Looking at 2-Chloro-m-Cresol: More Than Just a Chemical
Historical Background and Development
The chemical world always manages to bring us compounds that both fascinate and challenge. 2-Chloro-m-Cresol stands out as one such substance. For those of us who've spent time in a laboratory or chemical plant, names like this spark memories of the wild ride that organic chemistry has taken over the past century. Developed as part of a larger search for chemically versatile phenol derivatives, 2-Chloro-m-Cresol owes its roots to early 20th-century aromatic chemistry. Phenolic compounds, including cresols, caught the attention of researchers for their antiseptic power, paving the way for derivatives like this chloro-cresol to enter the scene. Over time, uses shifted from antiseptics and disinfectants to intermediates for dyes, resins, and agrochemicals. This legacy reminds us: the story of any chemical can only be told alongside the era’s technological advances and the push-pull between economic need and safety worries.
Getting to Know the Product
Among aromatic chemicals, 2-Chloro-m-Cresol carries a strong and medicinal odor, recognizable to anyone familiar with phenolics. In industrial settings, it's usually found as a solid that, thanks to its manageable melting point and solubility profile, offers up plenty of practical handling options. Chemists and product developers often favor it for its predictable reactivity. In my experience, tasks involving phenolic intermediates rely heavily on a compound's solubility in both polar and non-polar solvents, something this chloro-cresol delivers. It has found a home in products ranging from specialty resins to pesticides, and its value lies in allowing direct functionalization through that molecular scaffold—a phenol ring with both methyl and chlorine substituents.
Physical and Chemical Properties
Many of us remember learning to predict boiling points, solubility, and reactivity by just glancing at a chemical name. For this compound, chlorine’s presence on the meta position of cresol’s aromatic ring creates subtle but important differences from its ortho- and para-chloro analogs. Solid at room temperature, it typically forms colorless to pale yellow crystals. It melts below the boiling point of water, letting chemists manipulate it with standard heating methods. Moderate solubility in water gives it a practical edge for industry, though you’ll see it dissolve better in organic solvents. As for its reactivity, the combination of phenolic hydroxyl and electron-withdrawing chlorine atom makes certain reactions (like nucleophilic substitutions and various C–C coupling reactions) more straightforward, a benefit when trying to build more complex molecules or fine-tune a product's chemical profile.
Technical Specifications and Labeling
Precision matters in chemical production, but daily handling needs information that’s both clear and useful. 2-Chloro-m-Cresol ships with labeling that spells out concentration, impurity profile, and handling precautions. In factories and smaller operations, I’ve seen plenty of variation in how companies label strength, residual solvents, and moisture content, with each batch’s unique fingerprint determined by its preparation method. Regulations today force a closer look at trace contaminants, from polyhalogenated byproducts to unwanted phenolic residues, reflecting our growing concern for process safety and product purity. Clear labeling in line with hazardous chemical requirements—like GHS pictograms and risk phrases—pushes everyone in the chain to handle the compound with appropriate respect, underscoring the link between chemical literacy and workplace safety.
How Producers Make 2-Chloro-m-Cresol
The synthesis of 2-Chloro-m-Cresol isn’t a new trick, but it does show how simple reactions can offer up industrial value. Operators start by chlorinating m-cresol, making sure the process runs under controlled temperatures to avoid over-chlorination or byproduct formation. In older facilities, open semi-batch reactors powered much of the world’s cresol chlorination, but these days, safety-focused improvements use closed systems, better agitation, and sometimes catalysts to boost yield and keep impurity levels in check. I remember the sting in the air from off-gassing in cramped plants without modern fume management—a hazard the industry has mostly overcome with stricter environmental controls. Separation steps like distillation and recrystallization often follow, reflecting the ever-present balance between output, energy use, and purity.
Chemical Reactions and Modifications
Chemists prize 2-Chloro-m-Cresol as a building block, but not just for its own sake—the real excitement comes from its utility in upgrades and modifications. It can function as both an electrophile and a nucleophile, opening the door to a range of substitutions on the aromatic ring. The phenolic hydroxyl reacts in etherification, esterification, and condensation reactions, such as in the formation of protective groups or binding into more elaborate organic frameworks. The chlorine atom makes it handy for nucleophilic aromatic substitution, a path toward heavy-duty ligands, agrochemical precursors, or specialty pigments. There’s something satisfying about seeing a relatively simple molecule transformed with the right batch of reagents and just the right tweak of conditions—that moment of synthesis is what drew many of us to chemistry in the first place.
Synonyms and Product Names
Markets and research papers scatter alternative names across their pages. In literature, you’ll hit on 2-chloro-3-methylphenol, 2-Chloro-m-cresol, and sometimes more cryptic names like 3-methyl-2-chlorophenol. International markets occasionally reference older trade names or region-specific designations, but the basic phenol-cresol backbone stays easy to spot for anyone who’s spent time with chemical catalogs or Material Safety Data Sheets. For folks in research or procurement, those name variations can be a hassle—cross-referencing always becomes the best way to make sure you’re looking at the right molecule, especially with similar-sounding cresols in stock.
Safety and Operational Standards
Industrial safety demands more than gloves and goggles; it calls for a full understanding of what dangers a compound brings. 2-Chloro-m-Cresol can sting eyes, skin, and lungs, so workers receive targeted training and facilities enforce proper PPE use. Modern facilities install extraction fans directly over active reaction vessels, and routine air monitoring helps staff avoid chronic low-level exposure. Transport regulations for this substance reflect its potential hazards, with storage in sealed, correctly labeled drums away from strong acids, bases, and oxidizers. Containment and emergency response plans rely on clear spill protocols to limit both human contact and environmental escape. Drawing on my own years spent with chemical process audits, the rise of digital compliance tools and “near-miss” tracking apps has made a real difference in preventing incidents, but the most effective prevention always starts with honest respect for the chemical’s risks.
Application Areas Big and Small
The reach of 2-Chloro-m-Cresol extends farther than you might expect. Paint and coating manufacturers use it as a key intermediate, allowing for custom-tailored pigment dispersions and resins. Agrochemical producers incorporate it into herbicides and fungicides, because derivatives based on the cresol ring show activity against weeds and fungi. Occasionally, you’ll see it pop up in specialty antiseptics—reminiscent of cresol’s original role in public health, but generally replaced by newer, less hazardous alternatives. In polymers and resin production, it serves to help control molecular architecture and physical properties. Research settings value it as a probe for developing new synthetic routes or as a reference standard for analytical studies. For anyone who thinks industrial chemistry happens in some separate universe, the spread of this compound across markets is a reminder of how closely our daily lives mesh with chemistry’s invisible hand.
Ongoing Research & Development
Research doesn’t slow down, even for well-trodden molecules. Universities and corporate labs explore fresh ways to improve the green credentials of synthesis, cutting waste and using catalysts that leave behind fewer by-products. Work focuses on optimizing the selectivity of chlorination, as past production often led to multi-chlorinated residues or other phenolic contaminants. Some teams look to biological methods for future routes—microbial transformation of m-cresol, perhaps, over traditional chlorination. For products based on 2-Chloro-m-Cresol, research shifts toward better biodegradability and reduced worker exposure during processing. Seeing the environmental push coming from both regulators and supply chain customers, developers now prioritize greener alternatives and “benign-by-design” approaches. My own foray into collaborative projects found new value in teams pulling together chemists, engineers, and toxicologists—no one has a lock on all the best ideas, and cross-pollination pushes innovation further.
Toxicity Research: Facing Risks Upfront
Toxicology studies keep a close watch on phenolic and chloroaromatic compounds, precisely because of their known hazards. Direct skin or eye contact triggers acute irritation, and inhalation risks grow with dust or vapor. Chronic exposure worries public health officials due to potential liver and kidney effects, and occasional findings of bioaccumulation in fish habitats spark debate over wastewater management. Past research into mutagenicity and carcinogenicity gives mixed results, fueling regulatory caution. That's led to tougher exposure limits and requirements for containment, air scrubbing, and on-site decontamination. Workers don’t just trust to regulation—they want and need transparency about what they’re handling, making data-driven training and medical monitoring essential parts of daily protocol. Because environmental and occupational safety never stand still, ongoing toxicity research tells regulators, producers, and users how to adapt with new findings and fresh perspectives.
Looking Ahead: Future Prospects and Industry Direction
The future of 2-Chloro-m-Cresol walks the same tightrope as many specialty chemicals—balancing utility against mounting calls for sustainability and safety. Demand isn’t likely to disappear soon, given the ongoing need for custom intermediates in coatings, agriculture, and advanced materials. That said, growing public pressure to rein in persistent bioactive chemicals means producers and users will need to keep refining handling protocols, emissions controls, and life-cycle analyses. Demand for “greener” synthetic methods, circular chemistry, and compostable derivatives is forcing the industry to rethink everything from feedstock sourcing to final product use. Investment in real-time toxicity testing systems and digital supply chain tracing now play into decision making at every production stage. Speaking as someone who has watched regulations tighten decade after decade, the ability to adapt—chemically and institutionally—matters more than ever. Producers who embrace transparency, cooperation with researchers, and continual process optimization will shape the next chapter not just for 2-Chloro-m-Cresol, but for specialty chemicals as a whole.
What is 2-Chloro-M-Cresol used for?
Stepping Into the World of 2-Chloro-M-Cresol
For most folks outside a chemistry lab, 2-Chloro-M-Cresol sounds more like a tongue twister than a chemical you might run into every day. It's a chlorinated phenol — a mouthful, but important. Having worked closely with research teams in chemical safety, I’ve seen 2-Chloro-M-Cresol show up in all kinds of places that don't often attract headlines. This compound, with its sharp smell, pops up in several industries that touch lives in subtle but significant ways.
Keeping Products Safe and Mold-Free
If you’ve ever wondered why your paints and coatings last on a bathroom wall, or why cleaning products don’t break down in your kitchen cabinet, the secret sometimes lies with compounds like 2-Chloro-M-Cresol. As a preservative with antimicrobial qualities, it helps keep molds and bacteria out of household paints, adhesives, and even leather goods. In my time testing consumer products, preservatives like this one made the difference between a product you could store for months and one that spoiled quickly. Keeping goods mold-free cuts down waste and saves companies and families money.
Supporting Medicine and Personal Care
The pharmaceutical world relies on chemicals like 2-Chloro-M-Cresol for their disinfectant abilities. Take certain antiseptics and topical creams — this compound helps stop bacteria before it causes trouble. Hospitals and homes both count on stability and safety, and ingredients such as this add an extra layer of defense. From personal experience working with small clinics, any boost to the shelf life of a disinfectant or cream can stretch budgets and supplies, especially in rural areas.
Making Dyes and Specialty Chemicals
Industrial chemistry often needs starting materials that act as building blocks for more complex substances. 2-Chloro-M-Cresol often serves as an intermediate in the synthesis of dyes, pesticides, and other specialty chemicals. My first summer job at a dye company gave me a look into how crucial these intermediates are. Without stable, reactive compounds like this, creating reliable and bright dyes for textiles or effective crop protection chemicals would be far tougher.
What About Risks and Precautions?
While this compound extends the life and usefulness of many goods, handling it calls for some respect and caution. Prolonged exposure can irritate skin, eyes, and the respiratory tract. In the labs I worked, managers stressed gloves, goggles, and strong ventilation when dealing with chlorinated phenols. Regulations set limits to keep factory workers and the environment safe. These restrictions make sense — no cost savings are worth the risk to health or the waterways downstream.
Finding Safer and Greener Paths
There’s a growing movement toward reducing the use of tough preservatives and solvents in manufacturing. Newer biocides and greener alternatives are under study to limit environmental buildup and toxicity. Researchers look for chemicals that break down faster and leave fewer residues. Having followed these developments for years, I know industry shifts don’t happen overnight, but a steady push for alternatives can pay off. More investment in research, incentives for companies to switch, and stronger labeling rules all help tip the scales toward safer substitutes, without sacrificing the quality that consumers expect.
Conclusion
2-Chloro-M-Cresol may not be a household name, but the grip it has on daily comforts and industry cannot be ignored. It keeps goods cleaner, makes products last longer, and supports essential sectors. By understanding how it works and taking steps to manage its impact, society can enjoy the benefits while steering toward safer horizons.
What are the safety precautions for handling 2-Chloro-M-Cresol?
Understanding the Risks
2-Chloro-M-Cresol shows up in labs and industrial settings for good reason—it works as a disinfectant and as a building block for dyes, resins, and pesticides. Some call it 3-chloro-5-methylphenol. Don’t let its usefulness distract from the real risks it can pose. This stuff gives off a sharp odor, can irritate your skin and eyes, and isn’t something you want to breathe in or swallow. Years of chemical safety guidance keep repeating one thing: never underestimate how a compound like this can harm your health if you get too comfortable around it.
Personal Protective Equipment: The Unsung Hero
I’ve spent plenty of time working with volatile chemicals, and the rule never changes—gear up every time. Nitrile gloves seal off your skin from the liquid. Splash goggles stop vapors and drops from reaching your eyes. Lab coats and closed shoes keep splashes off your clothes and body. In places that get stuffy or don’t move air very well, putting on a respirator offers another line of defense for your lungs. No quick lifts of the goggles or skipping the gloves because “it’s just a small batch.” Accidents never warn you before they happen.
Ventilation Matters More Than You Think
Fume hoods should run every time you handle 2-chloro-m-cresol, whether it’s weighing powder, pouring liquids, or cleaning up spills. I’ve seen labs get headaches, dizziness, or trouble breathing because of poor airflow or broken fume hoods. OSHA data lists inhalation as a major risk—so don’t ignore a musty smell, and don’t believe you can “tough out” exposure. Routine checks on the hood’s airflow save trouble before anyone feels sick.
Storage: Keep It Isolated, Keep It Cool
Don’t keep this chemical next to acids, oxidizers, or food supplies. Use tight-fitting, labeled containers. If a label smudges or peels, replace it immediately. Avoid storing above shoulder height to prevent spills. Pick cabinets or rooms away from sunlight and heat since warmth speeds up decomposition and increases vapor release. A good storage area also needs chemical spill kits ready, not buried behind unrelated supplies.
Clean-Up and Dealing with Spills
Spills happen even for the careful ones. Trained teams with hazmat kits handle big leaks, but for a small mess: ventilate the area, gear up, and use inert absorbents like vermiculite or sand—never use sawdust or combustible materials, since 2-chloro-m-cresol reacts badly with strong oxidizers. Gather the waste in sealed containers for hazardous disposal, not down the sink or in the regular trash.
Medical Response: Know the Signs and Move Fast
Skin contact leads to redness or burning. Eyes sting and tear up. Breathing vapors causes coughing and nausea. Anyone exposed needs to rinse with water for at least 15 minutes and leave the area immediately. Call for medical help if irritation doesn’t fade or symptoms worsen. Keep emergency numbers near the work area, not tucked away in a drawer.
Prevention Through Training and Routine
Routine drill sessions build instincts, not just for handling but for responding to emergencies. New workers should never get left to figure things out on their own, and experienced staff need refreshers. Regular inspections of safety equipment—gloves, glasses, eyewash stations, ventilation systems—keep everyone one step ahead of accidents. Regulations like OSHA’s Hazard Communication Standard and guidelines from the CDC help workplaces set specific rules, but a safety culture grows through habits people carry into every workday.
What is the chemical formula of 2-Chloro-M-Cresol?
Understanding Its Chemical Formula: C7H7ClO
In everyday life, folks come across disinfectants, cleaners, and even some personal care items that promise to kill germs and keep surroundings safe. One of the ingredients showing up in these labels goes by the name 2-Chloro-m-cresol. Its chemical formula, C7H7ClO, may look like a random string of letters and numbers, but there’s a story behind every piece of it. This specific chemical belongs to the group of chlorinated cresols, and each part of its formula tells what atoms it holds together: seven carbon atoms, seven hydrogens, a chlorine, and an oxygen.
Researchers didn't pick this structure by accident. 2-Chloro-m-cresol’s arrangement lets it mess with the cell walls of bacteria and fungi, making it valuable as an active ingredient in some antimicrobial products. Without getting tangled in jargon, this means it hits the bad bugs hard and helps clean environments, from hospitals to household kitchens. This practical punch comes from the blend of chlorine’s reactive nature and the intrinsic antimicrobial traits from the cresol base. It’s been shown in studies, like those from the Journal of Applied Microbiology, that phenolic compounds with halogen substitutions—like a chlorine atom here—boost effectiveness against a range of pathogens.
Products, People, and the Balance of Use
People probably don’t ponder over C7H7ClO while using a cleaner or soap, but decisions about including chemicals like this steer health and safety outcomes. Chemical companies and regulators have to weigh benefits and risks, and that’s where real scrutiny comes in. Too much exposure or improper handling brings its own risks, not just for users but for workers who handle large scales of raw ingredients. It’s not all doom and gloom, but stories crop up every year about chemical sensitivities or skin reactions, reminding us of the real world impact.
Long before anything lands on a supermarket shelf, safety evaluations take center stage. Agencies like the Environmental Protection Agency (EPA) or the European Chemicals Agency don’t let ingredients like 2-Chloro-m-cresol slide in without toxicity and environmental fate studies. It’s not just about killing bacteria; run-off and air emissions from manufacturing or overuse might hurt aquatic life or even cause lasting changes in local water quality. Documented evidence links similar compounds to persistent residues in soil and water, creating a push for better disposal guidelines and safer alternatives.
Solutions and Responsible Handling
Training for those who make or use chemical-heavy products often gets overlooked. Clear labeling, wearing gloves, and sticking to recommended concentrations can prevent most problems for routine users. In my own line of work, I’ve seen how a simple tweak in packaging can make a big difference—like putting instructions front and center or using child-resistant caps—in avoiding accidents.
At the same time, researchers keep hunting for eco-friendlier antimicrobial agents that don’t stick around in the environment. Biodegradable alternatives are rising, some derived from essential oils or plant extracts, trimming the footprint without losing cleaning power. This shift brings a lot of hope, especially in sensitive settings like schools or clinics.
So, C7H7ClO isn’t just another puzzle piece in a chemistry textbook. It represents the balancing act between effective disinfection and human and environmental health. That formula guides decisions well beyond the lab, shaping the way we manage cleanliness, risk, and sustainability for today and tomorrow.
How should 2-Chloro-M-Cresol be stored?
A Chemical That Demands Respect
2-Chloro-M-Cresol pops up in many labs. Used in disinfectants, pharmaceuticals, and dyes, it looks pretty harmless to the untrained eye. But looks never tell the true story. Safety guides do not call for extreme caution just for the sake of bureaucracy—experience with chemical mishaps has shaped every rule, every detail. Anyone who’s spent years juggling jars and containers knows: one mistake sticks in memory.
Sensible Storage Starts With Temperature
No one enjoys surprises in the stockroom. Most chemical supplies store 2-Chloro-M-Cresol at room temperature, out of direct sunlight. UV rays can sometimes sneak in through windows and speed up degradation or cause unwanted reactions. On a warm summer day, temperatures in some warehouses creep up past comfortable levels. Inventory on metal shelves by a sunlit window—bad news written all over it. If you can, keep this chemical in a cool, shaded spot, ideally under 25°C. Refrigerators work, but only if they stand dedicated to chemicals, never shared with food.
No Shortcuts With Containers
Glass or high-quality HDPE plastic bottles keep this substance stable. Leaky, cracked, or reused jars spell risk. There’s rarely a good reason to risk a cheap fix, especially after seeing what a corroded cap can do in a storeroom. Lids must fasten tight, threads checked for grit and wear. Proper labeling using indelible pens clears up confusion and stops that famous last-minute guesswork. Hazard pictograms matter, especially for new hires or anyone having a bad day.
Keep It Dry, Keep It Isolated
Water and 2-Chloro-M-Cresol never get along. Humidity inside a bottle speeds up breakdown over weeks or months, generating heat or even toxic gas. If your workplace runs dehumidifiers in chemical storage, you know why. Keeping all containers away from water sources, including leaky pipes and mop buckets, helps avoid a slow disaster. Never place this chemical near acids, alkalis, or oxidizers. In my old workplace, one slip of the hand while reorganizing a shelf almost put incompatible drums side by side—a near-miss that sparked stricter routines.
Ventilation and Fire Protection
Fumes from 2-Chloro-M-Cresol sting the nose and throat. Small leaks in a tight space make breathing miserable, so a well-ventilated storage area really pays off. No one enjoys drawing the short straw to air out a fume-filled closet. Flame-proof cabinets matter too. This compound catches fire below 100°C—open flames or sparking equipment cannot be allowed nearby. One electrical fault, one spark, ruins more than just a few minutes.
Personal Responsibility, Shared Safety
Following procedures around hazardous chemicals protects everyone, not just the one person handling the jar. Staff training goes beyond safety videos—it sticks when taught by someone with years of practice and a few scars to show. Nothing replaces personal vigilance and honest communication in chemical storage. The best-run labs assign responsibility and run inspections, never assuming all is well. Families wait for folks to come home from work; colleagues count on each other to take common sense steps seriously.
What are the potential health hazards of 2-Chloro-M-Cresol exposure?
Why Knowing About 2-Chloro-M-Cresol Matters
For folks who work in chemical plants, labs, or manufacturing, coming across 2-Chloro-M-Cresol can be part of daily life. This compound pops up in disinfectants, dyes, and even in wood preservation. At first glance, it looks harmless—a white to yellowish powder that doesn’t give much away. The real trouble hides under the surface, and decades of industrial use bring a lot to unpack.
How 2-Chloro-M-Cresol Gets Into the Body
Most contact happens because of accidents or poor workplace hygiene. Skin exposure is easiest—the compound can soak in without much warning. Breathing it in, especially as dust or vapor, adds another layer of risk. Swallowing rarely happens, but it’s a nightmare case in settings where chemicals get stored without enough care.
The Direct Effects: Skin, Eyes, and Lungs
Many workers talk about the burning, stinging feeling after the powder touches skin or eyes. In mild cases, redness and irritation show up fast. More severe exposure leads to blisters, chemical burns, and sometimes lasting scars. Eye contact sometimes means blurry vision and pain, sticking around for days unless someone flushes the area right away.
When the dust floats in the air, the lungs bear the brunt. Inhaling even a small dose can trigger coughing, throat pain, shortness of breath, and sometimes asthma-like symptoms. NIOSH and OSHA post strict exposure limits for a reason—too much breathing with poor ventilation can cause serious lung damage. From what I’ve seen, those who don’t use masks or gloves face the harshest symptoms by the end of a shift.
Beyond the Obvious: Systemic Dangers
Scientists have studied 2-Chloro-M-Cresol’s deeper health risks. Animal studies flag damage in the liver and kidneys after repeated exposure. The body’s detox organs work overtime trying to break down and flush out this chemical. The EPA and European Chemicals Agency both list it as a hazardous substance based on this research. The worry, especially in developing countries, centers around weak regulatory controls and old equipment, which can turn a fixable problem into something deadly.
Some early studies hint at potential links between long-term exposure and disruptions to hormone function, though peer-reviewed data remain thin. For vulnerable groups—like pregnant women or those with immune disorders—the risks climb higher. My own time in a chemical factory taught me that workers will often keep quiet about chronic headaches or stomach pain until those symptoms reach a crisis point.
Managing the Hazard: Real Solutions
One simple fix stands out: better training. New hires seldom get more than a slideshow about chemical hazards. Real learning happens on the floor, with protective gear and hands-on spill drills. Industrial hygiene programs pay off when managers listen to the team doing the dirty work, not just the safety officer with a clipboard.
Companies need to invest in proper ventilation, regular air sampling, and sealed containers. Accidents tend to follow the cheapest shortcuts. From my own experience, the small step of providing barrier creams, eye stations, and visible reminders in the break room can cut the number of skin and lung complaints by half.
2-Chloro-M-Cresol’s dangers fade in the minds of those who use it every day. The real challenge is making health and safety a shared culture, where saying “I need better gloves” feels as normal as clocking in. That’s where lasting change grows.
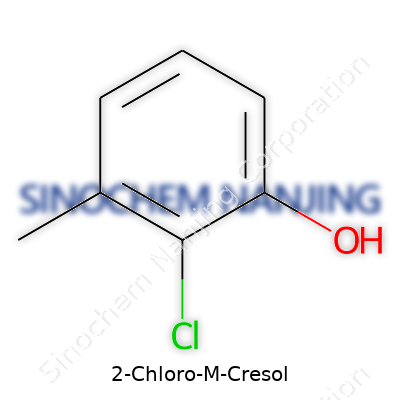
| Names | |
| Preferred IUPAC name | 2-Chloro-3-methylphenol |
| Other names |
2-Chloro-3-methylphenol 2-Chloro-m-cresol 3-Methyl-2-chlorophenol NSC 5016 |
| Pronunciation | /tuː-ˈklɔːr.oʊ em ˈkrɛs.ɒl/ |
| Identifiers | |
| CAS Number | 14806-19-4 |
| Beilstein Reference | 604619 |
| ChEBI | CHEBI:34232 |
| ChEMBL | CHEMBL374092 |
| ChemSpider | 214675 |
| DrugBank | DB14055 |
| ECHA InfoCard | 100.008.462 |
| EC Number | 201-204-4 |
| Gmelin Reference | 8063 |
| KEGG | C01736 |
| MeSH | D002765 |
| PubChem CID | 6979 |
| RTECS number | GO6475000 |
| UNII | CUU8M3LO5S |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C7H7ClO |
| Molar mass | 142.58 g/mol |
| Appearance | Colorless to pale yellow crystal |
| Odor | Phenolic |
| Density | 1.169 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 2.6 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | 9.47 |
| Magnetic susceptibility (χ) | -83.2e-6 cm³/mol |
| Refractive index (nD) | 1.585 |
| Viscosity | 10 cP (20 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 154.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -105.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3910.5 kJ/mol |
| Pharmacology | |
| ATC code | D08AE07 |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P310, P321, P330, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-Chloro-M-Cresol: 3-2-0 |
| Flash point | Flash point: 108 °C |
| Autoignition temperature | 120 °C |
| Lethal dose or concentration | LD50 oral rat 1830 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1830 mg/kg (oral, rat) |
| NIOSH | B0121 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | IDLH: 175 mg/m3 |
| Related compounds | |
| Related compounds |
Chlorocresol 4-Chloro-m-cresol 4-Chloro-o-cresol 2-Chloro-p-cresol 2-Methyl-4-chlorophenol 3-Chloro-o-cresol |

