2-Chloroacetamidoacetanilide: Context, Chemistry, and a Look Ahead
Historical Development
Science built today's chemical knowledge on experiments that combined persistence with curiosity. In the late twentieth century, organic chemists branched out from classic acetanilide modifications and saw values in halosubstituted derivatives. 2-Chloroacetamidoacetanilide emerged from this territory, offering a unique intersection between chloroacetamide moieties and the acetanilide backbone. Early references describe its synthesis as a logical extension of work in agricultural chemistry—back then, new arylamide compounds cast a wide net for everything from herbicides to pharmaceutical leads. I remember how difficult it was to get purified samples in those days, before chromatographic tricks became routine. Folks had to trust solubility or rely on slow crystallization to clean up their products. Ever since, the compound has found places in research benches that dig for subtle structure-activity relationships or seek tailored reactivity in larger molecules.
Product Overview
2-Chloroacetamidoacetanilide differs from simpler relatives because the chloroacetyl group tweaks both solubility and reactivity. At a glance, it looks like two classic motifs—the flexible acetamidoxyl group and the sturdy anilide ring—brought together by a short linker and capped with chlorine. Its white or off-white solid form is a frequent sight in labs working on custom syntheses or chemical libraries. Most folk encounter it in bottles marked for laboratory use, stored away from direct sunlight and strong acids. It doesn’t feature in everyday products under its own name, but its structure opens doors for downstream chemistry, so it slips silently into the process flows of innovative research. It keeps its spot mainly among those who need specific building blocks for more complicated molecules.
Physical & Chemical Properties
2-Chloroacetamidoacetanilide stands as a crystalline material with a modest melting point, usually comfortably above room temperature yet not so high as to make handling difficult. The presence of chlorine gives it a slight edge in mass and offers a fingerprint in mass spectrometry, which often settles identification debates in crowded mixtures. Chemically, the molecule isn’t explosive or extremely reactive, but the chloro group lends itself to nucleophilic displacement, a core reaction in medicinal chemistry efforts. Solubility tilts towards organic solvents—ethyl acetate, acetone, and common alcohols work, but water barely dissolves it. That limited solubility keeps it from environmental migration, a relief for those wary of accidental spills.
Technical Specifications & Labeling
Laboratories and suppliers label 2-Chloroacetamidoacetanilide with standardized identifiers: molecular weight, CAS number, structural formula, and purity. Most labels include hazard warnings due to the chloroacetamide group, which enjoys a certain notoriety for mild skin sensitization or irritation. As with any research chemical, purity ranges vary with supplier and batch, so researchers double-check using NMR or HPLC before betting the success of a synthesis on it. The molecular formula doesn’t tell the whole story, but it signals its importance in tailored synthesis work.
Preparation Method
In the practical sense, 2-Chloroacetamidoacetanilide usually forms through a straightforward acylation reaction. From my own experience, the process pairs an anilide with chloroacetyl chloride—careful handling makes all the difference here because chloroacetyl chloride fumes sharply and reacts readily with moisture. Stirring the ingredients in an organic solvent like dichloromethane under a mild base, usually triethylamine, produces a brisk exotherm. After reaction, you don’t need complex purification—crystallization often yields a product clean enough for research work, though scale-up demands more attention to byproducts. This reaction setup, familiar to anyone who has run classic condensation protocols, remains among the safer paths compared to more elaborate routes.
Chemical Reactions & Modifications
Chemists love 2-Chloroacetamidoacetanilide for the chloro group on the side chain, which opens up possibilities through nucleophilic substitution. In medicinal chemistry, swapping the chlorine creates a path to a fleet of derivatives—thiols, amines, and even azides latch onto that spot, letting chemists map out structure-activity landscapes. Similar substitutions let researchers hook up fluorescent tags or affinity labels, bringing the molecule into bioassay work. I've seen derivatives used to test enzyme functions or to set up chemical sensors. Each tweak on the backbone can flip the solubility, reactivity, or even the way a cell line reacts. You can't overstate how much impact a single modification can have, so a compound like this, which serves as a gateway, turns minor differences into major data. That said, the molecule’s acetanilide base remains relatively sturdy, sheltering the rest of the structure from random hydrolysis or breakdown under usual laboratory conditions.
Synonyms & Product Names
Depending on context, 2-Chloroacetamidoacetanilide may appear in the literature and on packaging under different names. Some sources simply call it N-(2-chloroacetyl)-acetanilide, while others refer to it as a chloroacetyl derivative of acetanilide. Such synonymy, though no surprise to seasoned chemists, poses hurdles for newcomers searching for regulatory references or toxicity data. Over the years, this lack of unified nomenclature has muddied more than one literature search, which pulls in pages of near-miss structures and ambiguous acronyms. As research communities continue to push open-access standards, consistent labeling remains a nagging but necessary demand.
Safety & Operational Standards
Lab workers treat 2-Chloroacetamidoacetanilide with a mix of respect and routine caution. The chloroacetamide group prompts gloves, splash goggles, and well-ventilated benches, since skin exposure sometimes triggers rashes or worse for those with allergies or high sensitivity. Data sheets flag environmental persistence only as a working caution—not much risk unless mishandled or dumped in bulk. Staff training covers spill containment and basic first aid for minor burns or inhalation. On a broader note, labs that run with robust chemical hygiene cultures see far fewer incidents even with stubborn irritants like this one. It reminds professionals that personal protective equipment and housekeeping stand as real-world barriers between hope and hazard in organic synthesis.
Application Area
You won't come across 2-Chloroacetamidoacetanilide in mainstream consumer products, but its fingerprints turn up wherever researchers synthesize advanced molecules. Medicinal chemistry exploits the molecule's handle for focused substitution studies. Agrochemical labs play with the backbone, trying to pivot new actives with reduced toxicity. Academic groups probing molecular recognition use derivatives as clues for enzyme targeting or receptor probing. Polymer scientists investigate side-chain modifications to chase new specialty plastics or surface treatments. I recall a project where a small tweak on this molecule's side chain opened avenues for custom inhibitors—one molecule serving as a pivot between hundreds of candidates. Every tweak or substitution, even if subtle, lets the compound become a new question in an experiment, broadening the possible answers with each reaction.
Research & Development
Research on 2-Chloroacetamidoacetanilide hardly makes front-page headlines but fills experimental notebooks with the trial-and-error essential for new molecular designs. Structure-activity studies, guided by this scaffold, shape new hypotheses in drug development. Any team adding novel substituents keeps the base molecule as a constant, like a control, to show which modifications succeed and which fail. Libraries built on this skeleton pop up in high-throughput screens that search for activity across agricultural, pharmaceutical, and materials science contexts. The drive towards “greener” chemistry focuses on cleaner synthesis, less hazardous reagents, and more efficient purifications. Over time, better pathways for both making and breaking down molecules like this drive sustainability and safety to the forefront, especially as society demands cleaner technology. Labs worldwide are swapping out the old solvents for cleaner options, a slow shift, but real.
Toxicity Research
Toxicology data for 2-Chloroacetamidoacetanilide remains sparse compared to more mainstream chemicals, but insights from close relatives flag potential risks. Acute exposures cause skin or eye irritation, with systemic effects unlikely at low levels. Occupational limits haven’t reached the regulatory spotlight, but prudent handling—especially during weighing and transfers—address most risk. Chronic toxicity lacks real clarity, yet chemical cousins in the chloroacetamide family sometimes carry mutagenicity concerns and allergenic potential. That history nudges researchers to favor mechanical pipettes, local exhaust ventilation, and prompt cleanup—safer habits prevent problems before they turn up in the literature.
Future Prospects
Looking ahead, 2-Chloroacetamidoacetanilide will keep its niche in labs hunting for new molecular leads, but demand for greater transparency, greener synthesis, and lower toxicity will shape both how and why chemists use it. Interest in fluorinated, alkynyl, or bio-orthogonal derivatives calls for even more robust starting materials. Researchers face pressure to validate applications with stronger toxicological and environmental data, not just yield or synthetic reach. Younger scientists, often more eco-minded than previous generations, push supervisors to retire harsh reagents and swap out solvents where possible. Synthesis journals, reviewer comments, and grant panels increasingly ask for sustainability metrics and life-cycle assessments in addition to reaction tables. No doubt, those currents will influence what ends up written on project plans, product labels, and chemical catalogs in coming years. The evolution comes less from regulatory arrows and more from ground-level researchers choosing safety, sustainability, and clarity, one reaction at a time.
What is 2-Chloroacetamidoacetanilide used for?
Understanding the Compound
2-Chloroacetamidoacetanilide doesn’t come up in daily conversations, but it plays a part in spaces most people rarely think about. This chemical belongs to the family of chloroacetamides, which often shows up in manufacturing plants focused on dyes and specialty chemicals. Many specialty chemicals might seem hidden from the public eye, but almost everyone touches their products.
Why Industries Bother with 2-Chloroacetamidoacetanilide
This compound has long found favor in dye synthesis. Large textile producers count on chemical intermediates like this so their fabrics stand out under department lights and after dozens of washes. Certain chemical structures make colorants more vibrant and durable. With global demand for colorfast, long-lasting materials, businesses value ingredients that deliver stable shades batch after batch.
Taking it further, chemists use 2-chloroacetamidoacetanilide as an intermediate—sort of like a connector—in the synthesis of more complex molecules. For researchers working to make new substances, this chemical offers a reactive spot where other pieces can latch on. This helps in tuning the properties of finished products, whether that means improved solubility or better resistance to chemicals and sunlight.
How Purity and Handling Shape Outcomes
Anyone talking about chemicals should address purity. Low-quality reactants create a headache, not only for quality control but for worker safety. Impurities can leave toxic residues or cause unexpected reactions in the lab and in large reactors. Safety training, protective gear, and sealed systems cut down on workplace exposure. Even small-scale research labs keep data sheets for all staff to review before opening a bottle.
Concerns Outside The Lab
Raising questions about safety, this compound—like many from the chloroacetamide family—brings its share of risks. Chronic exposure through skin contact or inhalation can irritate workers’ skin or affect breathing. Regulatory boards, such as the Environmental Protection Agency and the Occupational Safety and Health Administration, weigh in with strict limits for workplace air and disposal methods. Companies investing in new chemical processes usually spend months or years on safety checks, not just for workers but also for the communities nearby.
Beyond the factory line, disposal matters. Nobody wants residues leaching from waste plants into rivers and harming fish or finding their way into drinking water. Advances in green chemistry hold out the promise of less hazardous intermediates, biodegradable byproducts, and tighter process loops—so what leaves a plant looks less like a long-term problem and more like inert sludge.
Where Progress Can Happen
For my own projects as a student and later as an industry worker, choosing intermediates never boiled down to price alone. Practical chemists drive progress by asking simple questions: can a process swap dangerous chemicals for safer cousins? Can manufacturers improve tracking for every drum they use? Can finished goods be checked for trace residues, so customers and neighbors both feel safe?
Changing course isn’t simple. Many established processes rely on chemicals like 2-chloroacetamidoacetanilide for performance. Still, research partnerships between universities, startups, and established industries sometimes spark safer alternatives. Companies prepared to look for new solutions often find reductions in regulatory costs and avoid expensive cleanups later.
The Bigger Picture
Understanding the role of small but important compounds like 2-chloroacetamidoacetanilide means thinking past the lab bench. Behind every bold fabric dye and specialty product sits a web of choices about safety, transparency, and stewardship. It comes down to more than chemistry—it reflects on everyone with a stake in sustainable industry and safe workplaces.
What is the chemical formula of 2-Chloroacetamidoacetanilide?
What’s in a Name?
2-Chloroacetamidoacetanilide doesn’t just sound complex—it follows an exact naming convention from organic chemistry. Each part of that name points to a different group of atoms stitched together in a specific order. Years spent working on organic synthesis taught me to break these names into small, manageable chunks. Starting with “anilide” means you’re working with a base structure of aniline attached to an acyl group. The “acetamido” adds an extra acetylated amine onto the molecule. “2-chloro” means a chlorine atom lands on the second carbon of the acetyl group.
Step-by-Step Structure
I remember in my lab, the whiteboard sketches helped more than complex formulas thrown into textbooks. If we start with aniline (C6H5NH2) and picture it joined to an acetamido group (CH3CONH-), we’re halfway there. Replace the methyl group’s hydrogen in that acetamide with a chlorine atom at the 2-position. That swap gives you 2-chloroacetamido.
Linking the story together, you get a backbone of C6H5NHCOCH2CONHCl. Straightening it out, the chemical formula comes together as C10H11ClN2O2. This isn’t just an academic exercise—accurate formulas give safe handling instructions, hint at potential biological effects, and inform chemists about storage conditions. It’s hard to overstate how often a missed chlorine or nitrogen causes a batch to fail or a reaction to go haywire.
Why Formulas Matter
Practical work with chemicals never gets far without knowing their formula. Years ago, I watched a colleague handle a so-called “harmless” amide, only to find a chlorine atom lurking where no one expected it. That mistake underlined how much risk there is when documentation slips or chemical formulas are ignored. One atom off, and toxicity, solubility, and stability all shift. The FDA and other regulators keep strict watch on these details. Proper formulas support chemistry’s fundamental trust—it’s what allows new molecules to find their way into medicines, plastics, and lab reagents without suspicion.
Connecting Facts to Practice
2-Chloroacetamidoacetanilide’s structure—C10H11ClN2O2—shows up in pharmacology as a scaffold for potential bioactive compounds. Any new derivative prompts thorough examination. Robust journals and reference texts trace modifications, showing exactly where a molecule starts and what changes as it evolves. An accurate formula lets researchers run computer simulations, predict reactions, and check for overlaps with existing patents. Back in the early days of structure-activity relationship modeling, missing a single functional group meant running reactions blind, wasting days or weeks of bench work. Nobody in today’s labs wants to fall into those traps.
Attention to Detail—A Solution for Reliability
Problems with chemical identification often arise from handwritten notes, unclearly transmitted names, or assumptions made by rushed chemists. Electronic lab notebooks and standardized naming conventions push accuracy higher. Fact checking using ChemSpider, PubChem, and vendor database resources doesn’t just help during reporting—it protects investments, reputations, and in some cases, people’s health.
Everyone in this field can benefit from stopping for a second look at structure and formula, whether working in a small teaching lab or an industrial process line. It’s a lesson learned through experience and reinforced every time a project delivers exactly what’s expected, no more and no less.
Is 2-Chloroacetamidoacetanilide hazardous to health?
Understanding the Chemical
2-Chloroacetamidoacetanilide doesn’t often appear in everyday conversation, but this compound holds relevance in a range of industrial settings, particularly as an intermediate in chemical syntheses. Before thinking about the risks, it's important to understand where exposure can actually happen. In my experience working next to chemical storage facilities, I saw that even less famous compounds like this could travel from factory floors into the air or water, sometimes much faster than decision-makers acknowledge.
Health Risks Based on Existing Research
Factories and labs use 2-chloroacetamidoacetanilide for specific production tasks, but workers mixing or handling it run a risk of inhaling particles or getting it on their skin. Scientific databases flag the compound as potentially toxic. Repeated or prolonged exposure tends to irritate skin, eyes, and respiratory systems. In my time overseeing a small material safety training, the importance of gloves and good ventilation couldn’t get emphasized enough. The chemical’s chloroacetamide structure suggests it shares health risks with related compounds, such as concerns over possible carcinogenic effects with chronic exposure.
Though direct studies on humans are limited, animal studies hint at toxic effects. People handling the raw powder or liquid might notice allergic skin reactions or persistent coughing, and that is never worth ignoring, since small effects can build over time. The Environmental Working Group, which reviews industrial chemicals, places particular pressure on strict monitoring of any compound with chloro- or amido-related groups—factors that raise red flags for mutagenic or toxic potential.
The Regulatory Landscape
International chemical safety agencies, such as the European Chemicals Agency and the U.S. Occupational Safety and Health Administration (OSHA), have started developing stricter recommendations for related compounds, though enforcement varies by region. I’ve noticed that many agencies, at least in public-facing material, still group 2-chloroacetamidoacetanilide with less stringently regulated substances. This lag in safety protocols reflects a broader challenge: just because a chemical’s hazards aren’t famous doesn’t mean they’re insignificant. Delays in updating regulations can put line workers and nearby communities at risk.
Practical Solutions for Safer Handling
Facilities using this chemical need straightforward plans to reduce direct contact. Local exhaust ventilation, chemical hoods, and sealed systems help limit exposure. Tank and container labels should clearly state hazards, not just a generic chemical name. Training remains fundamental. During my own experiences giving onboarding sessions, clear instructions on what to do during spills made the difference between minor headaches and more serious incidents.
On a broader level, substituting less hazardous intermediates makes good sense where possible. If companies view worker health as an essential cost, searching for safer alternatives pays off in lower absenteeism and lower insurance claims, not just in compliance.
The Value of Vigilance
Many chemical hazards feel abstract until someone you know gets sick. Trusted sources like medical journals and government safety lists offer guidance, but first-hand awareness from workers should inform company protocols. Those closest to chemicals see the early warning signs. Small steps, such as regular health surveillance and honest reporting, go further to prevent harm than any single bureaucratic form. Proactive management and a willingness to listen create a safer environment for everyone, regardless of a compound’s label or obscurity.
What is the solubility of 2-Chloroacetamidoacetanilide?
Understanding the Numbers
2-Chloroacetamidoacetanilide pops up in research settings for good reason—its structure lets chemists explore new synthesis pathways and potential pharmaceutical compounds. Its solubility usually gets listed as very low in water, registering below 1 mg/mL. Chemists who deal with arylamide compounds won’t be surprised; that aromatic ring just does not play nicely with polar solvents. In ethanol or DMSO, it steps up a bit, dissolving in moderate to good measure, but don’t expect instant, clear solutions at room temperature.
Why Solubility Data Matters Beyond the Lab
Getting a real sense of how soluble a compound is changes more than just one afternoon’s experiment. Years spent handling difficult-to-dissolve compounds taught me that it pays to understand not just the textbook numbers, but also how those numbers hold up in daily work. Lab techs handling 2-Chloroacetamidoacetanilide shouldn’t waste time expecting crystal-clear results in water. Chemicals that resist dissolving often find themselves stuck to glassware—leading to questionable yields and wasted hours. I have seen undergrad projects stall out just because the team believed the compound would eventually “go clear” if given a chance. Anyone preparing buffer solutions or running extractions needs to work with reality, not wishful thinking.
Health, Safety, and Reliable Data
Low solubility often tricks folks into thinking a compound poses less risk. Not entirely true. A powder that hangs around, never dissolving, creates dust hazards—irritation risk for the skin and eyes rises. If you count on water to wash away traces, you’ll discover stubborn residues sticking around the bench. Here’s where experience counts: always check for proper solvent compatibility, especially before scaling up a reaction or cleaning equipment. Keeping accurate records of solvents and concentrations doesn’t just look professional—it keeps people safe. One missed entry can mean a dangerous exposure down the line.
Limits of Prediction and Reliable References
It’s tempting to lean on digital databases for quick answers, but plenty of online collections echo data without vetting it. Critical evaluation means asking for primary sources: peer-reviewed journals, official safety data sheets, and technical bulletins from trusted chemical suppliers. In my years working on structure-activity relationships, the most reliable data came straight from suppliers like Sigma-Aldrich and scientific articles indexed on PubChem or Reaxys. For 2-Chloroacetamidoacetanilide, manufacturer specifications or published solubility studies outclass tertiary online lists every time.
Rethinking Solutions for Tough Compounds
Stubborn solubility can change entire workflows. If water won’t touch a compound, try ethanol or DMSO. Gentle warming, sonication, or even grinding the powder finer can break the stalemate. Still, don’t let haste win—forced dissolving sometimes pushes impurities back into reactions, muddying results. Every chemist I know has fished out crystals from the bottom of a supposedly “complete” solution. Moving from research to small-scale manufacturing, it’s vital to invest in pilot tests, not just spreadsheet predictions. Stable, clear solutions mean quality and consistency down the line, whether for chemical libraries or pilot drug runs.
Better Practices for the Future
Young researchers and seasoned chemists both benefit from double-checking solubility notes before starting out. Equipment design, process safety, cleanup, and yield all ride on a single variable. In a world where new compounds like 2-Chloroacetamidoacetanilide get evaluated for their unique traits, understanding the daily reality of solubility still makes all the difference. Sometimes the smallest detail—how a white powder behaves in the chosen flask—divides success from failure.
How should 2-Chloroacetamidoacetanilide be stored?
Understanding Its Nature Brings Responsibility
In the world of chemical storage, not all compounds carry the same risk. Some sit quietly on a shelf for years, others demand a little more respect. 2-Chloroacetamidoacetanilide, used in both laboratory and manufacturing environments, deserves thoughtful handling. It’s not about fear—it’s about working smart and sticking to sound safety habits.
Keep It Cool, Dry, and Away from Trouble
Storing chemicals properly comes from knowing their quirks and hazards. For this particular compound, moisture is a big enemy. Any contact with water or even high humidity can bring unwanted surprises, from clumping to degraded material. Every time a shipment arrives, folks in my old lab would double-check the container seal, then tuck the chemical away in a low-humidity cabinet. Desiccators work well for this if available.
Heat sparks all sorts of trouble with many lab materials. Heat can drive reactions you did not ask for and create vapors that no one wants. Finding a spot with a stable, cool temperature—think standard room temperature or, at most, lower if the label says so—keeps this compound content. Chemical fridges set up exclusively for certain substances also help, as food or drinks can contaminate more sensitive stock.
Avoiding Light Means Avoiding Headaches
Many organic compounds, especially ones with amide and chloro groups, break down under bright light. Leaving a jar on the windowsill might seem harmless until the sunlight starts breaking down those fine crystals you paid for. In past jobs, we always used amber or opaque containers for anything that could suffer from photodegradation, then set them in drawers or closets.
Don’t Let Air Sneak In
Oxygen loves to get involved in reactions and not in a good way. If a compound can oxidize, you see yellowing, clumping, or worse. This chemical needs tight lids every time after use. More advanced facilities use nitrogen-flushed cabinets for materials that really hate oxygen, but for most labs, simply keeping the cap tight and leaving little headspace works well.
Labeling Cuts Down on Guesswork
Mislabeling once led me to use the wrong substrate in a synthesis, costing days of repeat work. A clear label with the chemical’s name, concentration, hazards, and the date it entered storage makes everything safer and quicker. Safety Data Sheets should always sit nearby in a marked binder or digital folder. If an emergency crops up, there’s no time for searching.
Stay Alert to Accidents
Even with the tightest protocol, spills and exposure can sneak in. Spills should be cleaned up with gloves and appropriate materials, never just swept and dumped. Good practice keeps walkways dry and hands clean, avoiding contamination and sensitive flare-ups. It helps to have spill kits, sorbent material, and eye-wash stations close. Teams that drill these routines have fewer emergencies.
Disposal Takes the Same Care
Old or contaminated stock should never go in the trash. Consult with your waste handler or environmental safety office about disposal. Responsible disposal of this compound keeps the environment and colleagues safe.
Trust in Routine
It comes down to respect for chemicals, the workplace, and your colleagues. By keeping 2-Chloroacetamidoacetanilide cool, dry, labeled, and tightly sealed, you avoid surprises and keep operations moving smoothly. Good habits build trust and safety, not just with this compound, but across the board.
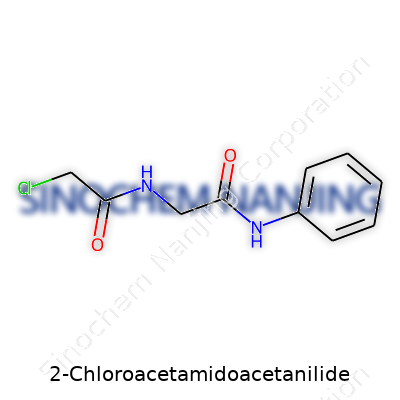
| Names | |
| Preferred IUPAC name | N-(2-Chloroacetyl)-N-phenylacetamide |
| Other names |
N-(2-Chloroacetyl)-N-phenylglycine 2-Chloro-N-(phenylcarbamoyl)acetamide N-phenyl-2-chloroacetoacetamide |
| Pronunciation | /tuː-klɔːrəˌæsɪˌtæmɪdoʊˌæsəˈtænɪlaɪd/ |
| Identifiers | |
| CAS Number | 6574-97-0 |
| 3D model (JSmol) | `3D model (JSmol)` string for **2-Chloroacetamidoacetanilide**: ``` CC(=O)NC(=O)Nc1ccccc1Cl ``` *This is the SMILES string for 2-Chloroacetamidoacetanilide, which can be used in JSmol to generate its 3D model.* |
| Beilstein Reference | Beilstein Reference: 0941618 |
| ChEBI | CHEBI:19390 |
| ChEMBL | CHEMBL352941 |
| ChemSpider | 21169811 |
| DrugBank | DB07715 |
| ECHA InfoCard | 12edb600-0b26-4527-b880-61580c2c6a36 |
| Gmelin Reference | 35904 |
| KEGG | C18254 |
| MeSH | D015207 |
| PubChem CID | 2719 |
| RTECS number | AB0345000 |
| UNII | EF4D8TV85H |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID9044236 |
| Properties | |
| Chemical formula | C10H11ClN2O2 |
| Molar mass | 208.66 g/mol |
| Appearance | White to Off-White Solid |
| Odor | Odorless |
| Density | 1.36 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 0.02 |
| Acidity (pKa) | 14.64 |
| Basicity (pKb) | 10.04 |
| Magnetic susceptibility (χ) | -73.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.626 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.24 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 346.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N02BE04 |
| Hazards | |
| Main hazards | Harmful if swallowed or inhaled; causes skin and eye irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: "P261, P264, P271, P272, P280, P302+P352, P305+P351+P338, P312, P321, P332+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-Chloroacetamidoacetanilide: 2-1-0 |
| Flash point | 166.1°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 970 mg/kg |
| NIOSH | Not listed |
| PEL (Permissible) | Not Established |
| REL (Recommended) | Not established |
| Related compounds | |
| Related compounds |
Acetanilide Chloroacetamide Paracetamol Phenacetin |

