2-Methyl-2-Pentanol: An Evolving Industrial Ally
Historical Development
Looking back, the story of 2-Methyl-2-Pentanol stretches across a long period of chemical discovery. Chemical workers first identified branched chain alcohols over a century ago as organic chemistry picked up steam. Industrial laboratories in Europe mapped out new alcohols, examining both their structure and reactivity, with 2-Methyl-2-Pentanol standing out as a promising target. Its unusual six-carbon chain with a methyl group resting on the second carbon brought early speculation about unique physical and chemical behavior. Through improved distillation and separation practices, especially those developed in the 20th century, chemists started to see clearer routes for isolating specific branched alcohols. Over time, as chemical manufacturing expanded, the demand for intermediate solvents called for materials with specific boiling and solubility properties, pushing 2-Methyl-2-Pentanol into larger-scale production.
Product Overview
2-Methyl-2-Pentanol emerges as a colorless liquid, with a mild, somewhat camphor-like scent that hints at its chemical nature. Its identifying number in the CAS registry is 625-33-2. Chemists in laboratories often make use of its moderate polarity and decent solvation capacity. Having a branched chain structure, the compound blends many features of lower and higher alcohols. Many know it best as a versatile intermediate, particularly in the paint, coatings, and solvent sectors. Each day in manufacturing plants, workers rely on it as a building block, integrating it in both finished goods and in process streams.
Physical & Chemical Properties
Boiling near 143°C and offering a melting point around -60°C, 2-Methyl-2-Pentanol sits comfortably as a liquid under ambient conditions. Those in the lab find its moderate solubility in water useful, but its real value shines through its ability to dissolve both polar and non-polar substances. The presence of a tertiary alcohol group means it’s less prone to oxidation than primary or secondary alcohols. This brings safety advantages and limits unwanted side reactions in certain applications. Its vapor pressure makes it less volatile than many lighter alcohols, offering better handling in large volume operations.
Technical Specifications & Labeling
Suppliers tend to grade 2-Methyl-2-Pentanol by purity, with the chemical trade favoring lots listed at 98% or higher. The liquid appears as a clear or slightly hazy substance if improperly stored, so maintaining sealed containers matters. Safety data sheets remind users to check flash points and compatible materials, since alcohols often bring both solvent power and flammability dangers to workspaces. Labels usually highlight storage temperatures and necessary personal protection due to its potential irritancy.
Preparation Method
Industrially, 2-Methyl-2-Pentanol comes from the hydration of alkenes. One common approach draws from the acid-catalyzed hydration of 2-methyl-2-pentene, using traditional strong acid catalysis. This reaction directs water across the double bond, sticking to Markovnikov’s rule, so the resulting hydroxyl group ends up at a tertiary position. Another way employs hydroboration-oxidation, which offers more selectivity, though it appeals mostly to smaller operations given the extra cost and reagent requirements. The chemical process fits well with existing infrastructure in plants already making branched alcohols for the flavor, fragrance, and industrial chemical industries.
Chemical Reactions & Modifications
Chemists prize tertiary alcohols like this one for their predictable reactivity. In the lab, dehydration comes easily, making it possible to produce the corresponding alkene. Its structure resists most oxidation attempts, since the tertiary carbon has no hydrogen available to remove, protecting it from transformation into a ketone or aldehyde. Under strong acids, 2-Methyl-2-Pentanol forms esters when paired with acid chlorides, expanding its reach into plasticizer and fragrance ingredient territory. Some research has explored halogenation at the methyl position, though steric hindrance keeps modification rates moderate. Its relative inertness to oxidation and mild reactivity keep it attractive as both a solvent and as a process intermediate for specialty syntheses.
Synonyms & Product Names
2-Methyl-2-Pentanol appears in chemical catalogs and papers under a roster of alternate names. The most recognized synonyms include tert-Hexanol, 2-Methylpentan-2-ol, and amyl methyl carbinol. In some uses, buyers know it simply by its molecular formula, C6H14O, or by roster listings for batch production. Local distributors outside of North America may supply it under varied translations or abbreviations, though regulatory agencies still enforce the core chemical identifiers.
Safety & Operational Standards
Working with 2-Methyl-2-Pentanol, people need to respect both health and fire hazards. The compound’s low to moderate vapor pressure means it can escape containment, and breathing its vapors can irritate the throat and eyes. Extended skin contact may strip oils and trigger sensitivity, so workers depend on gloves and local exhaust ventilation. In plant settings, managers review flammability risks, assigning safe work zones and equipping storage areas with grounding and appropriate extinguishers. Regulatory agencies from OSHA to the ECHA issue exposure limits and labeling guides to keep incidents at bay. Training staff on spill response, proper transfer, and personal protective equipment sets the foundation for safe operations, reducing both acute and chronic harm.
Application Area
Most demand for 2-Methyl-2-Pentanol flows into the coatings, paints, and ink sectors, where it acts as a specialty solvent. Its ability to thin resinous materials without causing phase separation solves problems many formulators encounter with more traditional alcohols. Product developers in adhesives, plasticizers, and even metal extraction identify 2-Methyl-2-Pentanol as a useful building block. In some specialty flavors and fragrances, the compound finds a limited role, though regulatory limits on toxicity keep its use tightly constrained. The production of esters and ethers, particularly for fuel and high-value intermediates, draws industrial buyers as the chemical sector searches for molecules that fill both functional and regulatory gaps.
Research & Development
Universities and company labs haven’t overlooked the value of understanding both safety and synthetic routes for branched alcohols. Projects focus on finding cleaner catalysts and milder conditions that reduce byproducts and streamline waste treatment. Chemists also scan for greener hydration methods, hoping to replace mineral acid catalysis with benign alternatives. On another front, process safety research examines temperature and pressure tolerances for bulk handling, developing safer vessel linings and transfer techniques. Toxicologists and environmental scientists contribute by tracing how these chemicals break down after use, mapping pathways to keep waterways and soils protected from accidental releases.
Toxicity Research
Toxicity studies of 2-Methyl-2-Pentanol underscore some real risks of exposure. Workers who inhale high vapor concentrations may suffer respiratory tract irritation, and contact with skin can lead to dryness or redness. Animal studies suggest moderate central nervous system effects at excessive exposures, underlining the need for careful engineering controls in workplaces. Regulatory bodies often draw a line for permitted airborne levels in occupational settings, keeping chronic effects in check for those performing repeated tasks. Environmental impact assessments focus on breakdown rates and aquatic toxicity, with most studies pointing toward manageable risks when releases get caught and managed by routine environmental controls. Research continues on understanding longer-term impacts, with current practices relying on containment and rapid response to spills.
Future Prospects
Industry eyes a future where 2-Methyl-2-Pentanol remains essential in processes requiring stable, non-reactive solvents. As regulatory standards tighten, chemical plants upgrade equipment and processes to recycle more solvent streams, cut emissions, and protect workers. Producers test new catalysts and fermentation-based production methods, hoping to lessen fossil fuel dependencies and drive greener supply chains. On the product side, formulators work to replace more hazardous solvents with tertiary alcohols like this one, aiming for comparable performance but with improved safety records. Ongoing research into its environmental fate and toxicology stands to shape where and how much 2-Methyl-2-Pentanol features in the next wave of industrial solutions. The chemical’s unique structure, robust performance, and broad compatibility keep it on the radar for both established industry players and startups working on next-generation materials.
What are the main uses of 2-Methyl-2-Pentanol?
Why 2-Methyl-2-Pentanol Matters in Industry
Tucked inside plenty of labs and factories is a clear, oily liquid with a faint, alcohol-like smell: 2-Methyl-2-Pentanol. Even if the name feels a bit forgettable, this compound keeps showing up in real work, not just chemistry books. My first experience with it came in an industrial coatings plant, where the technical lead explained how reliable solvents keep the lines moving—and avoid messes that slow us down. 2-Methyl-2-Pentanol played a part in that day, doing work that went unnoticed by most except when it ran out.
The Solvent Workhorse
This alcohol finds its groove as a solvent. In paint factories, ink production lines, or during the creation of specialty coatings, 2-Methyl-2-Pentanol helps dissolve pigments and resins, cleaning up the process and boosting consistency. Its molecular shape gives it a balance—strong enough to break down tough substances, mild enough to avoid damaging sensitive ingredients. That means manufacturers count on it during the blending of certain lacquers, adhesives, and various industrial and household cleaners.
Chemical Synthesis and Reactions
2-Methyl-2-Pentanol isn't just a background player—it’s an ingredient in making other chemicals too. Organic chemists in both small labs and large plants use it as a building block to shape more complex molecules. Producing plasticizers, fragrances, or pharmaceutical intermediates, this alcohol steps in to start, support, or speed up the reactions. It also acts as a reagent, meaning the chemistry would stall without a liquid that brings reactants together at the right speed.
Applications in Pharmaceuticals and Agrochemicals
In pharma and crop science, purity standards run high and process tweaks never stop. Manufacturers turn to 2-Methyl-2-Pentanol as both a solvent and an extraction agent, especially in drug formulation or when purifying active ingredients. Its performance makes it an option in agrochemical synthesis too, helping formulate and clean up the growth boosters or pest control agents that shape yields. Many people see only the finished pill or spray, but this alcohol had a hand in refining what ends up in the field or the pharmacy.
Learning from Experience: Safety and Sustainability
On the plant floor, safety talks always meant reviewing the Material Safety Data Sheet. When it came to 2-Methyl-2-Pentanol, skin protection and good ventilation stood out. The workers knew what dizziness felt like and shared their stories so newcomers understood the risks. Regulatory reviews track how companies store and dispose of such chemicals. For anyone looking ahead, greener solvents offer ways to cut emissions and protect workers. Experience showed us that a sharp focus on handling and substitution leads to better long-term results.
Room for Improvement
Every batch of coating or glass cleaner that uses 2-Methyl-2-Pentanol draws from resources and leaves a trace. Companies can look into reclaiming solvents on-site, investing in updated purification systems, or working directly with solvent producers to roll out alternatives with reduced toxicity. Training shifts and consistent audits keep chemical spills and exposure in check. Open conversations on the shop floor about chemical use spark ideas for less waste and safer environments.
Final Thoughts
2-Methyl-2-Pentanol quietly shapes industries behind the scenes. From dissolving tough compounds to holding its own as a reactant, its role stretches across products many of us rely on. Paying attention to how it’s handled—both for safety and sustainability—influences everything from worker health to product quality. The real work comes from people on the ground, taking what’s learned every day to build safer ways forward.
What is the chemical formula of 2-Methyl-2-Pentanol?
Understanding the Formula
2-Methyl-2-pentanol isn’t just another mouthful of a chemical name—it stands for a molecule many chemists and industry professionals might come across, even if most of us rarely hear about it outside textbooks and laboratories. The formula for 2-Methyl-2-pentanol is C6H14O. That's six carbon atoms, fourteen hydrogens, and a single oxygen. This specific layout means the molecule qualifies as a tertiary alcohol, rather than a primary or secondary one, so its structure and behavior offer something slightly different compared to other alcohols.
The Layout and Real-World Implications
Naming in organic chemistry works like a puzzle—every piece points to something specific. In this case, “2-Methyl-2-pentanol” tells you there’s a pentane skeleton (five carbons in a chain), with a methyl group stuck onto the second carbon and a hydroxyl group also attached to that same second carbon. Choosing the correct arrangement isn’t just for chemistry contests or university labs. It has real consequences for properties like solubility, boiling point, and reactivity. This is more than just trivia; small changes in a molecular structure, like shifting a methyl group, can make the difference between a harmless solvent and a dangerous toxin.
My experience working at a university chemical stockroom drilled home how easy it is to mix up compounds with nearly identical names. 2-Methyl-2-pentanol and its isomers can cause confusion, and that can mean mistakes in research or manufacturing. Effective labeling and precise language reduce risk in academic and industrial settings. Just imagine scaling up a reaction for a manufacturing process and using the wrong compound because of a simple oversight—that can cost time, money, and sometimes safety.
Industrial and Research Importance
This molecule isn’t made on a massive scale for the public, but it holds value for specific chemical syntheses and acts as a solvent or intermediate for certain reactions. Its tertiary alcohol structure gives it resistance to oxidation, unlike its primary or secondary cousins. Chemists appreciate this trait when they want a reaction that leaves the alcohol alone.
2-Methyl-2-pentanol acts as a good example of how structure steers function. Because the hydroxyl group connects to a carbon bonded to three other carbons, the molecule’s bulkiness means it dissolves differently in water compared to more streamlined alcohols. So, anyone working in a lab or designing industrial cleaning products needs to keep these details in mind. Even if mass production isn’t the norm, niche applications demand precision, which builds trust, especially in regulated industries.
Looking at Solutions and Smart Choices
Mix-ups with similar chemical names or formulas can seem small, but they spiral into big challenges for research teams, laboratory managers, and manufacturers. One way out involves double-checking stockroom inventories, using barcode or digital tracking, and giving ongoing training in both nomenclature and chemical safety. Labeling solutions should use bold, legible fonts and highlight any dangerous confusion possibilities.
Schools and workplaces can work together to highlight how these formulas affect everything from experimental outcomes to health and safety. It's not enough to memorize the chemical formula. Understanding what C6H14O means floored me with respect for the way chemistry shapes everything people design or manufacture. The next time someone sees a simple formula, it won’t just look like a collection of letters and numbers—it’ll represent a map of unique traits and choices, each with direct consequences, both inside the laboratory and out in the world.
Is 2-Methyl-2-Pentanol hazardous or toxic?
Real-World Contact Raises Real Questions
Most people never hear about 2-Methyl-2-Pentanol unless they work with solvents or specialized chemicals. It shows up in labs, industrial settings, and sometimes in products meant for cleaning or processing. Folks who handle it pay extra attention because it’s an alcohol, and like many chemicals in this group, it doesn’t come without caution labels. I remember seeing it on a material safety sheet and wondering how risky it really was.
Health Risks Stand Out
Anyone who’s read a label knows this stuff isn’t a soft drink ingredient. Breathing in its vapors can make people dizzy or give them headaches. More serious exposures don’t just mean discomfort; they might cause nausea, poor coordination, and worse if you keep breathing it or spill a lot on your skin. On one occasion, a colleague found, the burning sensation on exposed skin lasted hours. Nobody enjoys a chemical rash, and with solvents like this, that risk turns real.
Eye and Skin Contact Brings Trouble
Drops of 2-Methyl-2-Pentanol in your eyes sting fast. In factories, workers use goggles or face shields, which tells you something. The chemical seeps through skin as well, reaching the blood when there’s too much contact. Strong controls in workplaces point to the risks not being theoretical. Even the government’s chemical safety resources say long-term exposure hasn’t been studied enough to rule out other effects.
Breathing It in Isn’t Safe
The nose picks up its sharp scent before it does the lungs any good. In confined areas, fumes build up. Repeated inhalation can slow reflexes and cause clouded thinking. On job sites, ventilation matters much more than people realize until someone gets woozy one afternoon. Sometimes warnings about nausea or dizziness sound basic, but I’ve watched new workers learn the hard way. Safety data always stress proper airflow.
Drinking It Would Be Dangerous
Curiosity or error—either way, swallowing lab alcohols like this never ends well. As a close cousin to more notorious industrial alcohols, this one can hit the organs just as hard. Ingesting isn't just unwise; it brings real risk of nervous system depression and even organ damage. There are better ways to learn chemistry than gulping solvents that could cause permanent injury or, in worst cases, death.
Protecting Ourselves and Our Communities
Working around industrial chemicals demands focus and respect. For 2-Methyl-2-Pentanol, solid rules like gloves, eyewear, and ventilation become non-negotiable, not just suggestions from a safety poster. Companies ought to give better training, because a ten-minute talk can mean the difference between a close call and a trip to the ER. Stronger labels on both bottles and bulk containers also help. Most accidents I’ve seen came down to not realizing what was in a container or not having the right gear handy.
Room for Better Oversight
Regulators track chemicals with names like this one, since misuse or spills could sneak into the environment, with effects nobody fully predicts. Giving workers and neighborhood residents access to safety information is just as important as wearing gloves. Transparency puts pressure on companies to take exposure seriously, not just for workers but for everyone living near these facilities.
Informed Choices Matter
It’s easy to shrug off warnings or assume nothing bad happens with routine use. Real stories show otherwise. Those of us who’ve spent time around hazardous materials have seen what can go wrong and know that respect and vigilance should never take a day off, especially with chemicals where long-term effects remain uncertain.
How should 2-Methyl-2-Pentanol be stored?
Why 2-Methyl-2-Pentanol Demands Respect
2-Methyl-2-pentanol isn’t in every household, but anybody working in a lab or chemical warehouse will run across it sooner or later. This clear liquid comes with a mild, almost sweet odor, offering value for specialty coatings, fragrance blends, and solvents. Despite sounding benign, it actually brings a real risk if treated lightly. Exposure to high concentrations can irritate the eyes, skin, and airways, and some studies show central nervous system effects if mishandled. Even if you’re careful, one mistake can derail an operation, raise health questions, or spark an OSHA visit.
Storage Conditions Speak Louder Than Warnings
True safety starts with clean routines. A bottle on a cluttered shelf, a leaky cap, or storage near heat spells trouble for any alcohol, especially this one. My time in a chemical plant hammered home the lesson—someone always thought, “Just for now.” Soon enough, fumes leaked, confusion followed, and the blame circled. Keeping things simple works best: cool, dry, and locked away.
2-Methyl-2-pentanol demands a tightly sealed container, preferably built from high-density polyethylene (HDPE) or glass. Skip anything with unknown plastics, since alcohols eat through the wrong materials quicker than most expect. Always label containers—not with faded tape, but bold, clear writing. Anyone who’s walked into a room of crystalline and colorless chemicals knows how fast a mislabel turns into chaos.
Ventilation and Temperature: Non-Negotiable Parts
Stale air in a storage room shouldn’t be an option. Fumes build up quietly, especially when the temperature climbs. Store this alcohol away from heat sources and direct sunlight because high temps raise pressure inside the bottle, making leaks and even ruptures more likely. Don’t tuck it near oxidizing agents or acids—mix-ups create reactive messes and fire risks.
I still remember a minor incident in our storeroom where a heating vent malfunctioned one weekend, baking a shelf near hazardous liquids. The mess cost us hours of cleanup and some real anxiety. Spending a bit more upfront on temperature monitoring and ventilation pays for itself tenfold the moment something malfunctions.
Fire: A Real Risk, Not Theory
2-Methyl-2-pentanol takes a spark seriously. Its flash point hovers fairly high compared to other alcohols, but open flames or nearby equipment errors can get out of hand. Keep a fire extinguisher—Class B, preferably—close at hand. Post signs forbidding smoking or open flames, and run reminder drills. In my experience, most people overestimate their memory in an emergency; muscle memory matters more than policy posters.
Access and Oversight: Building Accountability
Keep keys tight. Only trained staff should handle storage areas. Rotate stock to ensure old containers don’t turn into time bombs. Log every movement—digital tracking works best in big facilities, but even a well-maintained notebook does the trick. Random checks catch forgotten leaks or inching containers before small problems grow.
Practical Training Beats Long Manuals
Reading about rules only goes so far. Hands-on walkthroughs, quick reminders, and encouraging questions keep safety living, not just written. If you notice someone skipping checks, call it out with respect. Consistent reminders show new staff what right looks like, even if the lesson gets repeated a half-dozen times.
Final Thoughts: Value Your Team’s Health
People trust coworkers with their safety every day, whether they think about it or not. By treating 2-methyl-2-pentanol storage seriously, you signal respect for those around you. Even in facilities pressed for time, skipping good storage and basic oversight only pays in regret. Simple habits and sturdy tools hold the line between routine work and accidents that change lives.
What is the boiling point of 2-Methyl-2-Pentanol?
Why Boiling Points Matter Beyond the Lab
Ask any seasoned chemist about reliable data, and the discussion always circles back to boiling points. These numbers aren’t just trivia for classroom quizzes; they guide major decisions, especially where safety and quality control take the spotlight. With so many chemicals floating around in industrial, academic, and even personal projects, if you can’t trust your boiling point—trouble isn’t far behind.
The Details: 2-Methyl-2-Pentanol's Boiling Point
2-Methyl-2-pentanol sounds like another anonymous compound. In reality, it fills a unique space as a branched-chain alcohol, a group of chemicals used for everything from organic synthesis to custom solvent systems. Its boiling point sits at about 120°C (248°F). Not the hottest number in the alcohol world, but hot enough to command respect.
This isn’t just a value pulled from a dusty textbook. Referencing sources such as the CRC Handbook of Chemistry and Physics, peer-reviewed journals, and the National Institute of Standards and Technology, you’ll see that number crop up with little room for debate. Bring a sample into a well-calibrated lab, and that figure holds up.
Real-World Impact
A boiling point of 120°C guides both handling and application. During my years managing a chemical storeroom for an underfunded university program, details like these separated safe storage from disaster. Its volatility steers the need for proper containers, good ventilation, and practical respect in the fume hood. Mix-ups—wrong data on a chemical sheet or a misread when prepping a separation procedure—invite both wasted time and safety risks. That kind of mistake doesn’t just cost money; it can put colleagues’ health on the line.
Manufacturers preparing solvents or specialty coatings benefit from a precise boiling point. Mismatched values for 2-methyl-2-pentanol could upset formulations, bake off at wrong temperatures, or trigger unsafe pressure shifts when using closed systems. I’ve sat with older machinists who prefer their data printed, circled in red, and taped above the distillation hotplate. The trust in numbers serves as a foundation for both productivity and peace of mind.
Improving Safety and Trust
Safe chemical handling depends on accurate, accessible reference data. Digital tools now make it easier for everyone from high schoolers to professional chemists to look up values instantly. Nonetheless, sometimes hardbound reference books still come out, especially during audits or when double-checking online resources for odd entries or outdated numbers.
Regular audits and proper labeling help prevent disasters. During a summer at local fire marshal’s inspections, we shifted every bottle into fireproof cabinets, checked against updated chemical inventory lists, and made sure any reference to 2-methyl-2-pentanol’s boiling point matched current standards. This tedious step turned out to be worth it—a mislabeled sample got flagged and corrected before it ended up in an incorrect experiment setup.
Finding Better Solutions
Training holds as much value as data. Building a team that respects boiling points and physical parameters often means fewer accidents and smoother workflow. Printed charts, hands-on demonstrations, regular safety meetings—these sit at the core of a responsible chemical workplace. Technology can help, but a mindful approach remains irreplaceable.
Boiling points aren’t lifeless statistics. They reflect years of careful experimentation, teamwork, and a genuine concern for human wellbeing. Whether in a multi-million dollar production facility or a cramped student lab, respect for numbers such as the boiling point of 2-methyl-2-pentanol saves both dollars and lives.
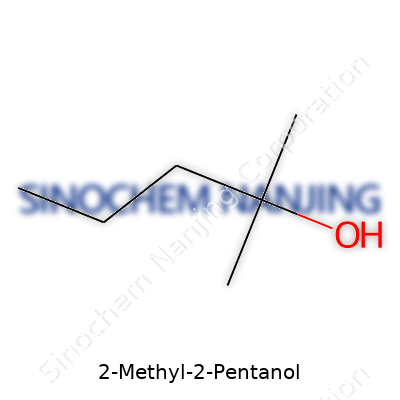
| Names | |
| Preferred IUPAC name | 2-Methylpentan-2-ol |
| Other names |
2-Methylpentan-2-ol Dimethyl n-propyl carbinol Methyl-2-pentanol Methylsecbutylcarbinol 2-Methylpentanol-2 |
| Pronunciation | /tuː ˈmɛθəl tuː ˈpɛntənɒl/ |
| Identifiers | |
| CAS Number | 625-18-5 |
| Beilstein Reference | 1720516 |
| ChEBI | CHEBI:34211 |
| ChEMBL | CHEMBL16337 |
| ChemSpider | 15410 |
| DrugBank | DB14634 |
| ECHA InfoCard | 100.118.246 |
| EC Number | 203-458-9 |
| Gmelin Reference | 7873 |
| KEGG | C06358 |
| MeSH | D010470 |
| PubChem CID | 11574 |
| RTECS number | SA9100000 |
| UNII | EC2M3L2ZOY |
| UN number | UN 2053 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Colorless liquid |
| Odor | mild odor |
| Density | 0.814 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.76 |
| Vapor pressure | 0.89 mmHg (at 25 °C) |
| Acidity (pKa) | 16.1 |
| Basicity (pKb) | pKb ≈ 15.2 |
| Magnetic susceptibility (χ) | -7.71×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.409 |
| Viscosity | 6.1 mPa·s (20°C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -389.75 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3905.9 kJ/mol |
| Pharmacology | |
| ATC code | N07BB03 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378 |
| NFPA 704 (fire diamond) | '1-3-0' |
| Flash point | Flash point: 49 °C |
| Autoignition temperature | 411 °C (772 °F; 684 K) |
| Explosive limits | 1.2–8.6% |
| Lethal dose or concentration | LD50 oral rat 2460 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 2500 mg/kg |
| NIOSH | SA9125000 |
| PEL (Permissible) | PEL: 100 ppm (410 mg/m³) |
| REL (Recommended) | REL (Recommended Exposure Limit) of 2-Methyl-2-Pentanol is "100 ppm (360 mg/m3) TWA". |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
1-Pentanol 2-Pentanol 3-Pentanol 2-Methyl-1-pentanol 3-Methyl-2-pentanol |

