2-Ethylaniline: A Detailed Look
Historical Development
Chemists first prepared 2-ethylaniline over a century ago. Laboratory notebooks from the early twentieth century mention aromatic amines and their simple derivatives, as researchers grew obsessed with coal tar and its chemical treasures. Back then, every tweak to the aniline skeleton opened paths to new dyes, drugs, and plastics. Working with those aromatic amines, folks discovered how bulk and position of ethyl groups shaped the behavior of these molecules. Over time, researchers refined methods, built safer protocols, and tapped into production techniques that turned small-scale reactions into reliable, scalable syntheses.
Product Overview
2-Ethylaniline lands in the organic chemistry toolkit as both a target and an intermediate. On the surface, it looks ordinary—a colorless to yellowish oily liquid with a distinct stable amine odor. Industry taps this compound to create dyes, pigments, and fine chemicals. In academic and corporate settings, scientists reach for 2-ethylaniline when they want to test the electronic and steric effects of aromatic amines bearing small alkyl groups. Its robust presence in dyes, pharmaceuticals, and agrochemicals highlights the nook it has carved in industrial chemistry.
Physical & Chemical Properties
With a molecular formula of C8H11N and a molecular weight of roughly 121.18 g/mol, 2-ethylaniline brings together the tell-tale features of primary aromatic amines. It boils close to 211°C and melts at a temperature generally below room conditions, staying liquid through most lab scenarios. Its moderate solubility in water and strong solubility in organic solvents reflect the balance of aromaticity and amine functionality. The ethyl group in the ortho position alters reactivity when compared to unsubstituted aniline, steering chemical reactions toward unique products.
Technical Specifications & Labeling
The commercial bottles of 2-ethylaniline often state assay numbers near 98% or higher and limit impurities such as moisture and related amines. Manufacturers use labels with CAS number 578-54-1, clear hazard symbols, and handling instructions in accordance with local and international guidelines. Strong labeling of hazards reflects the consensus that this compound requires attentive storage—cool, dry, and isolated from oxidizing agents.
Preparation Method
Classic methods for making 2-ethylaniline go back to the alkylation of aniline using ethyl halides under strong basic or catalytic acid conditions. Another option is the reduction of 2-nitroethlybenzene, first built by electrophilic substitution of ethylbenzene followed by nitration, and then hydrogenation using standard catalysts like palladium on carbon. Modern refinements scale these approaches by improving selectivity, cutting wastes, and steering clear of old hazards, such as overuse of mineral acids and dangerous side-products.
Chemical Reactions & Modifications
2-Ethylaniline takes part in a range of reactions, thanks to its amine donor and ortho-ethyl partner. Acylation transforms it into substituted amides, offering routes to medicinal chemistry and peptide synthesis. Diazotization brings out the full potential of aromatic amines, turning 2-ethylaniline into diazonium salts—the launching pads for azo-dye creation, aryl coupling, and halogenation. N-alkylation and sulfonation deliver further diversity, feeding downstream needs in pigment and polymer chemistry. Its reactivity is a playground for anyone studying electronic effects or exploring new types of molecular scaffolding.
Synonyms & Product Names
2-Ethylaniline appears under several labels, such as o-ethylaniline, 2-Aminoethyltoluene, Benzenamine, 2-ethyl-, and Ethaniline, 2-ethyl-. Industry catalogs and safety data sheets sometimes use these names interchangeably, but the structure always points back to the amine group at the ortho position relative to ethyl on the benzene ring. Awareness of synonyms helps when searching academic publications and global regulatory databases for information or compliance data.
Safety & Operational Standards
Working with 2-ethylaniline demands attention. Its aromatic amine structure has connections to health hazards, especially on chronic exposure. Short-term impacts include skin and eye irritation, and breathing in its fumes can cause headaches or discomfort. Evidence from studies on aromatic amines raises red flags about long-term carcinogenicity—even though definitive links for every derivative vary, responsible labs always treat spills, inhalation, and skin exposure seriously. Safe use depends on personal protective equipment, local ventilation, and prompt cleanup of any escaped material. Regulatory rules set exposure limits, require chemical-resistant gloves, splash goggles, and mandates for containment during transport and storage. These aren’t just checkboxes; they stand between staff and possible health consequences.
Application Area
Dye developers draw on 2-ethylaniline for its ability to form vibrant azo compounds, throwing sharp, lasting colors onto fabrics and plastics. Some drug molecules feature this scaffold, using the ethyl group to fine-tune biological interactions. Agrochemical developers also favor this template in the pursuit of efficient, selective crop protectants. Some of my peers in chemical engineering have pushed this compound into high-performance specialty resins, aiming for thermal resistance or unique electronic properties. Each application taps into a specific trait—reactivity, solubility, or structural contribution to a final product’s performance.
Research & Development
The academic world continues to wrestle with the electronic subtleties of 2-ethylaniline, especially in the hunt for greener synthesis routes and novel materials. Teams publish papers on selective C-H activation, for instance, hoping to build new derivatives in fewer steps and without relying on heavy-metal catalysts. Computational chemistry groups model the compound’s electron cloud, searching for activation energies and reaction pathways that beat current methods. Industrial R&D seeks to reduce waste, improve yields from crude feedstocks, and shrink the environmental footprint of manufacturing. This drive for innovation stems not only from competitive advantage but also from tightening environmental standards and public concern over chemical waste and toxicity.
Toxicity Research
Studies on 2-ethylaniline raise caution. Aromatic amines get a lot of attention for their links to methemoglobinemia—a condition where the blood can't carry oxygen efficiently. Chronic exposure, especially in workplaces without proper safety measures, raises the risk of disruptions to blood chemistry. Animal studies and short-term bioassays reveal routes of absorption, metabolism, and excretion, informing workplace safety guidelines. Regulators watch these numbers closely. The quest for lower hazard profiles spurs research into exposure thresholds, breakdown products, and less toxic structural analogs, responding to data showing that even minor changes in molecular structure lead to big differences in human health effects.
Future Prospects
The future of 2-ethylaniline looks dynamic. As green chemistry matures, demand rises for cleaner production and better process safety. There’s a strong current of interest in replacing hazardous solvents and reagents with benign options, cutting waste and exposure. Smart manufacturing and flow chemistry open up faster, more controllable syntheses, leading to cheaper and safer supplies for researchers and industry. Rising needs in electronics, dyes, and specialty chemicals hint at more engineered derivatives coming down the line—each pursuing performance, safety, or environmental improvement. Ongoing investment in health and toxicity studies keeps manufacturers and regulators on their toes, balancing innovation against safety in labs and on factory floors.
What is 2-Ethylaniline used for?
An Essential Ingredient in Chemical Synthesis
2-Ethylaniline stands out in chemistry labs and factories thanks to its basic amine structure paired with a subtle nudge from an ethyl group. This little tweak turns an otherwise common aromatic amine into a building block that chemists reach for. Labs use it when they’re piecing together dyes or pharmaceutical intermediates. As I’ve seen, reactions run cleaner and final compounds hold up to higher scrutiny when quality raw materials like 2-ethylaniline come into play. In dye factories, skipping on such intermediates leads to weak colors and batch inconsistency. For everyday items relying on vivid, colorfast dyes—think black t-shirts and food labels—a reliable source of 2-ethylaniline keeps product quality up.
Dye Industry’s Workhorse
Colorants soak through our world, from the blue jeans in your drawer to the plastics in your car. 2-Ethylaniline finds its way into the synthesis of azo dyes. It reacts readily, letting dye-makers tweak molecular structures for brighter reds and yellows. The history of aniline-based dyes goes back to nineteenth-century Europe, yet today’s shades owe their resilience in part to industrial chemicals like this one. Without them, fabrics would fade, paints might streak, and printed material would lose its crisp look long before wear and tear took their toll.
Stepping Stone to Medicines
Many pharmaceutical ingredients have roots in aromatic amines. 2-Ethylaniline isn’t an exception. Drug discovery relies on molecules that fit snugly into biological pathways, sometimes blocking, sometimes boosting, a signal in the body. Subtle changes make all the difference—a bit of ethyl here, a tweak there. Chemists use 2-ethylaniline to create small variations as they hunt for new treatments. This flexibility helps companies tackle painkillers, blood pressure medicines, or antifungals, all starting from the same basic backbone.
Link to Agricultural Chemicals
Anyone who works near fields or reads up on food safety knows how crucial crop protection has become. Weeds and pests adapt, so so do the chemistries we use to protect food supplies. Compounds built from 2-ethylaniline form the basis of some herbicides and fungicides, letting farms get better yields. Here, purity and reliability matter again, as regulators and scientists do not want unwanted byproducts lingering on produce. Strong starting chemicals mean safer, more predictable solutions for the food chain.
Risks and Playing It Safe
Handling industrial chemicals means respecting the risks. From my own lab days, gloves, goggles, and a good fume hood were standard when working with aromatic amines. Reports from regulatory bodies have pointed out the possible health hazards, such as skin irritation or potential toxicity from long-term exposure. Responsible producers and chemical firms stick to strict safety protocols to limit spills and airborne release. Regular training, good ventilation, and up-to-date equipment are the best defenses—including for those who live nearby manufacturing sites. Industry trends show a growing push for greener, safer processes and substitutes, but for now, careful use keeps workplaces and products safe.
Looking Forward: Smarter Solutions
Innovators in chemical manufacturing have started shifting towards more sustainable production. From renewable feedstocks to better waste treatment, the future for chemicals like 2-ethylaniline is not just about what they can build, but how cleanly and safely companies can do it. More robust oversight, smarter engineering, and industry transparency can keep hazards in check. Consumers deserve products that are both effective and safe—from the medicines in the cabinet to the colors on your clothes. Building safer supply chains starts here, with the ingredients at the very beginning.
What is the chemical formula of 2-Ethylaniline?
What 2-Ethylaniline Tells Us About Chemistry in Daily Life
Chemistry likes to look like a secret code. You see a name like 2-Ethylaniline and it might not mean much unless you’ve spent time with textbooks, safety goggles, or a lab coat. But behind a name, there’s a story about structure and function, especially with compounds that show up across industries and labs. The chemical formula for 2-Ethylaniline is C8H11N. This combination isn’t pulled straight out of thin air; it’s a map of how carbons, hydrogens, and nitrogen tie together to shape what happens next.
The Structure Shapes the Use
2-Ethylaniline comes from a group of molecules called aromatic amines. You get a benzene ring, which is a hexagon formed by six carbon atoms bonded together, each with alternating double bonds, plus one nitrogen-based amine group, and for this particular version, an ethyl group (two carbons, five hydrogens) sitting on the second spot of the ring. That little change—moving the ethyl group to the “2” position—creates properties different from other isomers. Structure lays the rules of reactivity and safety.
Importance Beyond the Classroom
A formula isn’t just for exams or brewing up a reaction in class. 2-Ethylaniline plays a real role out in the world. You find it in dye production, medicine synthesis, and sometimes as an intermediate in making pigments or certain agricultural chemicals. The ethylaniline family gets a job done that simpler anilines can’t handle, all because altering a carbon here or there creates unexpected abilities. Knowing the formula gives engineers, chemists, and environmental safety officers a clue about handling, transporting, and repurposing the material.
Health, Environment, and Responsibility
Working with aromatic amines isn’t without risk. Even one stray carbon moving around a molecule can change how it interacts with your body, or how it persists in soil or water. Understanding 2-Ethylaniline's formula—C8H11N—helps build safety protocols and develop better workplace rules. Over the years, research has tied certain anilines to health concerns, so clear knowledge about structure and reactivity isn’t just academic trivia; it forms the backbone of safety standards.
Education and Transparency
The way we teach chemical formulas often feels cold and remote. If students, technicians, or regulators approach these formulas as the roadmap to understanding what a chemical can do, communication improves. Workers learn what personal protective equipment makes sense. Regulatory agencies draft better guidance. Industry finds safer ways to recycle or break down old chemicals instead of dumping them. These changes rest on clarity—everyone in the chain benefits from knowing a chemical’s basic formula because it sets expectations for every next step.
Better Practices Through Knowledge
If chemistry shows up in the real world, it’s rarely abstract. Waste management, drug development, or color fastness in textiles all pull from the molecular building blocks that formulas reveal. For 2-Ethylaniline, those eight carbons, eleven hydrogens, and one nitrogen tell experts and learners what to watch for, what to expect, and how to adapt in the face of evolving science and regulation.
Is 2-Ethylaniline hazardous or toxic?
What We Know About 2-Ethylaniline
Working in a chemistry research lab years ago taught me that the real world doesn’t always stay in the textbook: solvents spill, fumes drift, and sometimes even small exposures leave a mark. 2-Ethylaniline often crops up in industrial settings, especially where dyes, plastics, and pharmaceuticals come to life. This molecule, an aniline derivative, can’t just be chalked up as another chemical on the shelf—it carries real risks if not handled with respect.
Health Effects: Real Concerns, Real Stories
Breathing in 2-ethylaniline fumes or letting it soak through the skin can make you dizzy, cause headaches, or bring on nausea. A few years back, a contractor I knew dealt with a spill during a pigment manufacturing job. He developed irritated skin and struggled to focus for the rest of the week. There’s a good reason behind all those warning labels in such factories—the stuff can trigger allergic reactions and mess with your blood’s ability to carry oxygen. Long-term exposure risks are more troubling. People who handle aniline compounds regularly can develop methemoglobinemia, which makes oxygen transport in the bloodstream much harder. Scientists have raised concerns about potential links to certain cancers, and lab tests show that chronic contact with aromatic amines contributes to organ damage.
Why Regulations Matter
Authorities like OSHA and the European Chemicals Agency keep 2-ethylaniline on their watch lists for a reason. Regulations set strict exposure limits and call for basic protective gear: gloves, respirators, goggles, proper ventilation. Following these rules isn’t just about liability. During one summer internship, I saw a co-worker ignore the guidelines on chemical gloves. After a month, he developed a persistent skin rash that took weeks to clear—a constant reminder for all of us. Safety is more than compliance; it’s about coming home as healthy as you left.
Environmental Impact: Out of the Barrel, Into the World
Making, using, or losing track of 2-ethylaniline doesn’t just hurt the worker. Pouring effluent containing this compound into water sources spells danger for fish and other wildlife. Small aquatic creatures are especially vulnerable to these aromatic compounds. Some local rivers near chemical plants show lower biodiversity where leaks or illegal dumping have taken place. Once this type of chemical leaks into the soil, it doesn’t break down quickly and can enter groundwater, making cleanup much more difficult and expensive for entire communities.
Better Habits, Safer Outcomes
The science is pretty clear: respect the hazards, and the risks drop. Workers and managers in chemical or dye plants need ongoing safety training, not just a safety sheet posted in the break room. Companies can invest in better ventilation or closed handling systems instead of open containers. Technological solutions like vapor detection systems or spill sensors can spot problems before they hurt people or the environment. At home, most folks won’t ever run into 2-ethylaniline, but staying informed about the chemicals in nearby factories and what kinds of emergencies could rise matters to every community. Change doesn’t always happen fast, but every smarter choice adds up in the long run.
How should 2-Ethylaniline be stored?
Why Storage Choices Matter for 2-Ethylaniline
If you spend a few years in any industrial or research lab, you come to know chemicals are only as safe as the rules guiding their storage and use. 2-Ethylaniline often finds its way into dyes, pharmaceuticals, and sometimes more specialized materials. While handy, it brings risks that demand respect. Long-term safety doesn’t hinge on luck—it follows good habits, honest vigilance, and smart organization.
Straightforward Rules for 2-Ethylaniline Storage
2-Ethylaniline gives off a strong odor, and even a small spill lingers in the air. This should remind anybody nearby: keep the container tightly sealed after each use. One afternoon, I watched an entire wing of a lab cleared because someone put a lid on loosely after weighing out their batch. The results stuck around long after the mess was cleaned.
Find a cool, dry spot away from any heat sources. This isn’t just about following safety data sheets. 2-Ethylaniline withers in strong light or if it sits anywhere hot—fumes grow stronger, and the risk for fire or health harm rises. Keep it away from direct sunlight and never leave it sitting on a window ledge or anywhere ovens or heaters heat up the room.
Don’t trust regular shelving if you want to avoid headaches. Chemicals like this belong in fire-resistant cabinets, preferably the yellow ones marked for flammable liquids. These cabinets stay locked unless they’re needed. Someone, somewhere, always tries to tuck a bottle on a top shelf to make extra bench space. Over time, bottles tip, leaks start, and then safety routines go out the window.
Why Labeling and Compatibility Count
A clear label with the exact chemical name, hazard symbols, date received, and the name of the person responsible makes all the difference. I’ve come back after holidays to see sticky notes barely hanging on serious chemicals. Proper printed labels help everyone—especially new staff or visiting maintenance workers—know what’s inside.
Never mix amines with oxidizers, acids, or strong bases. Stash different chemical families apart to cut down on accidents. Aniline derivatives react fast with the wrong neighbor; if 2-Ethylaniline shares a shelf with bleach or nitric acid, the results won’t end well.
Personal Stories from Mishandling
I once saw someone stack leftovers from a late-night experiment in the chemical fridge next to a leaking bottle of acid. By the next morning, the shelf liners had started breaking down and the fridge stank. Mistakes like that don’t just cost money—they put lives and research in danger, too. The most serious labs run weekly checks to catch rogue storage before it snowballs into a problem.
Daily Practices That Pay Off
Gloves and goggles are basic, but easy to forget when someone rushes. Keep a spill kit handy near the storage cabinet—absorbent pads, neutralizers, and a dedicated disposal bin reduce panic if a jar tips.
Routine goes a long way. It’s tempting to shrug off detailed record-keeping for “just another aromatic amine,” but experience teaches that old chemicals degrade and may build up pressure inside containers, leading to stubborn leaks that become dangerous.
The routines may feel like chores, but genuine safety in labs—homes for many of us—grows behind the scenes, one smart habit at a time.
What are the physical and chemical properties of 2-Ethylaniline?
2-Ethylaniline and Its Appearance
2-Ethylaniline stands out as a colorless to pale yellow liquid with a noticeable, pungent odor. Anyone who has worked in a basic chemical lab, or walked through an industrial facility, can recognize that aroma pretty quickly—it signals the presence of aromatic amines. Its oily feel isn’t surprising, given the ethyl group attached to the benzene ring, which adds a slight greasiness when compared to plain aniline. Dropping a bit on glass or metal often leaves a persistent film, giving away its stubborn, sticky nature.
Solubility and Reactivity Details
This compound doesn’t play well with water, dissolving just a little. In contrast, it’s a lot more comfortable in organic solvents like ethanol and ether. Lab workers often reach for these solvents when trying to clean up or dilute the substance. This low water solubility means accidental spills rarely run straight to the drain; they tend to linger and call for careful containment.
Chemically, 2-ethylaniline’s benzene ring packs a double punch. With both an amino group and an ethyl group, it’s reactive at the ortho and para positions on the ring. It undergoes typical aromatic substitution with surprising speed. Those working in synthetic labs can count on this property when making dyes or pharmaceutical precursors. Controlled reactions with acids lead to salts, while strong oxidizers break it down quickly—contributing to both its usefulness and its hazards.
Boiling, Melting, and Storage Insights
For folks in chemical plants, handling comes down to temperature as well. 2-Ethylaniline boils at around 210°C. Anyone who heats up this compound in glassware—or checks temperature during distillation—knows the importance of proper ventilation and temperature control. Its melting point, around -15°C, means it stays liquid at standard room conditions. If you work somewhere cold, you might find it thickening up but rarely freezing.
With a flash point not much higher than boiling water, storage turns into a real safety concern. Even experienced chemists double-check their storage tanks to avoid fire risks. Proper containment, decent airflow, and keeping ignition sources away turn into basic steps rather than afterthoughts.
Toxicity and Real-World Health Risks
No one in a lab wants to breathe in 2-ethylaniline or get it on their skin. Contact can irritate, sometimes cause sensitization, and even lead to more severe health issues with repeated exposure. It absorbs through the skin more quickly than you’d expect. The body metabolizes aromatic amines slowly, so chronic, low-level exposure can lead to lingering health risks. Workers need gloves, goggles, and a good fume hood. I once worked at a facility where a minor spill led to headaches and discomfort, reminding everyone that basic safety practices are never optional.
Potential Solutions and Industry Practice
Efforts should go into engineering controls—better fume extraction, spill containment plans, and clear labeling in shared storage. Substituting safer reagents for routine operations, where possible, helps lower risk to workers and the environment. Clear instruction and regular training keep otherwise routine handling from turning disastrous. It’s not about avoiding chemicals like 2-ethylaniline, but about building safer spaces so chemists and technicians can work with confidence and minimal long-term exposure.
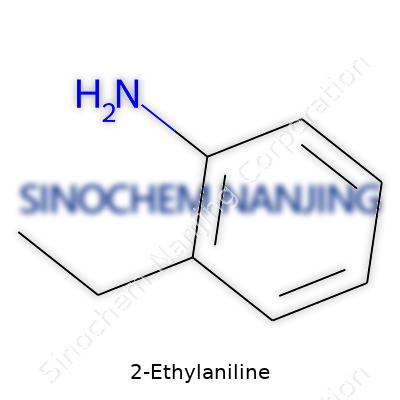
| Names | |
| Preferred IUPAC name | 2-Ethylaniline |
| Other names |
2-Aminoethybenzene o-Ethylaniline 2-Ethylbenzenamine |
| Pronunciation | /tuː ˌɛθ.ɪl.əˈnɪl/ |
| Identifiers | |
| CAS Number | 578-54-1 |
| Beilstein Reference | 607793 |
| ChEBI | CHEBI:50268 |
| ChEMBL | CHEMBL16215 |
| ChemSpider | 10966 |
| DrugBank | DB14265 |
| ECHA InfoCard | InChIKey=MBEKDGZQJUROIY-UHFFFAOYSA-N |
| EC Number | 202-424-3 |
| Gmelin Reference | 822741 |
| KEGG | C01881 |
| MeSH | D000463 |
| PubChem CID | 7466 |
| RTECS number | BZ9625000 |
| UNII | 2X6UO1XE5K |
| UN number | UN2273 |
| Properties | |
| Chemical formula | C8H11N |
| Molar mass | 121.18 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | aromatic |
| Density | 1.008 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.96 |
| Vapor pressure | 0.32 mmHg (25°C) |
| Acidity (pKa) | 4.5 |
| Basicity (pKb) | 10.67 |
| Magnetic susceptibility (χ) | -64.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.578 |
| Viscosity | 2.21 mPa·s (25 °C) |
| Dipole moment | 1.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 131.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3470.2 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H312, H315, H319, H332, H351 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P301+P312, P302+P352, P304+P340, P312, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | Flash point: 98 °C |
| Autoignition temperature | 615°C |
| Explosive limits | 1.3–7% |
| Lethal dose or concentration | LD50 oral rat 2050 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2025 mg/kg (oral, rat) |
| NIOSH | KN3325000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | Process chemicals, Intermediates |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
2-Nitroethaniline 2-Aminotoluene 2-Ethylbenzenesulfonic acid 3-Ethylaniline 4-Ethylaniline |

