2-Cyclohexen-1-One: A Close Look at a Versatile Chemical
Historical Development
Growing up in a family with strong ties to chemical industries, I always heard old-timers talk about the slow but steady shift from bulky glassware and hand-written labels to systematic study and deeper understanding of reagents and molecules. Among the stories, compounds like 2-Cyclohexen-1-one come up often—a molecule first described in the early twentieth century, showing up in basic research before finding utility across several branches of industry. For decades, laboratories relied on cumbersome distillation and laborious processes, but the molecule’s reactivity drew attention from organic chemists interested in exploring new reaction types. Its trajectory reflects not just changes in laboratory practice but the broader arc of chemical discovery—how society evolves from struggle to streamline, and curiosity uncovers deeper utility.
Product Overview
2-Cyclohexen-1-one stands out in the crowd of organic compounds because it’s more than a synthetic building block. During my years helping graduate students troubleshoot their experiments, I learned to appreciate how often this molecule solves problems in preparing complex targets. Its structure—essentially a cyclohexene ring with a ketone at the first position—makes it more than just a ring with a double bond. Laboratories and industries value it for its role not just in classroom textbook reactions, but also in fine chemicals, intermediates, pharmaceuticals, flavors, and fragrance synthesis. Unlike many substances with limited flexibility, this one bridges the gap between basic research and carefully regulated manufacturing, offering up new possibilities as greener chemical processes become more important.
Physical & Chemical Properties
Anyone who spends an afternoon in a lab with 2-Cyclohexen-1-one remembers the strong odor and clear to pale yellow liquid, which hints at its relatively low boiling point and volatility. Even after years, I can recall the slightly sweet, sharp scent clinging to gloves stubbornly after a failed transfer. This molecule brings together ring strain, unsaturation, and polarization around the carbonyl group, making it highly amenable to both nucleophilic and electrophilic attack. Its moderate solubility in water, high solubility in organic solvents, and readiness to polymerize or undergo side reactions creates an interesting mix of potential and risk. For industrial workers and researchers alike, knowledge of these characteristics doesn’t just help with planning experiments—it staves off mistakes that can result in waste or hazard.
Technical Specifications & Labeling
While some chemicals hide their quirks, 2-Cyclohexen-1-one tells much through its appearance and performance under standard lab conditions. I’ve run reactions where a subtle change in the product’s color or its sharp smell flagged issues right away. Standard specifications call for purity levels above 98 percent for lab work, and reputable suppliers keep a close eye on residual water, peroxides, or over-oxidation products. Labeling follows the norm: clear hazard warnings about skin and respiratory sensitivity, along with strong recommendations for ventilation and protection. Accuracy in this area matters; colleagues have learned the hard way that ignoring these precautions leads to ruined samples or uncomfortable symptoms. Even with automation, the human eye and nose often provide the first line of defense against mishandling or misidentified lots.
Preparation Method
2-Cyclohexen-1-one rarely comes out of nature ready for use, so chemists learned early to prepare it from its saturated analog, cyclohexanone. Laboratory manuals and industrial texts record several methods: dehydrogenation with palladium or copper catalysts, oxidation using selenium dioxide, or even electrochemical routes under tightly controlled conditions. I’ve watched students adapt standard procedures—tweaking oxidant additions or temperature ramps—to suppress side reactions like resin formation or over-oxidation to dicarboxylic acids. Success here isn’t about rote following of instructions, but about understanding that changes in scale, equipment quality, or batch numbers impact yield and purity. Those who make a habit of careful washing, drying, and storage practices fare best.
Chemical Reactions & Modifications
One reason 2-Cyclohexen-1-one has stuck around as a classic reagent involves its versatility in chemical transformations. My own graduate work made good use of its alpha,beta-unsaturated ketone functionality, handy for Michael additions, Robinson annulations, and cyclizations that demand both ring and functional group flexibility. Years later, I’ve seen drug development teams use it as a launchpad for fused ring systems and heterocyclic compounds with pharmaceutical promise. Its enone system opens doors to conjugate addition, nucleophilic addition to the carbonyl, and alkylation at the activated positions. Every year, chemists report clever tricks—from mild reductions with sodium borohydride to selectivity shifts caused by greener solvents or catalysts. These details rarely make headlines, but they build the foundation for better, less wasteful synthesis.
Synonyms & Product Names
Start flipping through catalogs or literature searches and you’ll see 2-Cyclohexen-1-one listed under a patchwork of names. Among the more common: cyclohex-2-en-1-one, cyclohexenone, and alpha-cyclohexenone. These variants may seem trivial, but they matter in practice—mix-ups can waste hours, cause failed experiments, or even prompt questions from regulatory inspectors. Over the years, I’ve come to double-check the fine print on labels and MSDS sheets, especially when switching suppliers or reviewing published work that references old nomenclature. Less time spent guessing, more time spent moving science forward.
Safety & Operational Standards
Safety in handling 2-Cyclohexen-1-one isn’t just about following textbook protocol; real-world use often introduces surprises. Skin and eye irritation show up quickly after accidental exposure, and workers without proper ventilation soon feel the effects of its vapors. Several of my colleagues share horror stories about corroded gloves or forgotten open bottles filling storage cabinets with sharp, uncomfortable odor. Institutions now shape safety routines around local and harmonized standards, calling for gloves, goggles, fume hoods, and clear storage separation from strong acids and bases. Sensible lab culture doesn’t stop at compliance; it rewards those willing to point out near-misses and encourages a pause to review spill management after every incident, no matter how minor. That approach not only prevents accidents but keeps teams learning and adapting.
Application Area
Through years in the lab and industry, I’ve watched 2-Cyclohexen-1-one weave its way from bench experiments to bulk commodity production. In pharmaceuticals, process chemists reach for it to create intermediates leading toward steroids or specialized antifungals. The fragrance world prizes its transformation into exotic musks or base notes, taking advantage of its complex, robust character. Researchers in advanced materials chemistry seek out its reactivity for new polymers, coatings, and cross-linked structures. Even undergraduate researchers get a taste for its flexibility in classroom exercises designed to introduce the intricacies of conjugated systems. Its role rarely shows up front and center in product advertisements, but those who dig into patents and academic papers see just how many pathways begin or branch off from this simple ring.
Research & Development
Progress in chemistry depends on curious, persistent researchers willing to get their hands dirty. Watching research trends develop, I’ve noticed that 2-Cyclohexen-1-one features in everything from exploratory reaction screens to highly optimized, multi-step syntheses. Universities and companies alike have ramped up efforts to use greener, cleaner methods for its preparation—electrocatalysis, selective biocatalysis, and alternative oxidants all find a seat at the table. Instrumentation advances now let analysts follow tricky intermediate formation in real time, improving both yields and selectivity. Interdisciplinary teams—combining organic, analytical, and process specialists—often revisit this molecule as they aim to optimize steps or troubleshoot persistent impurities. These efforts show that even classic molecules grow with us, responding to pressure for higher safety, better efficiency, and smaller environmental footprints.
Toxicity Research
Long before regulatory agencies required strict documentation, researchers and workers already knew that some chemicals—2-Cyclohexen-1-one among them—deserved caution. Early toxicity tests flagged its irritant properties, prompting the modern focus on low-exposure environments, careful storage, and swift response to accidental spills. Acute exposure brings headaches, coughing, and skin redness in unprotected users. Chronic effects receive attention in animal testing, which helps set occupational exposure limits and disposal rules. In community and worker education, transparent communication around chemical risk reduces anxiety and empowers people to act responsibly; simply putting up a sign does far less than open, ongoing dialogue. Toxicology studies keep evolving, with newer cell and animal models providing data on long-term risks, eco-toxicology, and breakdown products. This knowledge cycle—collect, review, update—keeps the science honest and helps protect both workers and the environment in a shifting industrial landscape.
Future Prospects
Looking into the coming years, practical experience tells me the demand for flexible intermediates such as 2-Cyclohexen-1-one won’t slow down. As pharma, fragrance, and specialty chemicals companies hunt for quicker, safer, and greener manufacturing, the need for robust building blocks stays strong. For research teams, the appeal of new transformations—driven by sustainable feedstocks, low-waste reagents, or continuous processing—creates incentives to revisit and rethink every step involving this molecule. Students will keep learning from its reactions, and regulatory changes will drive continued improvement in handling and disposal. With resourceful industry practices and conscientious science, this old standby will continue to serve as a bridge—connecting past, present, and future in the ongoing quest for better chemistry.
What is the chemical structure of 2-Cyclohexen-1-One?
Breaking Down the Molecule
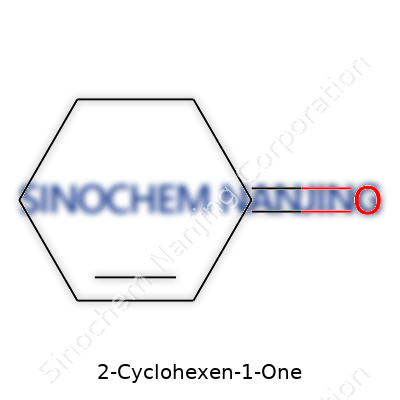
2-Cyclohexen-1-one looks simple at first glance, but like many organic molecules, its form tells a much bigger story. This compound belongs to the family of cyclohexenones, which people in both academic research and real-world industries cross paths with. The backbone consists of a six-membered ring that isn't quite as familiar as the classic cyclohexane you'll find in most basic chemistry sets. Instead, one double bond and a ketone (C=O) group transform this ring, making it far more reactive and interesting.
The carbonyl group on the first carbon and the carbon-carbon double bond between the second and third carbons work together to make the molecule more than just a ring with a twist. The atoms arrange themselves so that the planar nature of the carbonyl bond clashes a bit with the puckered ring. That angle between the carbonyl and the rest of the ring isn't just a quirky detail. It creates a hotbed for chemical reactions.
Why 2-Cyclohexen-1-One Stands Out
This molecule jumps out to researchers partly because it’s a neat example of conjugation. The double bond and the carbonyl group run just two carbons apart, letting electrons move between them. That small gap connects the chemical reactivity across the ring. The result is a molecule ready to take part in all kinds of syntheses, whether someone's building pharmaceuticals, studying biological processes, or designing next-gen materials.
Chemists who spend their days mixing and tinkering appreciate 2-cyclohexen-1-one mostly for its utility as a starting material. In the lab, it reacts in predictable yet versatile ways. Michael additions, Robinson annulations, and other familiar name reactions come to mind. Using this molecule, you can grow a simple ring into much bigger and complex frameworks. That flexibility matters when chasing new molecules for medicines or agricultural chemicals.
The Physical Reality
If you poured this compound into a glass vial, you’d see a yellowish oil. That color hints at the underlying chemistry, especially at the delocalized electrons in the structure. A molecule with that kind of electron flow tends to soak up certain wavelengths of light, sending out a unique hue. In practical terms, this color gives chemists a quick visual signal that the chemistry is working as it should.
With a relatively low boiling point around 155-157°C and a slightly sharp odor, the substance signals both volatility and the need for care. Safety data sheets warn about its irritant nature. Anyone working with it in the lab should wear gloves and goggles. Even something as seemingly minor as this oil reminds you of the real-world stakes behind each reaction.
Thinking About Impact and Solutions
2-Cyclohexen-1-one offers a launching pad for synthetic strategies in both research and industry. People making fine chemicals or testing new catalytic methods often partner with this molecule. Yet, as production and use go up, questions about environmental and personal safety follow. Chemists can’t just focus on output—they need to handle solvent recycling, minimize waste, and explore greener synthetic routes.
Teaching the next wave of scientists doesn't mean just explaining the molecular formula; it means emphasizing how these structures shape the world, for better or worse. Open discussions about alternative materials and safer reaction conditions help everyone keep progress ethical and sustainable.
What are the common uses of 2-Cyclohexen-1-One?
Essential Building Block in Organic Synthesis
2-Cyclohexen-1-one pops up almost everywhere in chemical labs and even large-scale manufacturing plants. For chemists, this compound ends up as a starting point for building much more complex molecules. Its structure—essentially a six-membered ring with a double bond and a carbonyl group—makes it attract a wide range of scientists dealing with synthetic organic chemistry, pharmaceuticals, and flavor industries. The presence of that carbonyl group allows researchers to add all sorts of atoms and groups onto it, helping them invent new molecules for medicine, agriculture, and advanced materials.
Key Ingredient for Pharmaceutical Development
Drug companies know 2-Cyclohexen-1-one as a reliable stepping stone when looking to create new compounds that combat disease. In drug research, it forms critical parts of active pharmaceutical ingredients, particularly those needing a cyclohexene backbone with various substituents. For example, derivatives of 2-Cyclohexen-1-one appear in early-stage screening for anti-inflammatory, antitumor, and antiviral properties. Scientists favor this molecule because it reacts smoothly with many common chemicals, letting them explore thousands of potential drug candidates rapidly.
Role in Fragrance and Flavor Chemistry
Step into the world of perfumes, and you’ll spot 2-Cyclohexen-1-one derivatives offering woody or musky base notes. Its subtle aroma makes it a popular intermediate in synthesizing flavoring agents and scented compounds. This cyclohexenone structure often opens doors to other fragrance molecules that aren’t easy to source from nature. The advantage? Consistency and reliability. Manufacturers can count on a stable supply, unlike natural extracts, which vary with climate and region.
Foundation for Agrochemicals and Dyes
Beyond medicine and fragrances, 2-Cyclohexen-1-one anchors many chemicals used in farms and textile industries. As a precursor, it leads to the production of pesticides, herbicides, and fungicides shaped specifically to target crop pests and weeds. In the field of dyes, the cyclohexenone core finds its place in the backbone of pigments and colorants used for plastics, inks, and fabrics. Its ability to bond with different functional groups lets chemists design stable, vibrant, and long-lasting colors.
Tool in Academic Research and Innovation
Academic chemists frequently turn to 2-Cyclohexen-1-one for teaching and discovery. Its straightforward reactivity and accessible cost make it a staple in laboratories. Students run experiments using this molecule to learn reaction mechanics, catalysis, and reduction techniques. Researchers engaged in finding greener chemical methods often test their ideas using this compound due to its familiarity and predictability.
Opportunities and Safety Concerns
Widespread usage brings responsibility. Handling 2-Cyclohexen-1-one safely makes a difference in both professional and academic settings. The chemical’s volatility and potential to irritate skin or eyes underline the need for solid protocols. As a writer who has seen both corporate and research labs, I know that clear safety labeling and proper ventilation protect everyone working with this compound. Moving forward, more research into sustainable production and waste recycling will address the environmental impact, making not only better chemistry but also a safer workspace for all involved.
Looking Ahead
The story of 2-Cyclohexen-1-one connects many fields: from cutting-edge pharmaceuticals to everyday products like scents and dyes. Its versatility speaks to human creativity in finding solutions with existing tools. Progress now relies on balancing innovation with safety and sustainability—goals that depend on sharing knowledge and supporting responsible research at every step.
What safety precautions should be taken when handling 2-Cyclohexen-1-One?
Understanding What You're Handling
2-Cyclohexen-1-One isn’t a household chemical you splash around without a second thought. It’s got a sharp, penetrating odor that serves as a built-in warning. That smell signals more than just a tough day at work; it signals risk. People who handle this compound in a lab or industrial setting come up against its effects fast. Exposure might feel like an itchy nose, watery eyes, or skin that stings for hours. Some even describe dizziness or headaches after breathing in its vapors for too long. So, anyone who spends time with it should respect both the chemical and the impact it delivers.
Personal Protective Gear Saves Real Skin
Forget the shortcut of working with bare hands. I learned early that wearing nitrile or neoprene gloves matters, not just latex. 2-Cyclohexen-1-One eats right through weaker gloves and leaves your skin exposed. Clothing choices count as much as gloves. A sturdy lab coat or disposable gown covers your arms and stops splashes from burning your skin. Chemical splash goggles won’t win any fashion points, but the shield they provide to your eyes lasts long after that momentary annoyance of fog or fit. Breathing this chemical’s fumes isn’t just uncomfortable. Over time, inhaling these vapors wears you down—leading to chronic irritation or worse.
Ventilation is More Than a Recommendation
No fan on a windowsill or cracked door cuts it with 2-Cyclohexen-1-One. Chemical fume hoods or proper extraction fans pull dangerous vapors away. I watched colleagues resist proper ventilation, then wind up with headaches that just wouldn’t quit—until they took this step seriously. Respirators rated for organic vapors become necessary if you have no access to a hood. One day spent working without one taught me a lesson that stuck. If a spill happens, leaving vapors unchecked in an enclosed space can build up to hazardous levels before you even notice something’s wrong.
Storage: Not Just Stashing Bottles on a Shelf
I once stored a bottle of 2-Cyclohexen-1-One next to acids out of sheer convenience, then spent days cleaning up a leak that scorched my sense of smell for weeks. Flammable storage cabinets work for a reason. This chemical burns, and storing it near oxidizers or acids creates a recipe for disaster. Tight-sealing containers with clear labels keep everyone on the same page. Never keep more than you truly need on hand, because every extra milliliter sits waiting to cause trouble.
Spill Response: Fast Action Beats Panic
A beaker tips, liquid runs everywhere. That freezing moment can mean the difference between a minor inconvenience and a serious medical emergency. You deploy an absorbent pad, then scoop up the remains with gloves on. Emergency showers and eyewash stations within a few steps of your workspace make cleaning up far smoother. If the spill spreads too far, don’t play hero—evacuate and call trained responders. Properly reporting and reviewing every incident strips away confusion during the next emergency, training everyone to act with muscle memory, not guesswork.
Training and Team Communication
Working alone or uninformed makes every one of these safety steps useless. Regular safety drills and keeping updated safety data sheets in sight saved more than a few of my former coworkers from making dangerous mistakes under pressure. Every team member brings value when they know the risks and stay prepared to act. Open conversations, lessons learned from near-misses, and respect for the chemical’s power help form habits that last through any shift.
What is the boiling and melting point of 2-Cyclohexen-1-One?
Real-World Data, Real-World Benefits
Chemists know the value of boiling and melting points on a level deeper than reference books suggest. For 2-Cyclohexen-1-one, an organic compound tucked into many synthetic strategies, the boiling point lands around 156-158°C, and the melting point usually hovers close to −38°C. These figures might seem dry on the page, but they guide daily practices in chemical labs and industries across the globe.
The Practical Significance in Lab and Industry
Take the boiling point. At around 156-158°C, 2-Cyclohexen-1-one offers a lot more than a technical hurdle. This range sets the operating window for distillation, purification, and safe handling. An organic chemist working at the benchtop always considers how volatile something is—if a product boils away at a low temp, you lose it, ruin your yield, or risk exposure. This is not just about efficiency; safety sits right beside. A boiling point in this range means you don’t need high-pressure setups, but you also avoid the fire risk seen in more volatile organics.
The melting point, dipping well below zero at −38°C, tells another story. It says this compound will be a liquid in most environments, even on a chilly northern winter day. Low melting points can complicate storage or handling in open containers for extended periods, but the upside is flexibility in solvents and compatibility with liquid-phase reactions. For people scaling up syntheses or running reactions in sequence, these features translate to lower costs, fewer hazardous procedures, and smoother production.
Boiling and Melting Points Steer Chemical Design
At the bench, those numbers help chemists shape strategies. Imagine measuring out a reagent. If it’s a low-melting solid, you chip a bit off, wait for it to liquefy, or warm it gently. If it flows at room temp, less fuss, quicker prep, and less chance of hazardous dust or accidental inhalation. With 2-Cyclohexen-1-one staying liquid in most lab conditions, it blends easily into solvent systems or reaction setups without extra steps.
In production, boiling and melting points feed directly into risk assessments. Plants using 2-Cyclohexen-1-one keep equipment and storage tanks at specific temperatures because they know the numbers. Any mistake—heating past boiling or chilling below freezing—can cause pressure build-up, leaks, or crystallization that blocks lines. Professionals rely on accurate, up-to-date data from trusted references like Sigma-Aldrich, Reaxys, or PubChem, all of which reaffirm these values.
Pushing Forward with Data-Driven Handling
Companies handling large volumes of 2-Cyclohexen-1-one face strict rules to prevent leaks or reactions gone wild. The boiling and melting points go straight into every safety data sheet. They help draft the protocols that keep workers safe. Those numbers even affect insurance rates and compliance with environmental standards.
There are always ways to improve how chemicals are managed. Controlling the environment, employing reliable temperature sensors, and having backup containment for spills mark the difference between a near-miss and a disaster. For an organic molecule like this, where reactivity and volatility play big roles, careful tracking of phase changes ensures operations stay smooth. Adjusting procedures according to the latest scientific data, ongoing safety training, and upgrading equipment deliver long-term benefits to labs and factories. These practices remind us that behind every number in a chemical handbook, a lot of hard-earned experience shapes the best way forward.
How should 2-Cyclohexen-1-One be stored to maintain stability?
Real-World Risks with 2-Cyclohexen-1-One
Few people outside research circles ever hear the name 2-Cyclohexen-1-One. Still, every time a chemist reaches for this chemical in the lab, misuse or poor storage threatens not only the compound’s effectiveness but also safety. Over years of working with reactive organic chemicals, I learned quickly that ignoring storage guidelines invites disaster, sometimes even before opening the container. At first glance, it seems like another routine bottle on the shelf. Yet exposure to heat or light changes more than just the makeup of the compound—it can shift outcomes, whether in a research setting or a manufacturing line.
Why Environment Matters
Any storage area worth using for 2-Cyclohexen-1-One has to fight light and heat. Experience told me that leaving the bottle near a sunny window—even for an afternoon—meant rolling the dice with both stability and purity. Commercial labs aren’t immune, either. Sometimes warehouses overlook the importance of steady, cool temperatures. The sweet spot sits below room temperature, usually between two to eight degrees Celsius. These temperatures help slow decomposition and side reactions. Fail at maintaining them, and the results often show up as strange odors, color changes, and unscheduled cleanups.
Sealing and Safety Precautions
Contamination often sneaks in from the most obvious source: air. Leaving caps loosely fitted, or reusing a crusty, old seal, means that every time you open the container, tiny amounts of moisture and oxygen join the party. Over time, these intruders degrade the chemical, sometimes producing peroxides. I once learned the hard way that peroxide formation does more than waste resources—it can spark hazardous reactions if left unchecked. Sturdy, airtight seals keep the drama at bay. Investing in amber glass bottles goes a long way, too. Amber cuts out much of the ultraviolet light that causes 2-Cyclohexen-1-One to break down before its time.
Labeling and Inventory Control
Nothing replaces a clear, honest label. In shared spaces, I’ve seen bottles pass from hand to hand, losing track of the purchase date, or even identity. A simple fix can be as unglamorous as keeping a sharpie nearby and writing down the open date. Freshness counts. After a few months, even under ideal conditions, quality can drift. When in doubt about the age or integrity of the contents, it helps to run a quick check using reliable analytical tools like NMR or GC-MS—two methods widely trusted in the chemical community for spotting unwanted impurities.
Handling Unexpected Spills and Routine Checks
Accidents always find their way in busy labs. Treat even a small puddle of this compound with care. Spills should get neutralized immediately and the area aired out, since vapors irritate eyes and the respiratory tract. Every lab or chemical storage room benefits from regular audits—routine checks of storage temperatures, condition of seals, bottle labeling, and the state of inventory. Skipping these steps can turn minor neglect into bigger headaches featuring both safety hazards and wasted investment.
What Works and What Still Challenges Us
The simple habits—cool storage, dark spaces, airtight containers, and honest labeling—continue to keep things running smoothly with 2-Cyclohexen-1-One. While automated sensor technology for fridges and improved bottle designs help, it’s daily human vigilance that makes the difference. Whether you’re in charge of a teaching lab or an industrial bulk warehouse, ongoing attention and clear protocols lower the risks and protect both people and product.
| Names | |
| Preferred IUPAC name | 2-cyclohexen-1-one |
| Pronunciation | /tuː saɪ.kloʊˈhɛk.sən wʌn oʊn/ |
| Identifiers | |
| CAS Number | 930-68-7 |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:19079 |
| ChEMBL | CHEMBL15329 |
| ChemSpider | 80715 |
| DrugBank | DB01939 |
| ECHA InfoCard | 100.011.132 |
| EC Number | 1.3.99.10 |
| Gmelin Reference | 69738 |
| KEGG | C01836 |
| MeSH | D003474 |
| PubChem CID | 7929 |
| RTECS number | GV7875000 |
| UNII | E695SDB53G |
| UN number | UN2312 |
| CompTox Dashboard (EPA) | DTXSID2034374 |
| Properties | |
| Chemical formula | C6H8O |
| Molar mass | 96.13 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | sweet, pungent |
| Density | 0.953 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.51 |
| Vapor pressure | 0.93 mmHg (25°C) |
| Acidity (pKa) | 19.3 |
| Basicity (pKb) | 7.75 |
| Magnetic susceptibility (χ) | -48.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | 2.56 mPa·s (25 °C) |
| Dipole moment | 2.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 312.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -45.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3084 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | `GHS02,GHS07` |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 58 °C |
| Autoignition temperature | 435 °C |
| Explosive limits | Explosive limits: 1.1–9.4% |
| Lethal dose or concentration | LD50 oral rat 370 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 500 mg/kg |
| NIOSH | GC8225000 |
| PEL (Permissible) | PEL: 50 ppm (OSHA TWA) |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | Unknown |

