2-Ethylthiobenzyl N-Methylcarbamate: A Closer Look
Historical Development
Scientists started testing N-methylcarbamate compounds sometime around the mid-twentieth century, driven by demand for new selective pesticides that offered alternatives to older chemicals like DDT and parathion. The advancement towards 2-ethylthiobenzyl N-methylcarbamate drew momentum from the success of carbamate pesticides, including carbaryl and propoxur, which gained trust for their comparatively quick breakdown in soil and less severe environmental accumulation. Chemical engineers tinkered with benzyl rings, sulfur substitution, and carbamate linkages, looking for ways to fine-tune insecticidal action without a sharp spike in mammalian toxicity. Small research teams shared new SAR (structure-activity relationship) insights, occasionally running up against regulatory walls or field setbacks. Not every lab discovery made it through, but compounds with balanced pest lethality and safer degradation paved the way for greater acceptance of modern carbamates like this one.
Product Overview
2-Ethylthiobenzyl N-methylcarbamate shows up in technical circles as a specialized insecticide, with an eye for certain greenhouse and orchard applications. Marketed under various trade names—factory records point to aliases like ETHIOCARB and BENZETHIOCARBAMATE—this compound rose in prominence for targeting sap-sucking insects. Its structure, a fusion of carbamate and thioether moieties, lets it act as a reversible cholinesterase inhibitor, disturbing nervous system signaling in bugs. In day-to-day use, those in the agricultural sector have handled the substance as a light tan powder, designed for mixing into suspensions or dusts. Unlike some broad-spectrum alternatives, this carbamate’s formulation chases precise pest targets such as aphids and leafhoppers, especially in crops including peppers, cucumbers, and berries.
Physical & Chemical Properties
With a molecular formula of C11H15NOS and a molar mass in the ballpark of 209 g/mol, 2-ethylthiobenzyl N-methylcarbamate stays solid at room temperature. It barely dissolves in cold water but finds greater affinity with common organic solvents like acetone and ethanol. The melting point hovers around 60–70°C. Chemically, the molecule’s carbamate group attaches at the benzylic position, buffered by an ethylthio side chain on the aromatic ring, which improves lipophilicity and biological uptake in target pests. Storage tanks keep this compound away from heat and oxidants to prevent gradual degradation or accidental release of noxious gases during mishaps.
Technical Specifications & Labeling
Bulk shipments of 2-ethylthiobenzyl N-methylcarbamate come labeled with hazard warnings and strict shelf-life guidelines. These specifications spell out minimum purity levels of 97–98% and maximum content for moisture, insolubles, and trace residuals (like free methyl isocyanate) below 0.2%. Drum labels also bear UN numbers and pictograms for toxicity and aquatic hazard, giving workers a heads-up on exposure risks. All labeling ties back to chemical inventory codes, which help track lot identity and monitor product drift in large operations. Instructions for dilution, mixing, and disposal stand right alongside English and local regulatory texts, making sure best practice information lands in the hands of supervisors and front-line handlers.
Preparation Method
Industrial chemists synthesize this carbamate through a multi-step process starting with 2-ethylthiobenzyl alcohol. First, the alcohol gets chlorinated to form the matching benzyl chloride. This intermediate then reacts with sodium N-methylcarbamate under controlled alkaline conditions. The substitution reaction relies on vigilant temperature and pH control, with workers using scrubbers or trapping gases to capture stray methyl isocyanate, which can present life-threatening risks. Final purification runs the crude reaction mixture through a series of washes, filtration, and drying steps—sometimes employing charcoal or silica to pick up stubborn contaminants. Technicians track progress by TLC and HPLC, tuning each batch to meet statutory limits for byproducts.
Chemical Reactions & Modifications
Once formed, 2-ethylthiobenzyl N-methylcarbamate can undergo a handful of practical chemical transformations. Exposure to base or acid leads to hydrolysis, splitting off the carbamate group and yielding the parent benzyl alcohol or amine. Chemists have looked at selective oxidation reactions to modify the thioether into sulfoxides or sulfones, occasionally seeking derivatives with new bioactivity profiles. On the analytical side, detection hinges on UV absorption and GC-MS fingerprinting, using characteristic fragmentation of the thio- and carbamate linkages. Modifications often focus on improving selective toxicity or on probing structure–function relationships with pests, giving research labs plenty of room to explore adjacent chemical territory.
Synonyms & Product Names
On the ground, chemical databases and manufacturers recognize this molecule by several synonyms: 2-ethylthio-benzyl N-methylcarbamate, ETHIOCARB, and sometimes as O-ethylthio-phenyl methylcarbamate. Registries like CAS or EC numbers solidify official tracking, but technical staff more often use short-hand product codes or brand names in day-to-day orders and application sheets. International trade can bring up Arabic, Mandarin, or other language variants, though regional safety sheets always list the IUPAC name for clarity during cross-border movement.
Safety & Operational Standards
Workplaces using 2-ethylthiobenzyl N-methylcarbamate must double down on PPE: gloves, goggles, and air-fed respirators take priority since the compound, like most carbamates, poses acute toxicity risks by absorption or inhalation. Spill control means easy access to absorbent pads and containment booms. Training regimes remind workers that, even with rapid environmental breakdown, direct contact or inhalation can inhibit their own cholinesterase, causing symptoms from headache to convulsions. Washing facilities and eye stations stay close at hand in all application zones. Disposal skips landfill; chemical incineration at high temps avoids contamination, required by both local and global regulations. Regular blood cholinesterase tests for workers act as a first-line indicator of overexposure, with retention programs keeping close tabs on cumulative toxic load.
Application Area
Growers turn to 2-ethylthiobenzyl N-methylcarbamate where standard organophosphates fall short or where soft-bodied bugs threaten to wipe out crops. Its selective knock-down lets it fit in with integrated pest management (IPM), sandwiched between less persistent options and more powerful, last-resort chemistries. Landscapers sometimes apply it for ornamental flowers and fruit trees under carefully managed permits. In the field, handlers mix batches just ahead of spraying, keeping a close eye on wind, humidity, and temperature to avoid off-target movement. Some regions restrict use during pollinator bloom or near watercourses, reducing the footprint on bees and aquatic invertebrates. Veterinarians generally skip this compound due to companion animal risks, but wildlife managers have experimented with it for locust or grasshopper suppression in emergency outbreaks.
Research & Development
Research teams in agricultural chemistry still study the breadth of carbamate compounds to outpace evolving pest resistance and regulate off-target impacts. Data from LC50 trials across aphids, thrips, and leafhoppers show wide variability depending on local genetic strains, climate, and stages of crop growth. Some newer work investigates slow-release capsule formulations, hoping to extend protection without raising residues at harvest. Analytical scientists scan water and soil for breakdown products, tracking half-lives and metabolite pathways using radioactive labels. This compound’s profile in rotational systems remains under assessment, as researchers weigh long-term impacts against immediate insect control. Research partnerships with universities and state extension agencies keep refining the technical playbook, mapping out new application patterns or resistance management protocols.
Toxicity Research
Toxicologists run comprehensive trials on acute, chronic, and developmental toxicity. For mammals, particularly lab rats and rabbits, oral LD50 data has carved a risk corridor, with rapid onset of poisoning symptoms confirming the need for tight exposure control. Studies show cholinesterase inhibition as the critical endpoint, tracked at both red blood cell and plasma enzyme levels. Long-term mouse studies probe for carcinogenicity or reproductive effects, but the fast hydrolysis of the carbamate bond usually limits tissue accumulation. Aquatic toxicity sits in the moderate-to-high range for crustaceans and fish, pushing regulatory bodies to regulate buffer zones and restrict direct water contact during spraying. Avian species see mixed sensitivity, with migratory risks tied less to persistent residues and more to accidental spray drift or improper storage. Medical doctors working with poisoning cases recommend atropine and pralidoxime, though early hospital admission gives the best shot at recovery.
Future Prospects
The future of 2-ethylthiobenzyl N-methylcarbamate hinges on the interplay of pest resistance, regulatory tightening, and evolving farm techniques. As policymakers worldwide limit the use of high-toxicity pesticides and ramp up surveillance of agri-chemical residues, this compound could face stiffer permit requirements, pushing demand toward even softer alternatives. At the same time, pockets of resistance to neonicotinoids or pyrethroids sometimes revive interest in rotational use of carbamates, preserving an escape hatch for outbreaks. Research may focus on safer analogs or delivery systems that cut operator and non-target risks, possibly through site-limited application or biopesticide blends. Cultivation trends toward organic or regenerative models will keep shrinking the chemical’s mainstream acreage, but niche use or stockpile deployment in regional crises may keep a specialized market alive. Field feedback loops—data from growers, wildlife monitors, and poison control centers—will drive both label changes and the pace of innovation as the industry responds to new challenges over the next decade.
What is 2-Ethylthiobenzyl N-Methylcarbamate used for?
Pesticides and Chemistry Collide
Walk into any field where farmers grow vegetables, and one thing looks the same around the world: the battle against bugs. 2-Ethylthiobenzyl N-Methylcarbamate falls smack into this struggle. It’s a chemical that kills insects and belongs to a group called carbamates. This class has been around since the 1950s, making headlines for both high-yield promises and environmental worry.
From my own time on a family farm, I learned pretty quickly that infestations wipe out months of work. You try everything—nets, crop rotation, even early morning handpicking—before grabbing a sprayer. Farmers value anything that saves those crops, keeping shelves at markets full. For this, carbamates, including 2-Ethylthiobenzyl N-Methylcarbamate, offer fast relief. They hit pests’ nervous systems, stopping chewing and sucking bugs in their tracks. Studies show these compounds can boost harvests in places dealing with heavy insect pressure. That’s no small thing when every acre counts.
Health Concerns: Not So Simple
Still, there’s always a catch with chemicals you use outdoors. Over years of reading research and watching regulations shift, I noticed one pattern: authorities don’t always agree about safety. 2-Ethylthiobenzyl N-Methylcarbamate, like others in its group, can harm more than insects. If exposure isn’t controlled, this stuff gets into waterways or lingers on food. Toxicologists warn that large doses harm the nervous system in humans just as they strike bugs. Even at low levels, farmworkers and those living near fields express worry, supported by findings of headaches, dizziness, and other symptoms tied to carbamate exposure.
Food safety groups remind us that washing doesn't always remove every trace. Regulatory agencies in various countries set limits for residues, but the line between “safe” and “risky” moves as new data appears. In some places, tighter rules or outright bans have kicked in to push farmers toward less synthetic use.
Alternatives and Moving Forward
Watching trends on farms and in gardens, I see more people turning to newer, “softer” pesticides or using smart technology for pest scouting. Integrated pest management (IPM) is gaining traction. Instead of blanket-spraying chemicals like 2-Ethylthiobenzyl N-Methylcarbamate, growers use weather data, crop rotation, and beneficial insects to cut down on spraying. Research shows this slashes risk for both the crops and the people eating and working around them. While total elimination of older pesticides moves slowly, these hybrid strategies deliver a middle ground.
Access to information matters. Producers, buyers, and eaters all want to avoid nasty surprises. Civic groups often press for transparent labeling and testing, helping buyers make choices that match their own comfort level. On the policy side, scientists and regulators continue to study how far residues travel and what risks they present to kids, wildlife, and people downstream from sprayed fields.
Why Paying Attention Still Matters
People tend to underestimate the balance growers juggle—growing food and keeping health risks in check. 2-Ethylthiobenzyl N-Methylcarbamate illustrates the tricky line between feeding more mouths and protecting the environment for tomorrow. By supporting research, practicing safer application, or choosing food that lines up with your values, each person can nudge the market. New answers won’t show up overnight, but sharing knowledge and pushing for progress keeps the issue front and center.
What safety precautions should be taken when handling 2-Ethylthiobenzyl N-Methylcarbamate?
The Reality of Handling Carbamates in the Workplace
2-Ethylthiobenzyl N-Methylcarbamate doesn’t show up in regular conversation, but for some of us doing field work, plant care, or research, it becomes a reality. This chemical belongs to the carbamate class, which means it can pose real health risks—specifically to skin, eyes, and the nervous system. Anyone who's ever picked up a bottle without reading the label or worked in a lab after hours knows how easily distractions can turn into actual danger.
Understanding the Risks—Because Shortcutting Safety Isn’t Worth It
Gloves matter much more than they look. Many people think touching a dry powder isn’t a big deal, or that a single splash of liquid won’t cause any harm. But chemicals like 2-Ethylthiobenzyl N-Methylcarbamate operate on the small scale—contact with skin doesn’t always burn or tingle right away, but over time these compounds can get absorbed and start causing symptoms no one wants. Headaches, dizziness, even breathing trouble—these are not the kind of workplace stories anyone wants to tell.
Steps for Real Protection—What Actually Works
Gloves and Eye Protection: Start with thick, chemical-resistant gloves. Nitrile types seem to work well against carbamates. Swap them out if they tear, get wet, or after a few hours. Eyes catch more than you think during mixing or spraying, so wraparound goggles work better than the cheap ones you just perch on your nose.
Ventilation Beats Guesswork: Many older labs or sheds still don’t have great airflow. If a fume hood is available, use it. Spraying outside helps dilute vapors, but closed spaces with poor air movement let toxins hang around. Anyone who has walked into an unventilated lab after a spill knows the nose stings and the throat scratches for hours. Regular checks and fans can make a big difference.
Labeling Everything: Black-and-white tape and a marker don’t cost much, and they prevent confusion. It takes under a minute to label a beaker or trigger sprayer. Spending that time means nobody accidentally pours or sprays something they shouldn’t. Experienced workers know that mystery chemicals on a bench mean wasted time and possible injury.
Covering Exposed Skin: Gown up. Aprons and long sleeves shield arms, and closed shoes keep spills away from skin. Folks working outdoors in warmer climates sweat under extra layers, but skipping them turns cuts and scrapes into easy entry points for toxins. Cotton works, but adding a chemical-resistant barrier over it makes cleanup a lot easier.
What Happens If Something Goes Wrong?
If a splash or spill happens, immediate action matters. Use water—lots of it. Trying to scrub or wipe off a chemical doesn’t do much, but running water over skin for at least fifteen minutes helps dilute and wash away residue. Eye-wash stations aren’t just a box-ticking exercise; they’re a real line of defense if exposure happens.
Solutions for Safer Workplaces
Regular training stops accidents before they start. Demonstrations drive the point home far better than pamphlets. Workers who share near-misses help everyone get better at spotting and fixing weak spots. Spill kits last longer than most folks realize, but keeping them stocked matters more than waiting for accidents to run out the clock.
A well-organized storage space with clear separation between chemicals makes everyone’s day easier and safer. If you’ve ever sorted through crowded shelves, you know why order beats chaos. Checklists and standard operating practices don’t just keep auditors happy—they save lives. No shortcut or saved minute justifies ignoring the risk.
What is the chemical formula of 2-Ethylthiobenzyl N-Methylcarbamate?
Getting Straight to the Structure
Every molecule speaks its own language. In the world of pesticides, 2-Ethylthiobenzyl N-Methylcarbamate brings chemistry and agriculture together. Its chemical formula, C11H15NO2S, reflects more than just numbers and letters—it stands for complex interactions with pests, crops, and the people working the fields.
Understanding the Building Blocks
This compound tells a story through its structure. The “ethylthio” group hooks onto a benzyl core, which links to an N-methylcarbamate group. The sulfur atom in the formula jumps out right away, signaling some unique chemical action. For a guy like me, who grew up around farming towns, I can tell you that every tweak in a molecule can mean big changes, whether that's how well it controls pests or how long it lingers on plants.
Why Farmers and Scientists Should Notice
The formula C11H15NO2S carries real implications. The presence of the carbamate group means this pesticide falls under the carbamate family, used widely for its ability to knock out certain pests and insects fast. The addition of sulfur gives it a twist: improved effectiveness against particular types of bugs that have developed resistance to older chemicals. Farmers have dealt with pests shrugging off older sprays, seeing their crops disappear before harvest. A compound with chemical backbone like this one offers new hope.
Talking Safety and Environmental Risks
Looking at the science, carbamates like 2-Ethylthiobenzyl N-Methylcarbamate target the nervous system of insects by blocking an enzyme called acetylcholinesterase. That same action, though, can affect humans if safety steps get skipped. The sulfur in this molecule means breakdown happens somewhat differently than with other carbamates—sometimes faster, sometimes slower, depending on field conditions.
I've watched neighbors set aside protective gear because of heat or hurry, only to deal with headaches or nausea later. Knowing what's in a pesticide down to its chemical formula sheds light on why safety matters so much. A mistake on the farm can turn into a hospital visit, not just some lost produce.
Shaping Smarter Choices with Information
A clear formula like C11H15NO2S allows researchers to predict how 2-Ethylthiobenzyl N-Methylcarbamate behaves in the field, what sorts of breakdown products might linger in soil or water, and how often it needs reapplication for effective pest control. This isn’t just book knowledge—my own experience digging in gardens and running tests as a lab assistant backs up what chemistry teachers like to say: structure drives solutions. The better the public understands these formulas, the easier it gets for growers to make smart, safe calls.
Solutions Rooted in Chemistry
We can push for better education on safe handling and application. Detailed labels, regular training, and community outreach programs have lowered accidental poisonings in places that take them seriously. Researchers tracking this carbamate keep looking for ways to reduce its downsides, whether that means breeding crops that need less protection or developing products that break down faster and safer after use. If you care where your food comes from and what goes into it, it pays to look behind the long names and examine each formula. The choices we make in the lab and on the farm ripple out to families and dinner plates everywhere.
How should 2-Ethylthiobenzyl N-Methylcarbamate be stored?
Understanding the Substance
2-Ethylthiobenzyl N-Methylcarbamate doesn’t sound like something you’d see on a supermarket shelf, and for good reason. This kind of chemical usually shows up in agricultural and industrial settings. Its properties make it useful, but it also means you can’t just stash it away like old paint or leftovers. Ignoring its handling and storage leaves people, animals, and the environment at risk. No shortcuts here—people working with these compounds need straightforward information, not generic warnings.
Practical Storage: Lessons from the Field
Deciding where and how to keep chemicals safely comes from more than just reading a label. Those in charge at storage facilities and company warehouses have a simple checklist: safety, security, and long-lasting stability. People who have handled pesticides or complex chemicals know spills or leaks often come from tight, dark, poorly maintained corners and forgotten containers.
2-Ethylthiobenzyl N-Methylcarbamate, in particular, stays stable if kept dry and cool. Moisture in the air or sunshine pouring through a window quickly shortens the shelf life. Humidity reacts with chemicals and can trigger dangerous byproducts. Experienced workers store it in closed, air-tight containers lined with the original manufacturer’s labels. No improvising with old food jars or anything that can’t handle chemical exposure. Locked cabinets or dedicated storerooms—insulated against temperature swings and away from busy foot traffic—make the difference between safe handling and a disaster waiting to happen.
Factoring in Human Error
Human error causes plenty of headaches in storage management. Many accidents start with one person assuming someone else tightened a lid, read instructions, or checked the expiration date. Team huddles before any project involving hazardous materials may sound old-fashioned, but hearing reminders directly from a supervisor or a peer keeps the basics from slipping through the cracks. Spill containment bins and clearly marked signs in local languages help avoid confusion. No one benefits from confusing jargon or faded labels; real-world experience shows color-coded warnings and bold, plain instructions on every shelf and package get far better results.
The Role of Training and Oversight
Formal training with hands-on practice beats slideshow presentations or endless handouts. Getting workers to wear gloves, face shields, and protective overalls calls for buy-in. I’ve seen management transform safety culture by rewarding vigilance—recognizing the sharp-eyed worker who catches a leaking bottle early on. It creates pride, not just compliance. Regular inventory checks, led by people who know the substances—rather than just checking off boxes—reduce blind spots where neglect seeps in.
What the Facts Show
Looking at reports from the World Health Organization and safety agencies, improper storage links to both workplace poisonings and environmental contamination. Keeping 2-Ethylthiobenzyl N-Methylcarbamate locked away from food, feed, and water sources protects not only staff, but the whole community. Inspections often uncover old drums rusting behind buildings or bags splitting open under rain; both invite a chain of dangerous consequences nobody wants to see on the evening news.
Building Safer Systems
Big improvements don’t always need fancy tech, but they do need consistency and a willingness to listen to those closest to the work. Sensors monitoring humidity and temperature now cost less than a single shipping error. Digital checklists, updated by the people moving and storing chemicals, keep information current. Direct reporting lines between staff and decision-makers brings real-world concerns to light before a crisis hits. Everyone at the facility—down to the weekend temp—needs to know what they’re storing and how to respect its risks. Real safety comes from more than just following rules; it comes from treating every drum and bag as if your own family lives downwind.
What are the potential health hazards of 2-Ethylthiobenzyl N-Methylcarbamate?
The Chemical and Its Common Use
2-Ethylthiobenzyl N-Methylcarbamate may sound unfamiliar, but it has played a role in pest control for decades. Fields and storage facilities often rely on it as an insecticide, turning to its chemical properties to protect crops and reduce losses from insect infestations. Against this backdrop of heavy agricultural use, the health hazards deserve a careful look that puts people first, not just productivity.
Understanding the Exposure Routes
Workers prepping or applying this compound handle strong concentrations, and farmworkers cross paths with residue. Accidental ingestion, inhaling sprays, or soaking up the substance through skin can all introduce it into the body. That risk also reaches consumers when produce has leftover traces, since washing sometimes only does so much. My own experience on family farms taught me pesticides have a knack for lingering in places you least expect, and practices for safe handling often lag behind modern safety data.
Health Risks that Can’t Be Ignored
2-Ethylthiobenzyl N-Methylcarbamate affects the nervous system. Carbamates share a knack for interfering with cholinesterase, which helps nerves function. This inhibition leads to muscle weakness, twitching, headaches, confusion—even seizures or death when exposure runs high and help comes too late. Mild symptoms may start with nausea or sweaty skin. Unless medical intervention steps in, the toxic build-up worsens quickly.
Reports gathered by health agencies don’t paint a pretty picture. Poison control centers and study groups note that kids, rural workers, and anyone living near application areas face a higher risk. Some learn this the hard way, with episodes of dizziness, labored breathing, and even comas. Data from the World Health Organization shows that low-income regions with less oversight deal with higher rates of pesticide poisoning—including from carbamates such as this one.
Growing Concerns Beyond Acute Poisoning
One-time poisoning scares send people to the ER, but long-term, low-level exposure stirs up its own trouble. Farmworkers are the canaries in the coal mine. Regular contact links to chronic nerve damage, an increased chance of developing Parkinson-like symptoms, and disrupted hormone systems. Research published in journals like Environmental Health Perspectives points to developmental effects in children, including lower IQ and delayed motor skills, when expectant mothers absorb carbamate residues during pregnancy.
Some animal studies found tumors linked to chronic exposure, though direct evidence in humans remains spotty. The International Agency for Research on Cancer keeps monitoring, citing gaps in knowledge and the slow pace of long-term investigations. This uncertainty keeps raising public health questions, since outdated safety limits sometimes let small doses through that add up over time.
Taking Steps Toward Safer Use and Protection
Regulators can demand stronger labeling and training for workers. Many health experts push for better personal protective equipment and for buffer zones between sprayed fields and homes. Stronger residue monitoring on foods shows promise, cutting down on hidden intake for people who don’t work on farms. Some countries look at alternatives—integrated pest management or less hazardous chemicals—since the price of human health tops short-term crop gains.
Farmworkers deserve easier access to health care and frequent checkups for early warning signs. Regular monitoring of cholinesterase levels in high-risk groups, along with education tailored to local languages and customs, gives people tools to look after themselves and their families. My time volunteering in migrant health clinics showed me that informed workers—supported by strong public policies—stand a better chance of guarding their well-being in the shadow of chemical risk.
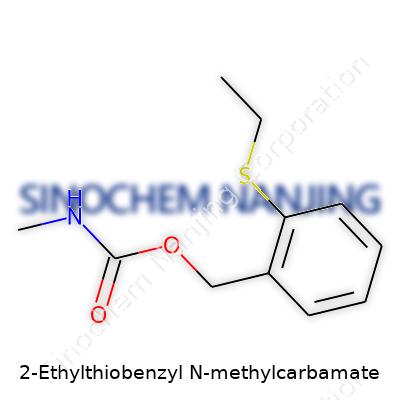
| Names | |
| Preferred IUPAC name | N-methyl-1-(2-ethylsulfanylphenyl)methylcarbamate |
| Other names |
Carbaryl-2-ethylthio NSC 65753 2-Ethylthiophenyl methylcarbamate |
| Pronunciation | /tuː ˈɛθɪlˌθaɪoʊˌbɛnˌzɪl ɛn ˈmiːθəlˌkɑːr.bəˌmeɪt/ |
| Identifiers | |
| CAS Number | 1129-41-9 |
| 3D model (JSmol) | `3Dmol '2-Ethylthiobenzyl N-Methylcarbamate' JSmol string:` ``` C1=CC=C(C=C1)CSC(C)N(C)C(=O)O ``` *This is the SMILES string representation, directly usable in JSmol and other molecular viewers to generate the 3D model.* |
| Beilstein Reference | Beilstein Reference: 2216166 |
| ChEBI | CHEBI:39105 |
| ChEMBL | CHEMBL2106413 |
| ChemSpider | 109545 |
| DrugBank | DB08676 |
| ECHA InfoCard | ECHA InfoCard: 100.102.608 |
| EC Number | 262-177-4 |
| Gmelin Reference | 100946 |
| KEGG | C18534 |
| MeSH | Dicarbocyanines |
| PubChem CID | 35219 |
| RTECS number | GV2975000 |
| UNII | C37O2N7B3F |
| UN number | 2810 |
| CompTox Dashboard (EPA) | DTXSID7043152 |
| Properties | |
| Chemical formula | C11H15NO2S |
| Molar mass | 241.32 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.132 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.01 |
| Vapor pressure | 4.2 x 10^-5 mmHg (at 25 °C) |
| Acidity (pKa) | 13.8 |
| Basicity (pKb) | pKb = 3.2 |
| Magnetic susceptibility (χ) | -62.01·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5890 |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -83.60 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7396 kJ/mol |
| Pharmacology | |
| ATC code | Pesticides |
| Hazards | |
| Main hazards | Harmful if swallowed. Toxic by inhalation, in contact with skin and if swallowed. Danger of serious damage to health by prolonged exposure. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Toxic if inhaled. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | Precautionary statements: P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P310, P330, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | Flash point: 138°C |
| Autoignition temperature | 360 °C |
| Lethal dose or concentration | LD50 oral (rat) 31 mg/kg |
| LD50 (median dose) | LD50: 3.2 mg/kg (mouse oral) |
| NIOSH | NM 2975000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Carbofuran Benfuracarb Aldicarb Methomyl Oxamyl |

