2-Chloroacetophenone: A Deep Dive into Its Legacy, Uses, and Future
Historical Development
2-Chloroacetophenone, more widely recognized by its designation “CN”, stands as a substance with a complex, controversial past. Back in the early twentieth century, chemists harnessed this compound’s properties to devise a tearing agent, leading to its notorious adoption as a riot control chemical. After its emergence in warfare and policing in the 1920s, law enforcement agencies across the globe leaned on it for non-lethal crowd management. Having grown up just blocks from a police supply warehouse, I heard plenty about “tear gas” canisters stowed away for emergencies. These stories weren’t just urban legend—in many regions, authorities depended on this substance to regain control during unrest. By the late century, the chemical fell out of favor due to safety concerns and stricter guidelines, yet its shadow still lingers in conversations around public order.
Product Overview
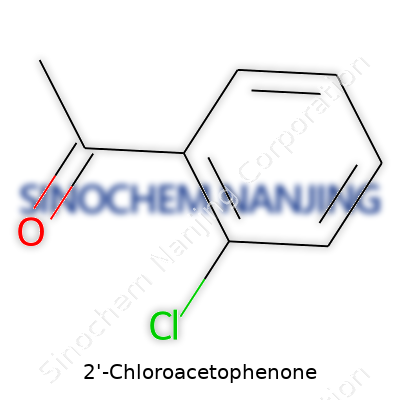
At its core, 2-Chloroacetophenone holds a simple structure—a benzene ring tagged by a chlorine and an acetyl group—but that simplicity belies a strong irritant legacy. Chemical manufacturers offer it as a pale, crystalline powder, often locked behind multiple layers of regulation. Its synonym list gives a sense of its notoriety: CN, phenacyl chloride, and tear gas among others. Whenever someone hears one of these names, they often think of crowd control before anything like research or industrial applications comes to mind.
Physical & Chemical Properties
This compound isn’t flashy in the lab. It carries a faint, sharp odor that hits the nose quickly. Part of why it captured police attention came from its volatility—2-Chloroacetophenone vaporizes around room temperature, making it easy to disperse. In powder form, it reveals a white to off-white hue. Solubility reads as limited in water, but dissolves well in many organic solvents. Its melting point, hovering around 54°C, shapes how it’s handled for both safety and formulation. Anyone who’s opened a bottle of the raw stuff knows immediately to keep a safe distance and protect their eyes.
Technical Specifications & Labeling
Labeling for 2-Chloroacetophenone carries heavy warnings. Chemical supply houses slap hazard pictograms front and center. Buyers must prove legitimate research or governmental need. Shipping containers must be sturdy, sealed, and clearly marked: “Irritant,” “Harmful,” and “Avoid inhalation.” In academic work, meticulous inventory tracking and specialized cabinets for volatile organics remain standard. I’ve seen entire lab rooms set aside for agents of similar pedigree, to isolate them from populated areas and reduce risk.
Preparation Method
Lab-scale preparation often involves the Friedel-Crafts acylation route. Chemists start with benzene, introducing chloroacetyl chloride using a Lewis acid catalyst, such as aluminum chloride. The reaction, while straightforward, gives off hydrochloric acid—a reminder to respect both chemistry know-how and personal safety. On an industrial scale, regulatory oversight increases, with process engineers enacting strict emission controls to prevent accidental release.
Chemical Reactions & Modifications
This acetophenone variant opens the door for a suite of downstream reactions. Given the reactive chlorine and ketone groups, organic chemists often use it as a starting block for building more complex molecules, whether in pharmaceuticals or materials research. For instance, substitution reactions swap out the chlorine, enabling new derivative compounds for test or specialty synthesis. In some circles, there’s hope that tweaking its backbone might reduce toxicity, perhaps allowing a safer derivative for use in instrumentation or tracer experiments—though progress remains sporadic at best.
Synonyms & Product Names
Over decades, 2-Chloroacetophenone has traveled under a bevy of names: phenacyl chloride lingers from the early organic chemistry literature, “CN” stands as a shorthand across law enforcement files, and the simple “tear gas” became common parlance. Marketing materials from chemical suppliers stick with the technical identifiers, intent on sidestepping political baggage.
Safety & Operational Standards
Regulatory agencies all but mandate best practices in every encounter with 2-Chloroacetophenone. Personal protection gear—a full set, not just gloves—becomes non-negotiable. Respirators, chemical splash goggles, and enclosed ventilated hoods cut down the risk in research settings. Local exhaust ventilation helps, too. Facilities that work with this compound update emergency response plans and spill kits annually, often in tandem with fire departments. I remember conversations with safety officers who spoke about the special paranoia that comes with maintaining stocks of substances known for crowd control, not just research.
Application Area
Historically, its reputation grew out of law enforcement circles—riot control, prisoner transport, and self-defense sprays. Over time, as public pressure mounted and alternatives entered the market, use narrowed and now remains controversial. In scientific spheres, 2-Chloroacetophenone finds work in organic synthesis—serving as a precursor to specialty compounds and as a reagent for analytical methods. A few researchers still use it to trace reaction pathways, exploiting its reactivity to study how certain molecules interact. Even so, ethical debates limit non-essential deployment.
Research & Development
In the past, researchers hoped to temper 2-Chloroacetophenone’s irritant kick while keeping its utility. Studies probed altered derivatives, using substitutions around the aromatic ring, aiming for compounds that retained signaling or marking qualities without inflicting such acute discomfort. Synthetic chemists eyed it as a convenient intermediate, drawn by the ease of functionalization. Despite its notoriety, a handful of groups continue exploring potential forensic and tracer applications, often with a dual focus on both scientific rigor and minimizing human harm. I’ve crossed paths with graduate students steered away from projects because of ethical review board concerns—not every promising chemistry direction survives today’s oversight.
Toxicity Research
Toxicological data paint a stark portrait. Exposure, even at low concentrations, causes severe irritation to the eyes, respiratory tract, and skin. Documented effects include lacrimation, coughing, and (at higher doses) temporary blindness or pulmonary complications. Chronic contact risks—tumor formation, respiratory illness—sparked sharp debate among public safety officials decades ago. Laboratory animal trials confirm these findings, which led to regulatory clampdowns and a push to phase out or restrict its use except in tightly controlled environments. Anyone who’s dealt with exposure, even secondhand, can attest: this chemical commands respect far beyond the beaker.
Future Prospects
Outlook for 2-Chloroacetophenone pivots on ethics and innovation. Public sentiment against chemical crowd control agents grows stronger each year—fueled by media coverage and human rights campaigns. In research, support tilts toward safer alternatives that model or simulate CN’s effects without the human fallout. Experts encourage green chemistry principles, nudging for molecules that degrade swiftly, present lower acute toxicity, or serve as more selective reagents. Regulatory frameworks keep tightening, sometimes driving labs to abandon use altogether in favor of less regulated analogs. Upcoming years promise more scrutiny, as policymakers weigh societal need against individual health. The tale of 2-Chloroacetophenone reminds us how a once-prominent compound can shape lives, both in the lab and on the streets, and why responsibility must walk hand in hand with scientific progress.
What is 2-Chloroacetophenone used for?
Beyond the Chemistry Lab
2-Chloroacetophenone might sound like something reserved for chemists and scientists, but its role stretches into some pretty familiar spaces. Most folks know it by another name: CN gas, or tear gas. Growing up during a time when news about protests flashed across the screen, people often saw officers using tear gas to control crowds. That thick white smoke? Often, it came from a compound like 2-Chloroacetophenone.
Why Law Enforcement Turned to CN
Crowd control in tense situations can push decision-makers to turn toward options they believe cause less harm than sticks or bullets. Researchers found CN gas effective at causing temporary irritation—burning in the eyes, nose, and throat. The burning makes it difficult for people to remain in an area. Law enforcement purchased it hoping to subdue crowds without deadly force. This shift away from harsher tactics spoke to a need for new solutions, even though the human cost of tear gas would soon show itself as not so simple.
Human Impact: Not Just “Non-Lethal”
During college, some of my classmates attended rallies and described their own run-ins with tear gas. Stories traveled fast—burning eyes, trouble breathing, panic as the gas spread. While classified as non-lethal, medical journals have documented that even brief exposure can cause lasting harm for people with asthma or other health conditions. Sometimes, cases of severe injury — and even permanent blindness — have been linked to misuse of these chemicals.
Other Uses: Farther from the Headlines
Let’s not forget, scientists first created 2-Chloroacetophenone for purposes outside of protest lines. The military adopted it as a chemical agent back in the early 20th century. Training exercises still use CN gas to help soldiers experience and respect the dangers of chemical exposure, preparing them for conflicts where they might face far worse threats. Industrial labs use it on a much smaller scale, usually as an intermediate in chemical synthesis, but outside specialized circles, trading in the stuff raises some red flags.
Concerns and Public Debate
Repeated incidents of excessive force involving tear gas have sparked heated public debate, both among neighbors and official circles. The World Health Organization has studied the risks of tear gas use in public settings and called for strict limitations or bans in crowded, poorly ventilated areas. Groups like Amnesty International have documented thousands of injuries worldwide resulting from careless deployment.
Calls for reform sound different in every city, but experts agree on one point: police forces and governments must train staff to use restraint. Some cities now publish statistics about chemical agents used in crowd control, and transparency helps the public hold officials accountable for safe practices.
Looking Ahead: A Smarter Approach
The bottom line comes down to respect for human experience. Health professionals suggest providing alternative methods like de-escalation training, investing in community liaisons, or simply avoiding confrontational tactics. Funding projects around mental health crisis response, education, and social services has brought incidents of violence down in many places. Rethinking the need for chemical agents can build trust between the community and those sworn to protect them.
What are the safety precautions when handling 2-Chloroacetophenone?
Understanding the Risks
2-Chloroacetophenone often finds its way into chemical research labs, and sometimes in riot control applications. It’s a mouthful, but the risks aren’t hard to understand. This compound acts as a tear gas. Exposure hits the eyes, nose, skin, and respiratory system hard. A splash brings burning and tearing, even in small doses. For anybody who remembers the first time they found themselves coughing, eyes streaming from an accidental chemical vapor, it’s not an experience you forget. Real safety starts with respect for how quickly things can go sour with this stuff.
Protection Means Layers, Not Just Gloves
I’ve seen new chemists think a good pair of gloves solves everything. Not with 2-Chloroacetophenone. Splash-resistant goggles belong on your face before the bottle even comes out. Contact lenses trap the chemical right on your eye, so those stay out. Think about the skin, too; cover arms with a lab coat made of chemical-resistant material, not just cotton. Nitrile gloves form the first line, but the stuff eats through latex and thin PVC. Double up on gloves if you plan to handle a lot or for extended periods. Always check gloves for pinholes and swap them at any sign the material feels compromised. This process doesn't feel like overkill once you see someone suffer a violent chemical splash.
Ventilation Beats Bravado
Chemical fume hoods aren’t extras—they’re basics. These setups pull vapors away and protect the whole crew in the room. A fume hood with a sash lowered to chest height takes the edge off accidental fumes and minimizes the chance of an airborne hit. Working outside a hood often feels convenient, but any convenience fades the moment the air turns acrid. Sudden headaches, coughing, or eye pain sometimes show up before you even smell much.
Spill Response, Not Panic
Forgetting to prepare for spills guarantees chaos. A small bottle knocks over faster than most people expect, and 2-Chloroacetophenone’s effects spread quickly. A specific chemical spill kit available within reach matters more than a regular mop and bucket. Granular absorbents, neutralizing agents, and waste bags meant for hazardous chemicals make cleanup safer. Any absorbent goes right into a sealed hazardous waste container since this stuff shouldn’t travel through normal trash or drains. Accident protocols drilled regularly keep panic at bay, and make the difference between a minor event and a major problem.
Personal Experience and Teamwork
During my graduate work, a buddy experienced a minor splash near his eye. We kept our calm thanks to prior drills. We used the emergency eyewash station for a full fifteen minutes—months of practice paid off. There’s no substitute for hands-on drills before an emergency; posters and written instructions fade from memory fast once real pain hits. Others in the room backed up with proper communication, called for further medical help, and made sure no one else got exposed. Experience taught us isolation of the affected workspace keeps the rest of the lab safe and avoids further spread.
Maintaining Good Habits
I’ve seen accidents happen right before end-of-day cleanups or in old labs where basic rules get ignored. Storing 2-Chloroacetophenone in tightly closed, labeled containers inside ventilated, locked cabinets prevents accidental mix-ups. Never use homemade or outdated labels. Before opening the bottle, check surroundings for any clutter. Proper waste disposal keeps janitors and trash handlers safe, too—hazardous waste storage requires clear labeling and prompt transport to authorized handlers.
Common Sense Goes Further Than Paperwork
People tend to focus on documentation, but in the heat of the moment, real safety depends on experience and minds-on alertness. Chemical handling lives up to safer standards only when everyone in the lab treats each moment with respect for potential danger. Training, teamwork, and personal attention make mishaps less likely and keep nerves steady when accidents strike. These aren’t distant worries—they come close to home any day you walk into a lab.
What is the chemical formula of 2-Chloroacetophenone?
A Closer Look at the Structure
2-Chloroacetophenone carries the chemical formula C8H7ClO. The name rolls off chemists’ tongues, but most of us recognize it by its role in tear gas. That single chlorine atom attached to the acetophenone backbone makes a big difference in how the compound acts and how people use it. I remember my first rundown with its molecular makeup during a college lab—watching experienced hands handle the white crystalline powder with absolute care. The lesson stuck: even a simple combination of carbon, hydrogen, chlorine, and oxygen can create something strong enough to change lives, for better or worse.
The Role of Experience in Understanding
No one forgets their first whiff of tear gas on the news, or in training, or during a security drill. 2-Chloroacetophenone doesn’t just sit in textbooks. Its formula shapes public life, whether in law enforcement, protest scenes, or debates around chemical safety. A deep understanding of its chemical nature—what those eight carbons, seven hydrogens, one chlorine, and one oxygen can do—matters in public discussions on safety and regulation.
Safety, Control, and Public Health
Chemicals like 2-Chloroacetophenone can bring people together in unexpected ways. People who work in security, healthcare, and emergency response all rely on good information about compounds like this one. The formula, C8H7ClO, isn’t just a string of letters and numbers. With it comes the weight of guidelines and safety measures because the effects on eyes and breathing can last long after clouds of gas clear.
The U.S. Centers for Disease Control and Prevention has detailed findings on the medical impact of 2-Chloroacetophenone exposure, showing how even brief contact can lead to persistent health problems. Knowing its exact structure supports the effort to build safer protocols and informs how public health teams plan for crowd management or industrial accidents.
Solid Facts, Real Consequences
Reliable chemical information comes from careful research. The formula C8H7ClO is referenced in trusted scientific publications and regulatory sources. For example, the National Institute for Occupational Safety and Health (NIOSH) has guidelines for exposure, since misuse leads to severe and long-lasting effects. Scientists and regulators look at more than just one molecule—they study breakdown products, environmental persistence, and the health profiles of exposed communities. This knowledge drives improvements in industrial hygiene and everyday workplace safety.
Searching for Solutions
Addressing the risks tied to 2-Chloroacetophenone starts with strong public awareness. Routine drills, clear labeling, and updated first aid guidance save lives. Investing in safer alternatives for crowd control should stay a top priority. Synthetic chemists and policy makers sometimes find themselves at odds, but every time teams work together to limit exposure, the public reaps the benefits. Open, honest science with clear data lets people make choices about safety and health. Nobody benefits from chemicals hidden behind obscure codes and formulas—the facts, spelled out in C8H7ClO, keep the conversation honest, and that keeps people safe.
How should 2-Chloroacetophenone be stored?
Understanding the Real Risks
2-Chloroacetophenone, often called CN gas, brings up some strong concerns for anyone working with it, even for folks outside the laboratory. Law enforcement, chemical handlers, and industrial workers deal with this substance regularly. The health risks are clear: toxic vapors can lead to coughing, burning eyes, and breathing trouble. Even a small spill or leak inside a storage area leads to costly cleanups and, more importantly, danger for anyone nearby.
Personal Experience: No Room for Carelessness
Years ago, in a university research lab, someone skipped labeling a secondary bottle and stashed it on the wrong shelf. The result? A tense afternoon scrambling to find the source of an acrid odor—a headache nobody needs. I learned that day: organization and vigilance matter just as much as technical knowledge. In my own workshops later on, I saw people get complacent with chemical storage. The folks who double check seals and practice good habits rarely face emergencies. Good procedures protect everyone; carelessness invites disaster.
Cold, Dark, Dry: Simple Rules Keep Us Safe
The science says it plainly. 2-Chloroacetophenone breaks down faster in heat and light. Warmth boosts its pressure, pushing more vapor into the air and stressing containers. Strong sunlight speeds up decomposition or even causes bottles to burst. Humid or damp storage lets water react with the powder or solution, damaging purity and sometimes forming new hazardous materials.
The everyday solution gets boring, but it works: Pick a cool, shaded spot in a secure chemical storage room. Go for a metal cabinet with clear hazard labels and a tight seal. Use personal protective equipment every time, especially gloves, goggles, and a NIOSH-approved respirator when handling containers. Never mix sources—don’t keep food, drinks, or flammables nearby. Simple separation helps avoid accidents that ruin careers and endanger lives.
Practical Solutions for the Modern Workplace
Many supervisors assume old steel cabinets or a roll of caution tape will do the trick, but chemical engineering teams keep storage controlled and compliant with safety codes. Use secondary containment trays built for acid and solvent resistance. If storing multiple chemicals in one room, separate oxidizers and bases. Automated sensors that detect humidity, temperature swings, or vapor leaks now come cheap and send real-time alerts to mobile devices. They give an early heads up before danger spirals.
Training saves more lives than any equipment upgrade. The most effective safety class I taught focused on the “why,” not just the checklist. If people understand the suffering chemical exposure causes—burns, lung problems, even blindness—they treat every bottle with respect. A well-practiced routine, strict checklists, and peer verification routines make storage much safer than crossing fingers and hoping nothing goes wrong.
Document Everything—Regulator or No Regulator
Every jurisdiction carries rules for hazardous chemical storage; OSHA, EPA, and local fire codes all matter. Even if regulations seem overdone, I’ve seen workplace audits save jobs and prevent injuries long-term. Maintain clear inventory logs and incident records. Keep up with safety data sheets—the latest version, not last decade’s printout stuffed in a drawer. Audit once each quarter, not just once a year.
Respect for Both the Science and Each Other
Storing 2-Chloroacetophenone goes beyond following a formula. It’s about developing a culture where workers look out for one another, share tips, and treat every risk as personal. There’s no shortcut—serious respect, good training, and standard precautions make all the difference.
What are the health hazards associated with 2-Chloroacetophenone?
What’s in Tear Gas—And What It Does to Your Body
Plenty of folks have heard of tear gas. Relatively few stop to wonder what’s really inside those canisters. One major component behind the pain is 2-chloroacetophenone. This compound isn’t just a tongue-twister; it causes a laundry list of problems for the human body. The folks working in law enforcement or military settings probably know its immediate punch, but you don't have to be on a protest line to worry about its health risks. Even brief exposure, without the proper protection, can knock your body off balance fast.
How 2-Chloroacetophenone Works on Human Senses
The sting in your eyes and the burning in your throat happen for a reason. This chemical works as an irritant for mucous membranes, which means it hits eyes, nose, mouth, and lungs almost instantly. Eyes go red, blink reflex kicks in, and vision gets blurry. You might try to breathe in, but the nose and throat can start swelling, with coughing and choking trailing close behind. Doses that get carried on the wind still force people to flee the area, usually gasping and in pain.
I spent time on a college campus where a protest spiraled out of control. One canister rolled across the lawn, and the impact was unforgettable. Within seconds, people coughed and gasped. A couple of students fell, clutching their faces. The chemical doesn’t discriminate—it catches children, the elderly, and anyone with asthma right in its attack.
Short-Term and Long-Term Health Risks
Short-term effects usually fade with clean air, but not always. Some unlucky people deal with severe eye burns, which can last for days. For anyone with pre-existing lung issues, exposure can tip them into a trip to the ER. 2-chloroacetophenone particles stick to clothing and skin for hours, so those running away from a contaminated area may take the irritant home without realizing it.
Long-term exposure brings even deeper problems. Studies run by the National Institute for Occupational Safety and Health (NIOSH) link the chemical to lasting respiratory trouble, skin rashes, and even permanent eye damage. There’s concern about delayed onset asthma, especially in children or the immune-compromised. Medical reports document scarring inside the lungs after repeated exposure. NIOSH sets its recommended exposure limit at the lowest possible level for a reason.
The Path to Safer Handling
Most people won’t ever choose to work with 2-chloroacetophenone, but still deserve protection. Workers in factories where this chemical gets manufactured or packaged have to keep strict protocols. Full-face respirators and gloves aren’t just a suggestion—they’re critical. Regular air monitoring helps catch leaks before they turn lethal. Employers need to step up on training, not tick boxes for compliance but actually explain the real risks and how to minimize them. In crowded environments, transparency around handling is key, especially if anything leaks or spills.
Communities could benefit from more education and transparency, especially in areas where law enforcement keeps these agents on hand. Emergency rooms and poison control centers should have clear guidance on quick treatment, because speed matters for minimizing damage. People exposed need access to clean water and non-contaminated clothes as soon as possible. The main lesson: the best defense is always knowledge, paired with the right gear and a plan that’s been tested, not just written down.
| Names | |
| Preferred IUPAC name | 1-chloro-2-phenylethan-1-one |
| Pronunciation | /tuː ˌklɔːroʊˌæsɪtəˈfiːnoʊn/ |
| Identifiers | |
| CAS Number | 99-04-7 |
| Beilstein Reference | 1214538 |
| ChEBI | CHEBI:2781 |
| ChEMBL | CHEMBL15821 |
| ChemSpider | 54670 |
| DrugBank | DB06715 |
| ECHA InfoCard | 100.005.076 |
| EC Number | 200-929-3 |
| Gmelin Reference | Gmelin Reference: 8331 |
| KEGG | C14343 |
| MeSH | D002770 |
| PubChem CID | 7411 |
| RTECS number | AB1925000 |
| UNII | Q6B4FI4DZA |
| UN number | UN1698 |
| Properties | |
| Chemical formula | C8H7ClO |
| Molar mass | 154.59 g/mol |
| Appearance | Colorless crystals or white crystalline solid |
| Odor | Aromatic odor |
| Density | 1.18 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.7 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 2.49 |
| Basicity (pKb) | pKb = 10.53 |
| Magnetic susceptibility (χ) | -52.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.557 |
| Viscosity | 2.08 mPa·s (at 25 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -45.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4994.6 kJ/mol |
| Pharmacology | |
| ATC code | N05CM03 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS06, GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H302, H315, H319, H335, H315, H317, H330 |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P308+P311, P330, P337+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1 |
| Flash point | 76°C |
| Autoignition temperature | 527 °C |
| Explosive limits | Explosive limits: 2.0–11% |
| Lethal dose or concentration | LD50 oral rat 595 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1400 mg/kg |
| NIOSH | CN6125000 |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 20 ppm |

