients to boxed medicine.
Flavor and Fragrance Foundations
2-Methyl-3-pentanol has a soft, faint aroma. This makes it a favorite for flavor and fragrance creation, where even minor players can lift an entire formula. Flavor scientists sometimes rely on it for developing food extracts. In these cases, the compound acts as a carrier that helps blend flavors, especially for products like chewing gum or bakery goods. Perfume makers occasionally tap its abilities when searching for a stable base that won’t overpower lighter floral or citrus notes.
Balancing Safety and Progress
Many chemicals come with a long warning label and a worry about their effects. With 2-Methyl-3-pentanol, safety should be top of mind. People who handle it at work follow strict rules: gloves, goggles, good ventilation. At high doses, it can irritate eyes and skin. Over time, breathing in too much can affect nerves, so regular training and careful workplace monitoring matter. The European Chemicals Agency and OSHA both give clear guidelines, and companies that value long-term success pay attention.
Future Possibilities
Sustainability demands keep changing the chemical supply chain. Researchers keep looking for solvents and additives that do the job without lingering in the environment. Green chemistry has picked up speed, and alternatives often grab attention. Still, until a replacement matches 2-Methyl-3-pentanol’s unique profile—stable, effective, and relatively mild—it is likely to remain important for chemists, product developers, and manufacturers. Tackling waste and recycling options could help make its use even safer and more responsible in coming years.
What is the molecular formula of 2-Methyl-3-Pentanol?
Getting to the Root of the Molecule
2-Methyl-3-pentanol pops up as a classic example in organic chemistry classrooms. Plenty of people studying chemistry recognize that name as a mouthful, but figuring out the molecular formula isn’t as tough as it seems. Looking at the name gives you almost everything you need to know. It tells you the number of carbons, their arrangement, and what functional groups are in play.
Demystifying the Structure
The molecule starts with “pentan-,” so we’re dealing with five carbon atoms in the main chain. The “3-ol” part says there’s an alcohol group (–OH) stuck to the third carbon in that chain. The “2-methyl” tells us that a methyl group (–CH3) attaches to the second carbon. Adding it up: you’ve got a backbone of five carbons, one extra carbon from the methyl side group, a single oxygen atom for the –OH, and enough hydrogens to fill out the rest.
From hands-on experience with drawing molecules, you know that every carbon in an alkane forms four bonds. Start your sketch with a straight chain of five carbons, then hook that extra methyl group onto the second carbon from the left. Attach the –OH to the third carbon. Now, fill in the hydrogens so that each carbon’s sticking to four things. Double-checking matters because even chemists who’ve spent years in laboratories still count up the hydrogens one by one to avoid silly mistakes.
Arriving at the Formula
It all comes together as C6H14O. Six carbons appear in total: five in the base, one from the methyl group. Hydrogens cover every available spot except those occupied by bonds to oxygen or other carbons. You count fourteen hydrogens, no more, no less. The –OH gives you one oxygen—simple as that.
Knowing Why the Details Count
Knowing the exact formula means more than passing a quiz. I’ve seen plenty of cases in research where a single missed hydrogen completely sabotaged a synthesis, throwing out weeks of work. It pays off in real-world labs to take these small details seriously. Chemists rely on precision since mixing up one formula with another can make the difference between a safe medicine and a toxic compound.
Textbooks and online resources usually back up this point. Reputable sources like PubChem and the Merck Index confirm the formula: C6H14O for 2-methyl-3-pentanol. Picking reliable databases matters because misinformation sneaks in between the cracks, especially on social media or forums filled with rushed answers. Chemistry has enough pitfalls that you learn quickly to double-check with sources that cite peer-reviewed studies or handbook data.
What It Means for Students and Researchers
Students sometimes glaze over formulas, treating them as trivia rather than the language of molecules. In the professional world, careful attention to naming and formulas forms part of the safety culture in labs and manufacturing. Even process chemists walking the production floor use molecular formulas to catch errors before they turn into safety incidents. I’ve watched good teams always do a quick formula check before signing off on new batches or test runs, not just to be thorough but to guard each others’ safety and reputations.
Sharpening Your Approach
Learning how to break down a chemical name and draw its structure sets you up for real understanding. Only then do the formulas start to make sense, clicking into place and connecting textbook theory to practice. The experience of working through each step—from naming, sketching, to spelling out the formula—helps you build confidence. In chemistry, as elsewhere, experience and double-checking catch mistakes, and precision keeps things running safely and efficiently.
Is 2-Methyl-3-Pentanol hazardous to handle?
What 2-Methyl-3-Pentanol Actually Is
2-Methyl-3-pentanol belongs to the family of alcohols that show up in labs and specialized industrial sites. It’s a colorless liquid, packs a mild odor, and doesn’t grab much attention outside chemical circles. This substance doesn’t seep into the news, but it finds a home in synthesis work and—as usual with alcohols—it isn’t entirely innocent.
Chemical Precautions Aren’t Just for Show
Many folks walk through labs thinking basic chemicals don’t ask for respect. That’s a costly mistake. I’ve watched grad students handle solvents while chatting about lunch, only to wind up learning why gloves and goggles stay popular. 2-Methyl-3-pentanol can easily irritate skin and eyes. Getting some of this on bare skin leaves behind stinging, dryness, or worse if you’re unlucky. Alcohol vapors have a way of sneaking into eyes and throats. Rushing a step, skipping those cheap nitrile gloves, that’s asking for red, itchy skin or worse, depending on individual response.
Science doesn’t exist in a vacuum. Industrial accidents don’t usually happen because someone was entirely ignorant. Sometimes it boils down to pressure, fatigue, or someone new to the scene who just hasn’t seen things go sideways yet. A splash to the eye with this liquid feels like a sharp warning. Not every chemical gets rehydrated in the emergency shower, but 2-methyl-3-pentanol has landed people there after small spills. Eye wash stations stand ready for a reason.
Breathing the Fumes Can Trip You Up
Workers sometimes think only strong-smelling chemicals are risky. Fatigue, stuffy rooms, and slow build-up of vapors change things. Even simple alcohols leave you with headaches, dizziness, or a foggy mind if you breathe enough. I once spent two hours working next to a poorly ventilated flask, forgetting that mild doesn’t mean safe. By the time the nausea set in, opening a window felt about as effective as bailing out a flooded basement with a cup. That kind of experience teaches respect for even minor-looking solvents. 2-Methyl-3-pentanol demands working under a hood and giving the bottle respect. OSHA lists this chemical as an irritant and recommends strong ventilation.
Fire Hazards Remain Real
Pouring alcohols near heat or open flames in a carelessly managed lab or plant risks flammable vapor clouds. 2-Methyl-3-pentanol won’t start fires just by looking at it, but it likes to catch fire if it finds a spark. I’ve heard more stories than I care to remember about solvent fires that started with a “just a quick transfer” over a hotplate or next to a careless Bunsen burner. Flammable liquids turn minor mistakes into property damage and sometimes scars, so safe storage and careful disposal make sense.
Simple Steps Make a Difference
Inexperienced or tired workers love to skip the basics. Gloves, goggles, lab coats, and ventilation serve a reason beyond rule-following. I keep an extra set at my bench. Fire extinguishers, labeled waste bottles, and fume hoods aren’t window-dressing. 2-Methyl-3-pentanol, like most chemicals, rewards respect with safety and punishes shortcuts with burns or worse.
What are the storage conditions for 2-Methyl-3-Pentanol?
The Practical Side of Chemical Storage
Working in a science lab means paying close attention to the small details, especially with solvents like 2-Methyl-3-Pentanol. This clear, flammable alcohol might not look like much, but it demands respect. Even if you’ve handled similar chemicals for years, getting too casual can come back to bite you. I’ve seen spills and near-misses from small lapses in storage practice. Most accidents stem from a moment’s forgetfulness: a cap not tightened, a bottle left too close to a heat source, or a label missed in the rush of a busy afternoon.
Prioritizing Safety to Protect Everyone
Accidents can escalate quickly, and 2-Methyl-3-Pentanol can release fumes that no one wants to breathe in, especially in a poorly ventilated space. The vapor gets into the air and can irritate the eyes or lungs, so even a routine transfer in a cluttered stockroom could lead to a problem. That’s why storage guidelines exist, not just as a formality, but to keep situations predictable and safe.
Ideal Storage Conditions
Store 2-Methyl-3-Pentanol in a cool, dry, and well-ventilated area. Heat is a major concern. Warmth encourages evaporation, and once those vapors build up, they can ignite with a spark. I always kept such flammable solvents in flameproof cabinets, away from any sources of ignition like hot plates or faulty wiring. Make sure that sunlight doesn’t reach the containers, either. Direct sun heats up bottles faster than most people expect.
Humidity plays a role as well. Water can creep into improperly sealed containers and begin altering the chemical or causing pressure build-up. I learned to check seals regularly, especially after a container spent a long weekend untouched. If a screw top seems loose or the interior looks foggy, it’s safer to replace it right away.
Container Choices and Maintenance
Reputable labs keep 2-Methyl-3-Pentanol in tightly closed glass bottles or specialty plastics rated for organic solvents. I remember situations where standard plastic began to soften or crack just from the fumes, so cutting corners never saves trouble in the long run. Clean and label every vessel clearly, using chemical-resistant markers or printed labels. Regular checks help avoid errors—old containers lead to confusion and increased risk, especially if staff changes or shifts get hectic.
Addressing Common Issues and Charting Solutions
Even with ideal storage, leaks and spills can happen. Immediate cleanup with absorbent materials and proper disposal means less risk. Sometimes people think “it’s just a bit,” but all it takes is one moment of carelessness. Better training helps, so I’ve always encouraged routine safety drills and open discussion. Getting everyone on the same page, from newcomers to old hands, keeps trouble out of the lab. Updating storage logs, labeling secondary containers if a transfer occurs, and reporting any broken seals or odd odors right away helps catch small problems early.
For storage rooms that struggle with heat, ventilation upgrades make life easier. Installing fans or better air exchangers keeps temperatures down and discourages vapor build-up. For small labs with limited budgets, even a simple thermometer and clear instructions about maximum allowable temperatures (usually below 25°C) improves safety.
The Value of Vigilance
I’ve learned from years of experience that small steps—tight seals, regular labeling, proper cabinets—matter far more than fancy technology or paperwork. Respect the hazards, take time to do things right, and 2-Methyl-3-Pentanol turns from a source of worry to a reliable part of the lab. Safety habits built up with routine, honest discussion, and shared responsibility make all the difference. Chemical work doesn’t forgive shortcuts, and proper storage of solvents is a lesson worth repeating.
Is 2-Methyl-3-Pentanol soluble in water?
What’s in the Mix: 2-Methyl-3-Pentanol and Water
Chemistry gives every molecule some character. 2-Methyl-3-pentanol, with its five carbon backbone and an -OH group shoved onto the third carbon, stands as an example. Picture this liquid as something with an alcohol functional group similar to isoamyl alcohol or isobutanol, but with a slightly different twist.Years of lab experience with small alcohol molecules have made one thing clear: structure dictates solubility. If an alcohol packs more carbons, it grows less likely to slip comfortably into water. Water’s got a thing for hydrogen bonding, so the more hydrocarbon chains stick out, the more water loses interest.
Solubility Isn’t Binary, and Here’s Why
Some online references toss out numbers, but anyone making solutions in real life knows that “soluble” isn’t always a yes-or-no situation. Take 2-methyl-3-pentanol. The methyl group bumps up its hydrophobic nature. That means its tail doesn’t mix well in a water crowd, unlike ethanol which gets along smoothly.
Lab manuals cite solubility values—sometimes around 12 grams per liter for similar branched alcohols. 2-Methyl-3-pentanol likely hovers near this mark. Compare that to 100% solubility for ethanol and you’ll see this alcohol falls closer to the “barely mixes” camp. In practice, try dissolving a spoonful in a glass of water, and you’ll notice oily droplets float to the top or sit at the bottom if you add enough.
Why This Matters Beyond a Textbook
Many researchers hit roadblocks with solubility every day. I’ve had plenty of reactions stall out or crystallize at the bottom of a flask, all because an alcohol like this didn’t play nice with water. Safety records matter, too. Mistakes around mixing compounds with low solubility lead to volatile layers or tricky extractions, not to mention headaches during cleanup.
Industries relying on precise chemistry—pharmaceuticals, flavors, specialty solvents—look at solubility charts before every batch. If 2-methyl-3-pentanol won’t dissolve, that signals design changes in both formulation and equipment. People handling flavor analysis know that something poorly soluble in water will struggle to deliver consistent results in a beverage or food sample.
Lessons and Workarounds for the Chemically Curious
Instead of wishing a molecule more soluble, chemists pull a few tricks. Sometimes a small dash of ethanol or another co-solvent lets 2-methyl-3-pentanol blend in just enough to do its job. Heat usually bumps up solubility, but too much temperature introduces its own risks: unexpected reactions, increased volatility, and safety worries.
On the flip side, if someone needs this alcohol not to dissolve, its moderate immiscibility can come in handy. Look at separation techniques, such as liquid-liquid extraction—the less it dissolves, the easier certain extractions run. This opens up flexibility in designing purifications for both research and production.
Connecting Chemistry to Real-World Decisions
Chemical information means very little until it’s tested on the bench or evaluated for product safety. According to PubChem and standard MSDS data, 2-methyl-3-pentanol only grudgingly blends with water. For everyday lab techs, this lays out the practical limits. Scale-up engineers, flavorists, and students can head into their work informed and ready to solve the solubility puzzle, one careful step at a time.
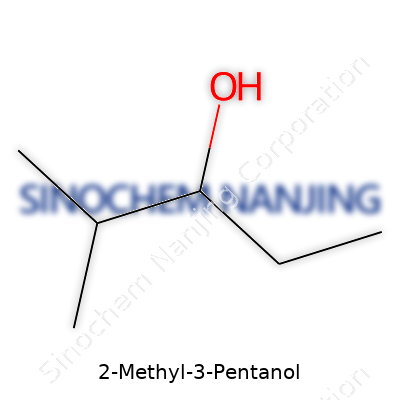
| Names | |
| Preferred IUPAC name | 3-Methylpentan-2-ol |
| Other names |
Ethyl isopropyl carbinol Ethyl isopropylmethanol 2-Methylpentan-3-ol |
| Pronunciation | /tuː ˈmɛθɪl θriː ˈpɛntənɒl/ |
| Identifiers | |
| CAS Number | 565-69-5 |
| 3D model (JSmol) | `JSmol|/HR5ESf5vCwP0QfMIXQZme1T6+V0t1Ik5+pUvNbK/CtYQgQki71R+xVQ1BM8DT6vKrrfE0kP7FpC18m0tHbdDg14Q6gttYVOlJawVSKuGInxjeRGna43EIBgzHuLlHotE5T7V6czS4QhIwTZPIYOvTo95OfzmiEJeZVrDCTnhgypKekJy5o+1OtSWT8g==` |
| Beilstein Reference | 1718731 |
| ChEBI | CHEBI:34240 |
| ChEMBL | CHEMBL15619 |
| ChemSpider | 6016 |
| DrugBank | DB04260 |
| ECHA InfoCard | 01c9b397-26a2-4aea-96ed-32f1e90aa155 |
| EC Number | 603-008-00-9 |
| Gmelin Reference | 80882 |
| KEGG | C06250 |
| MeSH | D010469 |
| PubChem CID | 11656 |
| RTECS number | SA9100000 |
| UNII | N9A422L09S |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID5050857 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Colorless liquid |
| Odor | mild alcohol-like |
| Density | 0.815 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 0.9 |
| Vapor pressure | 0.243 mmHg (at 25 °C) |
| Acidity (pKa) | 16.1 |
| Basicity (pKb) | pKb: 5.44 |
| Magnetic susceptibility (χ) | -7.46 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | n20/D 1.417 |
| Viscosity | 3.98 mPa·s (20°C) |
| Dipole moment | 2.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 335.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -345.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3906.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 56 °C (133 °F; 329 K) |
| Autoignition temperature | 411 °C (772 °F; 684 K) |
| Explosive limits | 5.3–10.3% |
| Lethal dose or concentration | Lethal dose or concentration: **LD50 (oral, rat) 5660 mg/kg** |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 4300 mg/kg |
| NIOSH | SA9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended): 100 ppm (360 mg/m3) |
| Related compounds | |
| Related compounds |
2-Methyl-2-pentanol 3-Methyl-3-pentanol 3-Methyl-2-pentanol 2-Pentanol 3-Pentanol 2-Methyl-1-pentanol |

