2-(Dimethylamino)Ethyl Methacrylate: A Closer Look at Its Evolution and Significance
The Pathway from Discovery to Ubiquity
Digging into the background of 2-(Dimethylamino)ethyl methacrylate—commonly known by the shorthand DMAEMA—sheds light on how the chemical landscape shifted when engineers and chemists began looking for smarter, tougher, and more versatile monomers. Early in the 20th century, synthetic resins and specialty plastics hit the scene, fueled by the search for alternatives to naturally sourced materials. Methacrylate chemistry promised flexibility. As soon as researchers managed to combine the reactive vinyl backbone with the dialkylamino side group, they opened up new doors in polymer science. DMAEMA brought a responsive, hydrophilic nature to the methacrylate family, which let industries play with both the physical and chemical landscape of their products. In labs across Europe and North America, this compound quickly moved from a research oddity to a reliable building block for everything from coatings to biomedical gadgets.
Unpacking What DMAEMA Offers
If you’ve ever looked at a bottle of clear nail polish, a water-treatment membrane, or a medical hydrogel, you’ve probably encountered polymers made from DMAEMA. The secret comes from the molecule’s split personality: tough enough to form a strong polymer chain, yet flexible and water-friendly because of that dimethylamino group. In liquid form, it brings a faint ammoniac smell and sports a clear-to-pale yellow tint. Get it near water or acids, and DMAEMA’s pH responsiveness takes over—making it a favorite for smart materials. The boiling point sits quite high, and it stays stable at room temperature with the usual care against heat, sunlight, and mixing with strong oxidizers. Markets care about these details because they translate to longer shelf lives and fewer surprises in the process line.
Technical Details That Matter
When labs receive a new tank of DMAEMA, analysts waste no time checking its purity, color, and stabilizer levels, as well as acidity and viscosity. Most suppliers provide material in concentrations that run above 99%—using inhibitors like MEHQ to keep premature polymerization at bay. Labels map out flash points, hazard symbols, and chemical registration numbers, not just to satisfy regulations, but because understanding these properties links directly to process safety and performance. My own experience in a polymer synthesis lab made it clear: a small hiccup in inhibitor concentration or trace water content will spiral into unexpected gelation or spoil the final material. For anyone dealing with DMAEMA, precision counts more than routine.
Manufacturing and Preparation: From Lab Bench to Ton-Scale
DMAEMA’s preparation usually starts with reacting dimethylaminoethanol and methacryloyl chloride, under strictly controlled temperatures and with careful handling of byproducts like hydrochloric acid. In large-scale operations, you’ll see closed systems and vent controls, since the fumes are corrosive and eye-watering. The method—sometimes switched up with methacrylic acid and dimethylaminoethanol under esterification—has been refined over decades, boosting yields and cutting down on waste. Even as the basic chemistry remains constant, every producer tweaks steps for efficiency, environmental compliance, and purity.
Inside the Reaction Toolbox
Once in the bottle, DMAEMA doesn’t just sit around—its value crops up through the reactions it enables. That amino group grabs onto acids and metal ions, letting chemists modify it for surface coatings, water purification agents, or smarter drug delivery vehicles. Chain transfer reactions let formulators adjust the size and branching of the polymers; crosslinking can make gels swell or shrink on demand. Some of the most creative minds in materials chemistry exploit the vinyl functionality to graft DMAEMA onto other polymer backbones, or tailor-make copolymers that respond to a shift in temperature or ionic strength. These aren’t academic details. In research on controlled drug release, being able to finetune polymer response with one small molecule like DMAEMA can decide the success or failure of a device.
Name Game and the Maze of Synonyms
Professionals in the field run into a tangle of names: DMAEMA, 2-(Dimethylamino)ethyl methacrylate, and sometimes its trade names in various languages. The situation echoes a common problem in chemistry—miscommunication caused by name confusion. In regulatory filings and patent literature, names might morph, so double-checking the CAS number (often 2867-47-2) helps sidestep costly mistakes during purchasing and compliance review. Experienced hands swap stories of production lines going awry just because a batch used a similar-sounding, but functionally different, compound. Clarity at every stage pays dividends.
Tackling Safe Handling and Operational Realities
Anyone who has ever uncorked a bottle of DMAEMA in a small space remembers the sharp, fishy odor and the immediate bite on the skin. Factories enforce gloves, goggles, and ventilated hoods not because of regulatory mandates alone, but because the amine content means that exposure leaves lasting effects on health. Inhalation can irritate the respiratory tract; spills sting on contact. Occupational guidelines limbo between local, national, and international regulations—but common sense and experience agree: DMAEMA demands respect in storage and during all reaction steps. Hoses, tanks, and pumps bear plastic or stainless linings, since metal corrosion can foul whole runs. Companies spend real money installing leak detectors, fire suppression, and continuous training. Accidents are rare in well-run shops, but they hang heavy in memory when corners get cut.
DMAEMA at Work: Where It Shows Up
Look outside the chemical plant, and DMAEMA-made polymers shine in water purification, haircare products, adhesives, or thickening agents in print inks. Contact lens hydrogels became a reality when chemists hit on methacrylate monomers like DMAEMA that balanced oxygen permeability with water-attracting groups. Paint formulations use it for strong, stretchable films; microencapsulation researchers blend it into smart coatings. In my own run-ins with product scale-up, the unique combination of solubility, pH responsiveness, and chemical reactivity made DMAEMA a go-to, letting us craft scaffold materials for regenerative medicine or tailor polyamines for wastewater treatment. The trick is in tuning the formulation—get it right, and the product sets new standards.
On the Frontier: Ongoing Research
Research labs worldwide keep pushing DMAEMA into new territory. One red-hot area: controlled radical polymerization. Scientists fine-tune size and functionality, preparing custom polymers for targeted drug delivery or biosensor surfaces. Techniques like ATRP and RAFT thrive on DMAEMA’s reactivity, allowing entry into precision medicine applications where every functional group counts. Teams explore using DMAEMA as building points for advanced hydrogels—and when pairing with other responsive units, create materials able to convert environmental stimuli directly into action, whether swelling, shrinking, or releasing cargo. The intersection of bioengineering and DMAEMA-based chemistry keeps spinning off novel dental materials, wound dressings, and coatings for cell scaffolds. The work involves more than academic curiosity—it lays the groundwork for the next generation of health and environmental products.
Weighing Toxicity and Exposure Hazards
Ignore DMAEMA's warning labels at your own risk. Toxicology studies point out clear risks: acute exposure irritates skin, eyes, and lungs; chronic contact links to dermatitis among factory workers. Rats and mice exposed to high levels show changes in organ weight and function—reminders for both industries and regulators not to get complacent. Water contamination risks loom large since DMAEMA’s solubility and reactivity mean it can move through natural and built environments fast. As much as companies tout their new polymer products, more research needs to fill knowledge gaps on low-level, long-term exposure—especially for biomedical applications. On-the-ground safety teams don’t just draw rules from government leaflets; old-timers in the business know the value of vigilance and the cost of letting small leaks or bad habits slide through unnoticed.
The Road Ahead: Where Could DMAEMA Go?
Looking forward, the potential for DMAEMA keeps attracting attention, especially in sectors chasing functional, adaptable materials. The trend toward green chemistry prods producers to clean up the synthesis, slash hazardous byproducts, and source more sustainable feedstocks. At the university bench, researchers work on biodegradable DMAEMA-based copolymers aimed at environmental sensors or temporary implants. The pH-responsiveness and polymerizability marry well with the demand for smart surfaces and environmental remediation agents that don’t stick around forever. From personal care to life sciences, DMAEMA’s story reflects the broader arc of chemicals carved into the fabric of daily life—not just by science, but also by the grit and care of the people who work with it, pushing boundaries and keeping one eye on safety at every turn.
What is 2-(Dimethylamino)ethyl methacrylate used for?
What Draws the Industry’s Eye to This Chemical
I’ve spent years watching how specialty chemicals shape everyday products, and 2-(Dimethylamino)ethyl methacrylate stands out for its role in the modern materials toolbox. Chemists lean on it in the world of polymers because its structure makes it blend seamlessly into a long list of custom plastics.
Real-World Products Built From DMEMA
Acrylic resins catch the spotlight thanks to 2-(Dimethylamino)ethyl methacrylate, known for its contribution to both toughness and flexibility. Walk past a display of contact lenses or hair gels and you’re staring right at its influence. Lens manufacturers turn to it since its chemical group attracts water, creating hydrogels that stay moist and feel comfortable on the eye. Medical adhesives, dental fillings, and gel nails also take advantage: the material bonds fast, holds tight, and doesn’t irritate tissues the same way harsher alternatives can.
Printing inks and specialty coatings also rely on the unique characteristics of this methacrylate. Printers care about how quickly an ink sets and how sharply it resists damage from heat or water. DMEMA brings a balance, letting coatings banish static electricity and toughen up against surface scratches. I’ve tested antistatic floor finishes that used it, and their performance held up during months of foot traffic.
What Sets This Building Block Apart
Plastics made from 2-(Dimethylamino)ethyl methacrylate show off a mix of softness and chemical strength. This is more than trivia—furniture producers, textile makers, and even water treatment labs benefit. If you’ve wrung out a water-absorbing sponge, there’s a good chance DMEMA helped it pull in and release moisture so efficiently. In textile coatings, it delivers a slick feel but still lets fabrics breathe, something clothing brands love to talk about.
Safety and Handling: A Key Piece of the Puzzle
Many of the practical concerns around DMEMA boil down to safety questions. This compound reacts fast during production, giving off vapors that will irritate skin and eyes. OSHA warns workers to keep it off their hands, eyes, and lungs. Anyone who works with it gets trained on protective gear and proper ventilation. I’ve seen labs lock it behind fume hoods and double-check every connection for leaks. Even trace spills demand immediate cleanup, since the acrid odor signals trouble before bigger problems develop.
What Could Change for the Better
Demand for greener solutions has led companies to revisit DMEMA processes. Some push to recycle more of the waste. Others dig into non-toxic alternatives, especially for medical products stuck against bare skin. Regulatory groups, especially in Europe and the US, keep a close eye on long-term safety data. Factories now log exposures and test waste streams with regularity.
Transparency plays a role in building trust. Manufacturers provide material safety data sheets and disclose how they’ve reduced environmental impacts. It makes a difference. Research teams work to swap petroleum feedstocks for bio-based options. These early wins set a precedent and show how specialty chemicals can meet health and safety benchmarks without giving up on performance.
Personal Take
Having tracked the rise of smart polymers, I’ve seen how chemicals like 2-(Dimethylamino)ethyl methacrylate make familiar products more effective and safer. New challenges keep engineers on their toes—especially questions about worker exposure and eco-friendly materials. Innovation hasn’t slowed. That balance between function, safety, and sustainability steers the direction of the industry, and it all starts with the details behind everyday names on a label.
What are the safety precautions when handling 2-(Dimethylamino)ethyl methacrylate?
Getting Real about the Chemical
There’s no way around it—2-(Dimethylamino)ethyl methacrylate shows up in a lot of industrial labs and manufacturing settings. Folks use it for its role in synthesizing specialty polymers. While it’s common to find this material in certain workspaces, the hazards involved shouldn’t slip anyone’s mind. I’ve worked with it myself, and if I learned anything, it’s that sharp safety habits matter far more than fancy protocols printed on a wall.
Personal Protective Gear Isn’t Optional
You’re handling a liquid that can strike fast. Skin burns, eye injury, breathing trouble—these are real possibilities. For me, pulling on nitrile gloves before opening any container of this stuff became as natural as grabbing my keys in the morning. I also made a habit out of full lab coats and chemical splash goggles. A simple face shield adds useful insurance if pouring or mixing is likely to cause a splash. One chemist I knew figured a pair of cheap gloves would do. Not long after, he spent the afternoon at urgent care. It’s not worth it to cut corners.
Good Air Means Good Health
Fumes from 2-(Dimethylamino)ethyl methacrylate seem mild at first. Lower concentrations already tickle the nose—but higher doses can set off headaches fast, even before you feel dizzy. I remember hearing about a colleague working in a closed room; air didn’t move, and nobody installed a fume hood. She landed with a bad asthma attack. Lab spaces must push out fumes. Strong local exhaust ventilation—especially a working fume hood—keeps air decent and lungs healthy. For anything bigger than a small batch, breathing masks help too, especially pointed to standards like NIOSH-approved cartridges.
Are You Ready for a Spill?
I never trusted bare counters or open glassware nearby. One slip, and a puddle forms, with vapors chasing you out of the room. Spill kits tailored for organic chemicals work best: absorbent pads, chemical-resistant scoops, and neutralizing agents. Sand or vermiculite stacks up well if budgets run tight. I saw a lab get lucky since they had a large chemical mat beneath every bottle—the liquid pooled, not spread. If a team drills and repeats basic spill drills, especially blocking it from drains and skin, no one panics when something tips over.
Storing for the Long Haul
Waste and leftover chemicals don’t mix well with sunlight, heat, or open flames. Over the years, I kept everything locked in the flammables cabinet, away from oxidizing agents or acids. At the wrong temperature, even a well-sealed bottle bulges. A friend once left some near a heat source—later, the cap popped, and the bottle leaked. Keeping the area dry and labeling every bottle beats relying on memory.
Training and Vigilance Always Win
Technical know-how only gets someone halfway. OSHA and hazard communication training fill in the rest. I stuck to the safety data sheet for every question that came up. Workers who ask questions get fewer surprises. Wear gear, guard your breathing, know your space, and keep emergency numbers visible. A culture of safety, piece by piece, keeps people healthy for more than one shift.
What is the chemical structure and formula of 2-(Dimethylamino)ethyl methacrylate?
The Nuts and Bolts of Its Chemical Skeleton
Everyday lab work draws us closer to the little details behind the materials and monomers we choose for designing better plastics, hydrogels, paints, or adhesives. 2-(Dimethylamino)ethyl methacrylate, often called DMAEMA, stands out for its chemical backbone and versatility. At the core, it carries the methacrylate group, known for undergoing polymerization readily—creating those strong, flexible connections in polymer chains found in many modern products.
DMAEMA’s formula: C8H15NO2. That’s eight carbons, fifteen hydrogens, one nitrogen, and two oxygens. Chemists reading structural formulas often spot two clear parts: the methacrylate fragment and the dimethylaminoethyl "tail." The methacrylate end sports that carbon-carbon double bond plus a methyl branch and a carboxylate ester function. The ester is linked via an ethyl bridge to a dimethylamino group. Looking at it, the molecule balances hydrophobic and hydrophilic elements—it brings together the water-loving amino with the oily nature of the acrylate core.
Why This Structure Matters in Everyday Science
I’ve seen DMAEMA in graduate school and industry projects both, not just as a building block but as something that can be tuned to interact with specific environments. That dimethylamino side makes the molecule basic. In simple words, it claims protons easily in acidic surroundings, and gives up protons in basic conditions—this property helps make "smart" polymers that react to their surroundings. Polyelectrolytes built from DMAEMA respond to pH changes, making them great in controlled drug delivery, gene therapy, and water treatment.
Beyond reactivity, that nitrogen brings an ionic edge when protonated. It attracts or repels other charged molecules. In coatings or adhesives, this can help bind to surfaces more tenaciously, especially those that have their own charge. Imagine an industrial floor coating that adapts to varying moisture or chemical exposure. Or a hydrogel that expands or contracts based on its acidity—this adaptability traces back to the clever placement of the dimethylamino group in the polymer backbone.
Real Impact, Real Challenges
Handling chemicals like DMAEMA offers lessons about both promise and caution. Without proper PPE, the substance can irritate skin and eyes. Small enough to soak in fast, it easily gets through gloves or unprotected hands, and it stings. From personal experience, a short splash on the arms is enough to drive home the lesson that even a simple molecule demands full respect in the lab. Always check the material safety data sheet and keep proper ventilation going.
Supply chain operators and workers down the line face risks too. Exposure levels need careful monitoring and process controls. Over time, regulations around handling, labeling, and transporting these monomers have improved safety on shop floors and in transport vehicles. Still, oversight can slip if it only stays on paper. Building a culture where staff know the dangers of chemical contact and see the reasons behind procedures—like chemical-resistant gloves and regular air checks—matters more.
What's Next for Safer, Smarter Uses
Research keeps churning out smarter formulas—DMAEMA co-polymers tailored for medical devices, self-cleaning membranes, or new adhesives. Cutting solvent use or developing greener synthesis reduces long-term health and environmental risks. Customizing polymers at the molecular level offers another way forward: switching up the side chains or using biobased sources to tweak biodegradability.
Stronger partnerships between researchers, plant technicians, and safety experts close the gap between the bench and the production line. Sharing incident reports between sites, updating training with real-life stories, and giving everyone hands-on demos of proper use go further than any poster on the wall. Real safety, like chemistry itself, comes from understanding down to the structure—and respecting how a few atoms make all the difference.
How should 2-(Dimethylamino)ethyl methacrylate be stored?
What Makes This Chemical Demanding in Storage?
2-(Dimethylamino)ethyl methacrylate stands out in the lab for more than just its mouthful of a name. This liquid reacts quickly with air and moisture and gives off strong smells. If left out, it can start to break down or thicken, causing problems for anyone using it in research or production. I’ve seen what spilled monomers do to a shared bench—sticky residue, wasted material, and safety paperwork nobody wants to fill out.
Many chemicals hide their hazards. This one makes its presence known fast, especially in humid weather. It doesn’t spark headlines like some famous toxicants, but it brings its own set of risks: skin and eye irritation, respiratory hazards, even fire risk around heat or open flame. Fact sheets from manufacturers and regulatory agencies underline this every time.
What Does Proper Storage Look Like?
Two key actions keep this chemical stable and workers protected: control the environment and secure the container. Leave it in a sunlit room, and you’ll see color changes and thickening in days. Store it close to an oxidizing agent, and you gamble with the real possibility of an exothermic reaction. Never a fun situation to explain to your supervisor.
In the labs I’ve worked, people stashed bottles in flammable-storage cabinets. Often, they missed labeling or forgot about shelf-life. Those mistakes can turn routine tasks into sticky, time-consuming messes or worse. A robust policy insists on tight lids, clear labels, age checks, and everything stowed far from sunlight, sparks, or humidity.
How to Build Good Storage Habits
Surviving years of handling volatile monomers, I keep three things in mind:
- Temperature stays low. Heat speeds up breakdown and increases fire risk. Chemical refrigerators or temperature-controlled rooms help a lot.
- Light stays out. Amber bottles block much of the harmful light. Opaque cabinets give extra protection. Excess light means more chances for unwanted reactions.
- Air stays out. Seal containers well. The less air exposure, the slower the material changes. Nitrogen blankets add another layer in large storage setups.
Labels tell stories, too. Marking bottles with the date received, opened, and recommended discard time has saved more than one batch from surprise gelling or contamination.
Raising the Bar: Safer Practices and Training
Training is worth every minute. Teams need to know exactly what makes chemicals like this tricky—fire drills don’t cover chemical mishaps. I recommend routine checks: look for bulging containers, color changes, or strange smells. If something feels off, don’t guess—check the product sheet or call the supplier.
Emergency gear, clear evacuation routes, and accessible spill kits keep workplaces prepared. Those steps don’t just protect employees—they keep projects on track. Nobody likes delay from accidents.
Why It Matters to Everyone
This isn’t just about dotting I’s and crossing T’s. Safe storage pays off in peace of mind and fewer headaches. Workers take less sick leave, labs avoid costly cleanup, and businesses feel confident about meeting regulations. NASA and chemical manufacturers document these lessons for a reason. Respecting the quirks of chemicals like 2-(Dimethylamino)ethyl methacrylate saves resources, reputations, and sometimes even lives.
What are the physical and chemical properties of 2-(Dimethylamino)ethyl methacrylate?
What Stands Out About This Compound
Anybody who’s spent time around a laboratory or a manufacturing plant might have come across 2-(Dimethylamino)ethyl methacrylate, or DMAEMA for short. It has a bit of a reputation in the world of specialty chemicals because of a handful of properties that make it both useful and tricky at the same time. DMAEMA doesn’t just pop into mind when people talk about methacrylate monomers, but that doesn’t mean it’s forgettable. In fact, the way it behaves physically and chemically explains why folks in polymer science give it a lot of attention.
Physical Nature: What You See, What You Get
DMAEMA shows up as a colorless to pale yellow liquid. It tends to carry a fishy, amine-like odor that’s unmistakable, which means you won’t mistake it for water. It is light enough to flow easily, which makes pumping and transferring it less painful than handling more viscous resins or adhesives. Volatility poses some challenges for bulk storage because its boiling point hovers around 165°C. Pour some out in a warm room and that smell will fill the air in no time.
One thing that stands out both in the research and in my practical run-ins: this monomer dissolves well in common organic solvents such as ethanol, acetone, and ether. It doesn’t shy away from water, either, because of the amine group attached to its structure. This dual compatibility opens up a lot of processing options, especially for folks looking to tweak polymerization in aqueous or non-aqueous setups.
Chemical Personality: Reactivity and Caution
DMAEMA carries that methacrylate group, so it jumps right into free-radical polymerization. You’ll find this compound grafted onto all kinds of polymer chains where flexibility, adhesion, or a little bit of hydrophilicity get the job done. The dimethylamino group brings a basic, electron-rich character, which isn’t just chemical jargon — it means you can use DMAEMA to create cationic polymers and increase compatibility with other functional groups.
Stickiness isn’t just a physical property—it’s a chemical one, too. DMAEMA tends to react with acids because that tertiary amine likes to form salts. I’ve seen people take advantage of this to create water-soluble polymers that change their solubility based on the pH. This ionizable behavior makes it a top pick for pH-responsive hydrogels, contact lenses, and coatings that need to interact with their environment.
Oxygen, light, and heat bring challenges. DMAEMA can polymerize spontaneously if you ignore stabilizers, so manufacturers always include something to keep those chains from linking up in the bottle. That’s a safety and storage headache. If you’ve ever seen a bottle of DMAEMA that looks a little thick or yellowed, you know it’s time to order fresh stock.
Hazards and Handling: Practical Lessons
Skin contact can feel like a burn after a few moments, especially with the raw monomer. Protective gloves and proper ventilation top the list any time people work with it. Inhalation over time can irritate airways—nobody wants that headache. As someone who’s dragged spill control materials across a lab floor, I can say this is one chemical you want to keep tightly sealed and labeled.
DMAEMA is flammable. That clear liquid can ignite if it finds a spark at the wrong time. Keeping it away from heat sources or open flames isn’t just a best practice—it’s essential. Used containers need to be triple-checked before tossing them anywhere near regular trash.
Pushing Forward: Safer and Smarter Use
Manufacturers keep fine-tuning formulations and handling protocols to reduce risks while getting the performance that DMAEMA offers. A lot of progress comes through better venting, automated dosing, and new stabilizers that keep it shelf-stable longer. Industry groups and research labs keep sharing lessons learned, helping newcomers avoid the bumps experienced hands have seen before.
It’s a compound that rewards respect for its quirks and hazards—handle it with care, and you’ll find it has a lot to offer.
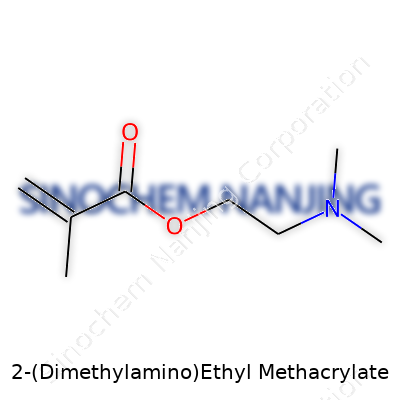
| Names | |
| Preferred IUPAC name | 2-(Dimethylamino)ethyl 2-methylprop-2-enoate |
| Other names |
DMAEMA Dimethylaminoethyl methacrylate 2-(Dimethylamino)ethyl 2-methylprop-2-enoate Methacrylic acid 2-dimethylaminoethyl ester 2-(Dimethylamino)ethyl methacrylate |
| Pronunciation | /tuː daɪˈmɛθɪl.əˌmiːnoʊ ˈɛθɪl mɛθˈæk.rɪ.leɪt/ |
| Identifiers | |
| CAS Number | 2867-47-2 |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:53005 |
| ChEMBL | CHEMBL12544 |
| ChemSpider | 11996 |
| DrugBank | DB13977 |
| ECHA InfoCard | 03beb33c-5e38-405a-9c9f-a15e2ae3fa60 |
| EC Number | 203-378-7 |
| Gmelin Reference | 84116 |
| KEGG | C05324 |
| MeSH | D008937 |
| PubChem CID | 8706 |
| RTECS number | DO3325000 |
| UNII | VT2XU30X55 |
| UN number | UN2522 |
| Properties | |
| Chemical formula | C8H15NO2 |
| Molar mass | 157.22 g/mol |
| Appearance | Colorless to pale yellow transparent liquid |
| Odor | fishy |
| Density | 0.90 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | 1.32 |
| Vapor pressure | 0.8 mmHg (20 °C) |
| Acidity (pKa) | 8.4 |
| Basicity (pKb) | 5.16 |
| Magnetic susceptibility (χ) | -7.55×10^-6 cm³/mol |
| Refractive index (nD) | 1.434 |
| Viscosity | 15 mPa·s (20 °C) |
| Dipole moment | 3.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 225.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -389.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3406.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H302, H312, H314, H332 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P264, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 51 °C |
| Autoignition temperature | 215 °C (419 °F; 488 K) |
| Explosive limits | 2.1-11.5% (in air) |
| Lethal dose or concentration | LD50 Oral Rat 1760 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 1850 mg/kg |
| NIOSH | MA8050000 |
| PEL (Permissible) | PEL: 5 ppm |
| REL (Recommended) | 5 ppm (18 mg/m3) |
| IDLH (Immediate danger) | IDLH: 220 ppm |
| Related compounds | |
| Related compounds |
Methacrylic acid 2-Dimethylaminoethyl acrylate Ethyl methacrylate 2-(Diethylamino)ethyl methacrylate Hydroxyethyl methacrylate Methyl methacrylate Butyl methacrylate Acrylic acid |

