2-Fluoroaniline: Looking Beyond the Molecule
Peeking Into Its Roots: Historical Development
Stepping into the story of 2-fluoroaniline takes us back to the broader search for aromatic amines that began in the nineteenth century, driven by the dye industry and the emergence of synthetic pharmaceuticals. Chemists understood early on that swapping a hydrogen atom on aniline’s ring for a halogen unlocked new chemical personalities. By the mid-twentieth century, as methods for controlled halogenation sharpened, making fluoroanilines with greater specificity became routine practice in organic labs. Over time, fluoroanilines, and especially 2-fluoroaniline, moved from curiosities to critical intermediates for modern industries. The recruiting power of that tiny fluorine atom drew in both academic researchers and chemical manufacturers, who saw in the molecule’s structure a shortcut to a slew of valuable end products.
Product Overview
At first glance, 2-fluoroaniline seems almost unremarkable: one part benzene, a dash of an amino group, a single fluorine set snugly next to it. But as with many so-called simple organic chemicals, those little tweaks mean the difference between something benign and something transformative for the end user. This compound finds itself poured into glassware as a staple intermediate, especially across pharmaceuticals and agrochemicals, where tweaking the structure sets off a cascade of changes in activity, stability, and even how a drug or pesticide reaches its target. The market’s appetite for derivatives of 2-fluoroaniline keeps growing, fueled by fresh patents and deeper investigation into new materials, all hunting for a competitive edge.
Physical & Chemical Properties
2-fluoroaniline shows up as a colorless to pale yellow liquid, managing a slight aromatic odor with a faint whiff of sharpness that reminds one of the fluoro compounds encountered in teaching labs. The compound’s melting and boiling behavior—melting just below room temperature and boiling well above 160°C—make it fairly easy to handle in the bench-top setting. Its moderate polarity, rooted in both the amino group and the electronegative fluorine, keeps it soluble in most common organic solvents and lends the compound just enough volatility to require careful storage. On a molecular level, that ortho-fluorine tilts the scales of reactivity, tightening the ring’s electron cloud and nudging the amino group into new roles during reactions.
Technical Specifications & Labeling
Purity always makes or breaks the application of a specialty chemical. Most labs demand 2-fluoroaniline upwards of 98% purity, especially since even minor impurities can introduce background noise in reactions or biological screens. In industrial shipments, attention swings to proper labeling, hazard warnings, and packaging resilience. It wears labels warning users about potential skin, eye, and respiratory irritation. Beyond that, a batch usually carries analytical tags—GC or NMR data—helping users track down even trace contaminants before larger-scale syntheses get underway. For anyone who has opened a suspicious reagent bottle, those little data sheets with clean numbers are worth their weight.
Preparation Method
Making 2-fluoroaniline on purpose means threading the needle between various synthetic strategies. One pathway starts from nitrobenzene, introducing the fluorine through electrophilic aromatic substitution, followed by reducing the nitro group back to the amine. Others prefer direct amination using 2-fluorochlorobenzene and aqueous ammonia, riding the power of nucleophilic aromatic substitution under high pressure. The chosen route depends on scale, access to reagents, and the value of by-products. Every chemist who has weighed up batches for synthesis knows that subtle tweaked conditions—temperature, catalysts, clean glassware—make the difference between respectable yield and troublesome muck.
Chemical Reactions & Modifications
With its two-point substitution, 2-fluoroaniline sits as a versatile parent for a whole host of further transformations. The amino group opens up acylation, alkylation, or diazotization routes, which can send the molecule down many synthetic branches. The fluorine on the ring digs its heels in, resisting some reactions yet enabling coupling by ortho-activation. For researchers, that stubborn fluorine both frustrates certain functionalizations and hands over a tool for medicinal chemistry—fluorinated aryl rings stretch drug half-lives and resist oxidative attack in the body. In industrial practice, these transformations form launchpads for everything from dye stuffs to specialty pharmaceutical scaffolds and agrochemical cores.
Synonyms & Product Names
Try searching chemical catalogs, and 2-fluoroaniline turns up under names like o-fluoroaniline or 2-aminofluorobenzene, all nodding to the same molecular skeleton. Early literature sometimes called it ortho-fluoroaniline, especially before IUPAC harmonized naming conventions. Its registry numbers hold constant across borders, helping buyers and regulators match chemical supply chains with proper paperwork. Old habits die hard, and many experienced chemists keep these synonyms in mind, knowing that a mislabeled bottle on the shelf could cause more headaches than a tricky reaction.
Safety & Operational Standards
Fluoroanilines, like most aromatic amines, come with a rulebook for safe use. Skin contact often brings irritation, and longer exposure can tinker with blood chemistry, especially when proper ventilation goes ignored. The standard for chemical hygiene—good gloves, chemical splash goggles, and fume hoods—always applies doubly for amines. Waste streams containing residual 2-fluoroaniline need proper neutralization and separation to keep things out of municipal drains. In my own experience handling similar compounds, a little too much comfort or a missed glove swap makes for a fast reminder about chemical respect. Industrial sites follow these lessons with strict operational standards, enforced by regulatory oversight.
Application Area
2-Fluoroaniline mainly shows up as an intermediate, which is where its utility really shines. Pharmaceutical labs value its unique substitution pattern for building blocks in kinase inhibitors or other targeted therapies. Agrochemical firms look to similar derivatives for crafting new herbicides or pesticides, where fluorine’s presence tweaks activity and environmental persistence. In the world of dyes, certain 2-fluoroaniline derivatives help fine-tune shade and lifespan, bridging performance gaps that unmodified aniline can’t handle. Specialty plastics and polymers also incorporate fluorinated anilines, searching for the sweet spot between flexibility and chemical resistance. Research groups still explore new uses, examining electronic properties for organic semiconductors and chasing the next big leap in material science.
Research & Development
Chemists working on fluorinated aromatics often marvel at how a single atom change redefines so much application potential. Research into 2-fluoroaniline constantly uncovers new synthetic pathways with fewer steps, lower energy demands, and cleaner by-product streams. Advanced analytical tools—NMR, mass spectrometry, crystallography—let scientists see and predict reactivity at atomic resolution, sharpening process safety and yield. Pharmaceutical research remains the biggest driver, testing libraries of fluorinated anilines for novel structural motifs that could unlock next-generation drugs. Collaboration between industry and academia, fostered by grant funding and patent races, ensures a constant churn of ideas tailored to evolving medical, technological, and agricultural challenges.
Toxicity Research
Toxicologists zero in on aromatic amines with extra scrutiny, especially given their history in occupational safety. Animal studies point to the risk of methemoglobinemia, a disruption of oxygen delivery in the body, after persistent or high-level exposure. Regulatory agencies require repeat-dose testing and environmental impact studies before any wide use or disposal pathway gets approval. Environmental scientists want to know how long traces of 2-fluoroaniline linger in soil or water, with fluorine making degradation stubborn. Personal stories from bench chemists—stinging skin or unexpected headaches—drive home reminders that lab coats and working hoods mean more than just ticking safety boxes.
Future Prospects
All signs point to more growth around the edges of fluorinated aromatics. As pharmaceutical design networks hunt for new molecular space, demand for unusual substitution patterns keeps 2-fluoroaniline near the top of the order list. Environmental pressures push chemists to design degradation pathways that leave no trace, pushing both synthetic method development and end-of-life treatment for these molecules. New reactions that use renewable catalysts or greener solvents promise to lower the environmental cost of making specialty chemicals like this. From the perspective of materials science, the quest for more resilient, high-performance polymers and electronic devices will ensure interest in fluorinated building blocks. The familiarity and challenge of this small molecule will keep it firmly in the laboratory toolkit for years to come.
What is 2-Fluoroaniline used for?
Chemistry in Action
Ask any chemist in the pharma space about 2-Fluoroaniline, and you’ll probably get a nod of recognition. This clear, slightly yellow compound pops up in labs wherever researchers and companies look for new medicines or specialty chemicals. If you’ve worked with organic synthesis, you see why: its aniline backbone, tweaked with a fluorine atom, brings some unique reactivity to the table. Changing a single atom sometimes upends an entire chemical’s personality. In 2-Fluoroaniline’s case, that tiny swap opens doors in drug discovery and material science.
Medicine and Molecules
Plenty of pharmaceutical companies turn to 2-Fluoroaniline as they experiment with ways to fight disease. This molecule doesn’t land on pharmacy shelves by itself, but chemists use it as a building block for more complex drugs. The fluorine trick often makes drugs last longer in the body, helps them hit their target, or simply makes them easier to manufacture. For example, some antidepressants, cancer medicines, and antibiotics owe part of their effectiveness to fluorinated pieces like this. The search for safer, more reliable drugs runs right through molecules in this family. Without these synthetic helpers, some medicine cabinets would look emptier.
Dyes, Pigments, and Everyday Color
Anyone who’s tinted fabric or worked in ink production can tell you fluorinated anilines make a difference. 2-Fluoroaniline serves as an intermediate in making dyes and pigments you find in everything from textiles to highlighters. The fluorine in the ring can sharpen color stability and help resist fading under sunlight or washing. I once worked in a lab that tested outdoor signage—dyes based on fluorinated compounds routinely outperformed the rest. No one wants a bright red banner turning pink by summer’s end, so these chemical tweaks make a visible difference in real life.
Crop Protection and Industrial Chemistry
In agriculture, 2-Fluoroaniline takes on another job as a precursor in manufacturing herbicides and pesticides. Farmers and companies look for agents that combat weeds and pests but break down without threatening crops or groundwater. Building new crop protection technologies often starts with finding a base molecule that delivers power but not persistent pollution. The same goes for specialty polymers and adhesives—engineers pull in fluorinated building blocks to add weather resistance or chemical stability. As global demand grows, the push for useful, more environmentally responsive chemistry depends on smart choices in molecular design.
Weighing the Challenges
Of course, success with 2-Fluoroaniline comes with responsibility. Fluorinated organics can resist breaking down in nature, raising questions about long-term safety and waste management. Labs need tools for handling, storage, and disposal that don’t risk workers or the environment. I’ve trained new researchers to respect every step, from measuring to disposal, because mistakes with aromatic amines can hurt more than just a project. Regulatory guidelines require careful tracking and documentation, especially as the industry faces tighter restrictions on hazardous chemicals.
Moving Ahead: Safer and Smarter Chemistry
People in labs and factories already search for less toxic methods to make and use 2-Fluoroaniline. Some teams explore greener solvents, improved containment, and closed-loop recycling. Funding for fluorinated compound alternatives is growing, led by concerns about environmental persistence. Digital modeling and automation also help spot problems earlier, keeping both workers and communities safer. The challenge remains: finding compounds that offer the benefits without unchecked risk. The right mix of innovation, training, and oversight keeps chemistry productive—and protects everyone involved.
What are the safety precautions for handling 2-Fluoroaniline?
Understanding What’s at Stake
2-Fluoroaniline is no everyday household chemical. It’s one of those substances researchers and professionals take seriously for good reason. This compound, used mainly in chemical synthesis and pharmaceutical research, can harm your health even after a short encounter. Over the years, I’ve seen colleagues overlook the importance of a few basic precautions, and the consequences have been anything but minor—skin rashes, headaches, and far worse reactions. Familiarity with the safety sheet is a start, but hands-on habits make the real difference.
Personal Protective Equipment: Not Optional
Nitrile gloves form a solid barrier between your skin and chemicals like 2-Fluoroaniline. Don’t swap them for cheap latex or skip them altogether. A lab coat or chemical-resistant apron helps keep your street clothes clean and your body covered. Eye protection isn’t a minor add-on. Goggles with side shields or a face shield matter, since splashes don’t announce themselves. A fume hood stands between you and harmful vapors. Without it, you risk inhaling something that can cause severe respiratory problems. Inhaling 2-Fluoroaniline vapors can irritate the airways, lead to dizziness, and even mess with your red blood cells—long-term exposure gets even riskier.
Exposure: Acting Fast Matters
Anyone who’s spilled something toxic knows those seconds after a slip-up can feel endless. For skin contact, wash right away with soap and lots of water. Remove any clothes that got contaminated; don’t leave them lying around. Flush eyes with water for a minimum of 15 minutes—heading to the eyewash station right away pays off. Ingesting or inhaling this chemical is serious. Head for fresh air or a medical professional right away. The Material Safety Data Sheet isn’t just paper—keep it within arm’s reach, because emergency responders benefit from having clear info.
Storage and Housekeeping
2-Fluoroaniline thrives on being left alone in a cool, dry, locked cabinet away from sunlight. Flammable cabinets are the right choice, not just any shelf. Never keep this chemical near acids, oxidizers, or bases. I’ve watched too many near-misses from storing incompatible substances together simply out of convenience. The risk isn’t theoretical. A faulty cap or open lid can fill the whole space with toxic fumes. Secure lids tightly after every use and check for leaks or cracks. Clear labeling reduces confusion, so use bold, legible lettering on every vessel. Don’t rely on memory, especially in shared labs.
Waste: No Cutting Corners
Dumping 2-Fluoroaniline in the sink poisons water supplies and endangers lives. Collect all waste in chemically resistant, clearly labeled containers. Coordinate with your institution or local hazardous waste facility for proper disposal. Take these steps seriously—not only because regulators say so, but because protecting others in the workplace matters as much as protecting yourself. Laboratory managers should set the tone by insisting on routine disposal checks and waste log reviews.
Situational Awareness
Fatigue, distraction, or rushing creates more hazards than any chemical alone. Regular safety drills, visible instruction posters, and open discussion about near-misses foster a culture where everyone looks out for each other. Training shouldn’t end at onboarding. Stay updated on new information, and never let routine breed carelessness. A sloppy moment might cause a lifetime of regret. Shared responsibility keeps everyone safer in the long run.
What is the chemical formula and structure of 2-Fluoroaniline?
Diving Into 2-Fluoroaniline
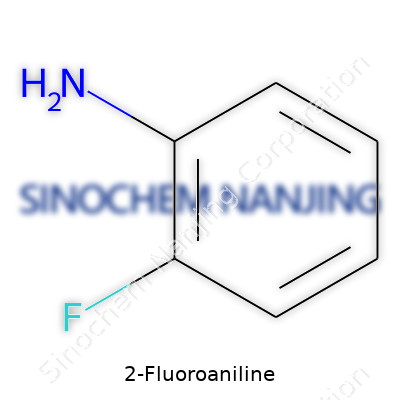
The name 2-fluoroaniline may not come up at the dinner table, but ask anyone who spends time in a chemistry lab about its relevance, and the conversation can spiral deep into aromatic chemistry. Its formula, C6H6FN, hints at its structure—a benzene ring carrying two important substituents, an amino group (-NH2) and a fluorine atom, at neighboring spots. The “2-” sets the fluorine next to the amine. The skeletal structure carries a simple logic: attach fluorine to carbon number two, stick the amino group on carbon number one, let the rest of the molecule finish the circle.
Why Chemists Care About This Structure
There’s a fascinating dance between structure and properties in chemistry. Take 2-fluoroaniline’s arrangement. The fluorine atom brings unique traits—it’s electronegative, it tugs at the electron cloud, and modulates the reactivity of the entire ring. Classic aniline reacts in all sorts of ways, but swap out a hydrogen for a fluorine, and new doors open. Synthesis routes for dyes, pharmaceuticals, and agrochemicals benefit from this kind of molecular tweak. For example, fluoro-substituted aromatics often yield better metabolic stability in drugs. Cost and performance both gain an edge.
Back in my student days, I ran a reaction to synthesize a substituted aniline. The plan called for a clean ortho-fluoro substitution. We learned quickly that the precise position of fluorine compared to the amine group influenced every step in the process. Even the color, solubility, and melting point changed—factoring in how we isolated and used the compound. Other chemists have found this, too. They see how the nitrogen and fluorine, sitting side-by-side, steer reactions differently compared to meta or para configurations.
Attention to Safety and Handling
2-Fluoroaniline holds more than academic curiosity. The amino group, once bonded to benzene, carries toxicity and potential risks for lab workers. Reports indicate possible skin and eye irritation, so gloves and fume hoods always come into play during handling. Chemists stress the need for strong procedures and protective gear—the same principles that underpin safety everywhere, whether working with solvents or sharp tools. Lax handling has led to documented cases of exposure, so organizations continue to adapt safety standards.
Supply and Real-World Uses
Suppliers worldwide offer 2-fluoroaniline in bottles sized for both research and mass production. Buyers look for high purity, fast delivery, and reliable documentation. Sourcing this compound goes beyond a simple internet search; labs vet vendors on quality control and regulatory compliance, as governments scrutinize aromatic amines due to their environmental impact. Disposing of waste requires special treatment, as simple drains don’t cut it in responsible facilities.
Toward Sustainable Practices
Chemists today keep an eye on sustainability. Demand for greener methods encourages the development of selective catalytic processes and alternatives that reduce toxic byproducts. Modern research continues to search for routes that use less energy and yield less waste, making compounds like 2-fluoroaniline more environmentally friendly. Some groups have published promising pathways that skip hazardous reagents altogether, showing that chemistry can innovate responsibly.
Understanding a molecule like 2-fluoroaniline takes more than knowing its shape or formula; it means tracking how it affects real-world products, worker safety, and environmental health. This compound sits at a crossroads of classic organics and modern needs, keeping researchers motivated to understand each atom’s place in the puzzle.
How should 2-Fluoroaniline be stored?
Understanding the Stakes of Storing Chemicals
Anyone who's worked around chemicals long enough knows that safe storage rules can’t be taken lightly, especially with compounds like 2-Fluoroaniline. This colorless to pale yellow liquid isn’t as famous as some industrial solvents, but its hazards are just as real. I've seen small oversights turn into emergencies, so paying attention to details can make a world of difference.
Risks Tied to 2-Fluoroaniline
Exposure to 2-Fluoroaniline brings a mix of concerns. The chemical can harm skin, eyes, and even someone’s respiratory system. Absorption through skin or inhalation can mess with red blood cells, leading to conditions that require serious medical attention. Some experts point to cancer risks with prolonged contact. These facts alone show why proper handling and storage practices really need to stick.
Key Storage Lessons from the Lab
In my lab days, we analyzed storage protocols for every chemical coming through the door. For 2-Fluoroaniline, a few principles always stood out.
- A cool, dry space — usually under 30°C — stopped chemical degradation.
- Sealed containers helped avoid spills and leaks, and made sure no fumes slipped out.
- Glass bottles or specific plastics proved safe, since aggressive chemicals like this one react with the wrong materials.
- Tight labels on every bottle, always with dates and hazard warnings, meant new folks could avoid rookie mistakes.
All these sound simple, but habits slip when workloads climb. I remember once a bottle with an old, peeling label started leaking. Only some quick thinking from a seasoned tech got everyone out before things got worse. That wake-up call turned into plenty of label checks and early reports to supervisors. It’s not paranoia — it’s practice.
Training and Routine: The Real Safety Net
No storage rule replaces a well-trained crew. Routine walk-throughs, supply checks, and emergency drills kept everyone sharp. In one facility I visited, a regular drill caught a ventilation issue nobody had noticed, preventing what could’ve become a major health hazard. Management recognized safe storage as a team effort, and every tech felt responsible for the chemicals under their watch.
Dealing With Spills and Leaks
With 2-Fluoroaniline, you can’t just wipe up a puddle and walk away. Any spill means clearing folks out, donning gloves and goggles, and following the spill procedure to the letter. Proper containers for hazardous waste make a huge difference, and easy access to material safety data sheets helps everyone respond the right way.
Policy Solutions and Forward Planning
Some companies still cut corners, storing sensitive chemicals near incompatible materials, or tucking containers away in crowded closets. Oversight and regular audits stop these habits before they catch up with someone. Digital tracking systems can flag expired or damaged stock, providing another layer of security. For operations working with 2-Fluoroaniline, clear policies and ongoing education should live at the heart of their safety plans.
Lasting Value in Safe Storage
Keeping 2-Fluoroaniline secure means fewer accidents, healthier staff, and a smoother regulatory path. In real workplaces, every safeguard gets tested, so forgetting the basics can carry a high price. Every time I’ve seen storage handled with respect and care, emergencies either shrank or never happened in the first place. It’s about setting a standard that lasts long after the training session is over.
What are the physical and chemical properties of 2-Fluoroaniline?
Getting to Know 2-Fluoroaniline
2-Fluoroaniline rings a bell for anyone in organic chemistry. It’s a colorless to pale yellow liquid, and the sharp, often unpleasant odor tells you quickly this isn’t your garden variety household chemical. 2-Fluoroaniline, as its name says, holds a fluorine atom attached to the benzene ring, right next to an amino group. This small change, that single fluorine atom at the ortho position, sets it apart from its cousins in the aniline family. The combination spells different reactivity, health concerns, and uses.
Physical Properties Worth Knowing
As a liquid at room temperature, 2-Fluoroaniline has a melting point at about 12 °C and boils near 184 °C. That makes sense—fluoro-substitution usually nudges boiling points up compared to the parent aniline. The density sits around 1.15 grams per cubic centimeter, a bit heavier than water. It doesn’t mix well with water, proving only slightly soluble, but it coaxes into most organic solvents without trouble.
That classic aniline stink floats out of the bottle, but with a sharper edge, probably thanks in part to the fluorine atom. Pure 2-fluoroaniline appears clear, but small impurities or traces of oxidation turn it yellow over time. Store it right, away from sunlight, and that color stays clearer much longer. Just from working with related compounds, even small accidental spills can clear a room, so good ventilation is a must.
Chemical Behavior: More Than Just a Ring and a Substituent
Amino groups kick up reactivity, but adding a fluorine atom alters electron flow across the benzene ring. In 2-fluoroaniline, the fluorine leans electron-withdrawing, pulling density away, while the amino group donates. That balancing act changes not just how the molecule reacts but also where other reactions take place on the ring.
Fluorine’s presence strengthens bonds, which makes it a stubborn target during chemical syntheses. Chlorinated or brominated versions swap their atoms more easily in nucleophilic aromatic substitution, but the C–F bond resists—chemists see this as both a blessing and a headache. For pharmaceutical companies and agrochemical designers, that means 2-fluoroaniline serves as a building block for robust, stable molecules. Drug developers have pursued fluoroanilines for precisely this reason: they last.
Health and Safety Aren’t a Sideshow
Anyone handling 2-fluoroaniline needs respect for its toxic potential. The molecule can impact blood and organs, like many aromatic amines. Absorption can come from skin, lungs, or ingestion, and symptoms might show late. Gloves, goggles, lab coats, and chemical fume hoods aren’t optional. Working in university labs, I saw even small spills treated seriously, and for good reason. Students who breathed fumes or got a drop on their skin reported headaches, dizziness, or worse.
Regulatory agencies, including OSHA and the European Chemicals Agency, flag substances like 2-fluoroaniline for strict workplace handling. Proper storage in sealed glass containers, away from oxidizers and sunlight, prevents dangerous reactions and slow degradation. Waste needs precise labeling, and sending it down the drain remains out of the question in any reputable lab.
Industry and Solutions
Chemists continue chasing greener alternatives and safer handling procedures for fluoroanilines. Research grows every year on cleaner synthetic methods, recycling, and real-time monitoring of airborne levels in the workplace. While 2-fluoroaniline brings crucial benefits to pharmaceuticals and material science, it deserves both technical respect and a practical eye. In my own practice, quick cleanup kits and digital safety training closed the gap between theory and reality. Fewer incidents mean smoother research and healthier labs.
Looking forward, building safer derivatives, installing better air scrubbers, and refining chemical education help keep scientists productive and healthy. 2-Fluoroaniline won’t leave chemistry benches soon, but awareness and preparation turn it from a hazard to a tool.
| Names | |
| Preferred IUPAC name | 2-fluoroaniline |
| Other names |
o-Fluoroaniline 2-Aminofluorobenzene 1-Amino-2-fluorobenzene |
| Pronunciation | /tuː fluːˈrɔːrəˌænɪliːn/ |
| Identifiers | |
| CAS Number | 348-54-9 |
| 3D model (JSmol) | `3d: c1=cc=cc(n1)F` |
| Beilstein Reference | 1209272 |
| ChEBI | CHEBI:50179 |
| ChEMBL | CHEMBL16024 |
| ChemSpider | 2037 |
| DrugBank | DB03784 |
| ECHA InfoCard | 100.012.336 |
| Gmelin Reference | 83204 |
| KEGG | C02573 |
| MeSH | D000773 |
| PubChem CID | 70017 |
| RTECS number | BX8575000 |
| UNII | Q4B83U8KH3 |
| UN number | UN1982 |
| Properties | |
| Chemical formula | C6H6FN |
| Molar mass | 111.12 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | ammonia-like |
| Density | 1.16 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.06 |
| Vapor pressure | 0.9 mmHg (25°C) |
| Acidity (pKa) | 3.98 |
| Basicity (pKb) | 9.15 |
| Magnetic susceptibility (χ) | -54.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.567 |
| Viscosity | 1.105 cP (20°C) |
| Dipole moment | 2.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –113.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2345 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause genetic defects; may cause cancer |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H412: Harmful to aquatic life with long lasting effects. |
| Precautionary statements | P261, P273, P280, P302+P352, P305+P351+P338, P310, P501 |
| NFPA 704 (fire diamond) | 2-1-0-Acids |
| Flash point | 71°C |
| Autoignition temperature | 615 °C |
| Explosive limits | Lower: 1.1%, Upper: 7% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 300 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 2-Fluoroaniline is "250 mg/kg (rat, oral) |
| NIOSH | RN2903 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 µg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Aniline 2-Chloroaniline 2-Bromoaniline 2-Iodoaniline 4-Fluoroaniline |

