2-Chloronitrobenzene: Uses, Hazards, and where Chems Go Next
The Long Trail of 2-Chloronitrobenzene
2-Chloronitrobenzene didn’t just drop into labs from nowhere. People started making it in the late 1800s, a time when chemists figured out you could add nitro and chloro groups to benzene rings and open up an entirely new toolbox for synthetic work. It’s always been a building block—something you put together with other pieces, never the centerpiece of the puzzle. Early on, dye makers grabbed it for its promise in making vibrant pigments. Over time, its reach spread into pharmaceuticals, agrochemicals, and all sorts of specialty chemicals. The story behind this compound reminds me that what seems just a stepping stone for one generation becomes a whole economic sector for the next.
Product Overview: Underappreciated but Essential
Nobody writes poems about 2-Chloronitrobenzene. It’s a pale yellow solid with a sharp, pungent odor. To the untrained eye, it’s just another slightly dangerous bottle on a dusty shelf. To industry, it’s vital. Companies use it for synthesizing downstream chemicals—think o-chloroaniline, which runs off into making rubber chemicals, pesticides, and dyes. This chemical fits right into the classic toolkit for anyone running large scale aromatic substitutions or trying to build a more complex scaffold. In my own lab days, it felt as reliable as a ball-peen hammer: unglamorous, but you’d miss it badly if it vanished.
Physical and Chemical Properties: Tough, Stubborn, and a Bit Nasty
Take a whiff—briefly, and only if you want a memory you can’t wash out. Boiling point, melting point, density—these numbers paint a picture of a tough aromatic ring that holds onto its functional groups like a bulldog. It’s only somewhat soluble in water, but dissolves easily in organic solvents. You look at the molecule and see a benzene ring, a chlorine atom, and a nitro group all jockeying for attention. Its ortho position matters—a seemingly small structural change can flip its reactivity or toxicity. In my chemistry days, bad handling meant ruined gloves and a smell that lingered. Too many people underestimate how the physical realities of certain chemicals force you to respect them, no matter how clever your synthetic plan looks on paper.
Technical Specifications and Labeling: Precision Over Hype
Handling this chemical safely requires knowing exactly what you’re dealing with. Most supply bottles list purity (often ≥99%), moisture content, melting point, and impurity profiles—essential details, not just for regulatory compliance, but for reproducibility in reactions. Labeling matters: hazard pictograms, recommended PPE, and risk phrases aren’t there to tick legal boxes; they save lives. I once saw a synthesis grind to a halt because a batch had too much o,p-isomer, gumming up the outcome. Nobody forgets that lesson. Upfront, clear information on a label lets chemists plan better and avoid ugly surprises.
Preparation Method: Industrial Chops and Chemical Know-How
Manufacturers typically fire up nitration of o-chlorobenzene by mixing it with nitric and sulfuric acids, kicking off an electrophilic aromatic substitution that’s hotter, trickier, and smellier than it sounds. Tight temperature control is critical—go too cold, you under react; go too hot, overnitration creates a stew of byproducts. Getting high selectivity for the ortho-nitro product, avoiding meta- and para-isomers, calls for operators who know their craft and their plant’s quirks. Poor separation or sloppy distillation can lead to products that are hard to purify downstream, sending off yields, efficiency, and margins in one easy misstep. Running these reactions at scale gives a true feel for chemical engineering, not just labwork on a shaker hotplate.
Chemical Reactions and Modifications: Many Roads Out
2-Chloronitrobenzene doesn’t just sit in a bottle waiting to be catalogued; it’s a starting point. Chemists draw on nucleophilic aromatic substitution to swap out the chlorine for amines, phenoxides, thiolates, and other nucleophiles. The nitro group can be reduced—chemically or catalytically—to an amine, releasing yet more synthetic options. Every modification leads somewhere else: rubber accelerators, veterinary drugs, corrosion inhibitors. In my experience, the real utility of this compound is versatility. You don’t need to reinvent the wheel, just bolt on new spokes where needed.
Synonyms and Product Names: Speaking the Many Languages of Chemistry
Look up 2-Chloronitrobenzene and you’ll see names like o-Chloronitrobenzene, 1-chloro-2-nitrobenzene, or just OCNB. This alphabet soup confuses newcomers, but actually reflects decades of competing nomenclature systems. Older chemists swear by their own short-hands. Fluency in these synonyms separates seasoned operators from those reaching for the index every five minutes. Having the right chemical but the wrong name wastes time, risks errors, and, worst of all, makes collaboration harder.
Safety and Operational Standards: No Room for Sloppiness
2-Chloronitrobenzene won’t forgive carelessness. It’s toxic by inhalation and skin contact, and prolonged exposure can lead to everything from skin irritation and eye burns to headaches, cyanosis, or nervous system effects. In regulated labs or plants, proper fume hoods, gloves, eye protection, and spill protocols keep incidents rare, but not impossible. I’ve seen poor practice result in trips to the medic. Modern safety data sheets don’t exist just to meet OSHA and REACH standards—they capture the lessons written in scars and near-misses. The best safety culture catches not only obvious missteps, but also the subtle lapses: labeling, incompatible wastes, storage off the floor, and working upwind of anything pungent.
Application Area: From Dyes to Drugs—and the Shadows Between
2-Chloronitrobenzene threads through multiple industries. Dyers leverage its chemistry for brilliant yellows and greens. Agrochemical firms harness its reactivity to add protection to fields at scale: making herbicides like phosalone starts here. Pharmaceutical researchers start with it to build chunks of painkillers, antihistamines, or veterinary drugs. Its intermediacy shows up in specialty rubber chemicals, blowing agents, and corrosion inhibitors. Each downstream sector finds its own value in the molecule, even finding use in less obvious areas like photographic chemicals and electroplating. In my days working in contract synthesis, the real surprise came with requests that sent this humble chemical off in entirely new directions.
Research and Development: Small Tweaks for Big Results
Chemists keep probing ways to use 2-Chloronitrobenzene more efficiently. Improvements focus on greener processes—changing solvents, lowering temperatures, exploring better catalysts, or recycling spent acids. Recently, researchers started pushing for bio-based or electrochemical methods for both nitration and subsequent modifications, hoping to cut down waste and energy consumption. Academic groups investigate alternative routes, not always for immediate industry use, but to build a foundation for the future. My own work on catalyst screening for cross-coupling found this compound stubborn in some reactions, yet surprisingly reactive in others when conditions aligned just right. The ongoing search tests both patience and creativity.
Toxicity Research: Balancing Value Against Harm
Nobody wants to sing the praises of 2-Chloronitrobenzene toxicity. Animal studies point to concerns over acute and chronic exposure—blood changes, liver effects, and possible mutagenic action. Environmental persistence raises flags: spill it or discharge waste carelessly, and it lingers in soil and water. Setting workplace exposure limits is a running battle between productivity and caution. Old stories about the “blue fingers” of nitration plant workers—caused by methemoglobinemia—remind us that industrial progress leaves trails in human experience, not always positive ones. Regulators keep raising questions and shifting the standards. Better monitoring, improved PPE, and a push for less hazardous processing have improved safety so far but won’t solve the dilemma entirely. In many ways, the greater challenge isn’t the molecule, but the systems built around it.
Future Prospects: Changing How and Why We Make This Stuff
The next chapters aren’t set in stone. Green chemistry enters the conversation: lower-impact syntheses, biocatalytic approaches, or even pathways that skirt around chlorination and nitration altogether. Though demand remains steady in legacy sectors—dyes, rubber, agriculture—pressure builds to reduce risks and environmental footprint. Circular chemistry, where waste becomes feedstock, sits just over the horizon. Governments and regulators drive changes through tighter controls and public scrutiny of what leaks into air, soil, and water. Insider knowledge whispers that even small shifts in global regulatory landscapes can upend supply, raise prices, and force companies to scramble for alternatives. At some point, substitution or elimination could gain ground as greener, safer pathways come online. Watching those trends in action teaches that chemistry doesn’t stand still. Today’s standard becomes tomorrow’s problem—or solution.
What are the main uses of 2-Chloronitrobenzene?
Understanding Where 2-Chloronitrobenzene Ends Up
Factories keep making 2-Chloronitrobenzene for one big reason: it serves as a handy building block. If you peek behind the labels in a typical chemical plant, dozens of things owe their existence to this compound. That starts with dyes, goes through pharmaceuticals, and ends up right at pesticides and rubber chemicals. My own experience working in the chemical supply chain taught me how one ingredient feeds a tangled web of industries.
Dyes and Colorants Lead the Way
Textile dye plants keep the demand for 2-Chloronitrobenzene steady. Workers there take it and transform it into intermediates for azo dyes—think bright reds, deep blues, and earthy browns you see in shirts and carpets. Just a small percentage of the compound ends up in a final product, but tweaking the color palette for next season relies on these intermediates. Without strong intermediates, dyes can fade fast with washing and sunlight. Reliable color in fabric isn’t just nice to have. It’s non-negotiable for global brands.
Medicines Trace Back to Simple Chemicals
Everyday painkillers and antibiotics get their start in places few consumers imagine. During my stint consulting on pharmaceutical sourcing, project managers often asked for assurances about the availability of 2-Chloronitrobenzene. Along the way, chemists turn it into important molecules like 2-chloroaniline. This one goes into making drugs that fight infections and inflammation. Some antihistamines and blood pressure medicines depend on this chemical family, so health outcomes can tie back to stable supplies.
Pesticides and Crop Protection
Farmers fighting bugs and weeds lean on modern crop science. Much of that science depends on chemical precursors, including 2-Chloronitrobenzene. Manufacturers use it to whip up herbicides like butachlor. Without herbicide intermediates, fields would be far less productive and food prices would soar. Critics of synthetic crop protection often forget that pest outbreaks can cripple harvests. Synthetic chemistry enables solutions on a huge scale that natural extracts rarely match.
Specialty Applications
Rubber products hide an overlooked role for 2-Chloronitrobenzene. Rubber factories use it in making vulcanization accelerators, which give tires and industrial rubber goods longer life. Car safety depends on tires gripping the road, no matter if it’s raining or sizzling hot. Sturdy tires, conveyor belts, and gaskets all start with chemicals like this.
Specialty chemicals outside these big groups also matter. Companies making photographic chemicals, corrosion inhibitors, and certain plastics occasionally turn to 2-Chloronitrobenzene to solve specific problems. For example, treating galvanized steel for construction needs compounds that help protect against rust and weather.
Risks and Responsible Handling
Dealing with strong chemicals means safety takes the spotlight. Exposure to 2-Chloronitrobenzene can harm health, affecting skin and sometimes causing more serious symptoms after inhalation. My colleagues in plant operations recall strict safety drills. Safe handling saves lives, and companies have a responsibility to keep workers informed and supplied with proper gear. In the past, some plants struggled with spills, so government rules now force better monitoring and storage methods.
Looking for Safer Alternatives
Interest in green chemistry solutions continues to build. Research teams test processes that use less hazardous chemicals or cut waste. Some dye and pharma companies now funnel R&D budgets into finding less toxic intermediates. Still, changing an entire supply chain takes time, careful study, and lots of real-world data. Until then, 2-Chloronitrobenzene stays important to several sectors even as safer replacements get developed and tested.
What is the chemical formula and molecular weight of 2-Chloronitrobenzene?
The Simple Details: Chemical Formula and Molecular Weight
The formula for 2-Chloronitrobenzene is C6H4ClNO2. Its molecular weight stands at 157.56 g/mol. Plenty of chemists know these numbers offhand, but it's the practical side where they really start to matter.
The Real-World Impact of These Numbers
Fact is, having the correct formula and molecular weight makes life smoother for everyone who works with this substance. Any time I walk into a lab, preparation hinges on reliable data. Messing up molar calculations can ruin an experiment, waste money, and even cause safety risks. At one point, during a college project, my group made a simple arithmetic mistake with a reagent that included a nitro compound. That blunder set us back days and left us with a pile of useless data. Nobody wants to repeat that, especially with chemicals like 2-Chloronitrobenzene, which are common in pharmaceutical and dye work.
Why Accuracy Matters for Health and Safety
It isn’t just about clean data tables. 2-Chloronitrobenzene can cause harm if handled incorrectly. A slight mix-up in weight means someone might inhale too much or end up with a spill that doesn’t get neutralized properly. National agencies like OSHA and the EPA list the dangers of chlorinated and nitrated aromatics for good reason. Getting the details right means the difference between a safe workplace and a health scare. I remember one supervisor who checked every label and formula twice—he avoided accidents for the whole decade I worked with him.
The Role of Standardization and Training
Industry relies on a shared understanding. If a tech in a factory needs 2-Chloronitrobenzene, she expects it to conform to C6H4ClNO2 and weight out at 157.56 per mole. Cutting corners or mixing up isomers means broken batches and higher costs. Training and double-checking with resources like the Merck Index, Sigma-Aldrich, or PubChem helps everyone stay on track. I’ve seen teams spend hours retracing steps after they discovered an order had the wrong isomer. All that waste just because someone made a typo in the ordering system. It pays to insist on precision at every step.
Improving Access to Reliable Data
Errors tend to snowball in operations where data isn’t accessible and clear. Digital databases and apps help today’s chemists pull up formulas and weights at a glance. The faster and more transparent that information becomes, the easier it is to avoid risks. Technology won’t fix carelessness, but it makes it easier for professionals to uphold standards. Building a workplace culture that values accuracy and supports people who double-check details means fewer mistakes and better outcomes for everyone involved.
What the Chemical’s Numbers Really Tell Us
On paper, 2-Chloronitrobenzene’s formula and molecular weight might look dry. Out in the field, those numbers back up safety, innovation, and efficient manufacturing. Keeping them close and paying attention to the details brings real results, both in the lab and in industry.
What safety precautions should be taken when handling 2-Chloronitrobenzene?
Real-World Hazards Don’t Get More Obvious Than This
If you’ve worked in a chemical lab or on the production floor, you probably recognize the sharp odor and yellowish appearance of 2-chloronitrobenzene. This compound doesn’t get much attention outside chemistry circles, yet a quick brush with it proves how risky things can get if you take shortcuts. You don’t want to rely on luck. Direct skin contact will easily burn you or trigger allergic reactions. Inhalation irritates your nose, throat, and lungs. People tend to underestimate how respiratory exposure takes its toll. I’ve watched a co-worker ignore their mask for just a minute and end up coughing for hours—take these risks seriously.
Effective Personal Protective Equipment Saves More Than Your Clothes
Standard goggles won’t cut it here. It’s worth buying a tightly-fitted face shield that covers everything above your collar. I prefer nitrile gloves, not just latex, because chemical penetration can happen fast. Lab coats or chemical-resistant aprons matter more than most folks think. I once saw a spill soak through a cotton lab coat within seconds. Stick with gloves in good shape—no rips or signs of wear. The right gear avoids a lot of pain and unnecessary doctor visits.
Ventilation: The Layer People Overlook
Fume hoods aren’t just there for heavy acids. The vapors from 2-chloronitrobenzene can build up and linger in still air. Even in production settings, inadequate ventilation means exposure creeps up over long shifts. Always test your fume hood before opening bottles or transferring powder. I recall our team running a monitoring test and finding levels above exposure limits, simply because the fan setting got dialed down. Don’t forget this detail.
Safe Storage Protects Everyone Around
Nothing ruins a day faster than a leaking bottle hidden at the back of a cluttered shelf. Small breaks can turn into a whole-room contamination problem. You’ll want to store this chemical in sealed containers, clearly labeled, on shelves close to ground level. I keep a chemical-resistant spill tray beneath bottles just in case. Store it far from heat sources, acids, and reducing agents—runaway reactions are nobody’s idea of a good time.
Always Have a Spill and First Aid Response Plan
Safety isn’t just about preventing incidents but being ready for them. Workers should get trained on spill kits specific to nitrobenzenes and follow step-by-step plans. Don’t improvise in a crisis; grabbing the wrong absorbent compound does more harm than good. Notify the right people and evacuate the area if the exposure risk jumps. Quick access to eyewash stations and showers means exposures remain minor instead of life-threatening. I keep the SDS printed and taped to the wall for easy reference. Getting these basics down keeps the workplace far away from disaster.
Fostering a Culture Where Speaking Up Matters
Some of the safest labs I’ve worked in encourage everyone to point out problems immediately, no matter how minor. If you spot a cracked glove or a loose seal on the vent, call attention to it fast. Management has to listen and act; near misses turn into hard lessons if ignored. Regular refreshers and honest debriefs after incidents turn mistakes into real improvements. This approach builds trust—saving faces, not just reputations or chemical inventory.
How should 2-Chloronitrobenzene be stored and disposed of?
Why Proper Care Means Protecting Health and Environment
2-Chloronitrobenzene sits on the shelves of labs and factories as a yellow, crystalline powder with a sharp, sometimes unpleasant odor. As someone who’s spent hours reading Material Safety Data Sheets, I can’t overlook how easy it is for routine handling to backfire if basic steps get skipped. This compound, used in dyes, pharmaceuticals, and agrochemicals, carries some real risks for skin, eyes, and lungs. Contact can trigger burning and irritation, and inhalation leads to cough or headache. The big issue—handling this chemical without discipline can cause harm that lingers.
Keeps Things Secure: Storage Rules that Matter
Walking through a facility, I see the wisdom in locking chemicals away from foot traffic and sunlight. 2-Chloronitrobenzene stores best in a dry, cool, well-ventilated room, tucked in airtight containers. Glass or certain plastics, sturdy and undamaged, keep the air out and block spills from spreading. Strong shelves haven’t failed me, especially those chemical-resistant ones that stand up to the occasional drip or jostle.
Fire is always on my mind with chemicals like these. 2-Chloronitrobenzene won’t burn easily, but if flames catch nearby materials, it makes toxic fumes. Stashing it far from heat sources, oxidizers, and acids adds an extra layer of peace. Each time I see easy access to a fire extinguisher, I breathe easier. Emergency plans for leaks or spills should hang in plain sight. Those steps build trust and confidence on the floor.
Labeling and Checking: Slow Down to Stay Safe
My habit always circles back to labeling—fat, clear letters say what’s inside, along with hazard warnings and the date it came in. Routine checks help spot cracks in containers or changes in color, warning of trouble before it erupts. I have seen more than one loose cap or faded label, and every shortcut invites disaster. Regular walk-throughs keep everyone sharper.
No Shortcuts: Handling Disposal Correctly
Leaving chemical disposal for the end of the day never pays off. Dumping 2-chloronitrobenzene down the drain destroys water safety and threatens soil and wildlife. Local laws hand out stiff fines for those who think landfills or regular garbage bins work for hazardous waste. In my own experience, working closely with licensed hazardous waste companies brings the best results. They have the gear and training for high-temperature incineration in controlled conditions, reducing chances of air or soil contamination.
Staff always need proper gloves, protective coats, and goggles, even for the routine. Forgetting one piece puts skin and eyes right in harm’s way. I have seen emergency showers used more than once after a minor slip. Every facility should run regular safety drills—these moments save lives and prevent costly mistakes.
Better Habits, Better Outcomes
A culture of responsibility grows stronger every time someone speaks up over a sloppy storage shelf or cut corners on waste pickup. Companies that invest in training see fewer accidents or violations. Sharing the why and the how behind each storage and disposal step turns routine tasks into acts of community protection.
As chemistry and manufacturing change, smart management of substances like 2-chloronitrobenzene does not just check a box. It keeps people healthy, water clean, and reputations solid. I’ve learned that every careful habit means one less close call down the line.
What are the physical and chemical properties of 2-Chloronitrobenzene?
Physical Profile: Not Just a Yellow Solid
2-Chloronitrobenzene is a yellowish crystalline compound with a strong chemical odor, typical of nitroaromatic compounds. If you’ve ever handled chemicals in a university lab, that sharp scent signals what’s inside the bottle long before you read the label. Its melting point sits around 53°C, making it easy to work with at room temperature. It melts quickly on a steam bath, so controlling temperature during experiments or production is crucial.
This material isn’t shy about dissolving in many organic solvents. Toss a bit into ether, benzene, or acetone, and it disappears easily. On the other hand, try dissolving it in water, and you’ll find most of it stubbornly floating around unchanged. The low solubility in water matters in industrial settings—wastewater coming from plants making dyes or pharmaceuticals often needs careful handling, or chemical residues end up in rivers and lakes.
Chemical Behavior: Why Position and Substitution Matter
Every ring structure tells a story, especially in aromatic chemistry. With a chlorine atom on the second carbon and a nitro group planted firmly next to it, 2-Chloronitrobenzene reacts differently than other isomers. The nitro group pulls electrons away with its strong electron-withdrawing force, reshaping how the whole molecule reacts.
Add heat, and this compound stands up well—it doesn’t break down easily, even when placed in demanding reaction conditions. Both the nitro and chloro groups are tough customers, making the molecule fairly stable toward mild acids and bases. Nucleophiles, though, can swap out the chlorine if given the right encouragement: a little extra heat and strong enough nucleophile, like ammonia or an alkoxide, and a substitution occurs.
Practical Consequences: Industry, Lab, and Environment
2-Chloronitrobenzene plays a big role in making dyes, agrochemicals, and some pharmaceuticals. I’ve seen it pop up plenty of times on synthetic schemes during late-night lab sessions. Its structure lets chemists build more complex molecules—sometimes it’s the starting point for pesticides like o-chloroaniline, and other times, it ends up in pigments used in coatings and plastics.
These advantages don’t come free. The very properties that make 2-Chloronitrobenzene useful pose challenges for environmental safety. A low breakdown rate means it can stick around in the ground or water, which is not ideal. Exposure risks increase during handling and disposal, so protective equipment and good ventilation are non-negotiable during work with this compound. Breathing in vapors or getting it on your skin brings health hazards—headaches, irritation, or worse with chronic exposure.
Building Safer Systems and Sustainable Chemistry
Chemists and environmental safety teams can’t afford shortcuts. Technology exists now for capturing and treating aromatic residues from manufacturing streams. Biological treatments show promise—some bacteria degrade nitroaromatics, but progress remains slow for compounds with both nitro and chloro groups. Tracking waste, treating spills fast, and keeping an eye on workplace exposure sharpen safety standards.
Research keeps pushing for greener alternatives. Developing catalysts and reaction setups that run at lower temperatures or use less hazardous chemicals makes a clear difference. During my years in a teaching lab, emphasizing responsible lab management went hand in hand with teaching students why every property matters—from melting point and solubility up to reactivity and toxicity.
Every detail about 2-Chloronitrobenzene’s structure and behavior informs safe handling, better engineering, and smarter environmental policies. Balancing its value to industry with responsibility takes more effort, but there’s no better way forward.
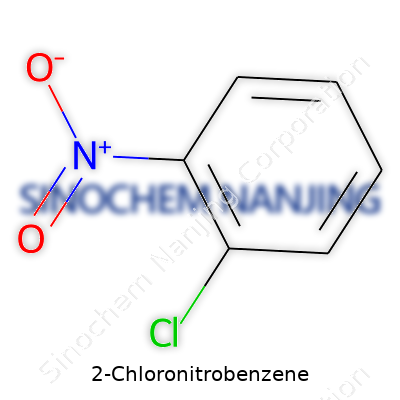
| Names | |
| Preferred IUPAC name | 1-Chloro-2-nitrobenzene |
| Other names |
o-Chloronitrobenzene 1-Chloro-2-nitrobenzene 2-Nitrochlorobenzene |
| Pronunciation | /tuːˌklɔːrəˌnaɪtrəʊbɛnˈziːn/ |
| Identifiers | |
| CAS Number | 88-73-3 |
| Beilstein Reference | 1209220 |
| ChEBI | CHEBI:140539 |
| ChEMBL | CHEMBL47359 |
| ChemSpider | 10320 |
| DrugBank | DB13518 |
| ECHA InfoCard | 100.005.440 |
| EC Number | 202-429-0 |
| Gmelin Reference | Gmelin Reference: 80253 |
| KEGG | C02645 |
| MeSH | D002859 |
| PubChem CID | 7419 |
| RTECS number | CZ9800000 |
| UNII | BO1W2YX80K |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C6H4ClNO2 |
| Molar mass | 157.56 g/mol |
| Appearance | Pale yellow crystals or flakes |
| Odor | aromatic |
| Density | 1.367 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.39 |
| Vapor pressure | 0.18 mmHg (25°C) |
| Acidity (pKa) | 23.7 |
| Basicity (pKb) | 11.10 |
| Magnetic susceptibility (χ) | -60.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.578 |
| Viscosity | 1.275 mPa·s (20°C) |
| Dipole moment | 3.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 170.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 106.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3522 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AJ04 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic if inhaled, causes skin and eye irritation, may cause respiratory irritation, suspected of causing cancer. |
| GHS labelling | GHS02, GHS06, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H335, H410 |
| Precautionary statements | Precautionary statements of 2-Chloronitrobenzene: "P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P362+P364, P391, P403+P233, P405, P501 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 554°C |
| Explosive limits | Explosive limits: 1.8–9% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 208 mg/kg |
| NIOSH | CN3675000 |
| PEL (Permissible) | PEL: 1 ppm (parts per million) |
| REL (Recommended) | REL: 2 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Nitrobenzene 2-Nitroaniline 2-Chloroaniline 2-Bromonitrobenzene 4-Chloronitrobenzene |

