2-Ethylbutyraldehyde: From Concept to Practical Use
Historical Development
Curiosity about chemicals led researchers down some interesting paths, and the history of 2-ethylbutyraldehyde speaks to persistence more than breakthrough moments. Over the past century, scientists figured out ways to selectively add and manipulate side chains on the aldehyde backbone, and by the 1920s, 2-ethylbutyraldehyde drew attention as a promising building block in flavor and fragrance synthesis. Early references linked back to investigations in petrochemical streams, mainly as distillation methods improved and allowed finer control over branched-chain aldehyde production. As chemical engineering got more sophisticated, the production of this aldehyde moved from small-batch laboratory curiosities into steady industrial practice. The story here isn’t about a single flash of insight, but a steady accumulation of know-how that made 2-ethylbutyraldehyde significant for chemists and manufacturers looking to expand their toolkit beyond simple, straight-chain molecules.
Product Overview
2-Ethylbutyraldehyde stands out by featuring a branched, four-carbon backbone with an ethyl group at position two and a terminal carbonyl characteristic of aliphatic aldehydes. Chemists rely on this profile to introduce complexity and variety in end products like flavors and fragrances—a sharp, distinctive aroma that gets used in compounds meant to mimic natural fruit and plant notes or to build more complex chemical architectures. Commercial suppliers deliver it as a clear to slightly yellow liquid, and it finds use both as an intermediate and as a final product in specialty applications.
Physical & Chemical Properties
Those who handle 2-ethylbutyraldehyde daily recognize its volatility and pronounced fruity odor, a feature that’s tough to miss in the lab. Its boiling point hovers around 120°C, and this manageable temperature means relatively simple distillation and purification processes. Its density lands at about 0.81 g/cm³ at 20°C. The aldehyde group renders it reactive, especially toward nucleophiles and oxidizing agents, so care and planning enter the picture immediately upon use. Water solubility stays on the low side, but it readily miscibles with common organic solvents—a factor that shapes decisions on storage and blending.
Technical Specifications & Labeling
Buying or selling 2-ethylbutyraldehyde involves strict attention to purity, usually specified at 98% or higher for most research and commercial uses. Suppliers report water content, acidity, and residue on evaporation—these matter for both downstream reaction yields and final product safety. Reliable labeling doesn’t stop at composition: hazard warnings, storage instructions, and transport compatibility tie directly to safe handling. Regulatory bodies expect clear hazard pictograms and fleet adherence to globally harmonized system (GHS) symbols, a move that gives plant managers and lab staff concrete information for Emergency Response Planning.
Preparation Method
Most industrial procedures source 2-ethylbutyraldehyde from the oxo process—hydroformylation of propylene with synthesis gas (a blend of CO and H₂) in the presence of a catalyst. Here, a mix of temperature, pressure, and carefully picked catalysts like cobalt or rhodium sets up the chemistry for adding a formyl group to the branched alkene, producing not only 2-ethylbutyraldehyde but often isomeric compounds requiring downstream separation. Most plants recycle unused gases, maximizing atom economy and bumping up efficiency. On a smaller scale, traditional organic synthesis routes—like selective oxidation of 2-ethylbutanol—still show up in specialty labs when precise isotopic labeling or ultra-high purity matters.
Chemical Reactions & Modifications
Once synthesized, 2-ethylbutyraldehyde becomes a versatile intermediate. The reactive nature of the aldehyde group sets the stage for a variety of transformations. Grignard reagents latch onto the carbonyl, giving access to alcohols with tertiary branching. Chemists often use it in aldol reactions, pairing it with other aldehydes or ketones to build larger molecules with unique backbone structures. Hydrogenation reduces it to 2-ethylbutanol, expanding options for downstream chemistry or usage as a solvent or flavoring agent. Reactions with hydroxylamine and hydrazine deliver oximes and hydrazones, providing both analytical and synthetic leverage.
Synonyms & Product Names
Those who search scientific literature or supplier catalogs often run into several names for the same compound. "2-Ethylbutanal" shows up in systematic nomenclature, while "Ethylbutraldehyde" or "alpha-Propylacetaldehyde" find use in older texts and some regional markets. Larger chemical suppliers sometimes assign trade names for batch tracking and product tracing, yet the molecular structure stays the same across brands and aliases.
Safety & Operational Standards
Long hours handling aldehydes breed respect for their reactivity and vapors. Exposure to 2-ethylbutyraldehyde causes mucous membrane irritation, and proper ventilation becomes mandatory for anyone working with larger quantities. Splash goggles, gloves, and fume hoods remain the mark of responsible labs. Ignition risk stays high around open flames or static discharge, and chemical operations crews rely on proper grounding, non-sparking tools, and local exhaust ventilation. Safety data sheets remind users about acceptable airborne concentration limits, typically linked to general aldehyde exposure standards. Waste management practices follow local laws since untreated aldehyde waste poses threats to waterways and soils.
Application Area
The reach of 2-ethylbutyraldehyde spreads well beyond the synthetic lab bench. In flavors and fragrance chemistry, its scent profile helps craft artificial fruit notes, often blended into confections, drinks, and personal care formulations. Paint and coating manufacturers use it as a starting material for branched-chain alcohols or acids, which then feed into specialty plasticizers and stabilizers. Research labs use it to probe reaction mechanisms and study carbonyl chemistry, where its branched structure brings new challenges and opportunities for catalysis and selectivity studies. Some pharmaceutical syntheses rely on it for making intermediates not easily accessed through simpler aldehydes.
Research & Development
Given the versatile nature of its aldehyde group, ongoing research into 2-ethylbutyraldehyde gravitates towards green chemistry. Scientists pioneer catalyst systems that push yields higher with less waste and milder reaction conditions, a vital goal as regulatory requirements edge toward lower environmental impact. Analytical chemists develop better detection methods, often using gas chromatography-tandem mass spectrometry, to track trace contamination or by-products in the supply chain and research environments. Modern labs also probe biological pathways affected by branched aldehyde exposure to understand both environmental breakdown and metabolism in living systems.
Toxicity Research
Researchers track both the acute and chronic toxicity of 2-ethylbutyraldehyde. High concentrations of vapor irritate eyes, skin, and respiratory tracts, while animal studies point to central nervous system effects at higher doses. Scientists test bioconcentration and breakdown in soil and water, tracking by-products that might threaten aquatic organisms. Regulatory agencies keep a close eye on worker exposure levels, often placing 2-ethylbutyraldehyde in the same risk bracket as structurally similar aldehydes, with recommended exposure limits set by occupational safety groups. Emergency medicine studies also examine best practices for treating overexposure, teaching first responders and workplace health staff how to deal with accidents quickly and efficiently.
Future Prospects
Looking ahead, 2-ethylbutyraldehyde offers clear opportunities and challenges. Green chemistry and environmental sustainability drive more efficient catalyst development and better waste reclamation protocols. Bio-based raw materials might overtake petrochemical sources as renewable feedstocks gain popularity and price parity, bringing new economic and environmental incentives. Research into selective oxidation and reduction also charts a path toward more specialized, high-value derivatives—extending its role in pharmaceuticals, agrochemicals, and specialty polymers. Pushes for stricter regulation and lower emissions will keep pressure on manufacturers to improve control, monitoring, and worker safety, further professionalizing handling and end-use sectors. Demand from the fragrance and flavor industry remains steady, but new applications may emerge as scientists find novel ways to harness this aldehyde’s branching and reactivity in advanced synthesis.
What is 2-Ethylbutyraldehyde used for?
Meeting a Niche in Chemical Manufacturing
2-Ethylbutyraldehyde doesn’t attract much attention outside of labs or industrial plants, but this small molecule turns up in more products than many folks realize. Chemists figured out a while ago it’s a handy building block. Its structure—simple, but just a bit quirky—lets it take part in all sorts of chemical reactions. I’ve spoken to researchers in fragrance and flavor labs, and they keep coming back to this compound because it’s got the right mix of volatility and reactivity. The strong, pungent aroma doesn’t hurt, either, if you’re looking to make that punchy note for certain perfumes or food additives.
Contributing to Flavors and Fragrances
The world of artificial flavors relies on molecules that pack a punch in small doses. 2-Ethylbutyraldehyde delivers that potency. Think about the kinds of fruity or nutty notes you might find in confections or soft drinks. Food scientists blend in trace amounts to mimic real fruit flavors or bring out richness in food. The perfume industry uses it to anchor more volatile scents, controlling how aromas unfold after you open a bottle or spray a fragrance. That characteristic smell, though overpowering on its own, fits right in once diluted and combined with other ingredients.
Stepping Stone in Chemical Synthesis
Many industrial processes depend on compounds that don’t show up in the final product but prove critical along the way. 2-Ethylbutyraldehyde gets converted into alcohols, acids, and other aldehydes for pharmaceuticals, pesticides, and specialty chemicals. Manufacturers use it in processes making solvents or plasticizers. In these facilities, careful handling and high standards keep things on track—exposure guidelines exist because inhaling too much can irritate the lungs. My years in research labs taught me that working with these raw materials demands both respect for their hazards and appreciation for their versatility.
Environmental and Safety Considerations
The flip side comes with risk management. 2-Ethylbutyraldehyde, like many aldehydes, isn’t benign. Strong odors often signal potential toxicity. I remember a factory safety seminar spelling out the importance of using it in well-ventilated areas, wearing gloves, and sticking to established exposure limits. Regulators track usage because spills or air releases can impact workers and surroundings. The good news comes from advances in containment and monitoring. Automated systems alert workers to leaks, while training programs walk employees through spill response and first aid.
Looking for Better Alternatives
Calls for greener chemistry urge companies to rethink raw materials like 2-ethylbutyraldehyde. Research teams, especially in Europe and North America, have started developing biosynthetic routes to make the same flavors and fragrances without harsh side effects. Some start with plant-based feedstocks, aiming to dial down environmental impacts. Even so, completely replacing it often means trade-offs—cost, performance, and purity all factor in. Innovators in synthetic biology and catalysis keep searching for safer ways to achieve the same end results.
Finding Balance
Many ingredients that play a behind-the-scenes role in manufacturing get little attention until problems pop up. 2-Ethylbutyraldehyde enables creative breakthroughs across flavors, fragrances, and industrial chemistry. At the same time, handling it responsibly, improving safety gear, and exploring alternatives keep it from becoming an unnecessary risk. Staying informed, supporting scientific research, and demanding transparency help hold everyone—from producers to users—accountable for its safe use.
What is the chemical formula of 2-Ethylbutyraldehyde?
Not Just Numbers: Understanding 2-Ethylbutyraldehyde
Most folks outside of chemistry labs rarely talk about 2-Ethylbutyraldehyde. Even in the industry, it tends to live quietly behind more famous molecules. What pulls it into the spotlight? Its chemical formula: C6H12O. More than a dry code, this formula tells a tiny story about how a small aldehyde can fit into bigger chemical processes.
Digging Into the Structure
Six carbons, twelve hydrogens, one oxygen—each atom counts. This formula points to an aldehyde functional group slapped onto a six-carbon chain with a little twist: an ethyl side group branching from the second carbon. If you’ve ever built a model with sticks and balls, you know the way a single extra group can change everything. Here, that second-carbon ethyl branch shifts physical and chemical properties compared to straight-chain aldehydes. In practical terms, it changes flavor and reactivity, making the compound handy in fragrance creation, manufacturing flavors, and as an intermediate in more complex organic syntheses.
Applications that Hit Close to Home
This compound can be found behind the scenes wherever you run into artificial flavors, some perfumes, even certain industrial solvents. Manufacturers don’t like to waste time or cash making things from scratch every day. They need reliable building blocks, and 2-Ethylbutyraldehyde fits the bill. Tools like this keep supply chains moving, letting flavor houses and chemical plants create consistent products without hiking costs. In my own home kitchen, I always reach for ingredients that save time. Coveting the same "go-to" in fields like chemistry seems only natural. The common chemical formula unlocks a repeatable, predictable path—no surprises, no breakdowns on the line.
Safety and Handling: Being Realistic
Working with organic aldehydes—2-Ethylbutyraldehyde included—calls for a respect for volatility and reactivity. The familiar smells and results might lull workers, but mishandling brings possible problems like skin irritation or inhalation risks. Reading up on the formula gives a clear sense of what to expect. Knowing it contains that reactive carbonyl group signals hurdles around storage and transport—flame, heat, and air exposure present serious risks. My time around workshops and home labs hammered in that hard rule: never underestimate flammable compounds, no matter how "routine" they seem on paper. Proper ventilation, personal protective equipment, and chemical training make a visible difference in workplace safety.
Making Chemistry Useful
Knowing the formula C6H12O isn’t about trivia—it's about laying a solid foundation for using and understanding 2-Ethylbutyraldehyde responsibly. If schools, hobbyists, and industry all fold quality chemical education into daily routines, accidents drop and productivity climbs. People who learn exactly what their chemicals do—just like cooks who learn their ingredients—end up wasting less, achieving consistent results, and staying out of trouble. Stronger chemical literacy isn’t just about passing tests; it’s how product quality and worker health improve across the board.
Is 2-Ethylbutyraldehyde hazardous or toxic?
Everyday Chemistry and Safety Blind Spots
2-Ethylbutyraldehyde doesn’t pop up in everyday conversation, yet its chemical relatives show up in perfumes, cleaners, and plenty of manufacturing settings. So naturally, workers might wonder: is this stuff dangerous? It's not a household name, but it shouldn’t be ignored. My time around industrial plants has taught me that anything with "aldehyde" in its name should be given a good look—too many years hearing stories about headaches, skin rashes, and worse from folks working without proper gear.
What Science Tells Us
According to data from the National Institute for Occupational Safety and Health, aldehydes are known irritants, and some cause severe reactions, even at low concentrations. 2-Ethylbutyraldehyde specifically does not enjoy a clean slate—a report from the European Chemicals Agency lists it as dangerous if inhaled. It irritates the eyes, nose, and throat. Anyone who’s been exposed to chemical vapors in a poorly ventilated workspace can recall how fast irritation can hit, sometimes before the nose registers the sharp scent. The risk goes up for workers who handle drums, transfer gallons, or mix raw chemicals by hand.
Real Harm or Regulatory Overkill?
It’s one thing to read safety data sheets—another thing entirely to see someone rushed to the eyewash station after a spill. 2-Ethylbutyraldehyde is not on the top shelf of global chemical hazards, but complacency works against us. Studies show that repeated exposure, especially through inhalation, may damage mucous membranes, hurt lung function, or leave skin with chemical burns. Some aldehydes have proven links to cancer and genetic mutations in animal studies, so prudent avoidance isn’t just bureaucracy—it's self-respect and responsibility.
How Risks Show Up at Work
Few companies purposely cut corners on worker safety, but poor labeling or worn-out PPE sneak up in unexpected ways. Mixing or heating 2-Ethylbutyraldehyde releases vapors, which, besides being flammable, can sneak past loose-fitting masks. Eye and skin contact usually comes from splashes that nobody plans for. I’ve seen accidents traced back to a single cracked glove or an unlabeled secondary container. People get used to the routine, and suddenly the burn sneaks up during a spill cleanup.
Steps That Make a Difference
Good engineering controls—like decent exhaust fans and air monitoring—beat wishful thinking every time. Training for workers can’t be an afterthought, especially for new hires who might not spot faint chemical odors yet. Besides basics like splash goggles and chemical gloves, clear labeling and spill kits at hand matter just as much as the chemical formula. Facilities need regular walkthroughs, not just annual reviews, to catch problems before they become injuries.
If disposal gets skipped over, those fumes won’t stay contained. Every workplace I’ve seen that takes the time for detailed logs and container checks ends up with fewer medical incidents. Employee involvement in safety planning makes a difference—a seasoned worker’s tip about a leaky valve or the wrong kind of gloves often saves more headaches than another memo from management.
Smart Handling Trumps Regret
2-Ethylbutyraldehyde isn’t the nastiest toxic chemical in the world, but it doesn’t deserve a shrug and business as usual. Up-to-date chemical hazard communication, hands-on training, and routine equipment checks create safer workplaces. That’s how families get home with fewer stories about burns and less time in urgent care. Whether in a research lab or a big production line, nobody benefits long-term by cutting corners with aldehydes. Direct, practical safety steps are worth every minute.
What are the storage conditions for 2-Ethylbutyraldehyde?
Why Storage Conditions Matter
Chemical storage gets overlooked until trouble shows up. I learned that during my own early days working in a university lab: a little neglect, a sticky cap, some unexpected heat — suddenly, you’re cleaning up more than just confusion. Take 2-Ethylbutyraldehyde. This stuff might seem straightforward, but letting down your guard can invite disaster. Flammable liquids have a reputation for a reason. If a container cracks open or vapors leak out, there’s a sniff of risk right behind.
What Science and Safety Officers Agree On
Forget high school assumptions. A clear shelf in the back room doesn’t count as “stored properly.” 2-Ethylbutyraldehyde calls for a cool, tightly sealed life, away from sunlight. Even in an ordinary storage cabinet, heat can make its volatility rear up. You wouldn’t leave milk out in summer — it spoils fast. In much the same way, 2-Ethylbutyraldehyde needs an environment below room temperature, ideally between 2°C and 8°C. A dedicated chemical fridge works best; keep it away from food fridges. Strong, shatterproof bottles or amber glass containers block light and resist accidental bumps.
Fire Hazards and Ventilation Concerns
Vapor builds up quickly with volatile aldehydes. Poor ventilation turns small leaks into big headaches, not just for the people in the room but for anyone walking by. One mistake I’ll never forget: a snap-top jar with no fume hood led to a strong, sickly-sweet odor rolling through the corridor. A safe storage room needs real ventilation and signs marking all flammables. Store 2-Ethylbutyraldehyde away from oxidizers, acids, or bases. Mixing those can trigger runaway reactions, and there’s no warning before they go off.
Labeling, Monitoring, and Simple Checks
Labels matter. Too many chemists rely on memory; nobody’s memory stands up after months away. Write acquisition dates, concentrations, and hazard codes right on the bottle. Check for leaks or cloudiness every time you grab a container. A sticky exterior usually points to escaping vapor. Employees should keep spill kits nearby, just as they’d keep a mop for a kitchen spill. Even small drips demand a full cleanup — not a half-hearted wipe with a paper towel.
Regulation, Disposal, and Environmental Responsibility
Local regulators — like OSHA and EPA in the US — don’t joke about negligence. Fines get steep, especially when flammable solvents escape track-and-trace logs. Used or expired 2-Ethylbutyraldehyde can't go down the drain or into the trash. Waste gets sent through special hazardous-waste streams, tracked, and incinerated to keep toxins out of groundwater. It costs money but pays off in public health and environmental safety.
Practical Steps That Make a Difference
Chemical safety draws on discipline, not just rules. Store 2-Ethylbutyraldehyde away from sparks and static. Avoid plastic shelving that warps or melts. Train everyone, not just supervisors — I’ve seen new lab members save the day by spotting a swelling bottle before it ruptured. Every workplace should keep emergency phone numbers posted, spill response materials on hand, and the simple habit of checking containers weekly.
What’s at Stake
Ignoring best practices means risking fire, poisoning, or regulatory violation. Storage is not about caution for its own sake, but real-world protection — for yourself, for your coworkers, and for the environment outside those lab doors.
Where can I purchase 2-Ethylbutyraldehyde?
Buying Chemicals Isn’t Like Grabbing Groceries
Most folks never hear about 2-Ethylbutyraldehyde unless they work in a lab, a manufacturing plant, or a research office. Its name alone can trip up experienced chemists. This chemical doesn’t show up on regular store shelves, not only because it’s a specialty compound, but also because chemistry always asks for respect—misuse or mishandling can go sideways quickly.
Specialty chemicals flow through channels built around trust and safety. Unlike cleaning supplies or plant food, sellers want to know exactly who they’re dealing with before opening their catalog. Most companies dealing with 2-Ethylbutyraldehyde or similar substances screen buyers, making sure there’s a legitimate need backed by paperwork and a paper trail. I’ve seen this myself when colleagues order for the lab—photo ID, business license, and details about the research go straight to the supplier before even talking price.
Compliance: Not Just Red Tape, Real Protection
Safety rules for controlled or industrial chemicals exist for plenty of good reasons. 2-Ethylbutyraldehyde isn’t something for the basement hobbyist. Many chemical suppliers need a clear business, research, or educational purpose. Laws keep up with new advances. In the U.S., the EPA, OSHA, and the Department of Homeland Security all keep eyes on distribution. In Europe, REACH rules set standards. Reputable sellers like Sigma-Aldrich, TCI, and Alfa Aesar work hard to confirm buyers aren’t using chemicals for harm.
A quick search online brings up suppliers, but an order doesn’t go through with a click. Legitimate companies use customer vetting, require proof of commercial or institutional use, and share safety data upfront. These steps protect everyone. Once a chemical leaves the shipping dock, the person ordering becomes responsible for safe storage, use, and disposal. No real supplier ignores these responsibilities.
The Myth of Easy Purchases
Online marketplaces sometimes pop up listed as sources for strange and obscure chemicals. Plenty of these listings don’t pan out. Some posts come from overseas resellers or individuals, and the quality is questionable at best, dangerous at worst. Regulatory bodies catch some of these sellers, but others slip through the cracks. If someone needs 2-Ethylbutyraldehyde and skips the verification steps, they’re putting research and safety in jeopardy.
Lab-grade chemicals aren’t consumer products for a reason. Reputable market leaders educate their staff, train for emergency response, and ensure handling meets specific requirements. Over my years working around scientists, most have stories of near-misses—spills, mislabeled bottles, or shady suppliers—none of which anyone wants to repeat. Cutting corners hurts everyone.
Building Stronger Access and Education
There’s a gap in public resources for people who want to learn about chemicals, their risks, or even who qualifies for access. Better outreach from suppliers and educational institutions can demystify the process. People who need 2-Ethylbutyraldehyde for legitimate projects should look up professional chemical suppliers, talk to institutional safety officers, and get training if there’s any doubt. Nobody fixes a lack of oversight with more rules—real results come from education, relationships, and a culture of responsibility.
Chemistry runs on trust. If a chemical stays behind a counter instead of out in the open, it’s not about gatekeeping—it’s about keeping people safe and making sure science moves forward without risk.
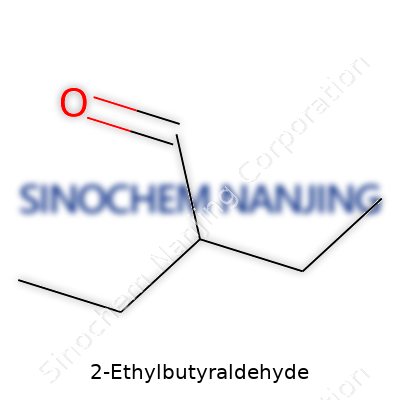
| Names | |
| Preferred IUPAC name | 3-Methylpentanal |
| Other names |
2-Ethylbutanal Ethylpropionaldehyde |
| Pronunciation | /tuː ˌɛθ.ɪlˈbjuː.tɪr.æl.deɪd/ |
| Identifiers | |
| CAS Number | 97-96-1 |
| Beilstein Reference | 582125 |
| ChEBI | CHEBI:49462 |
| ChEMBL | CHEMBL147338 |
| ChemSpider | 157372 |
| DrugBank | DB04106 |
| ECHA InfoCard | ECHA InfoCard: 100.004.341 |
| EC Number | 204-610-1 |
| Gmelin Reference | 173271 |
| KEGG | C08289 |
| MeSH | D008543 |
| PubChem CID | 11315 |
| RTECS number | EL8575000 |
| UNII | 8A7S3B46SL |
| UN number | UN1159 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.824 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 0.99 |
| Vapor pressure | 3.3 kPa (at 20 °C) |
| Acidity (pKa) | 14.89 |
| Basicity (pKb) | Basicity (pKb) of 2-Ethylbutyraldehyde: 12.89 |
| Magnetic susceptibility (χ) | -49.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.397 |
| Viscosity | 0.639 cP (20°C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -252.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2745 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0-ALQU |
| Flash point | 27 °C (81 °F; 300 K) |
| Autoignition temperature | 225 °C |
| Explosive limits | 1.3% - 10.2% |
| Lethal dose or concentration | LD50 (oral, rat): 3,287 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1780 mg/kg (rat, oral) |
| NIOSH | NA9375000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Ethylbutyraldehyde: "PEL: 100 ppm (360 mg/m3) TWA |
| REL (Recommended) | REL (Recommended Exposure Limit) of 2-Ethylbutyraldehyde is "5 ppm (18 mg/m3) TWA". |
| IDLH (Immediate danger) | IDLH: 200 ppm |
| Related compounds | |
| Related compounds |
Isovaleraldehyde n-Valeraldehyde 2-Methylbutyraldehyde |

