2-Ethyl-1-Butanol: Understanding a Versatile Chemical
Historical Development
Curiosity has always driven chemists to dig deeper into the structure and utility of alcohols, both for practical uses and pure science. Through the early 20th century, researchers kept stumbling upon branched-chain alcohols during studies of petroleum derivatives and fermentation. Among these, 2-Ethyl-1-Butanol stood out for its unique chain structure. Its emergence was tied closely to the petrochemical boom, as researchers found that by tweaking traditional synthesis approaches, they could build longer, more complex alcohols with specific branching patterns. Breaking away from the limitations of ethanol and n-butanol, the development journey of 2-Ethyl-1-Butanol highlights the chemical industry’s shift toward specialized intermediates that solve tricky formulation challenges in coatings, lubricants, and flavors.
Product Overview
2-Ethyl-1-Butanol brings a flexible backbone to its family of higher alcohols. The molecule appeals to manufacturers for its balanced mix of volatility, solubility, and a structure that resists easy evaporation, which opens doors in plasticizer synthesis, coatings, and as a solvent for nitrocellulose. This compound shows up in plant extracts and in industrial pipelines, embodying the merger of natural presence and tailored chemistry. In my own lab, the liquid’s faint odor and clear appearance often led to questions about its role compared to straight-chain counterparts, and its performance in blending paints hinted that even a small tweak to the carbon chain can change how materials behave in the real world.
Physical & Chemical Properties
A closer look at 2-Ethyl-1-Butanol reveals a colorless liquid with a mild odor, a boiling point near 146°C, and a density that sits just below water. It dissolves well in organic solvents but shows only slight solubility in water. The branched structure gives the molecule lower freezing and boiling points than straight-chain isomers. Anyone who has handled it in the lab knows it feels less volatile than n-butanol and keeps its cool when heated thanks to its robust molecular branching. Its vapor is heavier than air, a fact that shapes the safety approach around storage and ventilation. Chemically, the primary alcohol group reacts with common acids, halides, and oxidizers, leading to a swath of downstream products.
Technical Specifications & Labeling
In the chemical trade, labeling and purity are not negotiable. Producers often grade 2-Ethyl-1-Butanol by minimum purity standards, with technical grades above 98% and specific water content limits. The best practice calls for a clear mention of batch numbers, hazard pictograms, UN number (UN 1120), and signal wording aligned with global GHS standards. Packing gets special treatment too, with suppliers using steel drums or IBC containers lined to prevent corrosion. In the warehouse, labels jump off the container to warn about both flammability and the need to keep it away from oxidizers. My clients usually request full sheets with physical constants, shelf life, and instructions for spill cleanup, underscoring that plain labeling keeps both users and the environment safer.
Preparation Method
Industrial routes to 2-Ethyl-1-Butanol start with the aldol condensation of n-butyraldehyde and acetaldehyde, followed by hydrogenation. Factories optimize these steps using pressure reactors with solid catalysts, keeping yields high and byproducts minimal. The process favors economies of scale, and the materials flow from oil refining or synthetic gas reactions. In a smaller scale setting, chemists sometimes use Grignard reagent reactions with ethyl magnesium bromide and butyraldehyde, but this tends to be more expensive and slower. With green chemistry picking up steam, researchers keep searching for biocatalytic methods starting from fermentation feedstocks—no strong contender yet beats the classic petrochemical route’s cost. Each process has to keep purity in mind, as trace impurities can mess with end-product properties in sensitive applications like flavors and fragrances.
Chemical Reactions & Modifications
The reactive site on 2-Ethyl-1-Butanol is the primary hydroxyl, which plays the lead in esterification and etherification. Reacting it with phthalic anhydride gives plasticizers needed for soft PVC compounds. It picks up new groups easily, producing esters that serve as smooth-flowing solvents or as intermediates for specialty flavors. In my own experience, the selective oxidation to 2-ethylbutyric acid took patience but rewarded with strong-smelling acids useful in food industries. Manufacturers like to convert excess alcohol into ethers or longer-chain derivatives for surfactants. The compound’s moderate branching means it often sidesteps some of the harsh side-reactions that plague straight-chain alcohols, making it a quiet workhorse in blended systems. With proper temperature control, side products remain low, which keeps downstream purification simple and affordable in a busy plant setting.
Synonyms & Product Names
2-Ethyl-1-Butanol appears under several banners in scientific literature and industrial catalogs. You might see it called 2-Ethylbutan-1-ol, Ethylbutanol, or 1-Butanol, 2-ethyl-. In German and French datasheets, versions like "2-Ethylbutanol" or "Alcool butylique éthylé" often pop up. Certain brands market it as "2-EB" or as a component of solvent blends with minimal distinction. Smart purchasing teams keep these synonyms close at hand, as a missed label can derail supply chain orders or slow down compliance audits.
Safety & Operational Standards
Handling higher alcohols calls for a no-nonsense approach. 2-Ethyl-1-Butanol gives off flammable vapors and can irritate skin and eyes. Storage rules require cool, ventilated spaces, away from oxidizers and open flames. OSHA sets exposure standards and expects routine HVAC maintenance in sites using this chemical. Training matters most—operators wearing splash goggles, gloves, and flame-resistant jackets stand a better chance of avoiding accidents. The MSDS sheets prepared by reputable suppliers spell out first-aid procedures for accidental splashes, inhalation, or ingestion. Over the years, improvements in vapor detection and spill containment have reduced major incidents. Regular drills and proper PPE keep workplaces from letting their guard down, especially in small businesses or under-resourced labs.
Application Area
2-Ethyl-1-Butanol adds toughness and flexibility to PVC plastics through its role in making phthalate esters. Paint makers value it for its gradual evaporation profile, improving brush leveling and gloss in automotive finishes. Ink manufacturers go after its solubility for nitrocellulose-based toners. The food industry, guided by GRAS approvals in some markets, sometimes uses its esters as flavoring agents. Fragrance chemists depend on its backbone in building notes that last, either as an intermediate or a modifier in base oils. In my own dealings with specialty lubricants, the addition of 2-Ethyl-1-Butanol improved flow at subzero temperatures, solving client complaints about thickening in outdoor machinery. Its utility stretches into adhesives, textiles, and as a carrier in pesticide formulas.
Research & Development
Labs around the world keep testing this alcohol for both new chemistry and greener synthesis. Researchers look for biocatalytic routes, aiming to use waste biomass or engineered microbes as feedstock. Trials run with new hydrogenation catalysts have boosted yields and cut waste. In the coatings field, modified derivatives draw attention for their slow volatility and compatibility with water-based systems, offering help to paint chemists fighting VOC emissions. Public grant programs often target toxicity reduction and lifecycle analysis, focusing on both worker exposure and downstream breakdown in the environment. Recent patents highlight more selective oxidation processes and new ester formulations, hinting at a vibrant pipeline just under the industrial radar. Students in university labs also pursue experiments—sometimes less for market gain, more for the joy of exploring a versatile structure rarely taught in early chemistry courses.
Toxicity Research
The health effects of 2-Ethyl-1-Butanol don’t get swept under the rug. Animal studies reveal moderate acute toxicity, though not as high as some aromatic solvents. Inhalation at high concentrations produces symptoms like headache and dizziness; direct skin contact causes mild irritation but rarely burns. Regulators like the EPA keep updating reference doses, tracking new data on long-term exposure, environmental degradation, and metabolite profiles. Waste management guides send clear signals to incinerate or chemically neutralize waste before discharge. Lab studies on mutagenicity and endocrine activity continue, and so far, researchers haven’t flagged any major cancer risks, but the story keeps developing as new testing methods come online. Community right-to-know laws push companies to maintain robust chemical hygiene—something I’ve seen lag in smaller shops without active safety training.
Future Prospects
The journey of 2-Ethyl-1-Butanol stretches beyond synthetic rubbers and solvents. As plasticizer regulations tighten, demand grows for customized esters with lower migration rates and less toxicity, rewarding producers who invest in cleaner processes. Pushes in green chemistry and biomaterials set the stage for fermentation-driven supply chains, which could cut the carbon footprint and reduce reliance on oil. The steady rise in waterborne coatings and flexible electronics sends formulators searching for alcohols that blend smoothly and evaporate predictably in new settings—giving 2-Ethyl-1-Butanol a leg up. With public interest in workplace safety and environmental health running high, industry leaders partner with universities to close data gaps, boost recycling, and train chemists in sustainable manufacturing. Having watched these trends shift over decades, I see an expanding role for this subtle but powerful molecule both in tomorrow’s materials and in the broader push for smarter chemistry.
What is 2-Ethyl-1-Butanol used for?
Chemistry at Work: Why Manufacturers Trust It
Step into any modern factory making paints or coatings, and you’ll likely find 2-ethyl-1-butanol on the ingredient list. This chemical, created through hydroformylation of propylene and butanal, gets added because it blends well and works as an efficient solvent. Paint makers look for products that smooth on easy and dry right: this alcohol keeps pigments from clumping up, letting you roll out color with less streaking. It’s no secret the right solvent helps a fresh coat stick, shine, and last longer, especially on stuff that takes some everyday wear and tear.
Solvents: Not Just for Science Class
In labs and factories, solvents get all the attention. 2-ethyl-1-butanol doesn’t get much hype, but it handles grease, oils, and tough stains better than water ever could. Think of cleaning machines that spin through muddy jobs—removing residue can be a fight. This alcohol cuts through the grime and dissolves unwanted leftovers, saving time and keeping gears from breaking down. With many solvents, folks worry about dangerous fumes or explosive vapors. Research shows this one comes with a higher flash point, so there’s less risk when workers handle drums of it on busy factory floors.
Making Plastics and Rubbers that Stand Up to Life
Every device from your headphones to car dashboards uses plasticizers that keep things flexible and strong. 2-ethyl-1-butanol steps in here too. Plastic-makers mix it into the raw stuff so plastics won’t crack or shatter in sunlight or winter cold. Manufacturers like it because it unlocks more options for durable yet bendable parts. It also slips into the process of making synthetic rubber, giving tires and belts lasting power long past their first use.
Fuel Additives: Helping Engines Run Clean
Gasoline goes farther and burns cleaner with the right additives. 2-ethyl-1-butanol gets added to boost octane, which means the engine knocks less and runs smoother. Scientists and engineers study every molecule’s effect on emissions, since lower pollution matters for both city drivers and folks living along busy roads. Blending this alcohol into fuels can help cut down the nasty stuff coming out of the tailpipe, connecting back to public health and cleaner air. Regulations keep a close watch on what goes into gas tanks, but green chemistry research pinpoints this alcohol as an option that won’t gum up engines or corrode parts.
Safety and Sustainability: What Tomorrow Looks Like
As someone who has read plenty of safety data sheets, I know companies look for chemicals that bring less risk. Spills in a plant aren’t just a hassle—they threaten workers, neighbors, and the planet. 2-ethyl-1-butanol carries fewer dangers compared to some older solvents (for example, lower toxicity and inflammability), letting managers breathe a little easier. Researchers keep pushing for greener ways to make it too—switching from fossil fuels to plants as a feedstock. If this picks up, bottles on warehouse racks might someday show ‘renewable’ labels. Safety matters to me every bit as much as progress, since the people mixing chemicals in labs or tinkering with engines deserve the peace of mind that comes from cleaner, smarter choices.
What are the safety precautions for handling 2-Ethyl-1-Butanol?
Understanding the Hazards
2-Ethyl-1-Butanol brings plenty of industrial value, but it doesn’t like to stay in its lane. Without basic respect for its hazards, someone could end up harmed. It’s a clear liquid, often found in coatings, cleaners, and plasticizers. That mild odor hides a bigger story, though: this alcohol works fast to irritate skin and eyes. Too much contact, and you’ll remember. Workers should not go near it without understanding its potential for causing headaches, dizziness, and even respiratory distress at high vapor levels. Fire risk travels with it everywhere. This stuff catches fire even at a moderately warm room temperature. Every time I walk into a shop with open drums, I wonder if folks realize the risk sitting in those containers.
Personal Protection Comes First
Gloves and goggles sometimes sound like a nuisance, but with 2-Ethyl-1-Butanol you are just rolling the dice without them. Chemical-resistant gloves—nitrile fits the bill—save you from red, burning skin. A good snug pair of goggles will keep those painful splashes out of your eyes. Long sleeves don’t always show up in safety manuals, but more fabric between your skin and the chemical always buys you some insurance. In places that aren’t well-ventilated, a cartridge respirator adds an extra layer of safety for the lungs, especially during tasks like mixing or pouring.
Ventilation: A Key Line of Defense
Some workplaces downplay the value of solid airflow, but this is where problems start—especially with volatile liquids. Every shop needs exhaust fans pulling fumes away right at the source. Just cracking open a window doesn’t make much difference. I’ve seen coworkers drag their feet on using fume hoods because they take up space, but in the long run, avoiding one trip to the ER pays off.
Fire Prevention in Real Life
Workers too often underestimate flammability until something goes wrong. No one should use or store 2-Ethyl-1-Butanol near hot surfaces or open flames, not even for a minute. Static electricity sneaks up in dry, carpeted rooms, so grounding containers before pouring or transferring product matters no matter the scale. Fire extinguishers—even just one dry chemical type, clearly accessible—make everyone’s life safer. It helps to run checks monthly, not just dusting off the box twice a year. Spill kits, complete with absorbent materials, allow for a fast response if someone knocks a container over.
Training Changes Outcomes
Regular drills on emergency leaks, fire protocols, and PPE rules do a lot more than a stack of printed guidelines. Experience shows me that the staff who practice these drills act faster and more calmly when a real spill happens. Offering up short, targeted training sessions as new workers come in keeps old habits from breeding shortcuts. New hires should shadow supervisors to catch best practices in motion, not just read instructions.
Labeling and Storage Matter
People tend to slack off when containers aren’t labeled properly, leading to risky mix-ups. Workers need to use clear, durable labels for every drum and jug—no shortcuts with masking tape or faded marker ink. Storage works best away from sunlight, heat, and oxidizing agents. Flammable liquid cabinets are worth the investment for any shop using 2-Ethyl-1-Butanol on the regular. That signal yellow color reminds everyone these are not everyday supplies.
A Culture Around Safety
You can tell a lot about a workplace by how it treats chemicals. Whenever handling 2-Ethyl-1-Butanol falls into the daily routine, a strong safety culture keeps everyone accountable. Open conversations, feedback on near-misses, and encouragement for raising concerns go further than yelling out the rules. It’s true for any hazardous product: routines make or break safety, especially when the risks are invisible.
What is the chemical formula and structure of 2-Ethyl-1-Butanol?
Looking Closer at the Chemical Formula
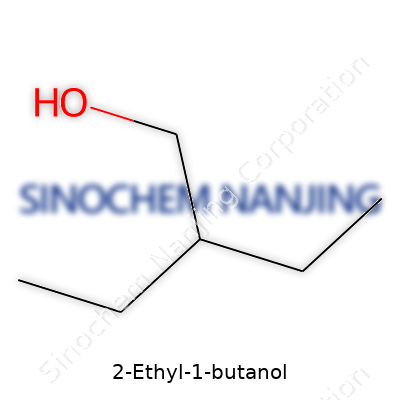
Walk into any lab supply catalog, and the name “2-Ethyl-1-Butanol” might not catch your eye. Strip away the branding, the numbers, and the fine print, and you see its simple chemical formula: C6H14O. Let’s break that down: six carbons, fourteen hydrogens, and a lone oxygen. On paper, it just looks like another page from a chemistry textbook. But what does this formula mean for the way it behaves and why do scientists keep returning to it?
Molecular Structure Tells the Real Story
Building 2-Ethyl-1-Butanol starts with the backbone of butane—four carbons in a row. The “2-ethyl” part means there’s a two-carbon branch hanging off the second carbon of that chain. Topping it off, a hydroxyl group (-OH) clings to the end carbon. Written fully, the structure appears as: CH3-CH(C2H5)-CH2-CH2OH. The backbone is pretty flexible, which helps when it slides into action as a solvent or intermediate.
Why This Structure Matters in Real Life
In my years hanging around labs, I’ve watched folks pick 2-Ethyl-1-Butanol over other alcohols because this little carbon branch changes everything. It brings a slightly higher boiling point, making it less volatile than many short-chain alcohols. For those mixing coatings or plasticizers, small tweaks like this keep mixtures stable and slow down evaporation. Safety routines always stress careful handling of alcohols; this one, with fewer fumes and a thicker texture, feels a bit easier to control during long days of blending or testing.
Let’s not skip over its role in chemical reactions. That “1-butanol” base offers reactivity where folks want it—plenty of esters in fragrances or pharmaceuticals begin here. Names on finished products rarely mention their building blocks, but without structures like this, modern manufacturing would jam up in a hurry.
Environmental and Health Perspectives
Chemicals don’t work in a vacuum. I’ve seen more labs turn an eye toward safety and sustainability. For 2-Ethyl-1-Butanol, this means careful monitoring of handling and disposal. Fact sheets from the European Chemicals Agency highlight its low acute toxicity. Still, careful ventilation is common practice thanks to any alcohol’s potential for irritation. Longterm, the structure doesn’t linger much in the environment, breaking down quicker than some of its larger cousins.
Room for Smarter Use
Talking to university researchers and production staff, the old ways of using solvents get challenged all the time. The push comes from two main fronts: making processes gentler for the people involved, and easing pressure on the environment. Increasingly, teams experiment with reducing the volumes needed or recovering and reusing what they can. Green chemistry groups search for ways to make production routes where fewer byproducts escape the process.
Sharing what’s learned about compounds like 2-Ethyl-1-Butanol—how its structure shapes its physical traits and its practical uses—builds trust from the lab up. Every detail, from chain arrangement to waste handling, shapes how safe and sustainable industrial chemistry can become in the long run.
Is 2-Ethyl-1-Butanol hazardous to health or the environment?
A Closer Look at This Industrial Alcohol
Walking through any chemical warehouse or reading the ingredient list of industrial solvents, 2-ethyl-1-butanol pops up from time to time. It has that characteristic scent you can't quite pin down, somewhere between sweet and pungent. Over the years, this clear liquid has shown up in paints, plasticizers, and as a chemical additive in manufacturing. Scanning the labels, you might wonder about the risks it brings to those who handle it every day or the broader impact on water and air.
What Science Shows About Health Effects
Direct exposure matters most here. If you’ve ever spent time in a factory that uses 2-ethyl-1-butanol, you'll know that even fleeting direct skin contact can cause irritation. Breathing in the vapors for lengthy periods doesn’t do the lungs any favors either. Based on what’s reported in toxicology literature and material safety data sheets, throat and respiratory tract irritation comes first. Workers sometimes report headaches and dizziness after regular exposure, usually at higher concentrations or in spaces where ventilation falls short. Here’s the thing: acute symptoms mostly clear up once a person gets away from the fumes, but long-term data stays limited. Animal studies mention possible impacts on the nervous system and liver after high doses, but translating those results to people remains tricky.
It makes sense then that the US Occupational Safety and Health Administration (OSHA) recommends limits for workplace air concentrations. Protective gear and proper exhaust systems help control risks, but not every workplace gives this enough attention. In my early days visiting facilities, I saw people lowering masks for comfort, a habit that’s tough to shake and might invite bigger problems later.
Environmental Footprint: Where Does It Go?
Factories that rely on this alcohol may worry less about personal exposure and more about what happens if a spill escapes into the environment. 2-ethyl-1-butanol breaks down in the air over days, especially in sunlight. That might sound reassuring, but concerns don’t vanish. If spilled or dumped down drains, this chemical heads into rivers, groundwater, or soil, where its persistence changes.
Aquatic organisms like fish and small invertebrates show toxic responses at certain concentrations. A 2018 review found higher levels slow growth in aquatic plants and algae. Though it doesn’t rank as “very persistent” or “highly bioaccumulative,” that short-term toxicity puts pressure on water treatment operators. Cities closer to heavy industry may see spikes in volatile organic compounds like 2-ethyl-1-butanol following storms or accidental leaks, and while filters catch a lot, trace amounts still leach into larger water systems.
Striking a Balance: Safer Use Makes All the Difference
Some companies are shifting toward safer solvents where possible and revamping storage systems to keep leaks and spills in check. Personal experience with training sessions on chemical handling taught me that real safety improvement comes from telling workers the “why” behind safety steps. Sticking hazard symbols on drums doesn’t go far unless folks know what skin burns or lung irritation feel like from mild exposure firsthand. Some businesses invest in closed-loop processes, so nearly every drop stays accounted for, or use alternative green solvents if the process allows.
National and local governments push for tighter reporting on any chemical with moderate toxicity, demanding more transparency from manufacturers. Friends working in water management talk about tracing compounds in the urban water cycle, working upstream with chemical suppliers to cut leakage and to boost routine water checks.
No single chemical wipes out the need for vigilance and a full look at impacts on health and the environment. 2-ethyl-1-butanol serves as a good reminder—what seems small in a lab flask can grow out of proportion once it leaves controlled spaces. Sharing the science, listening to those handling these substances daily, and demanding accountability from all sides steer us in a safer direction.
How should 2-Ethyl-1-Butanol be stored and transported?
Why the Details Matter
Many chemical plants and transporters handle alcohols every day, but some of them skip steps with 2-Ethyl-1-Butanol. I’ve seen what happens when this goes wrong. There’s an easy temptation to just treat it like ethyl alcohol, but that opens the door to leaks, contamination, and workplace incidents. 2-Ethyl-1-Butanol carries clear hazards—its vapors can irritate skin and eyes, it’s flammable, and any spills make both cleanup and insurance paperwork a nightmare.
Making Room for Safety
I've found that storage space decisions shape much more than one's inventory. A company I worked with used poorly labeled tanks for several years, and this led to confusion during audits and led to small spills during transfers. Only after clear training and strong labeling did safety improve. Tanks made out of carbon steel or stainless steel work best, and all signs should be obvious, clear, and weather-resistant. This isn’t just about ticking a regulatory box—it allows workers to respond quickly and avoid mistakes.
Storage spaces rely on solid ventilation. I’ve stood in hot warehouses where the air got thick and the smell of chemicals lingered; running extraction fans at all times changed both comfort and safety scores. Cool, shaded storage is essential. Keep the temperature below 25°C to slow chemical breakdown and stop pressure from building up. Every time a storage area gets too warm, the maintenance log grows with problems like expanding seals and pressure relief issues.
Transport Tactics That Work
Moving 2-Ethyl-1-Butanol pushes risk even higher. I’ve worked alongside drivers who rushed loading and closing valves, only to find leaks when they stopped on their route. One solution—constant supervision during loading and unloading—helped us slash both waste and incident reports. Transportation calls for more than just tank trucks. Secure tanks with vapor-tight seals keep product inside and people outside of harm. Local regulations demand clear hazard signs, and those should be resistant to rain and sunlight.
A driver once asked me why we double-check every manifest and drum seal. I explained, after a long shift, missing paperwork turns a routine road check into a hefty fine, and old seals can lead to product contamination. Everything builds up to one thing: tight recordkeeping saves headaches later and keeps everyone honest.
Learning From Others’ Mistakes
I’ve seen what happens if just one person leaves the spill kit tucked in the back of a cabinet: delays, panic, and in one case, major damage to painted concrete. Every employee should know where to find spill absorbents, gloves, goggles, and face shields. Routine training means less confusion, and it boosts morale—people trust their workspace when emergency gear stays in sight. Every year, update those trainings and rotate new staff through the drills, because nothing replaces muscle memory during a real emergency.
In the end, none of these precautions require expensive gadgets or revolutionary systems. A few well-placed warnings, consistent training, solid equipment, and a shared sense of responsibility work far better. Protect the product, the team, and the bottom line—simple steps go a long way, especially with a chemical like 2-Ethyl-1-Butanol.
| Names | |
| Preferred IUPAC name | 3-Methylhexan-1-ol |
| Other names |
2-Ethylbutan-1-ol Ethylpropylcarbinol Ethanol, 2-ethyl- 2-Ethylbutyl alcohol |
| Pronunciation | /ˈtuː ˈɛθ.ɪl wʌn ˈbjuː.tæn.ɒl/ |
| Identifiers | |
| CAS Number | 97-95-0 |
| Beilstein Reference | 1901216 |
| ChEBI | CHEBI:43699 |
| ChEMBL | CHEMBL43960 |
| ChemSpider | 6732 |
| DrugBank | DB03781 |
| ECHA InfoCard | 03b776fe-a42c-4331-9fe5-df2c6f3cb43e |
| EC Number | 203-306-4 |
| Gmelin Reference | 101821 |
| KEGG | C06327 |
| MeSH | D065710 |
| PubChem CID | 6309 |
| RTECS number | EL6475000 |
| UNII | B9S5T36694 |
| UN number | UN1120 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | characteristic odor |
| Density | 0.809 g/mL at 25 °C |
| Solubility in water | 8.7 g/L (20 °C) |
| log P | 1.76 |
| Vapor pressure | 0.34 mmHg (20°C) |
| Acidity (pKa) | 16.00 |
| Basicity (pKb) | pKb: 5.84 |
| Magnetic susceptibility (χ) | -7.51 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.419 |
| Viscosity | 2.57 mPa·s (25 °C) |
| Dipole moment | 2.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 241.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -345.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3505.8 kJ/mol |
| Pharmacology | |
| ATC code | 2-Ethyl-1-Butanol does not have an ATC code |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 77°C (171°F) |
| Autoignition temperature | 285 °C |
| Explosive limits | Upper explosive limit: 8.0%, Lower explosive limit: 1.2% |
| Lethal dose or concentration | LD50 oral rat 1230 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2,000 mg/kg (oral, rat) |
| NIOSH | EO1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Ethyl-1-Butanol: Not established |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1-Butanol 2-Methyl-1-butanol Isoamyl alcohol n-Hexanol 2-Ethylhexanol |

