The Complicated Journey of 2-Chloro-4-Nitroaniline: More Than Just a Chemical
Historical Development
Chemistry has always shaped industry, but few can claim a legacy quite like 2-Chloro-4-Nitroaniline. It first showed up in research labs around the tail end of the nineteenth century as scientists began tinkering with the interaction between halogenated aromatics and nitroanilines. Chemists wanted new dyes and better ways to make drugs, but the path wasn’t smooth—even in the early days, safety took a back seat to productivity, and plenty of folks got sick before anyone cared about personal protective equipment. Over decades, the manufacturing process matured. Additions in reactor technology, better control over exothermic reactions, and early hints of greener synthesis all shaped how 2-Chloro-4-Nitroaniline integrated into mainstream chemical portfolios. Its discovery and development marked one of those moments when pure laboratory curiosity transformed into everyday business.
Product Overview
To most people, 2-Chloro-4-Nitroaniline just looks like a clumpy orange or yellow powder. The real story lies behind it. This solid has built a quiet but important part in making dyes, pigments, and some pharmaceuticals. Thanks to a rigid chemical backbone, the compound resists breakdown in hot solutions and solvents. This eventually led to it getting picked for tougher industrial applications. Chemists rely on its predictable reactivity, but the uninitiated might not realize how many things in their world—plastic buttons, colored fabrics, sometimes even specialty inks—trace their origins to this odd-smelling crystalline compound.
Physical & Chemical Properties
At room temperature, 2-Chloro-4-Nitroaniline holds its shape, showing off a melting point in the high double digits Celsius. It acts stubborn in most solvents, staying put even when the temperature climbs. That inert behavior comes from having both a nitro group and a chlorine atom nestled on the aromatic ring, making the molecule less likely to jump into random side reactions. On its own, the powder gives off a slight bitter odor, and its intense yellow color makes it hard to miss in a laboratory setting. Those peering through a microscope see regular, needle-shaped crystals—evidence of an orderly, if toxic, structure. All these quirks affect how the compound performs during further synthesis and define its storage needs. It won’t break down just from sitting on a shelf, but improper handling or mixing with reactive agents can lead to sharp, unwanted reactions.
Technical Specifications & Labeling
Science-driven industries don’t mess around with ambiguity—clarity on chemical labels can mean the difference between a safe shift and an emergency. For labs and factories, clear details such as assay percentages, water content, and impurity thresholds must be easy to find on every bag or drum of 2-Chloro-4-Nitroaniline. Over time, standards shifted from vague descriptions to rigorous documentation. The modern era expects data from HPLC, GC, and NMR testing to reflect purity, while hazard labels need to square with global systems like GHS, including all those eye-catching pictograms for toxicity and irritancy. Workers who handle the material learn immediately the consequence of a missing hazard statement or mislabeled drum, so the stakes remain high.
Preparation Method
Nobody in the business glosses over the hazards of making 2-Chloro-4-Nitroaniline. Factories tend to stick with the tried-and-true method of nitration and subsequent chlorination using carefully measured reagents. The process always produces some harsh by-products, so risk management is more than a buzzword—operators can’t skimp on scrubbers or containment systems. Chemists fine-tuned the reaction temperatures and dosing rates over decades, but lower-yield modifications pop up in patent literature regularly. Some researchers chase greener synthesis, embracing catalytic methods with fewer emissions, but these don’t get much love from old-guard technologists sticking with reliability over novelty.
Chemical Reactions & Modifications
2-Chloro-4-Nitroaniline rarely shows up as a finished product. Chemists exploit its rich functional groups to build more complex substances. The nitro group opens the door for reduction to an amine, which then slides into azo dye manufacturing or high-performance pigment production. Chlorine, sitting on the ring, paves the way for nucleophilic aromatic substitution—a backbone for making all kinds of dyes and intermediates. Some reactions focus on coupling, while others aim to tweak the molecule for specific pharmacological properties. Outside of its famous transformations, 2-Chloro-4-Nitroaniline sometimes plays antagonist, causing headaches for those needing clean separation during multi-step synthesis. Yet, its value keeps it front and center in many chemical workflows.
Synonyms & Product Names
While 2-Chloro-4-Nitroaniline comes up in technical reports, nobody in the lab sticks exclusively to that mouthful. You’ll hear its formal-sounding name in regulatory circles, but common lingo swaps in labels like “4-Nitro-2-chloroaniline” or sometimes “CNA Yellow.” Some product listings group it under older trade names adopted by dye houses in the last century, confusing newcomers and making traceability across supply chains extra challenging. The abundance of aliases means careful vetting when comparing suppliers or reviewing safety data. The same goes for international trade; legal documents often pile on language variants, amplifying the chance of mixups unless the Chemistry Abstracts Service or IUPAC number appears. Real work teaches you that language always trails behind the pace of global chemistry.
Safety & Operational Standards
Nobody in manufacturing can ignore the health risks stemming from compounds like 2-Chloro-4-Nitroaniline. The bright powder might look benign, but direct contact or inhalation quickly leads to skin irritation, allergy-type reactions, and—if ingested—serious systemic effects. Regulatory standards forced factories to upgrade their engineering controls, adding LEV systems and insisting on sealed packaging lines. Gloves, splash goggles, and face shields now form the basic kit for any technician working near the compound. Spills trigger specific cleanup protocols, sometimes involving neutralizing washes and dedicated disposal containers. Training now stands front and center: every operator learns about acute risks from accidental exposure, chronic effects from low-level exposure, and the best escape routes in case of a process upset. Even the surrounding community pays attention, following news about accidental spills or fires and keeping a wary eye on compliance reports.
Application Area
Anyone who’s bought brightly colored textiles or owned a piece of vivid plastic has felt the impact of 2-Chloro-4-Nitroaniline, even if they never cracked open a chemistry book. Dye makers depend on the compound as a building block for azo dyes, which show up in everything from shirt fabrics to book covers. In pharma, it acts as a stepping stone for certain drug molecules, although its use gets limited by toxicity concerns. Some agrochemical processes pull this intermediate into their synthetic pathways, extending its reach into crop protection. With stricter regulations over the last twenty years, more industries demand cleaner derivatives with less lingering residue, yet the parent compound keeps its role as a go-to route for both affordability and reliability.
Research & Development
Science marches on, and 2-Chloro-4-Nitroaniline always finds itself under the microscope for fresh tweaks and applications. University chemists set out to craft milder reaction conditions, shaving off energy costs while trimming the generation of hazardous by-products. Industry research teams measure every variable, aiming to extract higher purity at lower costs. Some of the most forward-thinking work dives into alternative solvents and catalysts, trying to break away from legacy processes known to pollute waterways. In the search for safer alternatives, startups and big corporations jostle for new patents, hoping a clever modification will win market share for both chemical performance and lower environmental impact. As more industrial standards call for lifecycle analysis, innovators get pressured to demonstrate cradle-to-gate responsibility, not just bottom-line value.
Toxicity Research
Any conversation with environmental scientists or occupational health experts jumps quickly to the risks stacked against 2-Chloro-4-Nitroaniline. The chemical’s nitro and chloro pieces set off alarm bells for toxicity, especially after animal studies linked exposure to organ damage and carcinogenicity in certain contexts. Chronic exposure—low-level but consistent—gets most of the attention, especially in communities living near chemical plants or waste disposal sites. Every year, toxicologists publish new results on environmental breakdown rates and the possibilities for bioaccumulation, which influence not just industrial practices but also regulatory limits. Safer alternatives are catching on, but substitution often brings its own trade-offs. For now, the big-ticket solution comes down to tighter controls, better waste management and ongoing monitoring of both workplace air and water emissions.
Future Prospects
Looking ahead, nobody expects the demand for core chemical intermediates like 2-Chloro-4-Nitroaniline to vanish soon. A shift toward safer, greener alternatives trickles through technical conferences and academic publications, but widespread change moves at the pace of regulation and corporate risk-reward calculation. Some research spots new catalytic methods cutting both energy input and toxic output, a step in the right direction. Others speculate about emerging applications in specialty polymers or next-gen pigments tuned for unique applications in electronics or sensing materials. The bottom line holds steady: as long as the cost-performance equation tips in its favor, the compound stays in circulation. My own view, shaped by years sharing workspace with everyone from bench chemists to industrial engineers, says we’ll see steady but not seismic change—incremental improvements in safety, environmental controls, and possibly a resurgence of innovation the minute regulatory frameworks advance.
What is 2-Chloro-4-Nitroaniline used for?
A Closer Look at 2-Chloro-4-Nitroaniline
Every day, behind the scenes, chemicals like 2-Chloro-4-Nitroaniline shape the products we use and the processes we trust. For folks who don’t spend time in a chemistry lab, the name sounds daunting, but its uses touch everything from our clothes to the crops in our fields. I’ve seen its presence most clearly in the world of dyes and pigments, where it brings out colors in a way a home-mixed pigment just can’t match.
Colorants and the Story of Bright Fabrics
This compound plays a key part in making azo dyes, bringing brightness and lasting color to fabrics. Manufacturers value compounds like this because they deliver in terms of vibrancy and persistence. I remember touring a textile factory and talking with old-timers. They’d grown up in a world of plant-based dyes, then saw the switch to tougher synthetic colors. Without reliable intermediates, nothing looked right for long—faded reds, muddy oranges, weak yellows. 2-Chloro-4-Nitroaniline fixes that. Dyers rely on its chemistry to keep shirts, upholstery, and carpets looking lively through sun and wash.
Crop Protection and Agricultural Tools
Agriculture leans heavily on chemicals to keep crops safe and healthy. 2-Chloro-4-Nitroaniline acts as an intermediate in certain pesticide formulations, which help farmers fight off insects and blights. In my conversations with agronomists, they’ve been honest about the risks: weeds and pests cost them dearly, and having blockers that keep yields healthy changes the equation for struggling farms. Still, this is not just about boosting numbers; it’s about getting food on the table, keeping farms in business, and fighting the unpredictable blows of weather and insects.
Environmental and Health Questions
No chemical story is complete without talking about the risks. I’ve watched communities wrestle with what happens when industrial chemicals enter waterways or air. 2-Chloro-4-Nitroaniline is no exception. Its production and use need careful handling—long gloves, respirators, strict protocols in factories—because the risks linger in the air and, potentially, in the ground. Researchers have studied its toxic effects, not just for plant workers but for wildlife. If disposal or storage gets sloppy, the chemical could endanger nearby neighborhoods or water supplies.
Better Practices and Shared Responsibility
People sometimes ask if we could do without chemicals like this one. In a perfect world, maybe. But so much depends on responsibly managed chemistry. I’ve seen factories transform when managers pushed for cleaner processes: filters on smokestacks, scrubbers in wastewater pipes, tight training for everyone who handles hazardous goods. Many countries now require tracking and documentation for every batch of 2-Chloro-4-Nitroaniline, and that’s a good start.
For folks inside the industry, embracing alternatives—safer or more sustainable options—makes sense. Green chemistry brings new hope, but the reality is: change takes investment, research, and time. Consumers and communities need transparency about what makes it into their clothing or food systems, and to press for smarter regulation. Responsible chemical management protects more than the bottom line; it shields the people who make, use, and live with these products every day.
What are the safety precautions when handling 2-Chloro-4-Nitroaniline?
Respecting Chemical Hazards in Real Life
Years ago, I watched a coworker brush off chemical hazards and pay the price—one careless moment, and the emergency eyewash station got its first workout in months. The lesson stuck. Chemicals like 2-Chloro-4-Nitroaniline demand respect, not just from regulations but from what they can do to a person’s body. I’ve handled compounds in cramped, stuffy rooms with nothing but a fan, and I’ve also worked in well-equipped labs. I know which days I slept better.
The Red Flags of 2-Chloro-4-Nitroaniline
The substance looks harmless—a yellowish powder that doesn’t seem threatening. But beneath that mild look hides a toxic punch. It irritates skin, eyes, and respiratory tract. The chemical can enter the body just from touching or inhaling the dust, leading to headaches or even organ damage if you’re careless. Here’s what years in research have drilled into me: don’t assume the powder won’t travel. Gloves and masks offer real protection.
No Shortcuts: Personal Protective Equipment (PPE)
Nitrile gloves beat bare hands every time. Latex tears too easily, and cloth soaks up the substance. For eyes, wraparound goggles shield more than safety glasses do—chemicals find seams. A disposable lab coat keeps powder from sticking to clothes and hitching a ride home. For volatile fumes, a respirator with the correct cartridge filters out dust and vapor, keeping your lungs clear.
My colleagues and I check PPE for splits before every use and toss it after. It’s not wasteful—it’s self-preservation. Careful storage of gear means everyone finds what they need, even in a hurry.
Good Habits Save Trouble
My first months in the lab, I learned to treat every surface as contaminated. This worked better than a thousand warning signs. Lining up work in a fume hood controls exposure—those fans drag off stray particles, so mouths and noses don’t suffer. Good fume hoods also keep air moving in the right direction, away from faces. Keeping the lid on containers and scooping out only what you need cuts down spills.
Eating and drinking in chemical areas looks tempting—it’s also reckless. One sandwich next to a beaker could mean swallowing a tiny, invisible amount. The rules exist because mistakes last. Handwashing doesn’t just keep food safe later; it stops you from absentmindedly touching your face.
Tidy Working: Cleanups Make a Difference
Spills demand action, not panic. Absorb spills with appropriate material—never with paper towels or rags. Seal the mess in a proper hazardous waste bag and label it clearly, so the right people can handle it. Leaving a cluttered workspace triples accident chances; putting away powders and cleaning as you go helps everyone breathe easier.
Training and Teamwork
Safety isn’t a one-person show. In a decent lab, newcomers shadow experienced hands until they build muscle memory: donning PPE, disposing of waste, signing out chemicals. Regular drills and updates keep practices fresh. If something feels off—like a wrong smell or rashes—speaking up stops trouble before it spreads.
Respecting substances like 2-Chloro-4-Nitroaniline isn’t about fear. It’s about experience, common sense, and looking out for each other. Good habits, solid equipment, and shared knowledge keep us all standing at the end of the day.
What is the chemical formula and molecular weight of 2-Chloro-4-Nitroaniline?
Demystifying the Formula
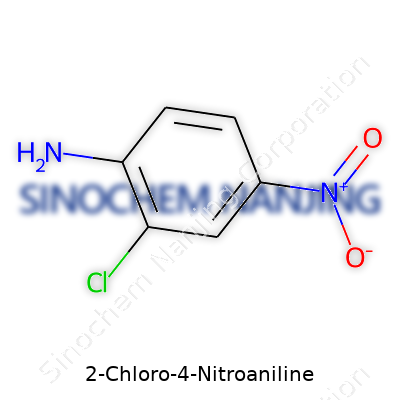
Chemistry reveals itself in the details, especially with a compound like 2-Chloro-4-Nitroaniline. Its chemical formula is C6H5ClN2O2. Visualizing the structure of something as simple and as specific as this helps ground the discussion beyond labels and charts. Molecular weight comes next, and you’re looking at 172.57 grams per mole for this molecule. The numbers don’t just matter on paper—they set expectations for how a sample will behave during storage, synthesis, and any downstream applications.
Why Exact Chemical Information Matters
Lab work demands accuracy and consistency. One wrong value in molecular weight, or a misjudged formula, leads quickly to ruined experiments or, even worse, risky results. Anyone who’s run a reaction only to watch the yields nosedive because of a data mismatch knows the frustration and waste in time and resources. With C6H5ClN2O2, clarity on numbers means the difference between good science and a failed batch.
Beyond the lab, clear chemical formulas and weights give manufacturers a reliable way to track and handle bulk shipments. Industries handling dyes or intermediates trust the formula to determine required quantities and to anticipate regulatory checks, shipping restrictions, and exposure risks. Errors in these calculations don’t just hit the bottom line—they pile hazard on top of logistical strain.
Applications: Where 2-Chloro-4-Nitroaniline Appears
Daily life doesn’t announce every molecule at work, but specialty chemicals such as this show up again and again. Factories deal with nitro anilines as building blocks for dyes and pigments that add color to fabric, plastics, or inks. Pharmaceutical firms sometimes tap this compound for its functional groups during synthesis processes. That nitro group and the chloro substitution offer flexible points for further chemical reactions, turning a simple aromatic ring into something far more complex and valuable.
Direct contact isn’t something many people seek, and most will never knowingly handle the powder. That doesn’t make it less important or less common. Trace residues and regulated amounts travel across borders and sit on regulatory registers. Global trade tracks every kilo, and safety checks focus hard on accurate labeling.
Challenges and Approach to Safer Handling
Exposure matters. Safety data for 2-Chloro-4-Nitroaniline calls for proper protective gear. Knowledge about this chemical—such as its formula and weight—equips workers to recognize risks. In practice, a label printed with the right numbers on every drum means that anyone down the line knows exactly what’s inside and how much to expect, avoiding surprises and dangerous misidentification.
Mistakes get expensive fast. Emergency responses depend on accurate identification. Response teams look at chemical formulas and weights before deciding what protective steps to take in a spill or accident. The right data cuts down on confusion. It’s worth taking the time to double-check every shipment and update inventory logs to reflect what actually sits in the warehouse, not just what’s on the invoice.
Steps to Take
Establish clear protocols for verification—compare supplier data sheets with trusted chemical databases. Stockrooms and labs need up-to-date safety information tied to every batch. Invest in staff training, not only for the “how to handle” aspect but also for reinforcing the “why.” Cross-checking chemical formulas and weights at every handoff along the supply line helps everyone focus on safe, productive work. Precise knowledge isn’t just a convenience, it’s protection.
How should 2-Chloro-4-Nitroaniline be stored?
Why Proper Storage Matters
2-Chloro-4-Nitroaniline isn’t a household name, but anyone who has worked in a chemical lab or manufacturing site knows this material well—yellowish, crystalline, and capable of causing serious harm if it’s not treated with respect. It only takes one moment of complacency for exposure incidents or contamination to strike. Chemical safety doesn't just protect product quality or equipment; every moment invested in good storage safeguards human health and the well-being of those nearby.
Direct Risks
Exposure to this compound may lead to skin, eye, or respiratory irritation. Prolonged contact or inhalation ramps up the risks, from headaches to longer-term organ problems. In case of accidental fire, toxic gases can enter the air, and once clean-up teams start suiting up, everyone wishes the storage protocols were stronger. The U.S. National Library of Medicine and the European Chemicals Agency both flag this substance as dangerous when safety basics get skipped.
Key Storage Principles
Keep the original, clearly labeled container. With so many yellow powders on a chemistry shelf, there’s too much room for error if containers get swapped. Place it in a dry location, away from sunlight and well away from heat sources. Exposure to moisture and heat isn’t just about product degradation—impurities or even a reaction can occur in poorly controlled environments.
Aim for a cool room with air circulation. Many chemicals can emit invisible fumes, and 2-Chloro-4-Nitroaniline fits into that category if left exposed or poorly sealed. Good airflow helps break up any accumulating vapors, reducing inhalation risks.
Never store the compound near food, drinking water, or personal items. Splashes and accidental cross-contamination pose dangers most people don’t consider until an acute incident reveals the gap in practice. In most labs I’ve seen, the simplest rule is: chemicals and calories never mix, no matter how organized the shelves might look.
Security and Segregation
Chemicals like acids, bases, and oxidizers shouldn’t share a shelf or cabinet with 2-Chloro-4-Nitroaniline. Accidental mixing through spills or even leaky caps can set the stage for unpredictable and sometimes violent chemical reactions. Using separate, locked, and specifically labeled cabinets for hazardous substances goes a long way here.
Limit access. Only trained staff should handle or even approach this material in its storage spot. This reduces theft, mishandling, or exposure—especially for new hires or visiting personnel who don’t recognize the risks at a glance. I always recommend a logbook at the storage site, forcing a pause for reflection and accountability each time the compound moves in or out.
Supporting Solutions
Routine inspections should be part of any chemical storage plan. Spot leaks, corroded containers, or faded labels before they lead to emergencies. It helps to assign a single individual to check the site each week, ensuring consistency and follow-up on any issues.
Post clear emergency instructions right next to the storage area. Include spill containment steps, use of personal protective equipment, and contact numbers for first aid or chemical safety professionals. Proximity and clarity can minimize panic and mistakes during real emergencies.
Keep clear documentation of every purchase, use, and disposal event. The EPA and OSHA emphasize tracking for both legal and safety reasons, and in any audit or spill situation, these records help piece everything together quickly.
Final Thoughts
Chemical storage sounds tedious until neglect turns into a crisis. It doesn’t matter if you work in a huge facility or a two-person workshop—the practices that keep 2-Chloro-4-Nitroaniline under control are the same. Trust in labeling, priority for segregation, and a culture that cares about every small detail remain the frontline defenses against disaster.
Is 2-Chloro-4-Nitroaniline hazardous to health or the environment?
The Substance in Industry
2-Chloro-4-nitroaniline lands on many chemical inventories because of its use in dyes, pigments, and some pharmaceuticals. In manufacturing, it plays a role in coloring textiles, plastics, and rubber. There’s a temptation to see something with such utility as safe, especially since it’s tucked behind factory doors and lab benches. Still, long-term experience with industrial chemicals forces anyone working with them to respect the risks.
Human Health Concerns
The structure of 2-chloro-4-nitroaniline means it brings both nitro and aniline groups—both notorious for their own risks. People can absorb these chemicals by inhalation, skin contact, or accidental ingestion. Many health agencies, including the International Agency for Research on Cancer, describe aniline compounds as probable or possible carcinogens. The nitro group can form metabolites in the body that damage blood cells, leading to methemoglobinemia. That’s not an abstract risk. Workers in dye shops and chemical plants often run into headaches, cyanosis, dizziness, or worse from direct exposure.
My time at a midwestern chemical plant taught one lesson above all: don’t underestimate skin and respiratory exposure. After seeing a co-worker land in the hospital from ignoring a few basic protocols, the point hit home. Not every hazardous substance gives off a sharp smell, but the threat remains all the same.
Environmental Impact
The molecule doesn’t just vanish after leaving a drum or reactor. 2-Chloro-4-nitroaniline finds its way into wastewater from dyeing operations. Bacteria and sunlight don’t break it down quickly, which means rivers and soil can hold onto contamination. Studies on aquatic organisms point to toxicity at relatively low concentrations. Fish and amphibians exposed to the chemical often show stunted growth, nervous system damage, or elevated mortality.
Communities relying on local water sources downstream from chemical factories must stay alert. Even trace amounts of persistent chemicals, especially those containing chlorine or nitro groups, can harm generations of wildlife and eventually show up in drinking water. A river in South Asia made headlines not for its color, but for the dramatic loss of biodiversity after years of aniline discharge.
Managing the Risks
No chemical with these hazards belongs around food, open water, or without proper controls. Personal protective gear, sealed handling, and air monitoring turn into basic requirements, not optional extras. Regulations push for stricter discharge limits and better wastewater treatment, yet loopholes and poor enforcement keep the problem alive. Investing in closed-loop manufacturing and safer alternative colorants makes more sense than doubling down on old tech.
On the home front, education does as much as regulation. Workers must recognize symptoms early and know their rights to safe working conditions. Suits, gloves, and respirators sometimes frustrate fast-paced production, but health always takes priority. Experience teaches that no one wants to trade years of life or well-being for a shortcut.
Fact-Based Decision Making
The risks of 2-chloro-4-nitroaniline are not theoretical. Decades of poison control data, case reports from hospitals near chemical clusters, and regulatory filings across continents all tell the same story. Firms using this chemical need clear protocols, full disclosure to their staff, and transparent records open for audit. Environmental monitoring should extend to nearby communities, not just factory gates.
A world that values progress shouldn’t ignore invisible dangers just because they wear a complicated chemical name. The health of workers, neighbors, and nature depends on real respect for substances like 2-chloro-4-nitroaniline.
| Names | |
| Preferred IUPAC name | 4-chloro-2-nitroaniline |
| Other names |
4-Nitro-2-chloroaniline 2-Chloro-4-nitrobenzenamine 1-Amino-2-chloro-4-nitrobenzene 2-Chloro-4-nitroaniline p-Nitro-o-chloroaniline |
| Pronunciation | /tuː-ˈklɔːr.oʊ-ˈfaɪ.nɪ.trəʊ-ˈæn.ɪ.liːn/ |
| Identifiers | |
| CAS Number | 89-63-4 |
| Beilstein Reference | 1207936 |
| ChEBI | CHEBI:34405 |
| ChEMBL | CHEMBL44448 |
| ChemSpider | 17702 |
| DrugBank | DB14611 |
| ECHA InfoCard | 03c9a7e1-d484-42ca-bcf7-bb9ac9177eb9 |
| EC Number | 202-200-1 |
| Gmelin Reference | 82273 |
| KEGG | C09743 |
| MeSH | D021106 |
| PubChem CID | 6924 |
| RTECS number | BX3850000 |
| UNII | 1I26Q93631 |
| UN number | UN3446 |
| Properties | |
| Chemical formula | C6H5ClN2O2 |
| Molar mass | 197.56 g/mol |
| Appearance | Yellow to brown crystalline powder |
| Odor | Odorless |
| Density | 1.41 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 1.25E-4 mmHg at 25°C |
| Acidity (pKa) | 1.68 |
| Basicity (pKb) | 9.88 |
| Magnetic susceptibility (χ) | -38.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.67 |
| Viscosity | 32 cP (20°C) |
| Dipole moment | 3.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −35.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −1662 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes skin irritation; causes serious eye irritation; may cause respiratory irritation |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335, H410 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P333+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-Chloro-4-Nitroaniline: 2-2-0 |
| Flash point | 120 °C |
| Autoignition temperature | 454 °C |
| Lethal dose or concentration | LD50 oral rat 934 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2,601 mg/kg |
| NIOSH | CN 9660000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
4-Nitroaniline 2-Chloroaniline 2-Bromo-4-nitroaniline 2-Chloro-5-nitroaniline 2-Chloro-4-nitrophenol |

