2-Methyl-1-Propanol: A Closer Look at Its Past, Present, and Future
Historical Development
2-Methyl-1-propanol doesn’t get a lot of attention outside the chemistry world, but dig a little deeper and you’ll find a useful compound with a slightly quirky past. Chemists started isolating and describing this alcohol more than a century ago, as advances in organic chemistry peeled back the layers of hydrocarbon structures. Its discovery traces back to the broader investigation of isomers, with early researchers examining how shifting just one methyl group could give rise to unique chemical personalities. Back then, most labs didn’t have fancy equipment or analytical software. Simple distillation, crystallization, and keen observation ruled the day, and someone noticed that this particular molecule offered distinct properties compared to its cousins. Over time, 2-Methyl-1-propanol morphed from a curiosity into a key ingredient in the development of solvents, flavors, and fuel additives.
Product Overview
Chemists know 2-Methyl-1-propanol as isobutanol. It’s a clear, colorless liquid with a characteristic smell that’s tough to forget once you’ve encountered it in the lab. Unlike its straight-chain relative n-butanol, isobutanol tacks on its methyl group at a different carbon, which shifts its boiling point and affects how it’s used. Factories keep tanks of this alcohol around for use in manufacturing flavors, fragrances, coatings, and plasticizers. As demand for biofuels and sustainable chemicals climbs, companies are looking at isobutanol as a more environmentally friendly building block compared to fossil-based constituents.
Physical & Chemical Properties
2-Methyl-1-propanol carries the formula C4H10O and weighs just over 74 g/mol. Pour it out and you see a liquid that mixes well with most organic solvents and some water. The boiling point sits around 108°C, so in my grad school days, you could distill it in an afternoon. The structure grants it lower solubility in water than n-butanol and a distinct place on the polarity spectrum. Flammability stays front and center, setting off the concern that comes with most lower-chain alcohols. Chemists appreciate how it resists unwanted side reactions compared to other butanols when used as a solvent.
Technical Specifications & Labeling
If you visit a chemical supply warehouse, you’ll notice bottles labeled with its CAS number 78-83-1. Expect clear labeling to include warnings about flammability along with purity percentages, often 99% or higher for lab use. Manufacturers test for the presence of water, acidity, and other organic contaminants using both gas chromatography and titration. Packaging typically ranges from liters in glass bottles for research to drums for industrial purposes, with each container covered by safety pictograms. Over the years, the industry has pushed for harmonized hazard communication—big, bold warning signs and standardized labeling formats reflect that trend, which deserves applause.
Preparation Method
Large-scale producers have moved away from old-school synthesis from petroleum fractions, focused instead on fermentation-based approaches using engineered microbes. These microbes convert carbohydrates—think corn or plant biomass—into isobutanol through engineered biochemical pathways. In the lab, simple hydroformylation of propylene followed by hydrogenation gives an efficient route. I’ve seen both methods firsthand, and the shift to bio-based production doesn’t just make sense for sustainability; it also helps buffer companies against oil price swings. The fermentation approach inspired plenty of start-ups promising “green solvents,” and though not all survived, it’s a sign of where the field is heading.
Chemical Reactions & Modifications
Isobutanol lends itself to a range of classic organic reactions. Chemists frequently oxidize it to isobutyraldehyde or isobutyric acid. Esterification brings out more commercial value, giving rise to isobutyl acetate—a common solvent and flavor component. In undergraduate labs, I watched folks react isobutanol with various acids to form esters with pleasant, fruity aromas. Alkylation and dehydration produce olefins, which lead to synthetic rubbers and specialty chemicals. The molecule’s branching means it doesn’t react the same way as n-butanol or sec-butanol, opening doors to tailored chemical products and innovative materials.
Synonyms & Product Names
2-Methyl-1-propanol isn’t the only name you’ll run across. The most familiar alias remains isobutanol, but dig into chemical catalogs and you’ll also spot terms like isobutyl alcohol and methylpropanol. These synonyms crop up in both academic literature and product invoices, which sometimes confuses newcomers unless they double-check CAS numbers. I’ve lost count of the number of times I’ve seen research students pull the wrong bottle, underscoring the value of education and clear labeling.
Safety & Operational Standards
In every chemical plant and teaching lab, isobutanol gets treated with the respect it deserves. OSHA classifies it as a flammable liquid, so static discharge, open flames, and heat sources stay far from storage areas. People working with it receive training on splash hazards and inhalation risks, with gloves and goggles as the norm. Engineering controls—fume hoods, vented storage, fire suppression systems—form the backbone of safe operations. Regulation continues to evolve, and I remember annual safety briefings emphasizing the importance of up-to-date safety data sheets and incident reporting. These standards don’t just protect workers; they keep operations running smoothly.
Application Area
Isobutanol plays a much bigger role behind the scenes than most people realize. Manufacturers use it to make paints, coatings, and lacquers more workable—cutting down on streaks and extending drying times for even coverage. The flavor and fragrance sector taps into its esters for fruit notes in chewing gum and soda. As the world searches for clean energy, researchers give it another look as a gasoline additive or direct fuel, thanks to high energy content and low vapor pressure. Its presence in plastics, pharmaceuticals, and adhesives reflects a reach that spans across industries. When companies want to reduce oil use, bio-based isobutanol offers an attractive, homegrown option.
Research & Development
Research on 2-Methyl-1-propanol has ramped up in recent decades, with national labs and private enterprises hunting for more sustainable chemical feedstocks. Advances in synthetic biology drive improvements in fermentation strains, making renewable isobutanol more competitive. Universities run pilot projects that measure both output and carbon reduction, pushing academic teams to break new ground in process optimization. On the applied side, paint formulators and coating specialists strive to reduce volatile organic compound content without giving up quality, and isobutanol anchors many of those efforts. Seeing the constant churn of patents filed each year reassures me that companies continue betting on its broad promise.
Toxicity Research
Toxicologists don’t take any short cuts when looking into isobutanol’s safety. Animal studies map long-term exposure risks, examining effects on the liver, kidneys, and nervous system. Regulatory bodies continue to update workplace guidelines based on these findings, limiting permissible exposure. While acute contact can bring on headaches, dizziness, or nausea, proper handling practices blunt most health risks. In my own work, structured training programs taught staff at every level to recognize early warning signs. Ongoing studies look out for any gaps in our understanding, keeping the focus on prevention, not just response.
Future Prospects
The future of 2-Methyl-1-propanol looks promising, especially with pressure mounting to decarbonize the chemical industry. With tweaks to microbial production methods and end-user demand for green chemistry, isobutanol seems set for broader use as both a feedstock and a biofuel. Upcoming research focuses on driving down costs, boosting output yields, and shrinking the environmental footprint from farm to factory. My gut tells me we’ll see even more creative uses for isobutanol descend from the lab bench to the warehouse. If governments keep tightening emission standards, products built on this versatile alcohol will find a solid place in everything from next-gen fuels to sustainable materials.
What is 2-Methyl-1-Propanol used for?
A Closer Look at a Common Chemical
Walk through a busy chemical plant, and the sharp scent of alcohol isn’t always a sign of beverages. In those halls, 2-methyl-1-propanol earns its place. This clear liquid, also called isobutanol, gets tucked into more products than folks realize, and there’s a reason big industries keep ordering more. From my time in manufacturing and working with chemical suppliers, I’ve seen it sitting next to the larger drums of solvents, waiting for its turn in the process. Yet most people outside the industry rarely hear its name.
Fueling Industry Beyond Gasoline
Isobutanol gets plenty of attention in the fuel business. Gasoline blenders pick it for its knack for boosting octane levels while helping to prevent engines from knocking. Energy companies see an answer to the ethanol blend wall, since this alcohol works in engines built for ethanol but doesn’t soak up water from the air as fast. Tests show it can blend into gasoline up to 16% by volume, higher than ethanol, without messing up engine performance. It stays stable sitting in storage tanks and pipes, which simplifies logistics.
Biofuel developers look at it as more than a gasoline extender. Some startups use microbes to brew it from corn mash or waste wood, creating a renewable fuel that matches the energy content of regular gasoline more closely than ethanol. That energy density matters, especially to businesses running fleets or anyone tired of filling up more often.
Solvents and Coatings: Where Versatility Counts
Artists reach for isobutanol without knowing its full story, as it helps paint flows finish smoothly. Paint makers rely on it in solvents and lacquers, since it helps other chemicals dissolve while evaporating at just the right speed. This balance keeps the paint workable—staying open long enough to spread but drying in time for second coats. The auto industry’s body shops and furniture businesses both appreciate those qualities, since unpredictable drying can ruin finishes.
Other coatings use it as a flow agent, helping to smooth out bumps and brush marks. In inks, it helps the pigment lay down clean with less clogging or streaking. Screen printers and packaging factories see fewer ruined runs, and that difference adds up over thousands of batches.
A Trusted Building Block for Manufacturing
Chemistry never stops at the first step. Factories use isobutanol to knit together compounds for plastics, pharmaceuticals, and synthetic flavors. It reacts predictably, giving companies confidence that batches will turn out with the same quality every time. In the world of plasticizers and esters, where reliability drives profit, that consistency pays off. Drug makers also use it to help create some medications during the synthesis stage, knowing it can be filtered out easily from the final product.
Flavors and fragrances companies use it in low amounts to shape the undertones of certain products. It pops up in fruit flavors, giving strawberry and cherry notes a little extra punch. I’ve tasted it myself during product trials—carefully diluted, of course. Its subtle sweetness and aroma round out the harsh notes in artificial blends, something flavorists have worked with for decades.
Looming Questions and Safer Handling
No one can ignore the safety side. Working around isobutanol means good ventilation and gloves, since it can cause headaches or dizziness at high doses, and spills can irritate the skin. That said, it hasn’t raised serious red flags about long-term health risks in industrial use, which reassures both workers and environmental regulators. It makes life easier on the shop floor as well as for transportation crews, since it doesn’t catch fire as easily as lighter alcohols like methanol.
All these uses rely on trust—trust that a barrel will always behave the same, and that quality will never slip. Earning that trust keeps the line running, keeps fuels cleaner, and supports products that people use without ever wondering about the chemistry behind them.
What are the safety precautions when handling 2-Methyl-1-Propanol?
Real Risks in Everyday Workspaces
2-Methyl-1-Propanol doesn’t smell dangerous, but that faint, almost sweet scent should not fool anybody. It’s flammable and causes irritation where you least expect it—eyes, skin, even airways if vapors get out of hand. My own background in industrial labs brought home how a simple slip in safety gear or sloppy ventilation could leave a coworker coughing for days or headed to the nurse with a red, swollen hand. It’s easy to underestimate clear liquids sitting in a glass bottle, but that’s when trouble usually starts.
Smart Handling Means Respect, Not Fear
Goggles are your friend. Spills happen, splashbacks are sneaky, and burn effects linger long after the surprise. I always reach for gloves—nitrile, not the thin plastic ones they hand out in cafeterias. A single drop turn fingers red, sometimes numb, so anyone pouring this alcohol into beakers or mixing with other reagents learns quickly. Clothes that cover skin also earn points—since this compound soaks through thin fabric. Just about every chemical splash I’ve seen on arms or legs became a painful lesson for someone in short sleeves.
Why Good Air Flow Matters
Too many small labs use up old fume hoods as makeshift storage, even though fume hoods protect lungs best. Vapors from 2-Methyl-1-Propanol drift quickly; I remember one colleague who ignored the hood for “just a quick pour” and lost his sense of smell for a week. Decent airflow isn’t just about following rules, it’s about everyone breathing normally at the end of the day. If there isn’t a working fume hood, open those windows wide and use fans to keep vapors away from faces.
Storage is Half the Battle
This stuff loves to catch fire if left near a heat source. I’ve seen more than one “clever” shortcut, with bottles shoved under benches close to radiators, or left in brittle plastic containers that crack under sun or light shocks. Choose metal safety cabinets, far from electrical panels, and mark them clearly. Labels need clear writing, not half-faded Sharpie lines—no one wants to grab the wrong bottle during a late-night cleanup.
Spill Plans that Actually Work
Many places stick a one-page “spill procedure” on the wall that no one reads. Real spill control starts with keeping the right absorbents nearby—vermiculite, not paper towels which can smolder after soaking up this alcohol. I keep a small spill kit in reach for my team with goggles, gloves, and proper waste bags. For bigger spills, evacuate and call in trained cleanup crews; trying to play hero with a mop often turns a small problem into a facility-wide hazard.
Training Builds Confidence
Annual safety refreshers aren’t just red tape. I’ve watched seasoned techs slip into bad habits, leaving bottle caps loose or cleaning bare-handed. Regular group reviews—walking through the process, using real bottles, simulating quick spills—give everyone a safer work environment. New staff always ask awkward, essential questions, and sometimes they remind the old hands of risks long dismissed.
Looking Ahead: Better Culture, Less Risk
Beyond the rules, it’s the work culture that keeps people safe. Open talk about sketchy storage or lazy spills improves things better than any rulebook. A safe workplace comes from every person knowing the risks of 2-Methyl-1-Propanol and taking real steps, daily, to lower them. Protecting our team means everyone goes home whole, which matters more than speed or shortcuts.
What is the chemical formula of 2-Methyl-1-Propanol?
Understanding the Structure
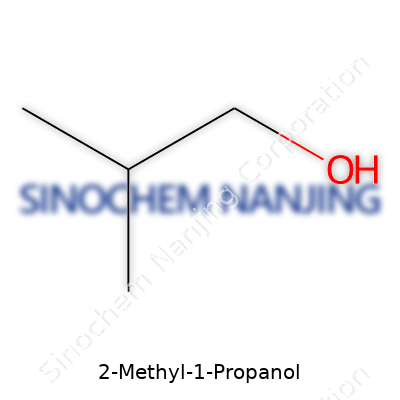
Ask anyone in a college chemistry lab about 2-Methyl-1-Propanol and a straightforward answer pops up—its chemical formula is C4H10O. Behind these numbers and letters stands a small, useful organic compound. The four-carbon backbone, with a methyl group on the second carbon and a hydroxyl functional group on the first, packs a subtle punch in laboratories and industry alike. Delving into its structure can help more than just students win quiz points; it uncovers how simple changes in a molecule’s configuration can completely shift its purpose and performance.
Real-World Uses Show Practical Chemistry
Many see chemical formulas as facts on a page, yet life has a way of bringing these chemicals into focus. Years ago in a university lab, I ran a set of experiments testing solvents for organic reactions. 2-Methyl-1-Propanol surprised me with its versatility. The molecule’s slightly branched backbone led to lower boiling points compared with straight-chain alcohols, affecting how it behaved as a solvent. These little differences can make or break an experiment.
Out in the world, C4H10O pops up in the manufacture of flavors and fragrances. The mildly sweet aroma makes it suitable for blending in food additives. Paints and coatings manufacturers also use it, relying on its ability to dissolve resins and dry evenly. Small molecule, big footprint.
Safety Demands Respect
Handling organics like 2-Methyl-1-Propanol brings safety front and center. Breathing in vapors causes irritation and working with it for hours leaves your hands dry and cracked if you skip gloves. The familiar scent in a work area quickly turns into an unwanted reminder to improve ventilation and stick to best practices. Data from workplace safety studies remind us that, at higher exposures, this compound can even impact the nervous system. Regulations covering storage and disposal need following, not just for compliance but for everyone’s well-being.
Environmental Impact Is Always Part of the Equation
Walk into any modern chemical plant and you hear talk about reducing emissions and waste. Even small-volume substances like 2-Methyl-1-Propanol play into these discussions. It’s biodegradable under the right conditions, but spills can pollute waterways and soil, harming aquatic life. Research teams and plant managers put heads together to develop better containment, spill response, and disposal methods. Some explore greener synthesis routes, seeking to lower the environmental toll from production through to breakdown.
Improving Outcomes: Education, Design, and Responsibility
Chemistry education stands at a turning point. Real stories in the classroom—ones involving specific compounds like C4H10O—show students why chemical structure matters. This approach also pushes future chemists to consider humanity’s broader relationship with substances they might one day create or regulate.
Designers of chemical processes and products have a responsibility to think beyond immediate results. Choosing safer solvents, improving recycling, and limiting exposure can transform a routine workflow into a model for health and sustainability. Each informed step, backed by solid science, brings clear benefits to workplaces, communities, and the environment. Real change starts with understanding what those numbers and letters represent in practice, not just on paper.
Is 2-Methyl-1-Propanol flammable?
Understanding 2-Methyl-1-Propanol
2-Methyl-1-propanol, often known as isobutanol, doesn’t come up in everyday talk unless you have a reason to care about chemicals. This compound ends up used as a solvent, in specialty fuels, and within labs all over the world. Flammability should matter to anyone dealing with this substance—whether you work in manufacturing, chemistry, or you’re just a curious student.
Recognizing Flammability in Daily Practice
Unlike table salt or water, 2-Methyl-1-propanol will catch fire if it gets near the wrong conditions. It has a flash point somewhere around 28°C to 37°C—well below many room temperatures, especially in summer or in areas lacking climate control. If you’ve ever stored paint thinner or certain cleaners in a garage, you know that these sorts of liquids don’t wait around patiently when a flame or spark comes their way.
Why the Flammability Risk Matters
Everyday stories from small businesses and home workshops show what happens when the risk gets ignored. Several years ago, I helped clear out a small auto body shop near my hometown. One shelf held a collection of bottles, one labeled “Isobutanol.” Someone had stored it next to a space heater. Nobody got hurt, but putting flammable chemicals near a heat source felt reckless once we saw the label. Not everyone gets lucky.
The National Fire Protection Association marks this chemical as a significant fire hazard. OSHA’s guidelines treat it seriously—not because rules exist for fun, but because plenty of fires started from one overlooked bottle, spilled by accident and ignited in a blink.
Common Uses Increase Exposure
People might not think about flammable liquids as often as they think about the open flame on a gas stove. Labs run rotovaps and distillations using isobutanol as a solvent. The printing and coatings industry grabs this chemical for its drying properties. Anywhere solvents work, fire risk follows.
Some farms even explore isobutanol as a renewable fuel. Industry hopes to use less gasoline, switching to bio-based alcohols. The more widespread these applications become, the more the average worker interacts with something capable of burning fast and hot.
Reducing Flammability Risks at Home and Work
Concrete steps can deal with these hazards. Personally, I always check for proper storage—flammable cabinets, tight-fitting lids, away from open flame or electronics. Ventilation keeps vapors moving out of a workspace; accumulation makes everything more dangerous, not just for a single worker but for everyone sharing the building.
Labels save lives. Even a fading, hand-written warning proves far better than nothing. Safety goggles seem obvious, but hearing about someone burned when splashing solvents reminds me not to skip basic gear. Supervisors and hobbyists should run short, memorable drills: How do you put out a fire started by this specific liquid? Water won’t work well in every case.
Taking Responsibility with 2-Methyl-1-Propanol
Ask anyone who has cleaned up after a chemical fire—they all say they wish someone had flagged the flammable bottle sooner. Treating 2-Methyl-1-propanol as a fuel source ready to burn stays crucial. With the right habits, small oversights no longer threaten whole projects, businesses, or lives.
How should 2-Methyl-1-Propanol be stored?
A Common Solvent with Big Responsibilities
2-Methyl-1-Propanol, also known as isobutanol, shows up in plenty of labs and plants, and it’s easy to brush aside as just another organic liquid in the lineup. I’ve seen it on benches and in warehouses right next to things that barely raise an eyebrow, but neglecting its safety quirks could open the door to trouble most people wouldn’t want to face. At room temperature, isobutanol smells sweet, but its pleasant scent hides a few headaches—mainly fire risk and direct health concerns.
The Hidden Hazards: More than a Flammable Warning
Drawn from my own days working around these chemicals, I remember early on how casual mistakes—keeping isobutanol near heating pipes, stacking cardboard boxes too close—almost always caused confusion or panic drills. One oversight, and fumes can catch. Vapors from isobutanol mix well with air, forming flammable environments even at modest concentrations. The flash point rests at about 28°C, so a summer day, a forgotten container, and a bit of static, create enough risk for a small fire.
Along with the fire risk, I’ve watched coworkers develop headaches or dizziness from breathing in those vapors longer than they should. The stuff doesn’t just stay put—without good ventilation and closed containers, it travels. I recall the sting of its vapors in a poorly ventilated storage room, which convinced me fast that airflow and proper sealing are non-negotiable for anything inside that room.
Packing and Placement: Simple Steps that Matter
Keeping this solvent secure boils down to a few direct rules I stick to every time. Store it in closed, labeled containers made of materials that can handle alcohols—high-density polyethylene or metal work just fine. Keep the storage area cool, away from sunlight or heat, since rising temperatures nudge those vapors out of the bottle and into the air. Never keep isobutanol near potential ignition sources, even things as small as a spark from tangled power cords or a hot piece of equipment.
It’s best to use grounded metal shelves and avoid stacking anything flammable nearby. Any spills always call for prompt cleanup using absorbent pads, with attention paid to proper disposal. For long-term storage, a dedicated flammable storage cabinet with fire suppression does double duty—cuts down on oxygen and makes accidental fires less likely to spread.
Training, Monitoring, and a Dose of Respect
Chemicals like isobutanol reward respect. Everybody who works with or around it should know basic procedures: why ventilation matters, how to quickly seal up a leaking bottle, what to do if someone feels faint. Relying on regular checks—both of the bottles and the ventilation systems—goes a long way.
Emergency gear like eyewash stations, fire blankets, and spill kits aren’t luxuries; they’re everyday tools. Personal protective equipment—gloves, safety goggles, chemical aprons—should be worn, because too many times, workers assume quick tasks won’t cause problems. Skin contact may not seem dramatic, but enough exposure can dry out skin, and eye irritation is immediate and painful.
Smart Storage is Good Business
Safe handling habits build a culture that keeps everyone at work healthy and productive. Regulations, like those from OSHA or local safety boards, exist for good reason. Following these guidelines, setting up clear documentation, and running refresher sessions do more than check off a legal box—they stop expensive clean-ups and real harm. For any team facing a storage decision about 2-Methyl-1-Propanol, a little diligence pays off every single day.
| Names | |
| Preferred IUPAC name | 2-Methylpropan-1-ol |
| Other names |
Isobutanol Isobutyl alcohol 2-Methylpropan-1-ol |
| Pronunciation | /tuː ˈmɛθɪl wʌn ˈprəʊpənɒl/ |
| Identifiers | |
| CAS Number | 78-83-1 |
| Beilstein Reference | 635164 |
| ChEBI | CHEBI:28885 |
| ChEMBL | CHEMBL43094 |
| ChemSpider | 7287 |
| DrugBank | DB02160 |
| ECHA InfoCard | 100.869.289 |
| EC Number | 200-746-9 |
| Gmelin Reference | 82233 |
| KEGG | C02310 |
| MeSH | D007374 |
| PubChem CID | 6560 |
| RTECS number | UJ5425000 |
| UNII | M3KAT1CAI6 |
| UN number | UN1120 |
| Properties | |
| Chemical formula | C4H10O |
| Molar mass | 74.12 g/mol |
| Appearance | colorless liquid |
| Odor | wine-like odor |
| Density | 0.805 g/cm³ |
| Solubility in water | miscible |
| log P | 0.76 |
| Vapor pressure | 3.2 mmHg (20 °C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 15.20 |
| Magnetic susceptibility (χ) | -63.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.395 |
| Viscosity | 2.9 mPa·s (20 °C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 130.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -330.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –2684 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H335 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P303+P361+P353, P304+P340, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 28°C |
| Autoignition temperature | 425 °C |
| Explosive limits | 2-11.4% |
| Lethal dose or concentration | Lethal dose or concentration: "LD50 (oral, rat): 2460 mg/kg |
| LD50 (median dose) | LD50 (median dose): **2460 mg/kg (rat, oral)** |
| NIOSH | SA9125000 |
| PEL (Permissible) | PEL: 100 ppm (300 mg/m³) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | IDLH: 800 ppm |
| Related compounds | |
| Related compounds |
Isobutyric acid 1-Butanol 2-Butanol tert-Butanol n-Butyl alcohol |

