2-Cresol: From Factory Floor to Frontier Research
Historical Development
Plenty of modern chemicals have roots in the heady days of industrial expansion, and 2-cresol carries that legacy. Chemists isolated cresols from coal tar long before the word "synthetic" dominated the lab. Early uses were pragmatic—disinfection, preservation, and a fast route to salicylic acid. Coal tar, oozing out of coking plants and gas works, presented raw material and headache at once. In crowded late-1800s Europe, scientists tackled cresols both to tame infection risks and squeeze value from waste. As techniques sharpened, industries learned to pull out the different cresol isomers: ortho, meta, and para, each with its quirks. The ortho form, 2-cresol, stood out for its chemical flexibility and sharp, phenolic odor, a far cry from any perfume—but a reliable building block for a changing world.
Product Overview
Anyone regularly working with solvents or disinfectants will recognize the sharp note of cresol. This chemical doesn’t look much: a colorless to pale-yellow liquid, sometimes with smoky tints. Its industrial role, though, can't be written off. 2-cresol sits as an intermediate in making resins, herbicides, and pharmaceuticals. It helps synthesize antioxidants and dyes, pop up in cleaners, and keeps cutting fluids from going rotten. In paint and coating world, it helps tailor drying times and surface finishes. Laboratories see it as a reagent more than a star, but in industry, it oils the gears of manufacture and innovation.
Physical & Chemical Properties
2-cresol doesn’t hide its chemical lineage. With a boiling point around 191°C, a melting point of about 31°C, and a respectable solubility in organic solvents, this compound slips between solid and liquid, water and oil. Under the hood sits a phenolic ring with a methyl group hanging off at the ortho position. That little tweak changes reactivity, giving it a different bite compared to phenol itself. That’s what makes it valuable: you want the familiar acid of phenol but need slightly different handling or reaction speed. Its pungent odor warns of its presence. It burns, mixes with water at low concentrations, and stands up to standard shelf conditions, though it will oxidize in air to give off that familiar medicinal scent.
Technical Specifications & Labeling
Anyone who’s rolled a drum of 2-cresol knows safety rules come first. Whether it's labeled by its CAS number 95-48-7 or its robust red diamond pictogram, the risks demand respect. Industries usually trade in technical or analytical grade—purity, moisture content, and residue matter when the product ends up as a pharmaceutical precursor. Labels read “2-methylphenol” almost as often as “2-cresol,” depending on geography and tradition. Regulations want clear hazard information, exposure limits, and compatibility warnings, not to coddle but because more than a century of experience shows what happens when corners get cut.
Preparation Method
Old-timers still recall the rich, tarry stink of the first fractions pulled from coal tar. That’s where cresols were born before petrochemicals offered synthetic routes. These days, production tends to rely on methylating phenol or refining through the selective distillation of coal tar’s complex brew. This approach continues because, despite decades of petrochemical innovation, it remains cost-effective and scalable. Catalysts, temperature regimes, and careful controls regulate yield and minimize byproducts. The choice between coal tar distillation and synthetic methylation can hinge on local infrastructure, feedstock cost, and downstream market demands.
Chemical Reactions & Modifications
Chemists keep coming back to 2-cresol for both its stubbornness and its willingness to transform. That little methyl group near the hydroxyl sets off unique patterns in substitutions and couplings. Nitration produces ortho- and para-nitrocresols, useful for dyes and explosives. When it goes through alkylation or sulfonation, factories bring out compounds for antioxidants and detergents. It doesn’t shy away from halogenation, either, giving rise to materials needed for wood preservatives and fungicides. These reactions matter not just in the fine chemicals world, but also in day-to-day essentials from crop protection to plastics.
Synonyms & Product Names
Language shifts around chemicals faster than many realize. 2-cresol might show up as o-cresol or 2-methylphenol, and if you’re searching in vintage documentation, “hydroxytoluene” may appear. Nomenclature often flips depending on which camp you stand in—manufacturing, scientific research, or regulation. Some brands lean on “ortho-cresol” to lock in the isomer, knowing that purity requirements started as a problem for dyes and now matter for electronics and pharma. Staying sharp on these names helps avoid mix-ups; mistakes can cost far more than embarrassment.
Safety & Operational Standards
Many chemicals mask their risks behind bland appearances, but 2-cresol lets you know it means business. Anyone handling it, even in a well-ventilated space, knows eye and skin irritation come fast, and inhaling vapors could lead to headaches, dizziness, or worse if you’re unlucky. Safety data sheets recommend goggles, gloves, aprons, and proper storage away from oxidizers. Disposal isn’t as simple as a trip down the drain—environmental rules demand incineration or licensed handling. Regulatory agencies from OSHA to REACH insist on exposure limits and monitoring, a testament to lessons learned the hard way on shop floors and in labs. Training, not just equipment, builds a safety culture capable of keeping incidents rare.
Application Area
Those who work in specialty chemicals or agriculture have likely run up against a use case for 2-cresol. In its earliest days, it kept hospitals cleaner and preserved wood from decay. Now, it anchors synthesis for herbicides and disinfectants, showing up in everything from cleaners to pharmaceuticals. Resin and plastics manufacturers depend on it for intermediates that give products the desired rigidity, flexibility, or resistance. As a reagent, it even has a role in small-scale organic labs training the next wave of chemists. Many laboratories use it in calibration or as a starting point for developing new analytical methods. None of these applications can afford to treat it as a mere commodity.
Research & Development
Industry never stands still, and neither does 2-cresol. While old-timers focus on bulk production, research teams keep spinning out new derivatives. The focus keeps shifting as market needs evolve. Green chemistry researchers aim to find catalysts that reduce waste, lower energy input, or use sustainable starting materials. In the pharmaceutical world, scientists tease out new ways to craft active molecules, sometimes building whole drug families from cresol scaffolding. Research into selective modifications continues to draw investment, especially for making electronics chemicals and advanced polymers. Collaboration between academic researchers and industry often spark breakthroughs that, years later, leave most people unaware 2-cresol sits somewhere in the background.
Toxicity Research
Toxicologists have scrutinized 2-cresol for decades. Early findings flagged acute toxicity if inhaled or ingested. Chronic exposure links to kidney, liver, and nervous system damage. Regulators took notice; occupational exposure limits became law in most industrialized countries. Researchers keep probing possible cancer links, reproductive toxicity, and how even low-level environmental exposure affects living things. Some studies show aquatic organisms are especially vulnerable, prompting wastewater limits and better technology for removing cresol before discharge. Yet the story is still evolving: new findings in molecular biology and environmental sciences may yet expose risks needing action.
Future Prospects
Looking to what lies ahead, 2-cresol faces a mix of opportunity and scrutiny. The push for greener synthesis methods could both reduce its environmental burden and expand its utility in specialized markets. Researchers exploring crop protection aim to use cresol derivatives that degrade safely and avoid bioaccumulation. Demand for advanced polymers and electronics chemicals also drives innovation—finding safer derivatives or more efficient synthetic routes puts chemists to the test. There’s talk in the regulatory world of ever-stricter exposure thresholds. That challenges traditional handling but spurs investment in process automation, containment, and safer-by-design chemistry. In the end, the chemical remains too useful to shelve outright, but the way it gets from lab and factory to final product will probably look very different ten years from now. The real test will be how industry, regulators, and researchers keep pace with the evolving science—and whether they ensure public trust along the way.
What is 2-Cresol used for?
Where 2-Cresol Shows Up
Growing up around workshops, that sharp, almost antiseptic smell always came from some chemical-based cleaners and wood preservatives stacked in dusty corners. Turns out, one of the main ingredients in those strong-smelling solutions is often 2-cresol. It’s a colorless or yellowish substance, and folks in all sorts of industries have leaned on it for over a hundred years.
2-Cresol in Everyday Products
Many people use products touched by cresol, maybe without even realizing. Manufacturers add it to disinfectants and antiseptics, which play a big part in hospitals and cleaning routines at home. Its strong ability to wipe out certain bacteria and fungi helps lower infection risks. Just the other day, I grabbed a heavy-duty cleaner from under the sink and a quick scan of the label listed cresol among the ingredients. Hospitals, too, use cresol-based solutions for cleaning surfaces. This isn’t about just killing germs, though; cresol breaks stubborn organic materials left on surgical tools and equipment, supporting the fight against hospital-acquired infections.
Protecting Wood and More
Wood preservation is another arena where 2-cresol pulls its weight. Nothing’s more frustrating than seeing a favorite deck rot, eaten by insects year after year. Pressure-treating wood with cresol-based preservatives makes wooden posts, sleepers, and fences last longer outside, even in harsh weather. Compared to leaving wood untreated, protective coats with cresol hold up well against fungus and termites.
The Chemical Industry’s Hidden Player
Big factories use 2-cresol as a starting block for other products. Chemical plants transform it into antioxidants and dyes, and it’s also a key ingredient in synthetic resins and plastics. These materials end up in electronics, car parts, and wire coatings—things we depend on for daily life. Agriculture leans on 2-cresol, too; pesticide producers use it to make certain herbicides and fungicides that help farmers save their crops.
The Downside: Health and Environmental Risks
All these uses come with responsibility. Breathing in cresol fumes at high concentrations irritates the throat and eyes and can damage the liver and kidneys over long stretches. Accidental spills in rivers or soil are toxic to fish and small wildlife. Personal experience on a job site showed me how one careless chemical leak can wipe out plants and insects in the patch of ground around it. Agencies like the EPA and OSHA step in to keep workplaces and neighborhoods safe by setting exposure limits and cleanup rules.
Balancing Benefits and Better Practices
People can’t ignore what 2-cresol brings to medicine, construction, and science, but safety should come first. Protective gear isn’t just a silly rule at work; gloves and face masks go a long way in keeping skin and lungs safe. When manufacturers improve their storage systems and educate staff, they prevent those costly leaks and spills before they happen. If you’re using cresol, know where to find a Safety Data Sheet and always follow the guidelines. Scientists look for greener alternatives, too, aiming to cut long-term risks while still meeting society’s needs.
Reflecting on the Future
Chemicals like 2-cresol won’t disappear from industry scenes overnight, but controlled use and practical invention can help keep health problems in check. My work and time in local communities have taught me how easy it is to take chemicals for granted, without noticing how they touch daily life or why we need to respect them. For now, common sense, careful handling, and a push for safer substitutes will keep people and the environment out of harm’s way.
What are the safety precautions when handling 2-Cresol?
Understanding the Risk
Working with chemicals like 2-Cresol feels familiar to anyone who has cleaned up a stubborn stain or tackled an engine leak. This substance, used in making disinfectants and resins, carries a heavy punch. It irritates the skin, damages the eyes, and stings the lungs if you breathe it in. In my years around labs and workshops, I found nothing keeps you safer than a blunt respect for what chemicals can really do—2-Cresol being no exception.
Personal Experience: Why Precaution Matters
I’ve seen what happens when safety rules get ignored. Colleagues have learned the hard way: minor spills turn into major emergencies without warning. Early on, I underestimated what a chemical like 2-Cresol could do. After a skin exposure left me with redness and a lingering burn, I overhauled my approach—never touching a bottle without gloves, never letting my mask slip. It’s easy to get comfortable, but this isn’t a chemical that forgives lazy habits.
Protective Gear Is Non-Negotiable
Gloves, goggles, and a lab coat aren’t just for show. Proper nitrile gloves shield the skin, while safety glasses stop splashes from sneaking into your eyes. Ordinary glasses won’t hold up. For those who transfer 2-Cresol or handle larger quantities, a face shield adds extra protection. Ventilated work areas lower the risk of breathing in dangerous fumes. Open a bottle in a cramped space, and you’ll know fast how strong those vapors get.
Good Habits Mean Fewer Accidents
Planning ahead saves trouble down the line. Anyone using 2-Cresol needs to know where the nearest eyewash station and safety shower sit. Spills can’t wait—grab a spill kit with absorbent pads, and clean up right away, wearing protective gear the whole time. Proper labeling stops confusion before it starts. I never set up my work without double-checking that every bottle clearly names its contents and hazard rating.
Ventilation and Clean Air
Anyone who’s worked with strong-smelling solvents will see the sense in a fume hood. The fumes from 2-Cresol irritate the throat and lungs in minutes. A working fume hood, or at least a well-ventilated space, makes all the difference. At home, opening windows wide is a bare minimum, but that only goes so far—industrial settings use real exhaust systems for a reason.
Storage and Disposal: Doing It Right
Keep 2-Cresol in a tightly sealed container, away from direct sunlight and strong acids. Heat or light makes it degrade, sometimes producing gases that corrode metal shelving. I always make sure to write storage instructions on each bottle. Waste disposal isn’t optional; 2-Cresol gets treated as hazardous waste, collected in marked containers, and sent to approved disposal centers—never poured down the drain.
Learning from Experience, Staying Prepared
Mistakes with 2-Cresol rarely get second chances. That creates a culture of vigilance. Newcomers and veterans alike benefit from regular safety training and refreshers. I keep an incident log and review it to spot patterns. If someone on the team slips up, we talk it through together, fixing the gap for next time. Keeping close to the facts, staying alert, and never letting routine become complacency—these habits save more trouble than any warning label ever could.
What is the chemical formula of 2-Cresol?
2-Cresol: What It Is and Why It Matters
2-Cresol shows up in my memory as that sharp-smelling bottle stashed on the bottom shelf of the chemistry lab. Plenty of us might remember its strong, medicinal scent. Chemically, this compound carries the formula C7H8O. It’s part of the cresol family, and this family comes from the basic skeleton of a benzene ring holding one methyl group and one hydroxyl group. It sounds simple, but the position of those groups changes everything. For 2-Cresol, also known as o-cresol, the methyl and hydroxyl groups sit right next to each other on the ring – a detail that shapes how it reacts, how it gets used, and even how it smells.
Chemistry Class Isn’t Just for Scientists
The world seems crowded with chemicals, but only a handful wind up mattering in real life. 2-Cresol is one of those. It’s found in coal tar, creosote, and can slip into antiseptics, cleaners, and some resins. Its sharp scent signals something strong, something that pushes bacteria out. Most folks don’t realize how many products lean on chemicals like this. I remember the pride in scrubbing a barn floor as a teenager, knowing the job got done right because the cleaner packed more than just good intentions–it had the punch of real chemistry.
The Impact of 2-Cresol on Health and Safety
This little molecule earns attention for another good reason. On one side, 2-Cresol works as a disinfectant and preservative in products that keep homes and workplaces clean. On the other side, there are risks. People can pick up skin or respiratory irritation from contact with it. The formula might look innocent, but the molecule doesn’t play around. Breathing too much or getting it on your skin leads to problems like headaches or burns. I’ve met folks in industrial settings who take those warnings seriously—nobody wants to learn the hard way about chemical safety.
According to the U.S. Public Health Service, constant exposure at high levels can harm organs such as the liver, kidneys, or lungs. Chemical spills involving cresols have forced evacuations in small communities because clean water and air come before anything else. Regulations step in because experience proves that safety should never be treated as an afterthought.
Connecting Chemistry With Real Solutions
Thinking about chemical compounds often gets lost in numbers and formulas, but real lives stand behind those formulas. Proper labeling, ventilation, and storage keep accidents from happening. The chemistry behind C7H8O—2-Cresol—reminds us to keep learning. Teachers who look past rote memorization and link chemical properties to health, environmental impact, and industrial use make a difference. Science isn’t just information for experts—knowledge about chemicals like 2-Cresol helps keep every community safer, healthier, and more prepared for whatever lands in tomorrow’s headlines.
Is 2-Cresol hazardous to health?
Understanding 2-Cresol and Its Uses
2-Cresol, a type of methylphenol, shows up in different chemical industries. It often gets used for resins, disinfectants, and as a preservative in some settings. In my time studying workplace safety, I’ve seen how chemical companies rely on its properties, but that use comes with a big responsibility. People dealing with 2-Cresol don’t always think about the risks involved, especially during storage and handling.
Health Effects on Exposure
Anyone who’s worked around strong chemicals knows that a lot of what you can’t see or smell can still harm you. With 2-Cresol, contact brings quick consequences. Skin feels irritation, and breathing vapors can make your throat and nose burn. A big spill or careless transfer might even cause burns or blisters if it touches your skin undiluted.
The scariest part kicks in once it gets into the bloodstream—either through the skin or if someone breathes in high concentrations. Short-term contact makes you feel nauseous and dizzy and might give you a bad headache. Longer exposure, which I’ve witnessed in older labs with poor ventilation, can stress the liver and kidneys. Documented medical cases link extended high-dose contact with organ damage. There’s always a risk of chronic effects when any chemical cleansers or solvents get tracked into common areas or absorbed into protective gear.
Reliable Science Weighs In
Regulatory groups, from public health agencies to research universities, spell out the risks plainly. The National Institute for Occupational Safety and Health (NIOSH), for example, lists 2-Cresol as toxic by inhalation, skin absorption, and ingestion. The Centers for Disease Control and Prevention (CDC) highlights its corrosive nature — burning and blistering skin without a lot of warning. Both long-term and high-concentration exposures have been connected to central nervous system problems and increased cancer risk in laboratory animals, which brings up the question of what happens to people over time.
How People Can Protect Themselves
Back in my earliest lab days, gloves and goggles weren’t always a priority on the factory floor. These days, strong policies about personal protective equipment (PPE) set a strong example. I’ve seen lives saved and medical visits avoided thanks to consistent re-training and proper gear — even in small workshops. Employers who provide solid ventilation, proper glove selection, and clear emergency showers make a huge difference in outcomes. Effective storage practices keep containers sealed and shield the wider community from accidental leaks.
Simple safety habits go a long way, too. Quick handwashing and avoiding other chemicals when working with 2-Cresol lowers chances of unexpected skin reactions or dangerous fume mixes. Good workplaces teach people how to find a safety data sheet and what to do if a spill happens. If more chemical users demanded cleaner, safer alternatives, progress would move faster, and cleaner substitutes might rise to the top.
Looking for Better Answers
Some problems don’t get fixed by wishful thinking. Cleaner production tech has cut a lot of the worst exposures over the last decade, but there’s room for better solutions. Research focuses on less toxic preservatives and greener solvents, and employers can stay ahead by keeping up with the latest guidance. Anyone handling chemicals at work or at home learns pretty fast that the right information, protective gear, and good habits spell the difference between a safe return home and a dangerous mistake.
How should 2-Cresol be stored and disposed of?
Why 2-Cresol Calls for Careful Handling
Some people run across 2-Cresol in labs, factories, or during industrial research. This chemical doesn’t belong on an open shelf or in an unlocked closet. Inhaling its fumes or letting it touch your skin isn’t safe, which means everyone needs a real plan for storage and disposal.
Best Storage Practices for Safety
On the job, I've seen facilities treat every potentially dangerous chemical with the utmost respect, and 2-Cresol deserves that respect. Store it in closed containers made of steel or glass, and mark the label clearly. A flammable cabinet away from direct sunlight and sources of heat keeps accidents at bay. Not every workplace maintains ideal temperatures, but with 2-Cresol, stable room temperature matters to keep the chemical from breaking down.
Even a strong lock on the door sometimes fails to stop the wrong hands, so a dedicated storage area with restricted access does the trick. Only those trained on how to handle chemical spills should grab that key. Proper ventilation lies at the core of good chemical storage. Even though nobody wants to deal with a leaking container, proper ventilation can help limit harm if one does give way.
Aware and Equipped: People Make the Difference
Handling training stays key. In my experience, the folks who actively check container seals and monitor expiration dates keep everyone safer. A shelf life isn’t just a suggestion with 2-Cresol; it actually affects stability and safety over time.
It pays to load up on reliable personal protective equipment: gloves made for chemical resistance, goggles, a face shield for splash risk, and a sturdy apron. Accidents with 2-Cresol can lead to serious burns and breathing problems, so shortcuts aren’t worth it.
Smart Disposal: More than Just Toss and Go
Throwing 2-Cresol in the regular trash or pouring it down the drain turns a workplace threat into an environmental crisis. Many major countries list this chemical as hazardous waste, and authorities don’t give exceptions. Nobody wants to end up with a fine or, worse, cause harm to their community’s water or soil. Filters, absorbents, and containers used with 2-Cresol become hazardous themselves—so always collect and manage them carefully.
I’ve seen companies work closely with licensed hazardous waste haulers. These professionals know how to neutralize and contain 2-Cresol, keeping the broader community safe. In some cases, specialized incineration at high temperatures gets used to fully break down the compound. Never mix it with other chemicals unless you’ve consulted an expert; unwanted reactions can make things worse.
Building a Culture of Responsibility
Good habits spread through a team. Leadership sets the tone, but everyone needs the know-how to act quickly in the event of a spill. Regular training drills, inspections of storage areas, and transparent rules keep people alert. Safe storage and disposal practices for 2-Cresol reflect a broader commitment to environmental responsibility and workplace safety.
Long-term, better chemical management usually pays for itself. Safer communities, healthier workplaces, and fewer emergency calls show what good chemical stewardship really delivers.
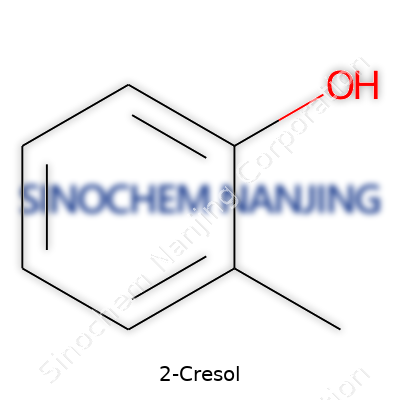
| Names | |
| Preferred IUPAC name | 2-Methylphenol |
| Other names |
o-Cresol 2-Methylphenol o-Hydroxytoluene |
| Pronunciation | /ˈtuːˌkriːsɒl/ |
| Identifiers | |
| CAS Number | 95-48-7 |
| Beilstein Reference | 1363380 |
| ChEBI | CHEBI:16998 |
| ChEMBL | CHEMBL1437 |
| ChemSpider | 546 |
| DrugBank | DB03752 |
| ECHA InfoCard | 100.029.278 |
| EC Number | 200-431-6 |
| Gmelin Reference | 822 |
| KEGG | C01406 |
| MeSH | D002454 |
| PubChem CID | 7048 |
| RTECS number | GO6475000 |
| UNII | N8D3KQ11L1 |
| UN number | UN2076 |
| Properties | |
| Chemical formula | C7H8O |
| Molar mass | 108.14 g/mol |
| Appearance | Colorless to yellowish crystalline solid or liquid |
| Odor | Phenolic |
| Density | 1.05 g/mL at 25 °C |
| Solubility in water | 24 g/L (20 °C) |
| log P | 1.96 |
| Vapor pressure | 0.11 mmHg (at 25 °C) |
| Acidity (pKa) | 10.26 |
| Basicity (pKb) | 10.29 |
| Magnetic susceptibility (χ) | -715.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5392 |
| Viscosity | 2.52 mPa·s (20 °C) |
| Dipole moment | 1.94 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −29.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3343.0 kJ/mol |
| Pharmacology | |
| ATC code | D08AE01 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS05, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H332, H373 |
| Precautionary statements | P261, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-2-0-Acad |
| Flash point | 86 °C |
| Autoignition temperature | 540°C |
| Explosive limits | 1.3% - 7% |
| Lethal dose or concentration | LD50 oral rat 207 mg/kg |
| LD50 (median dose) | LD50 (median dose): 121 mg/kg (oral, rat) |
| NIOSH | B016 |
| PEL (Permissible) | 5 ppm |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Phenol 4-Cresol Benzyl alcohol Benzaldehyde Benzene o-Toluidine |

