2-Methyl-1-Propanethiol: A Close Look at a Powerful Chemical
Historical Development
Years before chemists fully understood the structure of organic molecules, they found themselves face-to-face with odd-smelling compounds that changed the course of their work. 2-Methyl-1-Propanethiol, sometimes called isobutyl mercaptan, drew attention thanks to its potent odor and unique makeup. Early researchers, tinkering in labs without the spectrometers or analytical tools of today, followed their noses to uncover the sulfur-based backbone of these chemicals. The compound found its way into chemical registries soon after, as researchers mapped the tangle of thiols and started testing them across different reactions. Having been present in both industrial development and academic research, this compound’s name pops up throughout the literature of 20th-century organic chemistry. Over time, folks puzzled out its value not just as a chemical oddity, but as a practical ingredient in several applications.
Product Overview
2-Methyl-1-Propanethiol stands out because of its knack for introducing a sulfur atom where others don’t fit. With a structure built from four carbon atoms, a methyl branching, and a reactive thiol group, it gives chemists flexibility. The unpleasant smell, reminiscent of rotten cabbage or skunk spray, keeps most people at bay, yet for those who handle it, this very odor acts as a built-in warning sign. In industry, it doesn’t usually end up in final consumer products. Instead, it shows up in lab syntheses, gas leak detection, and as a precursor in specialty manufacturing. Over the years, refinements in distillation and handling have made it possible to keep the volatile fumes at bay, making storage safer and preparation more reliable.
Physical & Chemical Properties
A bottle of 2-Methyl-1-Propanethiol doesn’t look remarkable until opened. At room temperature, it holds as a clear, mobile liquid. It boils around 90°C, which means it evaporates quickly when left exposed. With a density just below that of water, it separates into a distinct layer in most mixtures. It dissolves in organic solvents like ether and alcohol far more readily than in water. Its sulfur atom, framed by carbon, gives the molecule a considerable reactivity, especially in the presence of oxidizers or when heated. In practical terms, workers treat it as both a flammable and a health risk, so proper ventilation and flame-proof settings play a sizable role wherever it is involved.
Technical Specifications & Labeling
Across chemical suppliers, 2-Methyl-1-Propanethiol usually appears labeled with hazard warnings and regulatory information tied to its toxicity and flammability. Storage instructions stress cool, shaded, well-ventilated spots, away from sources of ignition. Color-coded hazard diamonds often mark the packaging, emphasizing both the immediate risks and the need for personal protective equipment. Chemical purity usually lands in the 95-99% range for lab and industry stocks, with strict batch testing to maintain consistency. Shipping regulations frequently match those for other volatile organosulfur compounds, with limits on transport quantities and documentation requirements spelled out by both national and international guidelines.
Preparation Method
Although the earliest preparations leaned on multi-step routes, modern synthesis of 2-Methyl-1-Propanethiol often begins with isobutylene or derivatives of isobutane. By introducing hydrogen sulfide or sodium hydrosulfide under carefully controlled acidic or basic conditions, manufacturers attach the thiol group directly to the hydrocarbon backbone. Catalysts help steer the reaction toward the desired product, reducing waste and improving overall yield. In some processes, gas-phase reactions are preferred for speed and simplicity, even if this means more attention to ventilation and gas capture. Each approach gets refined over time, driven by scale, efficiency, and safety needs in local plants.
Chemical Reactions & Modifications
One of the most noteworthy things about 2-Methyl-1-Propanethiol has always been its versatility in chemical transformations. It participates readily in nucleophilic substitution, engages with alkyl halides to form thioethers, and serves as a building block in complex organic syntheses. Given the sharp reactivity of the thiol group, it reacts with oxidizing agents to generate disulfides, a step often used in polymer chemistry and protective coatings. In many reactions, its branching methyl group imparts selectivity, altering outcomes compared to straight-chain analogues like 1-propanethiol. Chemists working in pharmaceuticals, agrochemicals, and materials science draw on these features to improve drug designs, create new ligands, and tweak polymer backbones.
Synonyms & Product Names
Stepping into a chemical supply catalog, one will find 2-Methyl-1-Propanethiol listed under several aliases. Isobutyl mercaptan, isobutanethiol, and 1-isobutyl mercaptan all point to the same compound. In academic literature, the CAS number ensures clarity, especially for researchers bouncing between languages or regional naming fashions.
Safety & Operational Standards
Like most thiols, this one demands respect during handling and storage. Exposure to the vapors causes headaches, nausea, and irritation of the eyes and respiratory tract. Spills call for immediate containment, triple-checking ventilation and protective gear. Industry sets strict exposure limits, outlined by bodies like OSHA and confirmed by independent toxicological research. Fires pose a serious risk, as flammable vapors ignite at relatively low temperatures. For transport, only trained personnel work with the material, and facilities rely on regular audits and strict inventory management to keep risk under control. Every workplace involved in its production or use invests in emergency procedures, dedicated spill kits, and training sessions for staff, going beyond basic compliance because lessons from past incidents teach the value of vigilance.
Application Area
2-Methyl-1-Propanethiol’s role spreads across a handful of specialized sectors. Odorants in natural gas lines often rely on its aroma to alert users to leaks, making safety systems more effective than sight or sound alone. In laboratories, synthetic chemists use it as a versatile intermediate, leveraging its thiol chemistry for reactions that few other reagents can match. Its reactivity benefits the development of rubbers, plastics, and specialty coatings—places where the final product never smells but gains improved properties thanks to this sulfur source. Animal repellents and certain agrochemical mixtures have seen occasional use cases, with tightly controlled formulations minimizing environmental impacts. Industrial facilities measure emissions and manage waste streams to keep accidental exposure within legal and ethical boundaries, recognizing the sharp profile of this molecule in both urban and rural settings.
Research & Development
Researchers continue exploring new uses and safer handling of 2-Methyl-1-Propanethiol. Recent advances in analytical chemistry make it easier to track and model its behavior in complex mixtures, so environmental and health risk assessments progress much faster. Universities and private labs experiment with less hazardous derivatives, trying to expand function while shrinking exposure risk. Advances in predictive toxicology and green chemistry fill a growing body of peer-reviewed work, refining best practices and suggesting fresh methods of immobilization, neutralization, and recovery. Regulatory agencies lean on this research to guide updates in chemical safety standards, and industry adapts by investing in new equipment and training programs to keep pace.
Toxicity Research
Toxicological studies make clear that 2-Methyl-1-Propanethiol, despite its value, carries significant health risks even at low concentrations. Chronic exposure affects the central nervous system, and short-term inhalation at higher levels can result in serious symptoms. Researchers run a battery of tests in animal models, monitoring organ systems and carrying findings toward regulatory thresholds. Air sampling and biomonitoring in workplaces feed back into best practices, with monitoring data driving both legislative and practical changes for worker protection. More sensitive detection tools provide early warning, allowing operations to catch potential leaks before they escalate. Increasing emphasis on green chemistry prompts investigation into less harmful substitutions, but for now, strong safety controls remain the cornerstone of responsible use.
Future Prospects
Looking ahead, the story of 2-Methyl-1-Propanethiol shows both promise and challenge. Advances in containment, odor control, and remote monitoring could allow broader industrial and research use without raising health or safety risks. The quest for greener processes keeps pressure on chemists and manufacturers to find ways to minimize emissions, recycle waste, and reduce the hazard profile of every stage in its life cycle. Synthetic biology and advanced catalysis may one day offer routes that bypass the toughest hazards entirely. For those of us working on the frontlines of chemical safety and innovation, the lessons learned from decades of handling reactive thiols shape our approach—not just to 2-Methyl-1-Propanethiol, but to the ever-evolving toolbox of industrial chemistry. It’s a reminder that chemical advancement depends as much on human caution and curiosity as on technical progress.
What is 2-Methyl-1-Propanethiol used for?
Understanding the Role of 2-Methyl-1-Propanethiol
2-Methyl-1-Propanethiol brings a punchy odor to any environment it enters. Known to chemists and manufacturers for its sharp, sulfurous smell, this chemical finds its way into a surprising number of daily and industrial applications. The main appeal? Its presence gets noticed, which is exactly the point in its line of work.
Applications in Natural Gas and Leak Detection
Utility companies rely on our noses far more than most folks realize. Natural gas comes out of the ground odorless, which poses a safety hazard in millions of homes. By adding a pungent compound like 2-Methyl-1-Propanethiol, gas leaks invite attention before disaster strikes. This chemical, as a warning agent, plays a direct role in keeping neighborhoods safer. According to the U.S. Department of Transportation, thousands of leaks get caught every year thanks to these sulfur compounds in the gas supply. From my own experience growing up in a house with gas heat, nothing compares to the instant awareness when you catch that whiff—safety quickly becomes the only thing that matters.
Contributions to the Flavor and Fragrance Industry
Few people get excited about sulfur chemicals in their kitchen or beauty products, but without them, whole industries would lose some key notes. 2-Methyl-1-Propanethiol’s strong, savory aroma gets tamed and used in ultra-low concentrations in flavors and fragrances. It can mimic roasted or cooked food aromas at minute levels. Chemists know how to use a light hand: a dash enhances and brings out flavors in potato chips, soups, and savory snacks, helping processors deliver that cooked, umami punch—a little goes a long way.
Laboratory Uses and Synthesis
Synthetic chemistry relies on building blocks like 2-Methyl-1-Propanethiol to get creative with new molecules. In pharmaceutical research and agrochemicals, technicians use this particular thiol to add sulfur to larger molecules. The chemical structure offers a short, simple path to many other specialized organic compounds. New pesticides, fungicides, and even some medicinal products owe part of their creation to reactions involving this thiol. The more efficient the synthesis, the better scientists can respond to urgent needs, whether fighting crop disease or developing lifesaving medications.
Hazards and Responsible Use
With all its benefits, 2-Methyl-1-Propanethiol comes with downsides if used without caution. Inhalation can irritate eyes and lungs. Not many folks outside chemistry labs have to handle this material, but for those who do, a little negligence can turn a workday into a trip to the emergency room. Wearing proper protection, storing it away from heat, and using reliable ventilation eliminate many of these risks. Factories and labs must insist on strict protocols to keep workers safe, a principle I’ve watched respected operators enforce without compromise.
Steps to Improve Safety and Awareness
Better training and stronger regulations make a real difference with volatile materials like this one. Employers can minimize exposure by using proper containers and up-to-date detection systems. Teaching newcomers what to expect when they work with sulfur thiols helps everyone avoid careless handling. Government agencies and industry groups have learned from past incidents, leading to today’s more transparent guidelines for the use and transportation of hazardous chemicals.
Looking Toward the Future
Growing concern for workplace safety and environmental impact keeps pushing manufacturers to rethink their approach. Research into greener alternatives may cut down on the risks, though few replacements match this compound’s unique alerts for gas safety. For now, 2-Methyl-1-Propanethiol remains a behind-the-scenes player that many rely on, using its strength—an unforgettable smell—to keep everyday spaces safe and effective.
What are the safety precautions for handling 2-Methyl-1-Propanethiol?
Recognizing the Risks
Working with chemicals like 2-Methyl-1-Propanethiol brings back memories of standing in stuffy undergraduate laboratories. This compound stings the nose with a rotten, aggressive odor that’s impossible to ignore. The smell alone chases away half the bravado. But the hazards go deeper than discomfort. 2-Methyl-1-Propanethiol, with its potent volatility, lines up among those chemicals that command respect. There’s a reason workplace safety data consistently rate it as flammable and hazardous for skin, eyes, and lungs. A single careless moment turns a routine procedure into an emergency.
Ventilation Keeps You Out of Trouble
Every time I opened a bottle, the sharp fumes climbed into every corner. One strong whiff convinced me never to cut corners. Fume hoods are your best friend here. Open air alone doesn’t cut it—the gas is heavier than air and lingers near the ground. The right exhaust set-up drags vapors away before they reach your face or build up to explosive concentrations. Lab accidents often follow shortcuts or disabled hoods. I watched one technician rush and pay with burning eyes. Since then, I always double-check the airflow.
Ears, Noses, and Fingers: Protect the Body
Eye contact with 2-Methyl-1-Propanethiol will stop you in your tracks. A few splashes triggered red, watering eyes that took hours to calm down. Goggles with solid side shields matter more than ever. Thin latex gloves don’t stand up well to this chemical, so nitrile or even neoprene work better. Low-quality gloves left my hands hopping mad with irritation. Any exposed skin picks up the stench and delivers a sharp lesson in chemical hygiene. Changing into a chemical-resistant apron blocked those lingering odors from sneaking onto my clothes.
Fire Hazards: Where Caution Never Rests
This compound flashes at low temperatures. One lab session went sideways when a Bunsen burner set off a bottle cap dusted with residue. It sent everyone scrambling. Even basic static sparks hold real risk when vapors hover near ignition sources. Using non-sparking tools saved more than one project from disaster. Static-dissipative mats and grounding turn burrito-making into a necessary ritual, not bureaucracy. Storing the chemical far from heat cured several headaches. Strong warnings from safety officers ring true—never trust luck to substitute for proper storage.
If Things Go Wrong, Speed Saves
Chemical workers love routine, but nothing jolts you awake like an emergency flush after a splash. Quick access to eyewash stations made all the difference during a spill that caught a junior chemist by surprise. Training matters—a clear emergency plan lets people act, not freeze. Reporting symptoms way before they feel unmanageable lets medical staff prevent worse outcomes. Routine fire drills take on new urgency in places where flammable chemicals become part of daily life.
No Shortcuts Around Respect
2-Methyl-1-Propanethiol earns its fearsome reputation. Anyone handling it should reach for extra ventilation, gloves, and goggles. Regular training kept our lab safe, and a culture of reporting and double-checking small details built confidence. Dissolving the myth that familiarity brings less risk keeps everyone a little more careful. The best solution usually depends on real, lived experience—where mishaps drive home lessons that stick longer than any poster on the wall.
What is the chemical structure and formula of 2-Methyl-1-Propanethiol?
Unpacking the Molecule: What Makes Up 2-Methyl-1-Propanethiol?
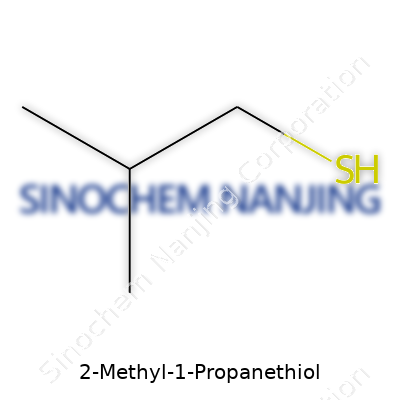
2-Methyl-1-Propanethiol might sound complicated, but its backbone tells a straightforward story. It’s an organosulfur compound, with a backbone of four carbons arranged as a propane chain, tacked with a methyl group and a thiol group. Its chemical formula is C4H10S. More specifically, the structure packs a sulfur-hydrogen group (—SH) at one end, while the other end carries a methyl branch. The actual lineup goes like this: start with a three-carbon chain, slap another carbon on the second carbon, and then anchor a —SH group at the first carbon.
Looking at Structure: Drawing the Skeleton
For someone peering at a stick-and-ball model or a Lewis diagram, you’d see something like this:
- A central backbone: CH3–CH(CH3)–CH2–SH.
- On the first carbon in the chain, the sulfur atom pokes out with a single hydrogen attached.
- The second carbon bonds to an extra methyl group, bulking out the molecule and impacting both its shape and smell.
So inside any bottle of this stuff sits a highly branched molecule. Sulfur’s placement packs a punch — that’s the source of the often sharp, unpleasant odor most people describe as skunky, or like fertilizer.
Why Knowing the Structure Matters
Plenty of people brush off organic chemistry as academic. Still, if you’ve ever done work in a lab, manufactured products, or even tried to identify what’s drifting in the air, you’ll bump into compounds like this. Emergency response teams, for instance, get trained to recognize the smells of mercaptans, which include 2-Methyl-1-Propanethiol. These compounds show up as odorants in natural gas, making leaks much easier to detect before catastrophe strikes.
There’s a practical side, too, in the way these molecules interact with other substances. Chemists have learned that the placement of atoms—exactly where each carbon, hydrogen, and sulfur lands—changes how a chemical behaves. Knocking the methyl group onto that second carbon, swinging the thiol to the first, and keeping the hydrogens in their places, makes this specific mercaptan different from its close cousins, like n-propanethiol or isobutanethiol. Each tweak to the structure brings a distinct boiling point, a different level of volatility, and a nose-tickling signature unique to the design.
Potential Concerns and Safer Handling
Working with 2-Methyl-1-Propanethiol, and other low molecular weight thiols, means dealing with very strong odors—sometimes even at concentrations you can’t see or taste but certainly can smell. Over the years, I've seen colleagues make the rookie mistake of uncapping a sample bottle outside of a fume hood, chasing off everyone in the lab for hours. Awareness comes from understanding the structure: that sulfur group just doesn’t hide well. The lesson runs deeper than a ruined afternoon—it highlights the value in respecting how a molecule’s structure leads to physical properties and consequences.
Proper ventilation, sealed containers, thoughtful storage, and focused handling keep exposures to a minimum. Chemistry sets the rules; experience proves them true, again and again.
How should 2-Methyl-1-Propanethiol be stored?
Why Worry About Storage?
Walking through any chemical storeroom, you can almost pick out the sections by the smells alone. Every bottle tells a story about its contents, risks, and the care—or lack of care—given to its storage. 2-Methyl-1-Propanethiol stands out in that lineup for its intense odor and its knack for raising safety concerns. I remember opening a cupboard in the lab—one whiff of that sharp, skunky smell and my day had already started off rough. This chemical has a way of making its presence known, even through a closed cap. That’s not just annoying; it’s dangerous.
Health and Safety Side of the Story
One of the biggest lessons I learned during my early years in the lab: short cuts in chemical storage almost always catch up with you. 2-Methyl-1-Propanethiol can irritate the eyes, skin, and lungs, even in small amounts. A leaky bottle stored at room temperature will spoil the air, but even sealed containers, left too warm or exposed to sunlight, can build internal pressure or cause chemical breakdowns. Stories keep popping up of workers getting sick headaches or worse just because someone decided to stash a container somewhere “out of the way.” Reliable data from the CDC and OSHA back up these firsthand stories—this thiol should never be stored like just any other bottle on the shelf.
Getting Storage Right
I’ve seen storage mistakes, but I’ve also seen what works. Locked chemical cabinets with good ventilation can make all the difference. Nobody wants a fire, and 2-Methyl-1-Propanethiol burns easily and gives off toxic fumes. Putting the bottle in a cool, well-ventilated spot, away from acids and oxidizers, isn’t just following the rules—it’s common sense if you’ve ever watched a near miss turn into an emergency. Flame-proof cabinets, spark-proof refrigerators (for temperature-sensitive stocks), and clear hazard labels go a long way to keep everyone safe.
Why Decent Containers and Ventilation Matter
A friend of mine used old glass bottles with loose-fitting caps—not his smartest move. That pungent thiol smell showed up in the hallway and, pretty soon, across the building. Airtight, chemical-resistant bottles with screw tops make all the difference. You keep the fumes in and cut down on leaks or accidental splashes. Storing these containers on spill trays and using secondary containment is simple, but it’s a trick that’s saved more than one storeroom from disaster. Good ventilation, like working fume hoods and regularly serviced air systems, helps keep people out of harm’s way, which always matters more to lab workers than anyone notices.
Checks and Training
In my experience, regular walk-throughs matter more than flashy equipment. Patients, nurses, scientists, even janitors always spot the “shelf of doom”—the one with all the problem bottles. Regular audits help catch leaks, corrosion, or labeling flubs before they become front-page news. Just as important is knowing what personal protective equipment is for which job—gloves, goggles, and the right lab apron aren’t optional when handling a container. Training sticks if it gets repeated, and not just covered in some onboarding slideshow. Sharing real-world close calls keeps everyone sharper than checklists alone.
It Draws a Line
Getting storage right isn’t fancy, but nothing beats the peace of mind that comes from walking into a chemical storeroom and knowing that every bottle is where it should be, labeled and sealed tight, in cabinets built to handle exactly what they contain. In a world where one small bottle can foul the air, cause injury, or worse, it’s on all of us to choose care over convenience every single time.
What are the physical and chemical properties of 2-Methyl-1-Propanethiol?
Getting To Know Its Nature
2-Methyl-1-propanethiol, known to chemists by the formula C4H10S, isn’t some rare laboratory curiosity. It’s a colorless or faintly yellow liquid, but its odor tells a different story—strong, pungent, reminiscent of skunk spray or rotten cabbage. People detect this scent at extremely low concentrations. Its boiling point sits around 90°C, so on a warm day, the chemical can evaporate and become airborne fast, which explains why even a small spill clears a room.
Flash point registers at around -20°C, making this compound flammable. It mixes with common organic solvents—think ethanol, ether, chloroform—but water has trouble soaking it up. That has real consequences for storage and safety. The density lands close to 0.81 grams per cubic centimeter, lighter than water. It forms vapors heavier than air, creeping along surfaces and potentially reaching open flames at ground level. That’s a real concern in workplaces handling large volumes.
Chemical Behavior That Demands Respect
The sulfur atom throws its weight around in this molecule. This spot is reactive, particularly toward oxidizing agents. Mix 2-methyl-1-propanethiol with strong oxidizers and you’re asking for hazardous byproducts or even fire. Sulfur compounds like this don’t play nice with nitric acid or hydrogen peroxide. Beyond lab hassles, this trait means sulfurous compounds must stay clear of reactive chemicals both in warehouses and in transit.
Light and air speed up breakdown. If left uncapped, the compound absorbs oxygen and light kicks off degradation. Over time, this process generates disulfides, which can smell even worse and change the properties of the original liquid. In my own experience, bottles forgotten under fume hoods shift color and stink up the entire room, proving that chemical carelessness lingers.
Uses and the Need for Caution
What do people use this smelly liquid for? It's important in organic synthesis and the flavor industry. Certain food flavors and scents rely on the compound’s unmistakable punch—even in tiny amounts—to get “meaty” and “savory” notes right. Working in analytical labs, I’ve seen 2-methyl-1-propanethiol sneak up on people: a misplaced drop can leave a persistent stench on gloves or clothes for hours.
Industrial and research workers wearing nitrile gloves and eye protection make a difference here. Testing for leaks with detection tubes and regular ventilation checks help cut down on health complaints and accidental exposures. The Material Safety Data Sheet suggests avoiding direct contact, since it may cause skin and eye irritation, not to mention headaches and nausea from fumes.
Handling Hazards and Looking Ahead
The handling risks aren’t theoretical. There have been well-documented cases of exposure causing acute symptoms, which shines a spotlight on the importance of proper training. Chemical fume hoods belong in every lab handling this liquid. Keeping stocks away from flames and oxidizers eliminates most major risks. Spill cleanup needs to be prompt and thorough, since residues can hang on to surfaces long after a mess appears gone.
If there’s a lesson from daily lab reality and chemical knowledge, it’s that working with distinctive-smelling, easily-ignitable chemicals calls for specific practices—ventilation, personal protection, and a little humility. Nitpicking storage, double-checking procedures, and not rushing in when something spills matter as much as any textbook property.
| Names | |
| Preferred IUPAC name | 2-methylpropan-1-thiol |
| Other names |
Isobutyl mercaptan Isobutanethiol Isobutylthiol 2-Methylpropane-1-thiol 2-Methyl-1-propanethiol |
| Pronunciation | /tuː-ˈmɛθɪl-waɪn-proʊˈpeɪn-θaɪˌɒl/ |
| Identifiers | |
| CAS Number | 75-33-2 |
| Beilstein Reference | 803873 |
| ChEBI | CHEBI:37968 |
| ChEMBL | CHEMBL135482 |
| ChemSpider | 7279 |
| DrugBank | DB11271 |
| ECHA InfoCard | 03d6c5d8-015a-404f-be69-2cad3e6df8e6 |
| EC Number | 200-860-9 |
| Gmelin Reference | 82552 |
| KEGG | C08202 |
| MeSH | D010499 |
| PubChem CID | 15604 |
| RTECS number | WN8575000 |
| UNII | LV856Q66FC |
| UN number | UN2396 |
| CompTox Dashboard (EPA) | DTXSID4038293 |
| Properties | |
| Chemical formula | C4H10S |
| Molar mass | 90.19 g/mol |
| Appearance | Colorless liquid |
| Odor | unpleasant, garlic-like |
| Density | 0.793 g/mL |
| Solubility in water | slightly soluble |
| log P | 1.77 |
| Vapor pressure | 137 mmHg (20 °C) |
| Acidity (pKa) | 10.62 |
| Basicity (pKb) | **6.66** |
| Magnetic susceptibility (χ) | -53.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.410 |
| Viscosity | 0.56 mPa·s (20 °C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 302.80 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -117.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2712 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Warning |
| Hazard statements | H225, H301, H311, H315, H319, H331, H335, H400 |
| Precautionary statements | P210, P261, P271, P302+P352, P305+P351+P338, P304+P340, P312, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | -18 °C (closed cup) |
| Autoignition temperature | 224 °C |
| Explosive limits | 1.9–9.5% |
| Lethal dose or concentration | LD50 oral rat 179 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 179 mg/kg |
| NIOSH | US3858400 |
| PEL (Permissible) | PEL: 0.5 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Methanethiol Ethanethiol 1-Propanethiol 2-Propanethiol 1-Butanethiol Tert-Butylthiol |

