2-Hydroxypropionitrile: A Closer Look at a Classic Chemical
Historical Development
Chemistry rarely stands still. Every molecule tells its own story, and 2-hydroxypropionitrile’s tale stretches back to the formative years of organic synthesis. The early studies tracked this compound alongside the emergence of nitriles in the nineteenth century, with German researchers cataloging it as part of broader investigations into cyanohydrins. Through decades, scientists worked out not just its structure, but how to produce it more reliably and keep it stable on the shelf. It’s clear from reading those old texts that the drive for innovation came less from theoretical pursuit and more from real industrial needs: improved dyes, safer solvents, and better chemical intermediates for making pharmaceuticals and plastics. Chemists in Europe and North America both contributed insights that shaped how industries use and think about this compound today.
Product Overview
Walking through a modern chemical plant, you can see where 2-hydroxypropionitrile fits in. It’s not just a lab curiosity; companies rely on it as a practical building block, especially for synthesizing other organic molecules. Its availability in industrial quantities brings an efficient route to lactic acid derivatives, even as greener tools nudge demand for safer alternatives. This molecule binds simple chemistry to complex real-world applications, and that's where I see its true value.
Physical & Chemical Properties
2-Hydroxypropionitrile stands out because of its mix of polar functional groups: the nitrile (-CN) and the hydroxyl (-OH), both connected to a propane backbone. The presence of both a strongly electronegative cyano group and a tangibly hydrophilic alcohol group lets it dissolve well in water, alcohols, and many organic solvents. Temperature affects its stability, so anytime I’ve worked with it, I’ve kept it under controlled conditions, out of strong acids and bases. Its clear, colorless liquid form keeps things practical during handling; volatility remains moderate rather than high. These core features make it a popular choice for chemical transformation, since it offers points of reactivity while lining up with common procedures in synthetic chemistry.
Technical Specifications & Labeling
Most suppliers specify purity by percentage, with trace water content and residual starting materials flagged for industrial quality control. Standard labeling follows familiar format: chemical names, hazard pictograms, and UN numbers, in line with international standards like GHS. The details help ensure safe transport and proper identification, making accidents less likely and supporting compliance with transport and storage regulations. My experience tells me that attention to these fine details, mundane as they seem, save big headaches; a wrong label in a busy storage room creates liabilities no one needs.
Preparation Method
Traditionally, making 2-hydroxypropionitrile involves nucleophilic addition of hydrogen cyanide to acetaldehyde (the cyanohydrin reaction). Despite the hazards of working with HCN, this reaction offers reliable yields and a clear method for scale-up. Over the past several decades, engineering advances introduced better ways to handle and neutralize HCN, while some coatings and catalysis research point to alternative routes—such as photochemical methods or less toxic cyanide sources. In practice, most facilities stick with the tried-and-true cyanohydrin route, since it balances cost with consistency. Working on-site, I’ve seen careful air monitoring, continuous-flow reactors, and scrubbing systems sidestep much of the traditional risk. No preparation is risk-free, but smart investments in process safety make large-batch synthesis a reality for many companies.
Chemical Reactions & Modifications
2-Hydroxypropionitrile doesn’t just sit on the shelf; chemists value its dual functional groups for building more complex molecules. The nitrile converts to carboxylic acids, amides, or primary amines with added catalysts or strong reagents. The alcohol group, on the other hand, reacts with acids, anhydrides, or alkylating agents. Pathways that combine reduction, hydrolysis, or substitution open up diverse application potential. In pharmaceutical work, I’ve found this flexibility helps in making precursors for amino acids and alpha-hydroxycarboxylic acids. Modern research expands into asymmetric synthesis: enantioselective catalysts aim to create optically pure intermediates. While not every reaction runs straightforward, the capacity to modify both parts of the molecule lets researchers tailor their routes, cutting waste and improving overall efficiency.
Synonyms & Product Names
Names carry baggage, especially in chemistry. 2-Hydroxypropionitrile goes by several synonyms: lactic acid nitrile, acetaldehyde cyanohydrin, and 2-cyanopropan-1-ol, among others. Industry tends to settle on whichever name lines up with major standards like IUPAC or common English usage. Picking up a bottle with a less-familiar synonym always makes me double-check since confusing one cyanohydrin for another can send a synthesis off the rails. Clear labeling and supplier catalogs help, but the maze of trade names and historic nicknames sometimes persists. It pays to keep an eye on synonyms and catalog numbers before using or ordering the material.
Safety & Operational Standards
Anyone who works with cyanohydrins knows the risk: cyanide toxicity and chemical burns top the list. Ventilation is a must, with air monitoring and personal protective equipment worn at all times. I’ve seen improper handling lead to hospitalized colleagues, which is why training and clear signage matter, even for seasoned staff. Emergency eye wash, spill kits, and fume hoods all help cut down exposure risk. Modern guidelines align with OSHA, REACH, and similar regulatory frameworks. Storage in tightly sealed, marked containers away from acids, heat, and incompatible substances keeps both people and the facility safer. Regular audits and drills reinforce good habits, because complacency leads to mistakes in chemical work faster than most realize.
Application Area
2-Hydroxypropionitrile finds itself woven through the tapestry of modern industry. The biggest share involves making lactic acid derivatives and specialty chemicals for pharmaceuticals, cosmetics, and polymers. It often serves as a starting material in preparing ingredients for synthetic flavors, fragrances, and some forms of biodegradable plastics. Some research efforts use it to produce nonracemic amino acids, important for drug development and protein engineering. Downstream, it finds roles as an intermediate for agrochemicals, stabilizers, and advanced coatings. The marketplace for cyanohydrins doesn’t get much public attention, but their quiet presence supports a surprising number of everyday products.
Research & Development
Research on 2-hydroxypropionitrile targets both classic and emerging needs. Much of it pursues more sustainable or less toxic synthesis routes, with project teams striving to sidestep traditional risks of hydrogen cyanide. Catalytic processes, including metal-free or enzyme-catalyzed reactions, get a lot of attention in the literature. Chiral chemistry scholars explore ways to create asymmetric forms, hoping for future pharmaceutical breakthroughs. Analytical chemists keep refining detection and quantification tools, which helps keep regulatory compliance straightforward and improves quality control. For industry, the drive continues toward continuous-flow setups and greener chemistry standards. In academic labs, I’ve noticed cross-disciplinary projects, where toxicology meets synthesis, helping students appreciate the full picture of what it means to work with—and improve—this molecule.
Toxicity Research
Toxicologists have studied 2-hydroxypropionitrile thoroughly, focusing especially on the release of hydrogen cyanide during metabolism or mishandling. Animal studies and in vitro tests document acute toxicity, prompting tight workplace exposure limits and standard operating procedures built around airborne monitoring. The seriousness of cyanide exposure keeps workplace safety officers vigilant, requiring rapid decontamination plans and robust medical response protocols. Research also tracks environmental persistence, so wastewater treatment and containment strategies form part of lab and plant design. Studies into chronic exposure remain ongoing, especially with shifting regulatory priorities worldwide. Knowing the risks keeps everyone sharp—safe handling comes from knowledge, not just rules.
Future Prospects
Standing at the intersection of old-school chemistry and new-era priorities, 2-hydroxypropionitrile sits poised for both traditional and innovative uses. As sustainability pressures rise, companies look for cleaner prep routes, less hazardous byproducts, and lower energy requirements. Advances in continuous processing and biocatalysis hint at ways to keep this molecule relevant, while fresh research in asymmetric synthesis opens doors to more valuable derivatives. Regulatory scrutiny shows no sign of letting up, so future-proofing processes means engineers and chemists need to keep adapting—balancing efficiency, safety, and environmental impact with every step. As the chemical sector retools for a world with tighter margins and more transparency, 2-hydroxypropionitrile reminds us how a single compound can bridge the gap between established practice and better, safer innovation.
What is 2-Hydroxypropionitrile used for?
What’s the Role of 2-Hydroxypropionitrile?
2-Hydroxypropionitrile, better known as lactic acid nitrile, turns up in a lot of factories, but most people never hear about it outside a chemistry class. This colorless liquid doesn’t look like much, yet it opens the door to creating all sorts of useful products. It steps into the spotlight mainly during the production of pharmaceuticals and specialty chemicals. If you’ve handled medications or ever wondered how drug factories create such precise active compounds, 2-hydroxypropionitrile quietly gets things started behind the scenes.
Giving Birth to Important Molecules
In the drug industry, the path from raw material to finished medicine often winds through stages where intermediate chemicals lay a solid foundation. 2-Hydroxypropionitrile acts as one of these building blocks. Chemists use it to form alpha-hydroxy acids and other vital intermediates. Some types of antibiotics and heart medications start their lives with a chemical cousin of this useful nitrile.
This molecule’s simple structure lets chemists modify it. Reactions with water, acids, or bases open up possibilities, so companies lean on it for making a family of compounds, including lactic acid and its derivatives. Many materials in synthetic chemistry rely on such versatile starting points—the easier it is to handle and transform, the more labs put it to work.
Contributing to Everyday Items
While medicine grabs the headlines, the story of 2-hydroxypropionitrile doesn’t end in hospitals and clinics. Factories producing pesticides and herbicides also make use of this compound. For example, its derivatives can help create formulations that increase crop yield or cut down fungal infections that threaten fields.
If you’ve ever used a cleaner with lactic acid, you’ve already benefitted from the chemical chain that 2-hydroxypropionitrile starts. On top of that, it plays a role in creating certain perfumes and flavors because it helps shape chemical groups that carry aromas and tastes.
Looking at the Safety and Environmental Side
Industrial chemicals never come risk-free. 2-Hydroxypropionitrile brings real hazards to both workers and the environment. It acts as a toxic substance that can harm health through skin absorption or inhalation. Its close relationship to cyanide chemistry raises the stakes—one careless step can have consequences.
This reality demands real caution, both from manufacturers and regulators. The U.S. Environmental Protection Agency (EPA) and the European Chemicals Agency monitor how companies handle and dispose of it. Simple negligence can lead to toxic releases, so training and facility upgrades make a difference. From my own experience around industrial labs, strong ventilation and proper handling procedures are non-negotiable; ignoring these steps only invites disaster.
Smarter Solutions for a Safer Future
Tighter rules and investments in engineering controls do pay off. Automated systems take people out of the most dangerous steps. More labs invest in closed-transfer technology, which helps keep vapors out of the air and off people’s skin. Better risk assessments and worker training lower the odds of exposure.
Alternative green chemistry methods keep growing. Some companies work on finding renewable starting materials or swapping toxic chemicals for safer options without losing performance. Cutting back on hazardous nitriles could make entire industries greener and shield future workers from the risks my generation still remembers. The next breakthrough will come from this drive toward cleaner, safer, and more responsible chemistry.
What are the safety precautions when handling 2-Hydroxypropionitrile?
Understanding the Chemical
2-Hydroxypropionitrile isn’t a name most people throw around their kitchen. Folks in chemistry and industry handle it for making things like pharmaceuticals, plastics, and specialty solvents. But it comes with risks you can’t ignore. Its vapors can fire up pretty easily, and it doesn’t mix well with skin, eyes, or lungs. I remember the heavy sweet smell from a lab years ago, which stayed with me for weeks. That was enough to teach me nobody should be casual around it.
Personal Protection: Not Just a Checklist
Before touching any bottle or container, the right personal protective equipment means the difference between a safe shift and a serious accident. Nitrile gloves, chemical splash goggles, and a sturdy lab coat do more than tick boxes for workplace safety—they keep you from painful chemical burns or long-term health issues. Once, a friend ignored his gloves just to make a quick adjustment; his hand paid for that shortcut with a nasty rash. The solvent slips right through ordinary latex, so cutting corners isn’t an option.
Air Safety, Not an Afterthought
Good ventilation doesn’t just make the room smell better. Proper fume hoods and exhaust systems pull those dangerous vapors away from your face and lungs. Some places install sensors for cyanide-based compounds because inhaling even a small dose is a genuine medical emergency. One lab tech I knew used a shed with no fan—the headaches and queasiness hit him harder than he ever expected.
Safe Storage Keeps Trouble at Bay
This chemical doesn’t play well with heat, flames, or sunlight. Locked, labeled cabinets in cool, well-aired storage make a huge difference. The memory of a cracked, sunbaked bottle nearly leaking on the shelf still makes me uneasy. Store it far from acids and oxidizers because accidental mixing can set off violent reactions. Manufacturers’ instructions usually warn against stacking the bottles or storing them near food and drinks. Common sense says: don’t let drinks or snacks close by, because even trace contamination can leave someone dangerously sick.
Spill Response: Fast and Decisive
Spills call for immediate action. Don’t grab paper towels or rags. Staff with proper training use absorbent materials rated for hazardous chemicals, plus they put on more layers of PPE in case the liquid splashes. After absorbing, the waste goes straight into a sealed, labeled hazardous container for disposal by a licensed professional. The worry isn’t only the chemical—it’s also the cleanup, since improper handling can make fumes stronger or catch fire. Emergency showers and eyewash stations have to sit nearby. I tested one last year in a drill, and the old line gushed rusty water—so regular safety checks are just as important as the chemicals themselves.
Confident Handling Comes From Real Training
Reading the safety data sheet gives you the facts, but hands-on training lets habits set in for the long haul. Supervisors need to test knowledge—not just hand out binders—before anyone handles the stuff. I gained confidence only after running through emergency drills and getting quizzed on the symptoms of exposure. Eyes watering, breathing trouble, skin turning red—everyone should know the first signs and how to respond fast.
Building a Culture, Not Just Taking Steps
Every safe procedure, from labeling containers to posting emergency numbers on the wall, comes from people caring about their coworkers. Stories of accidents don’t just fill the rulebooks—they remind us all to pay attention and watch out for each other. The right approach saves more than just property. It protects lives, eases everyone’s worries, and lets real work get done without fear or regret.
What is the chemical formula and structure of 2-Hydroxypropionitrile?
Chemical Formula of 2-Hydroxypropionitrile
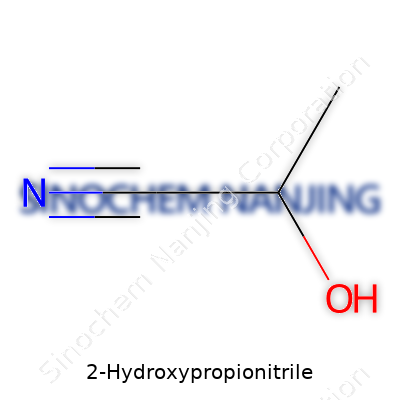
2-Hydroxypropionitrile, scientifically known as lactic nitrile, holds the chemical formula C3H5NO. This compound brings together three carbon atoms, five hydrogen atoms, one nitrogen atom, and one oxygen atom. The molecular weight clocks in at about 71.08 g/mol. The structure shows both a nitrile group and a hydroxyl group attached to a central carbon backbone—giving the molecule unique properties valuable in organic chemistry and industry.
Breaking Down the Structure
The backbone of 2-hydroxypropionitrile starts off with a basic propane chain. The first carbon bonds to the nitrile group (–C≡N), and the second carbon connects with a hydroxyl group (–OH). Chemists write its structural formula as CH3–CH(OH)–CN, highlighting the methyl group at one end and the nitrile at the other. The center, with its attached –OH group, isn't just for show—it plays a real role in how this compound reacts and what it gets used for.
Why This Structure Matters
Getting hands-on in the lab, I saw firsthand how the hydroxyl group changes the playground. The –OH group increases solubility in water, while the nitrile group can act as a building block for a stack of other organic compounds. This special blend isn't just a curiosity—it's practical for chemists making pharmaceuticals or specialty chemicals. Reactions involving 2-hydroxypropionitrile often tap into the electron-withdrawing nature of the nitrile, alongside the nucleophilic character the hydroxyl group brings to the table.
The dual functional groups offer a stepping stone for creating compounds like amino acids, amides, and other derivatives. In the world of research, that means fewer steps and greater flexibility. In practice, this translates into cost savings, improved yields, and streamlined syntheses. This is why its structure regularly shows up in chemical pathways that target higher-value, more complex molecules.
Safety and Handling
From experience, this compound doesn't play around—2-hydroxypropionitrile is toxic if inhaled or swallowed, mainly due to the nitrile group. Working in a well-ventilated lab, using gloves and goggles, stays non-negotiable. For anyone troubleshooting spills or accidental exposures, the standard cyanide protocols come into play. The best approach? Prevent exposure before anything goes wrong—education and equipment keep folks from paying a higher price.
Looking at Solutions and Improvements
One area where 2-hydroxypropionitrile could step up involves greener chemistry. Traditional production routes often require hazardous reagents, leading to waste and tough cleanup. Shifting towards methods using biocatalysts, safer solvents, or less toxic starting materials carries the potential to reduce risks and environmental impact. Green chemistry advances have made some headway, but industrywide changes call for collaboration among researchers, manufacturers, and regulators.
Folks in education can do their part, too. Driving home the facts around safe handling, clear storage practices, and emergency procedures helps reduce accidents. Developing new ways to synthesize 2-hydroxypropionitrile more safely and efficiently will keep this useful molecule relevant for years to come, especially as demand shifts with emerging technologies.
Real-World Impact
Understanding the formula and structure of 2-hydroxypropionitrile goes beyond books and classrooms. The molecule earns its keep in labs, manufacturing, and research settings thanks to its dual reactive sites. With a solid grounding in chemical safety and a push for sustainable methods, chemists put 2-hydroxypropionitrile to work across a surprising number of fields, from pharmaceuticals to specialty materials. Knowledge—backed by experience—makes a difference each step of the way.
How should 2-Hydroxypropionitrile be stored?
Honest Talk About a Hazardous Chemical
Anyone who spends real working days in labs or handles industrial chemicals knows extra care isn’t one of those “optional” steps. 2-Hydroxypropionitrile, also known as glycolonitrile, often turns up in synthesis lines. Its risks call for clear thinking and respect for basic safety. Mistakes here don’t ask politely. They bite.
What Makes This Stuff Tricky?
Let’s not kid ourselves: 2-Hydroxypropionitrile’s dangers stack up fast. This nitrile can release hydrogen cyanide if it breaks down or reacts with acids. Even small doses cause trouble. Its fumes will burn your nose and throat, but that’s the light side. Without enough ventilation or proper storage, one slip turns from a sore throat to a major emergency.
Most staff in properly run labs or plants stick to relevant guidance because they—and the people around them—want to get home safe. I’ve seen carelessness lead to avoidable hospital visits. Turning a blind eye? Not worth it.
Straightforward Storage Rules
Store 2-Hydroxypropionitrile inside a tightly sealed container, ideally made from glass or high-density polyethylene. Metal sometimes reacts. Place these containers in a well-ventilated location, away from places where heat or direct sunlight can sneak in and boost the risk of decomposition. Warm storage rooms bring trouble—temperatures need to stay under 25°C for best results.
Keep this chemical far away from acids—mixing means cyanide risk. I always recommend clear labeling, not just for legal compliance, but because no one needs to “guess” what they’re reaching for. Segregate it from incompatible materials. In my early days, a careless mix-up between storage shelves led to a scare that shut down half a facility. It left a lasting reminder: Don’t let shortcuts run the show.
Ventilation and Containment
Good storage setups rely on solid ventilation. Fume hoods matter, and I’ve seen how simple air exchange systems can save the day when a cap gets left loose. That’s backup you want, not just a line in a manual. Spill trays under containers help catch drips before they land on floors or mix with other chemicals during the wild rush of a busy shift.
Access Controls and Staff Training
Limiting who can access this material protects everyone. Only trained, authorized workers should get near it—no exceptions. Supervisors need to walk through storage rooms regularly. Stale air, strange odors, compromised packaging—small signs hint at big trouble. Document checks matter just as much as taking inventory. I remember a time when oversight flagged a slowly leaking bottle before chaos set in. Trust in double-checks, not blind faith.
Emergency Plans—You Want More Than a Poster
No one expects a spill, but anyone working with 2-Hydroxypropionitrile needs spill kits right next to where it’s stored. Quick access to eye washers and showers can mean the difference between a scare and a tragedy. Phone numbers for emergency teams must be posted on doors, not hidden on an office spreadsheet.
Wrapping It Up for Real World Use
Forget any idea of overdoing it. Taking these guidelines seriously means real people walk out of buildings healthy at the end of their shifts. Risk isn’t just an abstract concept. It’s what hangs around when easy steps go ignored. Safe storage of 2-Hydroxypropionitrile marks the line between business as usual and headlines no one wants to read. Safety isn’t a slogan—it’s the only way that works long term.
What are the potential health hazards of 2-Hydroxypropionitrile exposure?
Facing the Chemical Reality in Everyday Life
2-Hydroxypropionitrile, more commonly known as lactic acid nitrile, turns up in some surprising places. It helps make chemicals that show up in pharmaceuticals, pest control products, and even solvents used in industry. Without paying close attention, this clear, colorless liquid can slip quietly into workplaces or, sometimes, leak out into the broader environment.
What Happens After Exposure
Small doses don’t always ring alarm bells right away, but the body reacts all the same. The main worry kicks in with inhalation or skin contact. This stuff can irritate the eyes, nose, and throat fast, leading to headaches, dizziness, and confusion. People who work with it talk about burning sensations, nausea, and shortness of breath within minutes of poor handling.
One fact that stands out: 2-Hydroxypropionitrile converts quickly inside the body to cyanide. That’s a serious deal when talking about any chemical hazard, as cyanide poisons the machinery that lets cells use oxygen. The American Conference of Governmental Industrial Hygienists (ACGIH) and the Occupational Safety and Health Administration (OSHA) tag this compound as acutely toxic for a reason. People exposed in higher concentrations could experience seizures, loss of consciousness, or even sudden death from respiratory collapse.
Beyond Acute Threats—Thinking Long Term
Most headlines focus on immediate poisoning, but long-term health can hit the skids too. Regular, low-level exposures may set off chronic headaches, memory problems, and nerve pain. Stories from chemical plant workers describe aching joints and a kind of mental fog that doesn’t clear up once they clock out. Nobody wants to guess at permanent nerve or brain injuries, yet the science warns about real danger over time.
Beyond the obvious, there’s the possibility of genetic damage. Some studies with rodents suggest this chemical messes with DNA and cell division. There’s growing research, much of it from academic labs and government tox databases, suggesting repeated exposure around this compound links to a higher risk of miscarriages and birth defects. Regulatory bodies haven't put it on their lists of known carcinogens, but the evidence keeps stacking up for toxic potential elsewhere.
Real Solutions: Making Safety Non-Negotiable
Solutions boil down to respect—for both the science and the workers handling the chemical. The right gloves, goggles, and respirators go a long way, but proper training and daily routines matter even more. Managers who enforce good safety habits make an enormous difference. It shouldn’t depend on the mood of the boss or the size of the company.
Ventilation upgrades and closed systems for transfer and storage can stop leaks before problems start. Routine air monitoring and medical check-ups help spot trouble early. Reports of fish deaths or crop loss near factories show that spills hurt more than just those holding the containers. Good waste management prevents 2-Hydroxypropionitrile from sneaking into water sources, turning a workplace problem into a public health story.
Workers need ways to report symptoms and concerns, without fearing for their jobs. Whistleblower protections and strong unions often provide backup where regulation stumbles. Community pressure forces better practices, but it takes real follow-through on inspections and fines for sloppy handling.
Taking Action Matters
Plenty of chemicals in daily life call for this level of attention, but 2-Hydroxypropionitrile takes its place high on the list. The best way forward comes from learning what’s at stake and acting on it—constantly, not just after emergencies. I’ve seen communities change their tune after accidents wake everyone up. Safe practices and honest talk about risks win every time.
| Names | |
| Preferred IUPAC name | 2-hydroxypropanenitrile |
| Other names |
Lactonitrile α-Hydroxypropionitrile 2-Cyanoethanol |
| Pronunciation | /tuː haɪˌdrɒksi proʊˌpaɪəˈnʌɪtraɪl/ |
| Identifiers | |
| CAS Number | “78-92-2” |
| Beilstein Reference | 703660 |
| ChEBI | CHEBI:43077 |
| ChEMBL | CHEMBL168967 |
| ChemSpider | 10112 |
| DrugBank | DB11360 |
| ECHA InfoCard | 03b8ab9b-3eb2-4e33-867c-df3a8da18a12 |
| EC Number | 200-871-4 |
| Gmelin Reference | 7027 |
| KEGG | C00587 |
| MeSH | D016805 |
| PubChem CID | 7856 |
| RTECS number | SZ7875000 |
| UNII | 4JO66K408N |
| UN number | UN2627 |
| Properties | |
| Chemical formula | C3H5NO |
| Molar mass | 71.08 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Faint, bitter almond odor |
| Density | 1.023 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | -0.3 |
| Vapor pressure | 0.42 mmHg (at 25 °C) |
| Acidity (pKa) | pKa = 12.05 |
| Basicity (pKb) | 3.86 |
| Magnetic susceptibility (χ) | -45.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.401 |
| Viscosity | 1.023 cP (20°C) |
| Dipole moment | 3.5745 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 106.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -151.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -449.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H311, H331, H370 |
| Precautionary statements | P210, P261, P273, P280, P301+P310, P304+P340, P305+P351+P338, P308+P311, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-2-W |
| Flash point | 79 °C |
| Autoignition temperature | 644°F (340°C) |
| Explosive limits | Explosive limits: 5.4–23% |
| Lethal dose or concentration | LD50 oral rat 142 mg/kg |
| LD50 (median dose) | LD50 (median dose): 90 mg/kg (oral, rat) |
| NIOSH | NT8050000 |
| PEL (Permissible) | PEL: 3 ppm |
| REL (Recommended) | 290 K |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Acetone cyanohydrin Benzoin Mandelonitrile Glycolonitrile |

