2-Mercaptopropionic Acid: A Commentary on Its Journey and Role in Modern Chemistry
Unpacking the History and Development of 2-Mercaptopropionic Acid
Looking at the timeline of organic chemistry, few small molecules managed to carve out as many roles as 2-Mercaptopropionic Acid. Early chemists, searching for new ways to attach sulfur into carbon chains, experimented with this compound’s structure in attempts to unlock unique reactivity. The rise of analytical methods in the 20th century, including nuclear magnetic resonance and mass spectrometry, gave chemists better ways to understand its structure and reactivity. Over time, researchers learned to handle thiols—compounds featuring sulfur groups—with better control, allowing safer and more predictable use of molecules like 2-Mercaptopropionic Acid across research and industry. Its story grew alongside improvements in synthesis strategies, purification, and handling, reflecting the larger growth of organic chemistry as a field.
A Closer Look at the Molecule Itself
2-Mercaptopropionic Acid, sometimes called thiolactic acid or MPA, stands out with a simple yet distinctive structure: a three-carbon backbone hosting both a carboxylic acid group and a thiol (mercapto) group. The molecule brings a pungent, sulfurous odor—a hallmark of thiols. Unlike bulkier or more complex organic molecules, its water solubility sits in a practical range for both lab work and industrial use, without making it too volatile or unwieldy. At room temperature, it takes the form of a clear to slightly yellowish liquid. Its sulfur group is sensitive to oxidation, so storage conditions often require antioxidants or inert atmospheres to avoid unwanted byproducts. Basic chemical data, such as a molecular weight near 106 g/mol and a melting point below room temperature, keep it accessible for many applications.
Technical Aspects and Labeling Practices
Accurate labeling holds real importance for chemicals like 2-Mercaptopropionic Acid, especially given the variety of settings where it sees use. Labels typically highlight the potential for skin and eye irritation, both due to its acid group and its reactive sulfur atom. There’s no room for shortcuts in storage instructions since improper handling increases the risk of unpleasant smells, hazardous byproducts, and health concerns. Chemists trust suppliers who keep impurities—especially related organosulfur compounds—below tight thresholds. Industry standards often look for precise percentages of 2-Mercaptopropionic Acid purity, clarity on water content, and traces of oxidation byproducts like disulfides.
Synthesis and Preparation Methods
Laboratories and factories rarely settle for only one route to make 2-Mercaptopropionic Acid. A classic approach reacts chloroacetic acid with hydrogen sulfide or sodium hydrosulfide, taking advantage of the nucleophilic substitution that lets sulfur swap in where a leaving group once sat. Chemists may adjust solvent, temperature, and the order of reagents to tweak yields or lessen byproduct formation. In today’s effort to reduce hazardous waste and improve safety, greener alternatives leverage less-toxic thiol sources or find catalyst systems that cut down both waste and energy requirements. Every choice in preparation leaves its mark on purity and downstream usability, especially when the product gets used in sensitive fields like pharmaceuticals or electronics.
Exploring Chemical Reactions and Modifications
2-Mercaptopropionic Acid’s value often traces back to the unique chemistry of its sulfur atom. The thiol group gives access to reactions that link this molecule to metals, create disulfide bridges, or cap polymer chains for precise control over molecular architecture. Bioconjugation exploits this same functionality for linking to proteins or other biomolecules at defined sites. Oxidation, alkylation, and esterification reactions all play a role in transforming this small acid into countless specialized derivatives. In synthetic chemistry, it works as both a building block and a modifying agent, creating linkages that resist hydrolysis or foster stability in complex compounds.
Names and Synonyms
Chemists use a handful of different names for 2-Mercaptopropionic Acid, though the practical differences mostly stop at the label. Depending on what’s printed on the bottle or the language used in a paper, terms like thiolactic acid, 2-sulfanylpropanoic acid, and MPA refer to the same core structure. While these synonyms can cause confusion for those reading across different disciplines or regulatory frameworks, familiarity comes quickly to those who work with the compound over time.
Meeting Safety and Operational Standards
Staying safe with 2-Mercaptopropionic Acid depends on respecting the risk tied to its reactive thiol group. Short-term health impacts—especially irritation to skin, eyes, and membranes—make proper ventilation and personal protective gear a fixed part of every protocol. The strong, almost rotten odor signals leaks or spills, requiring immediate cleanup to avoid health risks and to prevent the smell from spreading. Standards from organizations like OSHA guide handling, storage, and emergency response, placing rigorous emphasis on spill containment and safe disposal. Modern labs and production plants keep thorough training for staff, and automation cuts down direct handling where possible. Anyone working with this molecule comes to recognize both its hazards and the steps that turn it into a manageable part of their daily work.
Pathways to Application: Where 2-Mercaptopropionic Acid Plays a Role
Think about industrial uses: the molecule acts as a chain transfer agent in the polymer industry, controlling molecular weights and branching in products like acrylics and related plastics. In the pharmaceutical sector, its ability to link to proteins or peptides supports drug delivery and biomarker research, thanks to the reliable chemistry of its thiol group. It provides corrosion resistance for metals by coordinating to surfaces, a trick used in both coatings and electronics. Analytical chemists use it to modify electrodes or to create self-assembled monolayers, giving them more flexibility over surface characteristics. The reach of this compound extends into agriculture too, where it modifies pesticide formulations for better delivery.
Deepening Research and Innovation
Recent years brought increased interest in eco-friendly synthesis and new applications. Research teams use 2-Mercaptopropionic Acid to stabilize metal nanoparticles, enabling advances in catalysis, sensor technology, and nanoelectronics. The molecule’s small size and high reactivity let scientists experiment with controlling material surfaces at the atomic level, key for next-generation devices. Biotechnology groups see it as a bridge for linking biological and synthetic systems, a step toward more effective biosensors and diagnostics. Academic partnerships, open-access databases, and global conferences all feed new directions for using and modifying this molecule. The growing focus on finding nontoxic, sustainable alternatives has pushed for improvements in its synthesis, not only in lab and pilot scales but all the way up to commercial production.
Toxicity and Human Health Concerns
Understanding the toxicology of 2-Mercaptopropionic Acid means facing the reality of working with a molecule that can irritate eyes, skin, and airways. Exposure above occupational limits prompts both short-term and chronic effects, underscoring the need for routine monitoring wherever it’s handled. Some animal toxicity studies suggest that higher doses can produce severe reactions, but routine lab and industrial controls—ventilation, gloves, and fume hoods—substantially cut risk when used correctly. Environmental questions focus on breakdown products and possible bioaccumulation, so regulatory agencies watch closely for contamination in water or soil near where it’s produced or used. Progress in green chemistry points toward safer analogues or modified practices, reflecting a clear direction for continued innovation.
Looking Ahead: Possibilities for the Future
Chemists, engineers, and regulatory bodies all take an interest in what comes next for 2-Mercaptopropionic Acid. Its role in creating smarter, more functional polymers will likely keep growing, especially as industries look for more sustainable plastics and specialty materials. The boom in nanotechnology puts further spotlight on this molecule’s surface chemistry potential. In biomedicine, researchers are pushing the limits in linking therapies or diagnostics to natural biomolecules in ever-safer, more controlled ways. The steady drive for green technology means every step—from raw-material sourcing to waste handling—will keep evolving. If the decades of experience with 2-Mercaptopropionic Acid have proven anything, it’s that its simple structure still has room to surprise and to enable solutions in unexpected corners of science and industry.
What is 2-Mercaptopropionic Acid used for?
A Niche Chemical Making a Big Impact
Walk through a chemical plant or glance around a modern lab, and you might spot a bottle labeled “2-Mercaptopropionic Acid.” Folks in the industry usually call it MPA. The name may not ring a bell unless you’ve worked with polymers, specialty plastics, or certain medical supplies. This sulfur-based compound smells sharp, not unlike rotten eggs—no mistaking its presence in a lab. Despite its modest profile, MPA plays several important roles in fields that affect our everyday lives.
Shaping Everyday Plastics and Resins
MPA grabs attention in the acrylics industry. Take acrylic glass: that sturdy, transparent stuff used for skylights, vehicle windows, and aquariums. Manufacturers rely on 2-Mercaptopropionic Acid as a chain transfer agent in making poly(methyl methacrylate) (PMMA) and other acrylic resins. By using it, they can fine-tune the strength, flexibility, and clarity of the end product. Cutting corners on this chemical means risking inconsistent results in plastic manufacturing—an issue I’ve seen lead to entire batches getting scrapped. According to market research from Grand View Research, the global acrylic resins market clocks in at billions of dollars. Even small quality issues can interrupt supply chains or result in product recalls.
Helping Science Push Boundaries
MPA doesn’t only work behind the scenes in plastics. Research labs use it for surface modification. Scientists add a layer of MPA to tiny gold or iron particles called nanoparticles. This tweak lets researchers attach different drugs or sensors to a nanoparticle, designing targeted treatments or clever diagnostic tools. For instance, in cancer research, MPA-coated nanoparticles can help deliver chemo drugs to tumors with greater precision. Journals like “ACS Nano” and “Journal of Biomedical Nanotechnology” point to research that uses this compound as a linking agent between medicine and delivery vehicle, aiming for fewer side effects and better outcomes.
Environmental and Health Concerns
No one wants toxic leftovers from manufacturing. MPA needs strict handling—its strong sulfur smell signals real risk, including skin and eye irritation. Accidental releases can harm wastewater systems, so factories install careful monitoring and filtering. Regulators like the Environmental Protection Agency (EPA) and the European Chemicals Agency (ECHA) publish guidelines on safe use and disposal. People who handle this compound benefit from safety training and protective gear. Having worked at a facility where improper handling forced a shutdown, I can say safety protocols must stay front and center.
Innovation and Responsibility Moving Forward
Making better plastics and advanced medical tools is great, but companies and labs have to act responsibly. Regular audits, investment in better fume hoods, and new training methods keep risks in check. Some labs experiment with “greener” transfer agents that reduce or replace hazardous chemicals like MPA. Collaboration between scientists and industry leaders speeds up safer alternatives. It’s not just about ticking boxes; each improvement helps protect people, communities, and local ecosystems. As sustainability becomes more valuable to manufacturers and consumers, demand will grow for safer and environmentally friendly production methods. My take: putting health and safety first never slows real progress.
What is the chemical formula of 2-Mercaptopropionic Acid?
Understanding the Chemical Formula
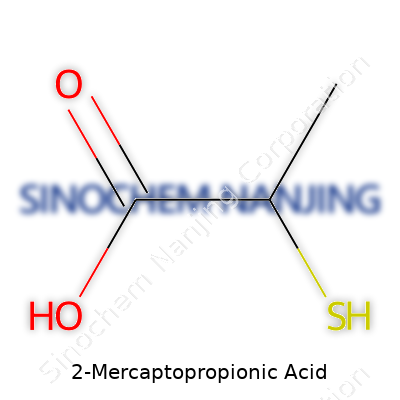
2-Mercaptopropionic acid grabs attention in the world of organic chemistry. Its chemical formula is C3H6O2S, which tells a pretty direct story: three carbon atoms, six hydrogens, two oxygens, and a single sulfur. It might sound dry, but that formula shapes how this compound behaves, how it interacts with other substances, and why it shows up in so many applications.
What Sets 2-Mercaptopropionic Acid Apart?
The “mercapto” group (–SH) is what gives this molecule its edge. That sulfur-hydrogen pair brings a strong and sometimes pungent character. Chemistry students might remember the smell that tells you a thiol is in the room. Combined with a carboxylic acid group, it unlocks reactions most single-function molecules can’t approach. The placement of these groups, two carbons apart, turns this compound into a ready tool for making more complex molecules.
Uses and Importance in Everyday Life
Labs never really run out of needs for 2-mercaptopropionic acid. In my years working around biotech research, this molecule kept showing up as an anchor in various syntheses. It helps make pharmaceutical intermediates, ties proteins to other molecules for diagnostics, and can improve the way plastics behave by linking their chains. Factories use it in making acrylic acid derivatives, which land in paints, adhesives, and even medicine capsules.
The mixture of a thiol and carboxyl means it can latch onto metals, especially in the purification of precious ones like gold. Environmental cleanup firms use this trait to separate out toxic metals in wastewater, making disposal much safer. These aren’t distant, laboratory-only uses. They end up touching healthcare, manufacturing, and environmental safety.
Handling Risks and Safety
With the benefits comes risk. The same chemical groups that make 2-mercaptopropionic acid valuable can create problems if handled carelessly. The thiol delivers a powerful odor, but more pressing is its ability to irritate or harm skin and lungs. I once watched a newly minted technician learn this the hard way; gloves, goggles, and a decent fume hood prevent more trouble than most warnings ever will. Safety data sheets matter, and they save grief. The chemical itself can form fumes, and a spill lingers far longer than anyone likes, so cleanup procedures stay important.
Environmental risk looms if it escapes into waterways. Extra training for anyone using it helps avoid costly mistakes. More companies update protocols, switch to closed systems for transfer, and tag storage containers with clear, bright labels. These steps keep both people and local ecosystems a bit safer.
Moving Toward Smarter Chemistry
Some researchers now seek new derivatives, aiming to keep the beneficial properties of 2-mercaptopropionic acid, but dial down the risk or the smell. Education helps here; the more people know about what goes into a reaction, the less likely accidents will happen. Looking back, the path from textbook formula to field use for this compound has been shaped by necessity and a big dose of caution. Smarter handling and sometimes, smarter chemistry, point the way forward for both labs and industry.
What are the safety precautions for handling 2-Mercaptopropionic Acid?
Why Take Extra Care with 2-Mercaptopropionic Acid?
If you’ve spent time in a chemistry lab, you know how something that seems harmless can still pack a punch. 2-Mercaptopropionic acid isn’t just a mouthful to say—it’s a chemical that smells like rotten eggs and can bite if you’re careless. This acid, important in pharmaceuticals and resins, brings risks worth respecting. A splash, a sniff, even a spill can cause problems for your skin, eyes, nose, and lungs.
Protective Gear Makes All the Difference
I’ve watched new lab workers handle acids with bare hands, thinking a quick drop here or there won’t matter. That approach leads to burned skin and ruined gloves. This acid burns and irritates on contact. Don’t skip the basics: Wear proper chemical-resistant gloves, such as nitrile or neoprene—not those thin latex types. Always suit up with goggles or a face shield and a lab coat that covers wrists and arms. Gloves stop burns, goggles protect your vision, and a coat prevents splash marks on your clothes and skin.
Ventilation: Breathing Easier
The stench isn’t just annoying; those fumes can trigger headaches, sore throats, or worse. Working in a fume hood keeps vapors out of your lungs. A simple room fan doesn’t cut it. Even after years in the lab, if I ever tried to shortcut here, I quickly regretted it. A well-maintained hood lets you focus on the experiment and not the air quality.
Smart Storage and Labeling
Storing 2-mercaptopropionic acid with flammable or reactive substances increases risk. It goes in a cool, dry spot away from sunlight, ignition sources, and anything it might react with. I’ve seen cabinets with poorly labeled containers; those mistakes lead to confusion and sometimes danger. Use sealed, clearly marked bottles. Add a hazard label with the chemical’s full name and date received. The right storage prevents accidental mix-ups and keeps emergencies to a minimum.
Spill and Exposure Response: Don’t Hesitate
Spills happen, no matter how careful you are. If the acid hits skin or eyes, rinse with water for at least 15 minutes. Use the eyewash station, not just a quick splash. For skin, take off gloves and rinse fully—no shortcuts. As for spills on benches or floors, contain them with absorbent materials meant for acids. Never try to mop it up bare-handed or with regular rags. If there’s a question about inhalation, get outside for fresh air.
Training Matters
Rules on paper don’t count for much if nobody reads them. Labs that put extra time into safety refreshers handle accidents better. A solid training program about chemical dangers, PPE, and emergency procedures turns “I didn’t know” into “I know what to do.” When I ran safety drills with my team, mistakes showed up in practice so that they didn’t show up under pressure.
Disposal: Keeping Communities Safe
Flushing chemicals down the drain isn’t just lazy, it’s illegal and dangerous. This acid needs neutralization and proper collection before disposal, following local hazardous waste guidelines. The right waste bin, picked up by specialists, goes a long way in protecting both workers and the environment.
Final Thoughts
A little care with 2-mercaptopropionic acid prevents most problems. The basic steps—personal protection, ventilation, smart storage, training, and responsible disposal—build a layer of safety that stands up to even the nastiest surprises. I’ve seen labs thrive or falter depending on these habits, and the right approach always makes life better for everyone on the team.
How should 2-Mercaptopropionic Acid be stored?
Understanding the Substance
2-Mercaptopropionic acid plays a significant role in various industries, from pharmaceuticals to polymers. Its structure features a thiol group, which means it brings along both a pungent odor and a strong tendency to react with oxygen. This isn't just a chemical detail; it changes the way you deal with this substance on a daily basis, especially when it’s time to put it away after a long day in the lab.
Storage Realities Faced by Professionals
Anyone who’s worked with mercaptans remembers that odor that doesn’t easily fade. Not taking storage seriously can mean a lab that smells for days, cross-contamination, or worse, chemical degradation. Left exposed, 2-mercaptopropionic acid oxidizes and might even produce compounds with higher toxicity. Ignoring best practices does not just frustrate co-workers—it messes up results and chews through budgets by ruining valuable supplies.
Oxidation stands out as a real enemy. In years of handling lab chemicals, I’ve seen enough experiments thrown off because someone screwed the cap loosely or left reagents in sunlight. The acid can form disulfides if exposed to air—that’s not just a theoretical risk, it’s a real headache for anyone relying on chemical purity.
Practical Steps for Safe Storage
The right container makes all the difference. Glass offers the most reliable storage option for this kind of acid, since it won’t react or degrade, provided it’s kept tightly sealed. Polyethylene containers offer another route, but I always lean toward glass for long-term peace of mind.
Not all spots on a shelf are equal. Keep it in a cool, dry spot, far away from sources of heat or direct sunlight. Moisture and heat speed up any reaction—just ask anyone who’s cleaned up a spill after a careless placement near a sunny window. Storing the acid at temperatures under 25°C helps maintain its stability. Refrigeration works, but frost-free freezers should be avoided due to periodic warming cycles, which can cause condensation inside containers.
Ventilation matters. In shared facilities, I always store 2-mercaptopropionic acid inside a well-ventilated cabinet, preferably one designed for corrosives. This approach does two things—keeps vapors from building up in case of leaks and avoids unhealthy work environments. Fume hoods offer an extra barrier during transfers, not just for lab safety requirements but for everyday peace of mind.
Risks Worth Taking Seriously
Ignoring the basics invites more than just bad smells. Accidental mixing with bases or oxidizers can trigger dangerous reactions, and spills can eat through certain floor coatings. Label the container with the date of receipt and opening. Keeping a material safety data sheet closeby is not bureaucratic overkill—people forget what’s behind a faded label.
Simple Routines for Long-Term Results
Store small quantities rather than big bulk bottles. This approach limits the impact of accidents and makes inventory checks much easier. Keep personal protective equipment within arm’s reach. I always double-check gloves and goggles before handling acids like this because it only takes one splash to learn a lifelong lesson about chemical safety.
What are the typical applications of 2-Mercaptopropionic Acid in industry?
Working with Additives in Plastics and Polymers
2-Mercaptopropionic acid shows up on many chemical inventories at plastics plants. It helps shape the features of finished products by acting as a chain transfer agent, especially during the creation of polymers like polyvinyl chloride (PVC) and acrylics. People on these production floors use small amounts to get a handle on molecular weight, which affects the way a finished sheet or pipe bends under pressure or stress. This single tweak has a ripple effect, offering manufacturers a tool to improve flexibility, lower brittleness, and hit performance targets that last through years of use.
There’s also a cost angle. By using 2-mercaptopropionic acid in these reactions, fewer problems show up with melt flow or processing errors at the plant. That means less waste, less rework, and lower energy use — benefits that anyone managing an industrial budget can appreciate.
Controlling Curing in Adhesive and Coating Production
Walk into a facility making specialty adhesives or protective coatings, and you’ll notice a focus on curing times and consistency. The industry counts on additives that ‘talk’ well with free radical polymerization systems. 2-Mercaptopropionic acid offers up a functional thiol group, making it a go-to for managing polymerization rates. If you ever struggled with adhesives setting up too quickly on a line, you’d welcome an ingredient that allows a more predictable cure time, improving batch-to-batch reliability.
That reliability leads to better shelf life and fewer callbacks from customers. Many global coating firms lean into this acid’s properties for clear coatings in electronics and auto parts. Every batch that resists yellowing or checks sets a higher standard for quality and safety.
Crafting Pharmaceuticals and Fine Chemicals
At the bench scale, chemists trust this molecule during the synthesis of active ingredients, specialty intermediates, or chiral auxiliaries. The thiol group in 2-mercaptopropionic acid helps introduce or modify specific chemical structures, steering reactions toward certain configurations. I’ve seen R&D teams use it to nudge a reaction toward a target enantiomer or block unwanted byproducts. In the pharmaceutical sector, that kind of control makes a difference between getting a few grams of pure compound versus weeks of troubleshooting and purification headaches.
Some medical companies also value it for its compatibility during the development of treatments that target certain enzymes, thanks to its role as a biochemical precursor.
Solutions for Wastewater and Heavy Metal Removal
Looking at environmental needs, 2-mercaptopropionic acid helps clean up contaminated water. Its chemical structure grabs metals like mercury, lead, and cadmium, forming stable complexes that can be filtered from wastewater. This application gets a lot of attention for industrial wastewater treatment in mining and electronics manufacturing, where tight rules on discharge demand cost-effective, proven treatments. I’ve worked with plant managers who use this approach to hit regulatory targets while keeping operating costs in check.
You see less disruption to local waterways, and the potential for recovered metals to enter recycling streams gives this application a clear environmental and economic upside.
Improving Production Safety and Sustainability
Industrial teams concerned with safety and sustainability benefit from the reliability and versatility of 2-mercaptopropionic acid. Its low volatility and manageable toxicity profile — when handled with proper training and protective equipment — skirt many of the headaches that come with more hazardous or volatile additives. Facilities that prioritize worker safety find it easier to train crews and avoid downtime for compliance reviews. Lower toxicity, well-documented handling protocols, and consistent quality from vetted suppliers are reasons it keeps a place in so many formulations across sectors.
| Names | |
| Preferred IUPAC name | 2-sulfanylpropanoic acid |
| Other names |
Thiopropionic acid α-Mercaptopropionic acid 2-MPA Propionic acid, 2-mercapto- 2-Sulfanylpropanoic acid |
| Pronunciation | /tuː mɜːrˌkæptəʊ proʊˈpɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 79-42-5 |
| Beilstein Reference | 3538735 |
| ChEBI | CHEBI:35987 |
| ChEMBL | CHEMBL14183 |
| ChemSpider | 6793 |
| DrugBank | DB04183 |
| ECHA InfoCard | 100.007.858 |
| EC Number | EC 244-627-9 |
| Gmelin Reference | 8220 |
| KEGG | C00441 |
| MeSH | D008770 |
| PubChem CID | 7821 |
| RTECS number | TZ7350000 |
| UNII | 1F8W7J16QE |
| UN number | UN2967 |
| CompTox Dashboard (EPA) | JT500S94SR |
| Properties | |
| Chemical formula | C3H6O2S |
| Molar mass | 106.14 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | unpleasant, strong |
| Density | 1.21 g/mL |
| Solubility in water | Miscible |
| log P | -0.54 |
| Vapor pressure | 0.07 mmHg (25°C) |
| Acidity (pKa) | 3.6 |
| Basicity (pKb) | 10.20 |
| Magnetic susceptibility (χ) | -48.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.507 |
| Viscosity | 1.672 mPa·s (25 °C) |
| Dipole moment | 2.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -467.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -669.5 kJ/mol |
| Pharmacology | |
| ATC code | D10AX01 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H301, H311, H314, H331 |
| Precautionary statements | P210, P261, P264, P273, P280, P302+P352, P305+P351+P338, P312, P321, P330, P362+P364, P370+P378, P501 |
| NFPA 704 (fire diamond) | 2 2 2 |
| Flash point | 105 °C |
| Autoignition temperature | 185°C |
| Lethal dose or concentration | LD50 Oral Rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 206 mg/kg |
| NIOSH | MAK67980 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Mercaptopropionic Acid: Not established |
| REL (Recommended) | 0.1-1.0 |
| IDLH (Immediate danger) | IDLH: 250 ppm |
| Related compounds | |
| Related compounds |
Thioglycolic acid Dithiothreitol 3-Mercaptopropionic acid Cysteine Mercaptoacetic acid Glutathione |

