2-Ethyl-1-Butene: Beyond the Laboratory
Historical Development
Chemistry always leans on stories that stretch back several generations. 2-Ethyl-1-butene started to shape commercial chemistry as petrochemical production ballooned in the twentieth century. Once crude oil refining technologies matured, olefins like 2-ethyl-1-butene began arriving in bulk. Refiners saw early on how branched alkenes could replace older raw materials, especially as demand for high-octane fuels swelled after World War II. By the sixties, synthetic lubricants and additives needed new building blocks, and this molecule landed on the shortlist. Today, heavy industry depends on it because past engineers spotted its utility and charted ways to make it at scale.
Product Overview
This clear, colorless liquid rarely makes headlines, but most people use something that traces back to it. Petroleum refineries crank it out, then specialty chemical producers funnel it into products from polymer modifiers to syntheses that demand accessible double bonds. On the scale of global chemistry, 2-ethyl-1-butene stands out as a vital intermediate, often coming in steel drums or bulk tankers, depending on market needs.
Physical & Chemical Properties
The molecular formula C6H12 reflects a backbone with real character: six carbon atoms, a double bond most often at the end, plus an ethyl branch that locks in some quirky behavior. Typically, 2-ethyl-1-butene boils around 63-65°C, and the flash point sits near -23°C, which spells out flammability concerns that everyone handling it must respect. The faint, sweetish odor would ring a bell for anyone who has spent time in a refinery. Most users prize its reactivity at the double bond, which speeds up addition reactions or modification with acids, halogens, or peroxides.
Technical Specifications & Labeling
Commercial shipments get labeled with purity grades, usually tracking residual isomers and lighter alkenes, since small impurities can derail downstream synthesis. Buyers look for specifications like minimum 98% purity, total sulfur below detection limits, and moisture under 100 ppm. Labels carry UN numbers for flammable liquids, plus hazard statements driven by international chemical transport protocols. Every container comes with batch numbers and production site codes so traceability never slips, which helps a lot when inspecting supply chains or investigating any problem batches.
Preparation Method
The most common process starts with oligomerizing ethylene, sometimes leveraging nickel or titanium catalysts that encourage branching right at the first step. Next, hydroformylation and other organometallic routes help fashion the short-chain, branched skeleton. Refineries also can forget a path via isobutene alkylation with ethylene, chasing yield with tweaks in pressure, temperature, and catalyst support. Even small shifts in those levers promise big payoffs, so process engineers keep countless logs and pilot batches humming.
Chemical Reactions & Modifications
Chemists reach for 2-ethyl-1-butene thanks to the double bond that opens doors. Hydration gives alcohols—key in plasticizer or resin production. Halogenation introduces chloro or bromo groups, tuning building blocks for agrochemical synthesis. Polymerization routes let manufacturers feed its carbon skeleton right into specialty elastomers or high-end lubricants. Oxidative cleavage or epoxy modifications grow all kinds of downstream products, pushing the molecule from simple hydrocarbon toward bespoke compounds with market value.
Synonyms & Product Names
2-Ethyl-1-butene sometimes comes under familiar aliases in old reports or supplier lists. Folks who worked in plants or labs a generation ago might mention Ethylisobutylene or 2-Ethylbut-1-ene. Some European documentation calls it α-Butylene, 2-ethyl. No matter the name, suppliers rely on CAS number 3334-83-6 for unambiguous orders, since translation across languages or regions can jumble project communications.
Safety & Operational Standards
A liquid eager to vaporize and burn demands respect. Industrial sites lock in explosion-proof pumps, retrofit loading bays with vapor recovery, and train crews to avoid any static discharge. Personal protective gear—sometimes respirators, always gloves—keeps exposure below established limits. The acute health risks come mainly from high vapor concentrations: headaches, dizziness, or worse if accidents disrupt containment. Any spill drills focus on keeping vapors from reaching ignition sources, and even small leaks prompt immediate cleanup. Modern buyers look for suppliers who keep safety training up-to-date and maintain emergency response teams drilled on their sites.
Application Area
Few consumers realize how often 2-ethyl-1-butene powers modern manufacturing. In lubricant plants, it delivers the backbone to synthetic esters that keep jet engines running clean at high temperatures. Coatings factories turn it into resins with specific flexibility and cure profiles, sometimes feeding it into automotive finishes that handle road salt or UV without flaking. Some pharmaceutical intermediates start with this branched alkene, using careful chemical steps to protect sensitive functional groups. As an intermediate, it enters performance chemicals unseen by the public eye, but chemists know the chain wouldn’t run without reliable sources on hand.
Research & Development
People working at chemical companies watch for improvements in catalyst life or conversion efficiency, as even minor enhancements stack up over months in continuous operation. Some recent patents describe new catalysts that sharpen selectivities, reducing need for final product distillation. As sustainability pressure heats up, companies invest in processes that squeeze out more product while generating less waste. Green chemistry labs probe bio-based feedstocks or recyclable catalysts, hoping to break dependence on fossil fuels, but scaling lab tricks to commercial outputs always takes time and funding. Networking with universities or tech start-ups lets established firms pull in new ideas faster and balance risk.
Toxicity Research
Anyone tasked with worker safety management takes toxicity data seriously. Short-term exposure studies show irritation at inhalation levels above safe thresholds, and literature surveys point out that long-term effects remain under-explored. Older animal studies indicated low acute toxicity, yet chronic exposure—especially in combination with other hydrocarbons—remains a topic needing solid research. Tracking metabolites in exposed test subjects showed limited bioaccumulation, but regulatory agencies prefer more robust evidence, so global standards remain guarded. Health physicists and industrial hygienists look for continued surveillance in facilities, drawing on real-time sensor data when available.
Future Prospects
Branched alkenes like 2-ethyl-1-butene play a critical role for anyone plotting out next-generation fuels, plastics, or specialty chemicals. Some R&D teams hope to build drop-in equivalents from renewable sources, a goal that depends on bio-catalysts robust enough for industrial cycles. As stricter emission rules sway procurement, buyers put pressure on suppliers to cut losses and recover more product per kilogram of input. A shift toward high-throughput experimentation, paired with machine learning for process optimization, signals concrete shifts ahead. If global supply chains continue to absorb renewable carbon, synthetic routes to 2-ethyl-1-butene could pivot to feedstocks outside the oil barrel. Markets rewards those who update infrastructure and rethink long-term risks along with short-term profits. So, for all its modest appearance, this compound could lay groundwork for much broader sustainability shifts in the industry.
What are the main applications of 2-Ethyl-1-Butene?
Fueling Innovation in Chemistry
Walk into any modern petroleum refinery, and you’ll find all sorts of specialty chemicals. Among these, 2-Ethyl-1-Butene plays a quiet but important role. It’s a clear, colorless liquid with the kind of double bond chemists love to experiment with. I’ve watched teams in the lab harness its reactivity for a range of synthesis projects, turning a simple molecule into something vital for many industries.
Production of Lubricant Additives
Every car engine, whether in Los Angeles traffic or hauling freight across the Midwest, counts on lubricants to keep moving. 2-Ethyl-1-Butene shows up early in the production process for these engine oils. It helps create alkylated phenols and other compounds that improve how oils work under heat and pressure. The big benefit? Engines last longer, need less maintenance, and pump out fewer emissions. Long-haul truckers and city drivers both notice when oils break down too fast or form sludge—2-Ethyl-1-Butene helps guard against that.
Boosting Polymer and Plastics Manufacturing
Plastic manufacturers often look for building blocks that can handle a heavy workload. 2-Ethyl-1-Butene works as a comonomer in making specialty polyolefins. These aren’t the cheap plastics you toss after one use. Think about sturdy consumer goods, reliable packaging, and medical tubing that takes a beating without falling apart. During my time working alongside polymer researchers, I saw how a tweak in the starting materials, such as adding a molecule like 2-Ethyl-1-Butene, changes a product’s feel and strength. I’ve held prototype plastics that stood up to repeated twisting and stretching compared to their brittle, outdated cousins.
Creating Fragrance Intermediates
Synthetic fragrance companies always seek efficient, safe chemicals to fill out their palette of scents. 2-Ethyl-1-Butene steps in as a precursor in the production of aroma compounds. It doesn’t smell much like anything on its own. Still, after a couple of steps in the reactor, the molecules spin into synthetic musks and fresh, persistent notes used in laundry detergents and air fresheners. As someone who breaks down ingredient panels for fun, I keep spotting the fingerprints of this compound in everyday consumer staples.
Facilitating Pharmaceutical Synthesis
It may not end up in your medicine bottle, but 2-Ethyl-1-Butene often helps build more complex pharmaceutical molecules. Medicinal chemists need clean, reactive starting materials to reduce byproducts and inefficiency. I’ve worked with teams who use alkene chemistry to create intermediates that get shaped into antivirals, anti-inflammatories, and other treatments. Without reliable raw materials such as this, the cost and time for drug development would shoot up.
What Challenges Do We Face?
Every industrial chemical brings questions about safe use and environmental impact. 2-Ethyl-1-Butene requires careful handling due to its reactivity and flammability. Facilities using it invest in vapor controls and fire prevention. Firms also explore bio-based routes and greener production methods, aiming to use less energy and generate less waste. As someone who’s seen the cost of cleanup firsthand, I believe tougher standards and stronger oversight make a real difference in protecting workers and neighborhoods.
Looking Forward
The applications for 2-Ethyl-1-Butene keep expanding as manufacturers look to improve performance and create more sustainable products. Whether it’s a longer-lasting motor oil, a safer medical device, or a less wasteful chemical process, this compound keeps earning its place in the toolkit. Industry insiders and outsiders alike should keep an eye on the legacy of innovation built from small but mighty molecules.
What is the chemical formula and structure of 2-Ethyl-1-Butene?
Chemical Formula
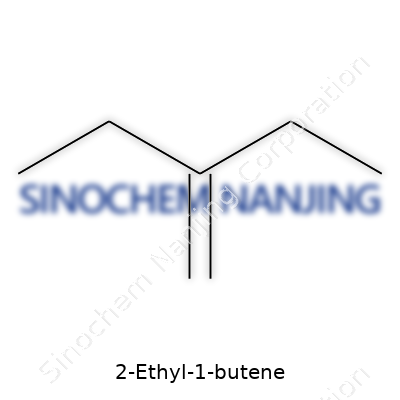
2-Ethyl-1-butene packs its atoms into the formula C6H12. Six carbons and twelve hydrogens team up to form this hydrocarbons family member. For folks who work in labs or enjoy a bit of backyard chemistry, it belongs to the alkene family. That double bond always changes how a molecule acts. In its name, “butene” hints at four carbons in the longest chain, and the “2-ethyl” bit points to a branching sidekick off the second carbon. This name carries all the info about how the molecule is pieced together.
Structural Layout
Picture the main backbone as a straight chain of four carbons. Attach two hydrogens to the first carbon and stash a double bond between carbon one and two, which introduces reactivity at that point. An ethyl group—two carbons long—hangs from the second carbon in that chain. Lay it out and you’ll see: H2C=CH–CH(C2H5)–CH3. That extra carbon chain swinging from the middle changes pretty much everything about how this molecule mixes or reacts with other chemicals.
Relevance in Industry
In my early days scrubbing beakers and running small-batch reactions, 2-ethyl-1-butene kept cropping up on order sheets. This wasn’t just because folks loved its name. It’s useful as a starting ingredient for making lubricants, plasticizers, and specialty chemicals. Those branches not only jazz up the molecule—they give processed materials more stability and flexibility. You’ll find traces of this compound’s handiwork in synthetic motor oil, making engines run smoother and last longer.
Producers favor this compound for alkylation reactions, where it becomes the backbone for many high-octane fuels. Refineries blend it to boost gasoline’s quality or turn it into alcohols and detergents. That double bond acts like an open door for other molecules, letting chemists attach all kinds of useful groups during production. This versatility hooks into a wide range of applications outside the lab: things we use every day, from the plastics in household goods to the solvents in detergents.
Health and Safety Questions
Like a lot of alkenes, 2-ethyl-1-butene doesn’t exactly mingle well with lungs or skin. Encountering the sharp, somewhat sweet scent in a workroom, I learned early to never skip proper ventilation. Breathing in too much vapor leads to headaches or dizziness. Long sleeves and gloves aren’t optional. Spills and leaks call for fast action—most companies keep tight rules on storage to keep workers safe and reduce risks of fire or chemical burns. Its flammability isn’t a small concern, either. One spark near a loose drum can spell big trouble, especially given its low flash point. Being careful with storage temperature and using spark-proof tools makes a world of difference.
Potential for Greener Chemistry
Newer production tech leans toward fewer emissions and smarter waste management. In my own work with process teams, we swapped out harsh catalysts for milder, recoverable ones. Teams now look for ways to recycle leftover streams or recover unused alkene for new batches. Other solutions involve containment and fume handling upgrades, keeping both workers and air quality in better shape. Research always chases after biodegradable alternatives or just cleaner recipes for making the same useful molecules—proving that small tweaks in chemical structure and production routes can ripple through into a safer, more sustainable industry.
What are the storage and handling recommendations for 2-Ethyl-1-Butene?
What Experience Teaches Us
Working around chemicals like 2-Ethyl-1-Butene means respect and clear routines matter more than fancy science language. If you’ve spent some time in a warehouse or blending facility, you've seen how fast things can go wrong with flammable liquids. I remember back in a smaller plant, a loose cap on a drum led to a lingering smell no one could ignore. That day, we shut the place down for hours. Nobody wanted to risk a spark. This sticks with you.
Ventilation Isn't Just for Comfort
2-Ethyl-1-Butene is an alkene with a pretty low flash point — around -42°C. Storing it in a cool, well-ventilated space keeps vapors from collecting. Most workers don’t always notice vapors until someone complains of a headache or dizziness. What we learned early: if you smell it, ventilation isn’t doing its job. Reliable exhaust fans and regular air checks pound that lesson home.
Temperature and Sunlight
Leaving drums near a loading dock, open to summer sun, always seems easy until pressure swells make a mess or create a hazard. These drums want shade and steady, low temperatures. In many shops, that means picking the coolest aisle, away from windows, with rigid routines for temperature checks. Fluctuating temps stress containers, and that’s what leads to leaks.
Fire Risk: Don’t Cut Corners
Flammable chemicals demand a respect for grounding — from metal drum racks to static-resistant scooping tools. Early on, a co-worker learned the hard way; a plastic scoop caused a small spark that charred a drum label. No one got hurt that day, but every safety meeting after got a bit more serious. Proper signage, up-to-date fire extinguishers, and grounded drums now shape daily routines in any responsible facility.
Container Details Make the Difference
You see a lot of variety in equipment. In bigger plants, 2-Ethyl-1-Butene often gets stored in sealed steel drums or specialized tanks. Labels get printed right, seals checked twice, and everyone working the line must know which wrench fits the bung cap. Those practices save headaches down the road. Leaky caps or old gaskets never get ignored.
Training and Emergency Readiness
All the written rules in the world sink fast if people skip safety drills. Whether in a new spot or the same job you’ve held for years, the right response to a spill gets drilled in. If material splashes, it isn’t just a wash-and-go moment — people need protective gear and quick cleanup with absorbent pads or sand, plus well-marked disposal bins. The goal is to keep incidents small and predictable.
Straightforward Solutions
Routine inspection of storage areas and containers keeps surprises away. Investing in proper training saves money — and time lost from unexpected shutdowns. Good lighting and uncluttered walkways matter, too. In every operation I’ve worked, simple, steady routines matter more than complicated new tech or fancy buzzwords.
Wrapping Up Personal Lessons
Every chemical tells its own story. 2-Ethyl-1-Butene rewards respect and consistency. Safety depends on honest routines, clear communication, and gear that does its job. All that prevents drama nobody wants to see on the evening news.
Is 2-Ethyl-1-Butene hazardous and what safety precautions should be taken?
The Real Risks of 2-Ethyl-1-Butene
2-Ethyl-1-butene is more than just a name on a chemical drum. This clear liquid, widely used as an intermediate in synthetic manufacturing, packs its own set of risks. Most people never handle it in daily life, but for those of us working in labs, refineries, or chemical plants, knowing how it behaves can make all the difference. The experience of a close call in a student lab showed me how easy it is to underestimate common substances until you get that sharp vapor in your nose or a warning flare on a detector.
This material smells faintly sweet, which gives an early warning if it's in the air, but that’s no reason for complacency. Like many low-weight alkenes, 2-ethyl-1-butene catches fire fast, even from sparks or static. The vapors hug floors and can travel a surprising distance, creeping unnoticed until meeting an ignition source. Once, I watched a small spill create panic as vapors found a pilot light across the room. Flames raced back in an instant. That memory stick with me, and I read about similar incidents in accident databases: not rare at all. There’s real firepower in these vapors.
Health Hazards: More Than Immediate Effects
Even without a fire, trouble lurks. Breathing in 2-ethyl-1-butene can irritate the throat and lungs. Extended exposure or high concentrations overwhelm your nose and chest, causing coughing and headaches. Several studies suggest long-term inhalation could harm the nervous system, though evidence is less clear-cut than for other industrial solvents. Nobody needs a headache or lingering cough just for doing their job. I’ve seen co-workers underestimate this—until discomfort forced them outside for air.
Skin contact doesn’t give much warning, either. It may not burn right away, but it strips oils, leaving skin dry, cracked, or red. Splashing eyes brings pain and can damage vision. Even a small mistake—wiping sweat from a brow with a contaminated glove—can leave a lasting stinging reminder.
Safety Practices That Matter
Solid workplace safety relies on more than just posters on the wall. From experience, gloves made of nitrile or neoprene block most splashes—latex offers less protection with many hydrocarbons like this one. Always wear goggles or a face shield when transferring or pouring. Good ventilation isn’t just a guideline; local exhaust hoods or reliable open airflow make sure vapors never hit dangerous levels. Monitors and alarms catch what your nose misses.
Static discharge can turn a routine transfer into an emergency. Grounding metal containers and using non-sparking tools lower the odds. Working alone increases the risk—I always make sure someone knows what I’m doing in case something goes wrong. Labeling every container and keeping storage away from heat or open flames helps prevent small mistakes from spiraling out of control.
Solutions for Safer Handling
Switching to less volatile or less flammable alternatives can cut out the worst risks, but that's not always possible. Routine drills on handling spills, using absorbents, and knowing evacuation routes make a difference. I’ve seen people freeze up in emergencies from lack of practice—regular hands-on practice builds confidence. Clear communication between supervisors and staff helps catch changes or problems early. Safety gear means little if it’s left on a shelf or used improperly, so keeping it cleaned and ready helps everyone.
Investing in these steps costs less than dealing with fires, medical visits, or lost productivity. Protecting people—myself included—is reason enough to take 2-ethyl-1-butene seriously every single day.
What is the purity specification for commercial 2-Ethyl-1-Butene?
The Numbers Behind the Label
Commercial 2-Ethyl-1-butene rarely flies off the shelves at your average hardware store, but folks in the chemical and plastics industries know it has a specific job to do. Purity sits high on the priority list. Producers sell this compound with a minimum assay of 97%. The remaining 3% consists mostly of similar hydrocarbons, like 2-ethyl-2-butene or trace butadienes, plus a few heavier or lighter alkenes, depending on the production method. A 99% benchmark pops up in certain applications, especially where tighter specs mean fewer process surprises.
Why Chemical Purity Never Feels Optional
Impurities change downstream results fast. A little extra 2-ethyl-2-butene, even at a fraction of a percent, can throw off catalyst performance, foul up polymerization reactions, and trigger unwanted color or odor in end products. Polyethylene makers and surfactant suppliers value reliable feedstocks for that exact reason. Even in non-polymeric applications, like the production of specialty lubricants or fragrance intermediates, stray molecules can punch above their weight.
I once worked with a group optimizing catalyst yields for a major olefin producer. Any shift in feedstock purity, even a single percent, meant testing days lost and barrels wasted. Not every batch gets accepted, especially if the customer wants a traceable process or must meet REACH or TSCA compliance checks. Losing predictability means losing money—something no plant manager takes lightly.
Testing, Fact-Checking, and Transparency
GC analysis tells the truth about what’s in a shipment of 2-ethyl-1-butene. Suppliers list minimum purity on their certificates of analysis, but smart buyers ask for chromatographs, impurity profiles, and stability data. Reputable producers run regular tests and keep production lines tight, limiting cross-contamination and removing excess water or peroxides. Water content usually stays below 0.05%, and acid numbers must not register beyond trace levels—both factors help keep side reactions low.
Fractions with a true 97–99% active molecule fetch a better price thanks to this extra attention. Any supplier without regular batch testing or who can’t trace impurity origins puts customers at risk.
Improving Consistency and Raising Standards
Many countries hold chemical suppliers to ISO 9001 or similar quality certifications. In practical terms, this makes random out-of-spec shipments less likely since every lot links back to a consistent process and checks by trained staff. Some facilities switch from batch to continuous production to shave off variability, while others refine their capture and purification techniques with better distillation columns and in-line analyzers.
Buyers sometimes demand higher purity for their processes, driving innovation along the whole supply chain. Investment in better equipment, stricter protocols, and transparent documentation builds confidence with auditors, regulators, and end-users alike.
The Takeaway
Purity specs for commercial 2-ethyl-1-butene rarely drop below 97%, but the real test is whether every bottle and drum lives up to that number once it reaches the customer. Industries depending on this chemical refuse to roll the dice with performance or compliance. Only detailed analysis, honest reporting, and continuous process improvement separate truly reliable suppliers from the rest.
| Names | |
| Preferred IUPAC name | 3-Methylhex-1-ene |
| Other names |
2-Ethylbut-1-ene 2-Ethylbutene-1 Ethylisopropylene |
| Pronunciation | /tuː ˈɛθɪl wʌn ˈbjuːtiːn/ |
| Identifiers | |
| CAS Number | 5332-23-4 |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:52236 |
| ChEMBL | CHEMBL45392 |
| ChemSpider | 69260 |
| DrugBank | DB14096 |
| ECHA InfoCard | ECHA InfoCard: 100.011.742 |
| EC Number | 205-491-7 |
| Gmelin Reference | 7758 |
| KEGG | C06507 |
| MeSH | D017697 |
| PubChem CID | 12314 |
| RTECS number | EL8575000 |
| UNII | R24M9J1187 |
| UN number | UN2459 |
| CompTox Dashboard (EPA) | DTXSID1039222 |
| Properties | |
| Chemical formula | C6H12 |
| Molar mass | 84.16 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet odor |
| Density | 0.676 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 85 mmHg (20 °C) |
| Acidity (pKa) | 15.7 |
| Basicity (pKb) | 2-Ethyl-1-Butene does not have a relevant pKb value as it is not a base; it is an alkene. |
| Magnetic susceptibility (χ) | -9.72×10⁻⁹ cm³/mol |
| Refractive index (nD) | 1.398 |
| Viscosity | 0.404 mPa·s (20°C) |
| Dipole moment | 0.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 349.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -52.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4097.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P303+P361+P353, P304+P340, P312, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0-アル |
| Flash point | -18 °C (closed cup) |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Explosive limits | 1.2% - 7.7% |
| LD50 (median dose) | LD50 (median dose): >5 g/kg (oral, rat) |
| NIOSH | RN0130700 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Ethyl-1-Butene: Not established |
| REL (Recommended) | Not established |
| IDLH (Immediate danger) | IDLH: 800 ppm |
| Related compounds | |
| Related compounds |
1-Butene cis-2-Butene trans-2-Butene Isobutene 2-Methyl-2-butene 2-Methyl-1-butene 1-Pentene 2-Pentene |

