Looking Deeper into 2-Ethylhexyl Chloroformate: More Than Just a Chemical Reagent
Historical Development
Some of the most impactful industrial reagents have roots stretching back to the rapid chemical innovation of the twentieth century. 2-Ethylhexyl chloroformate never made headlines like DDT or aspirin, but its story reaches into shifts in organic synthesis methods. Chemists looked for reagents bridging raw compounds and increasingly specialized intermediates, searching for safer, more targeted ways to modify functional groups. The development of this compound reflected those pressures and priorities, ultimately carving out a niche in both research and large-scale applications.
Product Overview
Most chemists don’t keep 2-ethylhexyl chloroformate on the same shelf as their everyday solvents, but its value in selective synthesis feels unmistakable once you’ve used it on the bench. The molecule forms part of the chloroformate family—a class of chemicals celebrated for their ability to introduce carbonate and carbamate groups cleanly, often under milder conditions than old-school alkylating agents. You’ll find this product presented as a clear to pale yellow liquid, chosen because of its specific balancing act between reactivity and manageable volatility. Industrial users notice its stability during shipping and handling, so long as the environment avoids too much light or moisture.
Physical & Chemical Properties
With a molecular formula of C9H17ClO2 and a molecular weight above 192, 2-ethylhexyl chloroformate holds a key spot among mid-weight reagents. It boils between 195-196°C, making it workable under typical laboratory conditions, but its fuming behavior and sharp odor quickly remind you of its volatility. Splashes and spills don’t just make a mess—they send fumes that demand good ventilation or a fume hood. Its relative density ensures it won’t evaporate as fast as diethyl ether, yet its reactivity with water—sometimes violent—puts respect into even seasoned synthetic chemists. The molecule contains a chloroformate functional group attached to a branched alkyl chain, which defines most of its downstream chemistry.
Technical Specifications & Labeling
On the bottle, labeling requirements reflect both regulatory caution and a track record of incidents involving chloroformates. Hazard symbols spell out corrosivity and toxicity, and safety data sheets insist on strict storage and handling protocols. In labs and industrial spaces, containers need a tight seal, away from heat and moisture. Specifications for purity fall above 98% for high-end research applications. Impurity levels must be tracked, since even small amounts of hydrolyzed byproducts or unreacted alcohol can disrupt sensitive downstream reactions.
Preparation Method
Preparing 2-ethylhexyl chloroformate involves reacting 2-ethylhexanol with phosgene or safer phosgene substitutes in the presence of a base and an organic solvent. This step must be done with full attention to process engineering—the toxicity of phosgene has driven many improvements in closed-system synthesis and alternatives for small-scale runs. Chemists in both industry and academia often use batch reactors with careful degassing and vent management. The resultant compound undergoes thorough purification, sometimes including distillation or column separation, to reach the necessary purity for either research or manufacturing.
Chemical Reactions & Modifications
This reagent earns its keep for its smooth reactivity with a range of nucleophiles—especially alcohols and amines. Reactions tend toward formation of carbonates and carbamates, prized in custom polymer work or for temporary protection of functional groups during complex syntheses. Beyond those uses, researchers have found value in modifications that explore its reactivity with different catalysts or under greener solvent systems. For example, careful control of temperature and order of addition can steer reactions toward different protected intermediates, making the compound remarkably versatile in synthetic planning. But every reaction comes with the unchanging risk posed by its corrosive fumes, pushing innovation toward closed systems, better in-line monitoring, and improved scrubbing of exhaust streams.
Synonyms & Product Names
You’ll find 2-ethylhexyl chloroformate listed in technical literature by several names, including octyl chloroformate and 2-ethylhexyl carbonochloridate. These designations point back to its carbonochloridate structural core and the branching on its carbon chain. While product catalogs differ in how they describe grades or purity, the underlying compound always carries those distinct chemical signatures.
Safety & Operational Standards
Most chemists and engineers learn about this reagent in courses that focus on meticulous safety practices. The fume risk isn’t hypothetical—I’ve seen even seasoned colleagues scramble after an unexpected puff from a bottle cap. Gloves and goggles aren’t negotiable, and splash hazards make having a good eyewash station close at hand a must. The compound hydrolyzes quickly with water, releasing hydrochloric acid and carbon dioxide, so fire safety crews drill on how to suppress and clean up small spills rapidly. Proper air handling systems in workspaces keep exposure well below regulatory limits. Waste handling protocols—especially incineration at high temperatures—keep it out of local water and soil, preventing persistent contamination.
Application Area
Outside the lab, this reagent serves a range of chemistries, especially in pharmaceutical intermediate production and specialty polymers. Carbamate and carbonate protection strategies in complex organic synthesis lean heavily on it. Some manufacturers use it in the production of custom coatings or advanced plastics, while others rely on its unique properties for constructing dendritic materials or fine-tuning properties of specialty surfactants. Emerging methods that demand precise protection and deprotection cycles during large molecule assembly often rely on robust performance and high yield transformations supported by this compound.
Research & Development
Recent years have pushed researchers to explore how to make and use chloroformates with fewer risks. R&D teams look for greener production routes, replacing phosgene with milder carbonylating agents, sometimes investigating on-water or solvent-free approaches. Automation and closed-loop synthesis protocols focus on precise dosing and minimizing vapor release. Material scientists test how changes to the alkyl chain alter properties, pushing toward application-specific derivatives with less volatility or greater selectivity. The collaboration between safety researchers and synthetic chemists continues to shape how this and related reagents adapt to new industrial pressures.
Toxicity Research
Concerns over worker exposure and end-product residues have kept toxicity research near the fore. Inhalation, skin exposure, and accidental splashes can cause acute harm. Animal studies have illuminated the dangers of repeated exposure, so workplaces track air and surface levels closely. Chronic exposure risks to lungs, skin, and eyes drive strict air monitoring and medical surveillance for frequent users. Wastewater from production sites gets tested to confirm breakdown before discharge, with ongoing toxicology studies guiding regulatory standards and reformulations. Recent initiatives seek new scavengers for accidental releases, aiming to neutralize spills and vapors safely in real time.
Future Prospects
The future for 2-ethylhexyl chloroformate moves alongside the larger trend toward cleaner chemistries and safer workplaces. Green manufacturing chemists and engineers seek fresh catalysts, new protective technologies, and source chemicals that reduce overall hazard in the lab and the plant. As new synthetic methods pick up steam, the hope lies in finding balance—maintaining the versatility and efficiency that made these reagents essential, while cutting down on the risks that have shadowed their use for decades. Education, technical refinement, and legislative action all shape what the next chapter holds, promising a safer and more sustainable role for this member of the chloroformate family.
What is 2-Ethylhexyl Chloroformate used for?
Understanding Its Main Job
2-Ethylhexyl chloroformate pops up a lot in chemical plants, especially where companies make medicines and specialty chemicals. It's a reactive liquid, which means it sparks changes in other substances. Most folks in chemistry circles know it for its usefulness in pharmaceutical manufacturing.
The backbone of this compound’s value sits in creating carbamates and urethanes. People aren’t talking about foam padding for car seats here—these carbamates are usually essential steps in making certain medicines work. For example, in cancer drug synthesis, 2-ethylhexyl chloroformate plays a part in attaching protective groups to molecules, which stops them from falling apart or changing too early during the manufacturing process. That small, initial addition lets chemists control reactions and push only the right change at the right time.
Why It Matters in Medicine and More
Drug developers count on predictable chemistry. If they need to shield a sensitive spot on a drug molecule, they reach for reliable chemicals that won’t react with bystanders or toss in weird byproducts. By using 2-ethylhexyl chloroformate, the chance of ending up with mystery leftovers slims down, which supports both drug purity and patient safety. That’s not a minor detail. Contaminants in medicine can trigger severe side effects or cause a treatment to fail.
This chloroformate also lands in the lab toolbox for peptide synthesis, the method used to piece together the building blocks of new antibiotics or diabetes treatments. It lets researchers lock down molecular structure, steering clear of mishaps that could burn through weeks of progress in seconds.
Potential Troubles and Smarter Choices
2-Ethylhexyl chloroformate doesn’t just float around harmlessly. It reacts quickly, and you don’t want to mishandle it. The substance releases toxic and irritating fumes that damage lungs and eyes, so open-air work is out of the question. Any chemical that puts people’s health at risk needs respect and tight controls. I’ve met chemists who won’t even open the bottle outside a fume hood. Mistakes with chloroformates can cost more than just failed experiments—they can put someone in the hospital.
Using this stuff comes with responsibility. Companies have a legal duty to shield workers with protective gear and rigid training. Where possible, process automation and sealed systems cut down human exposure. From what I’ve seen, firms that invest in proper safety gear and teach good habits lose less time to accidents and face fewer lawsuits. In a country like the US, OSHA regulations keep a tight leash on its use. Meanwhile, waste management can’t fall by the wayside. Without proper disposal, this chemical could seep into waterways and harm wildlife.
Room for Safer Chemistry
Green chemistry isn’t a pipe dream anymore. Efforts push toward alternatives with less toxic fumes or easier waste handling. Developers look closely at which protective-group chemicals match or beat the results of 2-ethylhexyl chloroformate, but without the fallout. It takes time and effort, but the pressure from safety advocates and new science drives change.
Every tool in science brings trade-offs. Refusing to look for safer methods freezes innovation. By questioning long-held traditions and focusing on safety, chemists help keep workers healthy and the environment intact. That’s the best way to make sure chemicals like 2-ethylhexyl chloroformate remain a help, not a hazard.
What are the safety precautions when handling 2-Ethylhexyl Chloroformate?
Why Understanding This Chemical Matters
Working around chemicals with long names and tricky reputations never gets easier. I’ve seen what happens when people cut corners. 2-Ethylhexyl Chloroformate pops up in making plastics, coatings, and pharmaceuticals. It’s clear, almost oily, and doesn’t give much away until it makes your eyes water or your lungs feel tight. Folks treating this stuff like it’s just another ingredient soon learn its risks the hard way. Accidents in the lab or factory often stem from overconfidence, not lack of warnings.
Risks That Deserve Respect
A splash can burn skin. Vapors sting your eyes and can scar your lungs. This chemical reacts fierce with water, turning into hydrochloric acid, which brings its own set of hazards. I remember once a friend rinsed a spill with water—turned out worse than starting with a dry absorbent. Inhaling even small amounts triggers coughing, tight chest, and headaches. Just opening a bottle without protection can create enough vapor to irritate throats across the room.
Keep Exposure Low: Practical Precautions
Personal protection comes first. Always wear safety goggles that seal around your eyes. Heavy-duty gloves resist both chemicals and punctures—nitrile holds up better than latex here. Lab coats and splash-proof aprons help, especially around careless hands. I double-checked my gear even for quick transfer jobs.
Good ventilation saves trouble. Fume hoods don’t just sit there for show—draw the nasty stuff away before it can land in your lungs. In places without built-in hoods, local exhaust fans do the job. Open windows only move vapor around. Respirators with organic vapor cartridges work as backup but don’t rely on them as the first line.
What to Do If Something Goes Wrong
Even careful folks run into trouble. Getting this liquid on skin or eyes stings right away. Rinse the area nonstop with water for at least fifteen minutes and get medical help fast—don’t wait for a “good sign.” If you breathe in vapors, step outside or get to fresh air fast. Some symptoms build slowly, so don’t shrug off a nagging cough.
Spills need quick, smart action. Skip the mop and grab absorbent pads, making sure to wear the right gear. Don’t pour anything down the drain. Collect waste in sealed containers—label them and get them out of the workspace right away.
Training, Labels, and Communication: No Shortcuts
Nearly every mistake I’ve seen came out of someone skipping training. Regular refreshers make sure no one forgets why the steps matter. Labels need to stand out—scratched-out writing or faded stickers cause confusion, fast. Safety data sheets hide in dusty binders too often; keep them handy and short enough that people actually read them.
The Real Key: Teamwork and Vigilance
It’s never a one-person job. Whenever I’ve worked with 2-Ethylhexyl Chloroformate, a buddy system made the difference. People notice each other’s gear, catch errors, and give the nudge you need to follow through. Culture around chemical safety comes from mutual habit and shared memory of close calls. Trust built on day-to-day caution keeps everyone returning home healthy—something rules alone can’t guarantee.
What is the chemical formula of 2-Ethylhexyl Chloroformate?
Getting Familiar with 2-Ethylhexyl Chloroformate
As someone who has spent years around chemical labs and industrial settings, certain names bring back a mix of caution and respect. 2-Ethylhexyl chloroformate is one of those. Its chemical formula, C9H17ClO2, barely hints at the range of applications and safety demands tied to it. This molecule turns up mostly in processes where precision and responsibility take the lead. Seeing this compound’s role gets easier by breaking down what each part of its formula means for daily work.
Why 2-Ethylhexyl Chloroformate Matters
One of the first things you notice about 2-ethylhexyl chloroformate is its versatility. In manufacturing, it plays a significant role as a reagent, especially where people create specialty chemicals and pharmaceuticals. Its use isn’t about profit alone. Mistakes with this chemical create real risks, like exposure that can harm skin, lungs, and the natural world if safeguards slip. It’s one of those raw materials where one hand washes the other: care for the process, and you care for the people involved.
Many chemical workers will tell you: the devil really hides in poor storage and handling. Leaks or spills call for quick, trained responses—think eye washes, neutralizers, and airtight procedures. Having an emergency kit for 2-ethylhexyl chloroformate isn’t paranoia—it’s practical, grounded thinking.
Supporting Facts and Responsible Handling
The formula tells a story about the atom types—carbon, hydrogen, chlorine, and oxygen. Those who’ve handled it directly recognize the strong, pungent odor right away. Chlorine’s presence in the structure signals the potential for eye and lung irritation. While that might seem like just another job for protective gear, it’s also a reminder about environmental impact. Chloroformate compounds show toxicity toward aquatic organism populations, which can ripple through food chains and affect local water safety.
Regulatory agencies such as OSHA and the EPA emphasize strict guidelines for handling, transport, and disposal. People in the chemical trade often keep a written log of usage and spills. Records don’t just keep regulatory agencies happy; they help workers know the real situation on the ground.
Looking Toward Safer Solutions
Seeing the impact of mishandling dangerous chemicals changes how you work and teach others. Instead of thinking “good enough,” industry leaders focus on training, investing in updated equipment, and regular site audits. Small improvements—like better ventilation systems, spill trays, or routine drills—save headaches and, sometimes, lives.
There’s also a push for greener chemistry. Some companies invest in finding safer substitutes with lower environmental footprints, hoping to cut down toxic byproducts and operator risk. These advances don’t happen overnight. They rely on steady commitments, company-wide communication, and open feedback from the folks who actually mix, load, and transport these substances.
Respecting Chemistry in the Real World
My years of working with specialty chemicals, including 2-ethylhexyl chloroformate, taught me to take nothing for granted. Understanding the formula, C9H17ClO2, isn’t just about memorizing symbols. It means recognizing how chemistry shapes safety culture, business decisions, and community health. When knowledge meets practical steps and real accountability, people and places stay safer. That approach, more than the formula on paper, makes all the difference.
How should 2-Ethylhexyl Chloroformate be stored?
Understanding the Risks
Nothing grabs attention like a chemical that poses real hazards. Working with 2-Ethylhexyl Chloroformate taught me respect for its reactivity and toxicity. This ester reacts sharply with moisture and strong bases. Any slip can lead to toxic fumes or unpredictable outcomes. Safety comes from recognizing risk before ever opening a drum.
Keep It Cool, Keep It Dry
Heat speeds up unwanted reactions. Even a few degrees above room temperature can do more harm than expected. Many industrial safety sheets place maximum storage temperature at 25°C, and I’ve seen how that advice plays out in practice. Slight temperature rise turned a sealed flask foggy faster than anyone liked. So, a dry, cool store room isn’t an option—it’s essential.
No Room for Water
Humidity leads to hydrolysis. Even minor leaks or sweat inside a cap will break down the chloroformate, releasing phosgene-like fumes. I’ve watched seasoned chemists swap out metal tools for dry plastic widgets to minimize stray moisture. Desiccators and moisture indicators, often overlooked, act like silent sentries. If you spot any pink dots turning blue, act—don’t wait.
Sealed Containers Matter
Leaks and vapors aren’t just chemistry class horrors—they’re regular worries in the real world. Standard screw tops won’t cut it. Seal integrity never remains a guessing game. The industry leans hard on tightly sealed glass or teflon-lined containers, each with gasketed lids. Tampered seals underline more than minor loss; they suggest exposure, even contamination. Nobody should ever decant this chemical into wide-mouthed bottles or makeshift jars.
Segregation from Reactive Substances
It hides in plain sight, but incompatibility kills. Any shelf shared with strong acids, alkalis, water sources, or oxidizers invites trouble. Splash from a lye spill next door can trigger a violent reaction. I remember a shelf collapse that stacked incompatible containers on floor level. What seemed like a storage shortcut spiraled into a costly cleanup. Lesson learned: chemicals with problems sharing space get their own place, far from firefighting equipment that sprays water.
Label Everything—Every Time
In one cluttered storeroom, faded labels created confusion and close calls. Every bottle of 2-Ethylhexyl Chloroformate should show content, date, and hazard info at a minimum. Make language clear enough that no one guesses. Newer regulatory standards now call for pictograms, signal words, and clear risk phrases. Old hand-written tags don’t meet today’s safety needs. Pull outdated inventory, check for leaks, and update quadrennially (if not sooner).
Prepare for Emergencies
Personal experience reminds me: chemical risks multiply without readiness. Spill kits with activated charcoal, absorbent pads, and neutralizing agents need to stay nearby. No one in their right mind handles this material without goggles, gloves, and air flow. Respiratory protection—no matter how hot or uncomfortable—beats the alternative. Eyewash and safety showers require clear paths, never blocked by boxes or bins. Training refreshers and routine drills shouldn’t feel optional.
Compliance Isn’t a Paper Drill
Regulators care for good reason. The Environmental Protection Agency and Occupational Safety and Health Administration consider 2-Ethylhexyl Chloroformate hazardous. Inspection-ready documentation doesn’t just avoid citations; it keeps workers informed and careful. I’ve watched places that skip checklists turn sloppy, raising risks for staff and even the neighborhood. Treat every container like a promise to uphold safe practices—cut corners and it’s not just your signature on the line.
Is 2-Ethylhexyl Chloroformate hazardous to health or the environment?
Understanding the Risks of 2-Ethylhexyl Chloroformate
Walking into a lab, it doesn’t take long to realize that handling chemicals brings risks, even if the labels look harmless at first. 2-Ethylhexyl chloroformate isn’t as well-known as some hazardous materials, yet its hazards don’t stay in the background. This chemical often serves as a building block in making pesticides, pharmaceuticals, and dyes. Few people outside chemical plants have heard of it, but for those who work with it, exposure is a genuine worry.
Direct Health Hazards
Chemical safety data tells a clear story. 2-Ethylhexyl chloroformate acts as an irritant. Touching it can burn the skin and eyes. Breathing its vapors irritates airways and lungs. Safety manuals place it in the same hazard class as other chloroformates, which means it can also release toxic gases like phosgene if it reacts with water or humidity in the air. Phosgene turned up in World War I as a chemical weapon, so hearing it connected to any workplace chemical should catch attention fast.
Personal experience with chemicals like this has taught workers to respect not just the clear and present burns, but also the unseen long-term effects. Respiratory problems, skin damage, and eye injuries don’t always show up right away, but they add up.
Environmental Impact
It’s not just direct contact that worries me. 2-Ethylhexyl chloroformate does not break down quickly in soil or water, which puts aquatic species at risk. Spills don’t disappear overnight. Runoff can wash into streams, threatening fish and plants long after the original incident. Even small leaks, when repeated, have a way of building up and stressing local ecosystems in ways that can’t be fixed just by stopping use later on. There isn’t any silver bullet for remediating chloroformate contamination. That means the emphasis always lands on prevention.
Why Proper Handling Matters
I have seen that strong rules for protective gear—goggles, gloves, masks—are not just about ticking a box. With substances like this, skipping those steps changes the outcome. Ventilation systems need to be strong enough to pull chemical fumes out of the air. Emergency showers and wash stations don’t look like much until you need them. In labs or factories where chemicals move around every day, even a brief mistake can mean weeks out recovering from burns or breathing trouble.
Pushing for Safer Alternatives
The best answer in industry often points to substitution. Green chemistry leaders push for building blocks derived from less hazardous materials or, in some fields, from renewable sources. Scientists and companies can pay more upfront to move away from chloroformates, but the savings come in fewer injuries and less cleanup in the long run. Regulatory agencies sometimes set legal limits or require special permits, which raises the bar for safe handling and drives research for replacements.
Building a Safer Future
Transparency goes a long way. Sharing real data on incidents helps convince managers and operators to invest in better training and systems. Every improvement lowers risk for workers, nearby neighbors, and entire watersheds. For people who handle chemicals every day, real progress means not just reading safety bulletins but living by them and pushing for safer, cleaner options wherever possible.
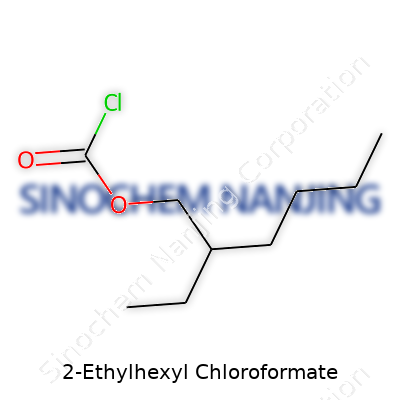
| Names | |
| Preferred IUPAC name | 2-ethylhexyl carbonochloridate |
| Other names |
2-Ethylhexyl chloroformate Chloroformic acid 2-ethylhexyl ester 2-Ethylhexyl chlorocarbonate 2-Ethylhexyl carbonochloridate O-Chloroformylcaprylic acid |
| Pronunciation | /tuː ˌɪθ.ɪlˈhɛk.sɪl klɔːr.oʊˈfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 24468-13-1 |
| Beilstein Reference | 1739460 |
| ChEBI | CHEBI:51436 |
| ChEMBL | CHEMBL4292346 |
| ChemSpider | 212275 |
| DrugBank | DB14485 |
| ECHA InfoCard | 100.076.337 |
| EC Number | 202-590-5 |
| Gmelin Reference | Gmelin Reference: 83628 |
| KEGG | C18721 |
| MeSH | D017217 |
| PubChem CID | 11803 |
| RTECS number | GG9625000 |
| UNII | J364A4W38E |
| UN number | UN2735 |
| Properties | |
| Chemical formula | C9H17ClO2 |
| Molar mass | 184.68 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Mild; ester-like |
| Density | 1.014 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 3.6 |
| Vapor pressure | 0.14 mmHg (20 °C) |
| Acidity (pKa) | pKa = 1.5 |
| Basicity (pKb) | 11.1 |
| Magnetic susceptibility (χ) | -6.08E-6 cm³/mol |
| Refractive index (nD) | 1.4300 |
| Viscosity | 3.3 mPa·s (20 °C) |
| Dipole moment | 2.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 527.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -589.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -726.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H331, H334 |
| Precautionary statements | P210, P261, P273, P280, P301+P330+P331, P305+P351+P338, P309+P310 |
| NFPA 704 (fire diamond) | 2-1-1-W |
| Flash point | 85°C |
| Autoignition temperature | 385 °C |
| Lethal dose or concentration | LD50 oral rat 960 mg/kg |
| LD50 (median dose) | LD50 (oral, rat) 544 mg/kg |
| NIOSH | SN 2100000 |
| PEL (Permissible) | PEL: 0.1 ppm |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | IDLH: 2 ppm |
| Related compounds | |
| Related compounds |
Methyl chloroformate Ethyl chloroformate Isobutyl chloroformate Phenyl chloroformate 2-Ethylhexanol |

