2-Methyl-1-Pentanol: More Than Just a Chemical Name
Digging Into the Past: Historical Trajectory
Chemistry textbooks often mention 2-Methyl-1-Pentanol as a minor player in the wide world of alcohols, but laboratory benches and factory floors tell a different story. I remember in my first organic chemistry lab, tucked away between rows of bottles with faded labels, 2-Methyl-1-Pentanol filled the air with a subtle, not-quite-fruity odor. Synthetics researchers back in the early-to-mid 20th century learned its quirks through hard knocks, using older distillation methods that lacked the precision of today’s equipment. These early tinkerers didn’t just contribute recipes—they built a basic scaffolding for solvent chemistry. The molecule gradually found its place in the shadow of its more famous cousins, not for lack of utility, but for not boasting the headline-grabbing effects of higher alcohols. As industries went from dyes and perfumes to complex coatings and lubricants, 2-Methyl-1-Pentanol continued to sneak its way into formulations that demanded a balance between volatility, solvency, and compatibility with modern processes. For a long stretch, it stayed a chemist’s secret, surfacing in patents and process write-ups only when someone probed just outside the familiar.
The Nuts and Bolts: A Product Overview
2-Methyl-1-Pentanol is more than a formula on a sheet. With its molecular layout—an isomeric alcohol with a methyl substituent branching from the pentanol backbone—it has a structure that grants certain flexibilities and restrictions across different applications. It sits as a colorless liquid, sometimes carrying a faint scent that reminds one of fresh solvents, hinting at its use in fragrance intermediates and specialty flavors (in carefully regulated amounts). The real stories crop up in resin synthesis, where technicians lean on this alcohol to control viscosity and volatile organic content, two much-discussed talking points for compliance officers and production managers alike.
Physical and Chemical Character: Not Just Numbers
Most datasheets will spit out boiling points, density, and flashpoints, but working with 2-Methyl-1-Pentanol reveals traits that don’t always fit into columns. Its boiling point floats in the range for medium-chain linear alcohols, making it useful for progressions in fractional distillation when separating close relatives. Its moderate miscibility with water shapes how people blend it into two-phase systems or use it as a co-solvent where complete mixing sets up unwanted side reactions. The viscosity, neither syrupy nor too thin, keeps it at home in many industrial paints and plasticizer blends, minimizing dripping or unpredictable flow during application. Handling this fluid reminds me of the time we adjusted cooling settings in a teaching lab; temperature control made the difference between a clean run and an off-spec product.
Technical Specifications and Labeling: What Matters in Practice
Labeling in a modern facility strikes a balance between clarity and regulatory pressure. For 2-Methyl-1-Pentanol, identification relies on CAS numbers and standardized naming, but real-world shipments include hazard communication for flammability and exposure. Recent years brought stricter labeling for both storage and transport, enforced by global safety frameworks. As a regular observer of compliance audits, I’ve noticed companies now lean more on QR codes, letting handlers dive quickly into up-to-date material safety data—not just to fulfill regulations, but to tap into the collective industry wisdom on accident prevention and incident response.
Preparation Methods: Classic and Current Approaches
The backbone of 2-Methyl-1-Pentanol’s preparation traces back to classic organic synthesis from petrochemical sources through hydroformylation of olefins, typically 2-methyl-1-pentene. The choice between catalytic systems—cobalt-based versus newer rhodium variants—sets up trade-offs between yield, selectivity, and downstream processing waste. In an era chasing sustainability, labs have begun exploring biobased feedstocks, though the hiccups of scaling and feedstock purity remain stubborn limits. I’ve watched teams debate the merits of greener routes; often the timeline and budget win, but interest in fermentation and enzymatic pathways is growing, especially as regulatory pressure mounts to cut carbon footprints.
Chemical Reactions & Modifications: Tools in the Kit
2-Methyl-1-Pentanol, like other alcohols, slots into esterification, etherification, and oxidation reactions. In practice, it helps produce specialty esters for flavors or plasticizers, sometimes surfacing in formulation notes for flexible polymers or advanced coatings. Chemists value the secondary nature of its alcohol group, striking a useful balance between reactivity and stability. One memorable project involved tweaking reaction conditions for customized surfactants, relying on subtle temperature adjustments to boost selectivity—every degree matters, especially with industrial cost margins squeezed so tightly. Its ability to undergo further functionalization helps R&D folks chase niche targets for everything from lubricants to next-generation solvents.
What’s in a Name: Synonyms and Product Variants
2-Methyl-1-Pentanol travels under plenty of aliases, thanks to the patchwork history of chemical naming conventions. References might call it hexanol, isohexyl alcohol, or even 2-methylpentan-1-ol, depending on regional norms or supply chain translation quirks. Years of consulting in global supply networks taught me that clear naming on paperwork matters as much as technical specs—confusion over synonyms can cause regulatory delays, border hold-ups, and even wrong deliveries, especially with multilingual labeling requirements. For manufacturers and downstream buyers alike, clarity in naming cuts down on costly mistakes.
Safety & Operational Standards: From Theory to Shop Floor
Working with 2-Methyl-1-Pentanol, safety lines up as top priority. Common themes repeat: equipment rated for flammable materials, scrupulous ventilation, PPE ranging from gloves to goggles, and an eye on spill containment. The chemical’s moderate volatility means a well-run facility won’t lean only on fume hoods but checks for long-term exposure with air monitoring—particularly in older plants where modernization often lags behind standards. Regulatory bodies put 2-Methyl-1-Pentanol in flammable liquid categories, and incidents on record underscore the importance of staff training, robust spill kits, and clear hazard communication. Industry best practice emphasizes not just reaction conditions, but also the routines that keep teams safe during cleaning, maintenance, and transfer.
Where It Goes: Application Footprint
Formulators in paints, coatings, and adhesives circles use 2-Methyl-1-Pentanol not just for its solvency, but for how well it influences drying times and flow control within finished products. The molecule pops up in specialty plasticizers, tweaking flexibility in everything from cables to soft-touch polymers found in consumer goods. Flavor and fragrance chemists reach for it in the quest for nuanced, tenacious notes, but only after rigorous toxicological review. I once helped a team hunt for substitute solvents compliant with new VOC caps—2-Methyl-1-Pentanol made the shortlist because it balanced performance and cost without derailing entire product lines.
R&D and The Search for New Avenues
Research professionals keep probing 2-Methyl-1-Pentanol’s molecular potential, both in basic academic work and behind closed doors in corporate settings. Patents pile up for custom esters, hand-tailored surfactants, and solvents tuned for engineered materials. Academic collaborations cross national borders, pushing for greener, smarter, and safer syntheses. Emerging fields like green chemistry attract grant money and startup investment, promising cleaner synthesis or bio-based derivatives. At conferences or through journal alerts, it’s common to see papers on catalytic advancements or bio-alternatives, as the field pursues both lower environmental impact and broader industrial flexibility.
Toxicity Research: Looking Out for Risk
All chemicals surface questions of exposure and toxicity. For 2-Methyl-1-Pentanol, data gaps from decades past gradually shrink as new studies roll in. Acute toxicity sits in ranges comparable with similar medium-chain alcohols, with respiratory irritation and skin contact standing out as practical hazards in case of spills or poor air handling. Researchers study metabolic breakdown in animal models and in vitro systems, giving regulators evidential grounding to set workplace exposure limits and permissible concentrations in consumer products. Modern toxicity studies use advanced in silico models and high-throughput screening, speeding up detection of subtle or long-term effects—crucial as regulatory scrutiny tightens and companies chase approvals for emerging uses.
What Comes Next: The Forward View
Looking around chemical manufacturing today, there’s a clear urge to clean up origins, shrink carbon intensity, and catch up to ever-stricter standards. 2-Methyl-1-Pentanol sits smack in the middle of this wave. Process developers look for better catalysts, less waste, and biorenewable outcomes, eyeing both cost and regulatory targets. Tightening environmental rules in markets like the EU push companies to consider not just what goes into a drum, but how it gets there, who handles it, and what footprint is left behind. End users in paints, plastics, and flavors keep pushing for tailored solutions, nudging suppliers to deliver more, with fewer compromises on safety or sustainability. This molecule may not make headlines, but its performance and adaptability keep it embedded across countless supply chains, quietly reminding us that the chemistry we overlook often drives the products we touch every day.
What is 2-Methyl-1-Pentanol used for?
Everyday Impact of a Lesser-Known Alcohol
My early years working in a small chemical plant showed me that many compounds stay out of the limelight, yet they're quietly essential in everyday products. One such chemical, 2-Methyl-1-Pentanol, can be found behind the scenes in industries that keep the world running smoothly. It’s a colorless liquid with a faint odor, and even though few people talk about it, some important sectors rely on its unique set of qualities.
Solvent Power in Industry
2-Methyl-1-Pentanol’s main job lies in its use as a solvent. In paint factories, I watched this alcohol mix with pigments and resins, helping everything blend in a way the human eye never notices. Its molecular structure gives it strength for dissolving substances that water can’t touch. Companies producing coatings and inks count on it for consistent results and smooth texture. In my days packaging specialty paints, I noticed the difference a good solvent made, especially in drying times and finish quality.
Creating Flavors and Fragrances
Some forms of 2-Methyl-1-Pentanol also support the creation of flavors and fragrances. Synthetic chemistry teams turn to compounds like this alcohol to help generate esters, which provide fruity or floral tones. These esters end up in perfumes, cleaners, or even chewing gum. Anyone who’s worked in a flavor house remembers the delicate balance of ingredients needed to capture natural scents or tastes—and 2-Methyl-1-Pentanol often stood in as a building block for their formulas.
Pharmaceutical Synthesis
In the pharmaceutical world, this alcohol helps create active ingredients or intermediates for medicinal compounds. Chemists appreciate the way it reacts under controlled conditions. I recall discussions with colleagues about the importance of purity standards, especially in any material destined for use in medicine. Here, trace impurities could mean stopping a whole production batch, so sourcing good-quality 2-Methyl-1-Pentanol meant fewer headaches for everyone involved.
Lubricants and Plasticizers
Automotive workshops and plastics labs also use 2-Methyl-1-Pentanol as a starting material for lubricants and plasticizers. These additives make products last longer and work better under tough conditions. Heavy machinery, conveyor belts, and flexible plastics often need a touch of this compound to reach the right balance between stiffness and flexibility. After years spent watching production lines break down from cut-rate materials, I’ve learned the hard way that choosing a solid plasticizer source helps prevent a mountain of maintenance bills.
Safety and Environmental Attention
Government regulations guide chemical production and use, so health and safety must come first. Despite its usefulness, mishandling this chemical can cause health problems. Strong odors sometimes linger in closed rooms, and even small spills demand quick action. Laws in the US and Europe outline rules on exposure and disposal. The American Conference of Governmental Industrial Hygienists posts regular updates on safe levels, and any responsible employer keeps Material Safety Data Sheets within reach.
Finding Solutions for the Future
It takes teamwork between manufacturers, regulators, and research teams to reduce risks while preserving the benefits of 2-Methyl-1-Pentanol. Training programs teach workers about PPE and handling. Labs keep searching for even safer substitutes. Green chemistry initiatives push for less hazardous processes and ingredients.
So much relies on compounds that most people never hear about. 2-Methyl-1-Pentanol, for all its obscurity, helps industries create better products every day—quietly shaping everything from the color on your walls to the scent of your favorite soap.
What are the safety precautions when handling 2-Methyl-1-Pentanol?
Pay Attention: Why 2-Methyl-1-Pentanol Demands Respect
2-Methyl-1-Pentanol isn’t some everyday household cleaner. The liquid might look harmless, but chemical exposure can flip a normal workday upside down fast. I’ve seen co-workers rush a project, splash solvents across their hands, and end up with weeks of skin irritation. Respiratory complaints aren’t rare at all either. These issues crop up in labs, warehouses, and maintenance shops, not just chemical plants. People often think, “I’ve done this job for years.” Yet accidents pick anyone who skips the basics.
Protection Starts with What You Put On
Splashing a chemical like this on your skin almost guarantees trouble. Nitrile gloves beat latex every time; 2-Methyl-1-Pentanol can seep through cheap material. Go for long sleeves, chemical-resistant aprons, and always put on goggles. Face shields block unexpected sprays during transfers or pouring. Simple tasks – moving a drum or rinsing glassware – can go sideways if you don’t suit up right.
Air Quality Makes All the Difference
This liquid gives off a strong odor for a reason. Inhaling the vapors day in, day out leads to headaches and dizziness. I’ve worked in cramped storage rooms where fans made the difference between a regular shift and a splitting headache. Fume hoods and proper exhaust systems don’t need fancy budgets – even a basic local vent or open window can help. For big jobs or longer exposures, fit-tested respiratory protection isn’t just for show – it’s the difference between safe handling and a health risk.
What Spills Teach Us
Spills can happen during delivery or mixing. Fast response prevents a slippery mess from turning into an emergency. Absorbent pads, sand buckets, and spill kits placed near workstations make sense. People forget that 2-Methyl-1-Pentanol burns easily. No open flames, no smoking, and no welding near storage or open containers. I once watched a workshop boss lose half a day because someone tossed a used rag into a regular trash can. Containers should stay sealed, clearly labeled, and stored in cool, ventilated rooms away from direct sunlight.
Training Should Stick
No one expects to memorize every data sheet. Still, regular safety training gives teams the confidence to spot trouble and fix it before it snowballs. Eye-wash stations and shower points should actually work, and everyone should know where to find them. I’ve seen new hires and old hands alike freeze during emergencies. A few minutes of hands-on practice saves hours of panic when something really happens.
Smarter Use Begins with Respect
2-Methyl-1-Pentanol is useful in making flavors, fragrances, and specialty plastics, but safety never takes a back seat, whether blending batches or cleaning up glassware. Respect for these chemicals creates a work culture where people pay attention to their health and never treat shortcuts as time savers.
Nobody pulls these habits out of thin air. It takes real reminders, regular checks, and a willingness to look out for co-workers. That attitude turns chemical safety into everyday practice, not just lab talk.
What is the chemical formula of 2-Methyl-1-Pentanol?
Digging Into the Compound
2-Methyl-1-pentanol pops up in the world of organic chemistry, neatly tucked in the family of alcohols. It holds a chemical formula of C6H14O, which means it has six carbons, fourteen hydrogens, and an oxygen. Although numbers and letters on paper don't raise eyebrows, this little molecule carries weight in lab settings and the production line. I still remember my early lab classes, scribbling such notations and wondering why a single atom shift mattered. Over time, I realized that a subtle swap in position sets apart two chemicals in ways that can impact everything from scent to how the compound reacts.
Why Structure Matters
In 2-Methyl-1-pentanol, the methyl group branches off the second carbon of the pentanol chain. This tiny shift changes how the molecule behaves. For anyone who dabbles in synthesis, structure matters. C6H14O isn’t just something for the textbook—it shapes how manufacturers get the blend and performance they want from flavors, fragrances, or even solvents. Chemical structure touches on things like boiling point, miscibility, and how the body processes it, which in turn touches research safety and regulations. EPA and ECHA databases keep tabs on details for precisely this reason, flagging data sets for compounds with close relatives but different safety profiles.
Day-to-Day Relevance
I’ve seen people roll their eyes at chemical jargon, and maybe that’s because it rarely seems to cross paths with daily life. In fact, these alcohols blend into consumer products, specialty chemicals, and fuel research. 2-Methyl-1-pentanol can enter the spotlight as a building block in organic synthesis, or as a niche solvent where something less common fits the bill. Scientists look at its relatively low toxicity and find ways to work with it safely, whether in industrial environments or labs. Understanding chemicals by name and formula tells a story about where and how they show up, not just in beakers, but in finished products on store shelves.
The Bigger Picture—Safety and Synthetic Applications
Paying attention to the structure also has practical consequences for safety engineers and environmental chemists. Products and by-products with similar formulas—often called isomers—don’t always behave the same. C6H14O could mean hexanol, 2-hexanol, or 3-methyl-1-pentanol, each with quirks that show up in flammability, toxicity, or even environmental persistence. These details go straight into the discussion when designing safer chemical workflows or developing data sheets for industry. Producers, regulators, and researchers share the same goal: keeping people and places safe while getting consistent results.
Improving Chemical Literacy
Molecules tend to stay out of sight, out of mind. Still, learning the pieces that make up 2-Methyl-1-pentanol can help demystify the universe of products, research, and risks. Educational tools and digital databases can close that gap. I’ve leaned on open-access platforms and hands-on learning to see how formulas translate to real-world impacts. Individual curiosity supported by solid sources may not spark instant community change, but it gives people power to navigate daily decisions about safety, sustainability, and science. Every chemical story, even if only a formula, has a role in shaping a safer, more informed society.
How is 2-Methyl-1-Pentanol stored?
Keeps Things Running, But Needs Respect
Anyone who’s spent time managing chemicals in a lab or industrial setting learns quickly that storage isn’t just an afterthought. Especially with substances like 2-methyl-1-pentanol, a flammable alcohol, getting storage right prevents headaches that go way beyond bad smells or wasted material—safety sits on the line. Flammable liquids can change a regular day into a fire drill faster than you can grab a spill kit.
The Safety Baseline
First up, flammability stands out as the core hazard of 2-methyl-1-pentanol. The flash point, at roughly 60°C, pushes it right into a risky category. Watching out for ignition sources—open flames, sparks from bad wiring, even static discharge—matters every single day. I’ve seen times when a “temporary” container left on a cluttered workbench led to trouble nobody needed.
Experience in labs tells me: a proper safety cabinet, marked clearly for flammables, saves a lot of trouble. Metal cabinets with self-closing doors and spill containment in the base make sense. Too many folks underestimate just how quickly a small leak or a turned-over bottle becomes dangerous when vapors build up. Ventilation isn’t an optional extra in spaces where vapors from alcohols can collect.
Containers Matter
Original, tightly sealed containers matter as much as location. Manufacturers sell 2-methyl-1-pentanol in specialized drum or bottle designs that slow evaporation and resist corrosion. I always check that caps actually thread all the way down—crystalized residue or overtightened lids lead to cracks or air leaks. If the container looks compromised in any way, it doesn’t belong near storage for the rest of the stock.
Glass or high-grade HDPE plastic works best, but storage should never be above eye level or somewhere containers can drop off a cluttered shelf. Once, a simple knock sent a bottle rolling off a high rack. The scramble to clear and ventilate after that left us much warier of shelf placement. The lesson: always use solid shelving that gives bottles their own clear space.
Temperature and Light: Subtle Saboteurs
Heat kicks up vapor pressure, ramps up flammability risk, and can even degrade the chemical over time. Cool, dry storage—often 15-25°C for most labs—wards off these problems. Humidity sneaks in as a problem too, especially if a seal loosens, since water can affect both purity and reactivity. Sunlight does more than fade labels. UV can spark reactions, so a dark cabinet or storeroom counts for more than people realize.
Keep It Separate, Know Your Neighbors
I’ve walked into too many shared storerooms where acids, bases, and solvents crowded a shelf. Even among alcohols, 2-methyl-1-pentanol has no business next to oxidizers or strong acids—mixing those up risks fires or toxic fumes. Storing chemicals by hazard class, not by alphabet or convenience, kept my team from making expensive mistakes more than once.
Training and Labels Save Lives
Clear labeling—chemical name, hazards, date of receipt—beats guessing and accidental misuse. Everyone on the team, from interns to senior staff, understands what sits behind yellow doors. Routine training and regular audits catch mistakes before they become emergencies. On one shift, a missing hazard pictogram could have cost hours, if not more. Catching it on a walkthrough kept things trouble-free.
Real-World Solutions
From my years around students and professionals, storing 2-methyl-1-pentanol safely boils down to four points: respect fire risk, guard against leaks, keep things cool and dark, and never slack on labeling or training. Simple steps; real-world payoff in safety and peace of mind.
Is 2-Methyl-1-Pentanol hazardous to health or the environment?
What is 2-Methyl-1-Pentanol?
Anyone who’s spent some time in a chemistry lab, or even reading labels on industrial products, might spot the name 2-Methyl-1-Pentanol. It’s one of those colorless liquids with a faint scent, often tucked away in the “organic solvents” section. It finds use across several manufacturing processes, from coatings and lubricants to plasticizers and flavors. At first glance, it appears as another cog in the factory machine, but behind its usefulness are questions that matter to everyday people: Is it risky to handle, and should we worry about what it leaves in our world?
Health Risks: Skin, Eyes, and More
No one wants to walk away from a day’s work with skin burns or eye irritation. Spills of 2-Methyl-1-Pentanol can sting your eyes or redden your hands. Even short exposure on bare skin causes discomfort, and breathing in its vapors can lead to headaches, dizziness, or a scratchy throat if you’re stuck in a stuffy space. Longer-term exposure hasn’t been studied like household chemicals or common pesticides, so doctors and safety folks usually play it safe. Workers get gloves and goggles. They keep the room air moving, partly because memories of chemical headaches linger longer than most safety briefings.
Human Experience and Workplace Safety
I remember a friend who worked around industrial alcohols. He always made sure to keep lunch away from the workbench. Spilled liquids and hidden residues have a sneaky way of finding unwashed hands. We joked about “chemical cologne,” but no one wanted to take chances with solvents. This kind of real-world risk—skin or eye contact, or accidental inhalation—matters more than a technical data sheet. Factories call for safety signs and ventilation, but home hobbyists and small-scale users sometimes rely on hope rather than gloves. That’s where stories of careless exposure usually start.
Environmental Impact: Nature Never Forgets
The fate of chemicals doesn’t end with the drain. 2-Methyl-1-Pentanol breaks down in air after a few hours to days, but spills in rivers or soil tell a different story. Fish and bugs in streams can’t handle many solvents. Even mild concentrations can disrupt tiny water life. For years, reports out of research labs have pointed to the risk of aquatic toxicity. Wastewater treatment handles most city-scale discharges, but accidental leaks at refineries or spills during transportation slip through holes in the system. The result feels small until you see a stream lose its shimmer—or hear about a dead zone downstream from an industrial park.
Better Practices—Room for Change
Plenty of safer solvents exist, but swapping them in isn’t always possible—or cheap. Simple steps cut down risk. Ventilation systems aren’t glamorous, but they keep vapors out of lungs. Tight-fitting lids and handling training help prevent spills. Proper glove use stops skin rashes. Spent solvents need collection and high-temperature incineration, not backyard dumping. Local rules often lag behind best practices, so individual responsibility carries real weight. Sometimes curiosity and caution—asking “what does this do to me, what does this do to the creek?”—get overlooked for speed or cost savings.
Choosing Safety Today
2-Methyl-1-Pentanol works as intended in many industries, but without thoughtful handling, its risks to people and planet stack up quickly. Balancing business needs, worker protection, and nature calls for real attention—not just box-checking and paperwork. The best solutions start with asking how each spill or whiff might ripple out far beyond the factory gates. Care in today’s choices shows up in better health and cleaner water tomorrow.
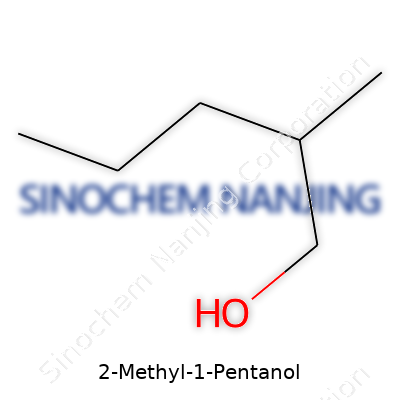
| Names | |
| Preferred IUPAC name | 2-Methylpentan-1-ol |
| Other names |
2-Methylpentan-1-ol isohexyl alcohol 2-Methyl-1-pentyl alcohol 2-Methylpentanol-1 |
| Pronunciation | /tuː ˈmɛθ.əl wʌn ˈpɛn.tə.nɒl/ |
| Identifiers | |
| CAS Number | 626-89-1 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:34219 |
| ChEMBL | CHEMBL15362 |
| ChemSpider | 14519 |
| DrugBank | DB14036 |
| ECHA InfoCard | 100.009.067 |
| EC Number | 1.1.1.1 |
| Gmelin Reference | 79239 |
| KEGG | C06007 |
| MeSH | D010465 |
| PubChem CID | 12016 |
| RTECS number | SA9100000 |
| UNII | 737I9S86FA |
| UN number | UN2282 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Colorless liquid |
| Odor | mild alcoholic odor |
| Density | 0.814 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.83 |
| Vapor pressure | 0.48 mmHg (25°C) |
| Acidity (pKa) | 16.1 |
| Magnetic susceptibility (χ) | -7.82×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 4.6 mPa·s (20 °C) |
| Dipole moment | 2.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 334.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -368.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3899.6 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P305+P351+P338, P304+P340, P403+P235 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 72 °C |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Explosive limits | 1.1–6.4% |
| Lethal dose or concentration | LD50 oral rat 3300 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 3000 mg/kg |
| NIOSH | SA1750000 |
| PEL (Permissible) | Not established |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
1-Pentanol 2-Pentanol 3-Methyl-1-butanol 2-Methyl-2-pentanol 2-Hexanol |

