2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate: Beyond the Label
Historical Development
The world of agricultural chemistry changed a lot after World War II. Synthetic pesticides grew out of the urgent drive to boost food production and control pests that once devastated fields. Back then, folks rarely gave a second thought to what would happen to farm chemicals after they did their job. 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate came about during deep dives into carbamate chemistry. Research teams—searching for pest killers with surgical precision, less persistence than organochlorines, and less price volatility than organophosphates—found carbamates promising. Interest spiked in the 1960s and 70s. That era’s textbooks and trade journals show how this compound caught the attention of chemists who aimed to walk the line between pest control and human safety. Years of incremental tweaks brought this carbamate into the toolkits of both scientists and growers, even as the public learned more about what actually happens, chemically and biologically, after the spray fades.
Product Overview
This carbamate’s structure packs a punch: a phenyl ring with two methyl groups at the 4 and 5 positions, a chlorine at the 2 position, and a methylcarbamate chunk joined to the ring. This particular combination set out to knock back pests with dose control and reasonable breakdown rates, in contrast to legacy compounds that lingered far longer than anyone expected. Labeling standards evolved, guided by both scientific study and mounting regulatory frameworks that demanded clear identification of active ingredients and detailed use guidance. Through my own conversations with agronomists, I've heard the same story: this molecule found its place in a competitive roster, each product vying for a blend of knockdown power, selectivity, and acceptability to markets wary of pesticide residues and environmental side effects.
Physical & Chemical Properties
Handling this particular carbamate means dealing with a colorless to light-crystalline substance. It doesn’t dissolve much in water, but common organic solvents like acetone and alcohols take it up pretty well. Its melting point and thermal stability encourage storage in dry, cool areas. Chemists learned early that even small tweaks to a molecule’s structure shift how it behaves on a plant, in water, or under sunlight. Exposure to strong bases, oxidizers, or moisture over long periods can degrade this carbamate—sometimes into far less understood byproducts. Careful formulation helps delay that process, but no system is perfect. From lab bench to field tank, the physical quirks and chemical reactivity of this molecule have driven changes in how it is packaged, transported, and stored.
Technical Specifications & Labeling
Current labeling goes far beyond the minimal templates seen in early years. Requirements have become clear: percentage of active substance, application rates, safe intervals, signal words, and clear pictograms show up on every shipment, large or small. Few people who use these compounds ever read the full technical bulletins, but the details shape how regulators and stewardship programs think about safety and transparency. I’ve sat through regulatory workshops where staff pore over every word, knowing that full disclosure paints a more accurate—and ultimately safer—picture for users and communities. That scrutiny highlights a bigger shift: traceability and transparency no longer stand as abstract ideas, but form the backbone of public trust in agricultural chemistry.
Preparation Method
Producing 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate takes more than an afternoon in a lab. Synthesizing this compound involves introducing a carbamate moiety onto a selectively chlorinated and methylated phenyl base, a sequence needing both patience and strict temperature control. Chemists prepare the aromatic base via targeted substitution reactions before coupling with methyl isocyanate—an ingredient itself notorious for safety risks. Stories from the industrial side stress just how important it is to keep reaction vessels sealed, waste managed, and operators protected by rigorous protocols. Changes in synthesis routes over the years reflect both the push for higher yields and the drive to cut hazardous byproducts. These choices at the bench scale ripple out to affect the sustainability, cost, and risk profile for every kilogram produced.
Chemical Reactions & Modifications
The molecule’s reactivity allows chemists to carry out further substitutions or even build hybrids with new biological targets. In early days, teams attempted to swap out the methyl groups or tweak the carbamate moiety to maximize pest specificity. Those studies helped explain why only certain bugs fall, and why others show resilience. The chlorine group seems critical for disrupting particular enzymes in pests; try to change it, and sometimes the compound just fizzles. Still, process chemists keep searching for cost-effective adjustments that maintain potency while trimming production steps or simplifying waste management. Pretty soon, future modifications may tailor activity profiles for specific geographies or climates, keeping one step ahead of shifting pest populations and new resistance patterns.
Synonyms & Product Names
This carbamate goes by a handful of names in scientific literature and on the farm. Many industry insiders use common trade names, which can shift by region. For those digging into academic articles or regulatory filings, the full IUPAC name or a concise system name often appears. These labels matter. They cut through confusion, especially as importers, exporters, and handlers coordinate through global supply chains. Over time, mislabeling controversies have reminded everyone that the stakes rise with cross-border trade. Transparency and standardized nomenclature work together, protecting both public health and business interests, as I’ve seen during audits at border warehouses.
Safety & Operational Standards
Modern agricultural work depends on following strong safety measures. I’ve seen firsthand how field workers change their routines when handling carbamate pesticides, donning gloves and respirators, sticking to strict re-entry intervals, and undergoing regular health checkups. The stakes weigh heavier for those closest to the compounds—farm laborers, chemical mixers, and applicators face the biggest hazards from mishandling or leaks. Stories of exposure underscore the need for well-maintained protective equipment, clear signposting, and ready access to clean water for decontamination. Many of the worst-case scenarios result from cutting corners: poorly ventilated storage, mixing near water sources, or failing to train new hires. Strong operational standards, grounded in hard evidence and backed by clear, enforceable rulebooks, keep workers safer and cut down on emergencies and long-term health problems.
Application Area
This compound found a place mainly in commercial agriculture, especially where persistent pests shrugged off older pesticide options. Growers working with a variety of crops—such as fruits, field vegetables, and ornamental plants—chose it for both foliar spray and soil treatment. Conversations at extension meetings often revolved around the latest data: what crops benefit most, which insects show resistance, where residue concerns drive buyers’ decisions. Urban settings rarely see this compound, but its reputation spreads through news on food safety and occupational hazards. Regulatory tightening and consumer demands for residue-free food are nudging many operations to reconsider or even phase out certain carbamate applications.
Research & Development
The research community keeps trying to outthink both pests and the public’s shifting standards. Academic labs and private firms run battery after battery of assays testing analogues for broader spectrum, shortened breakdown times, and reduced non-target toxicity. Technology marches forward: powerful analytical equipment tracks trace residues, and big data platforms model pest resistance trends. Many teams are searching for the holy grail—targeting only the intended pest, sparing everything else, and disappearing quickly after doing the job. Improved understanding of enzyme pathways and gene targets guides new synthesis strategies. Sometimes these advances trickle out slowly, caught in patent disputes or cost barriers, but once in a while a breakthrough shapes the landscape overnight.
Toxicity Research
Public concern about chemical exposure runs deep, and for good reason. Acute toxicity in mammals led researchers to test a wide range of endpoints, from enzymatic inhibition to eye and skin irritation. Results published in toxicology journals show carbamates inhibit acetylcholinesterase not just in target pests but in exposed humans and wildlife too. Case studies from poison control centers, along with careful animal testing, direct policymakers to set exposure limits and re-evaluate permissible uses. Chronic exposure studies and environmental monitoring continue, especially as regulators adjust what levels they consider safe in food and water. I've heard from colleagues in pesticide monitoring that regular testing now forms a non-negotiable step for both domestic producers and importers aiming to reassure cautious consumers.
Future Prospects
Uncertainty trails every legacy agrochemical compound. Changing weather patterns, globalized food markets, and the rising cost of regulatory compliance push everyone—growers, scientists, companies—toward more selective, less persistent alternatives. Development of resistance in key pests threatens the effectiveness of even well-established products, and stricter residue regulations from large markets raise the bar. Advances in biopesticides, RNA interference, and precision application hint at a new era where broad-spectrum carbamates may fall out of favor for many crops. Still, some regions lack affordable replacements, and the deep experience growers and applicators have with carbamates adds inertia to any shift. Achieving meaningful progress calls for open data sharing, investment in safer chemistry, and policy that balances food security with health and environmental protection. Based on decades of watching this sector adapt, what comes next will draw on scientific creativity, government oversight, and real-world wisdom from those closest to the field and the lab bench.
What are the main uses of 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate?
Why Farmers Rely on This Carbamate Compound
Ask a row crop farmer or a citrus grower about their biggest problems, and at least half will bring up insects chewing through all their hard work. 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate steps into the story as a trusted carbamate insecticide, working against those pests that refuse to back down. This compound, known in the agricultural community as Propoxur, has built a reputation as a reliable solution when resistance to other materials creeps in.
Out in the fields, crops go up against a steady line of beetles, weevils, caterpillars, and aphids. Propoxur helps by hitting their nervous systems. It interrupts acetylcholinesterase, an enzyme bugs depend on to keep their nerves firing properly. Once exposed, insect muscles flop instead of twitching, and soon, pest populations take a noticeable dip. Many fruit growers, particularly those raising apples and citrus, use it for that quick knockdown when pest counts spike. Even public health workers put it to work gunning for disease-carrying cockroaches and mosquitoes in urban settings.
Public Health: Beyond the Field
Farmers aren’t the only ones who see results from Propoxur. Cities with cockroach or mosquito trouble turn to it for controlling outbreaks. Urban centers with limited options after older chemistry fades out sometimes reintroduce Propoxur into rotation. Health departments treat sewage works and drains to tame mosquito populations. The Centers for Disease Control recognizes carbamates like this one as a valuable part of integrated vector management, especially when focusing on areas plagued by insect-borne diseases like dengue or West Nile virus. My own county’s vector control office relies on it during peak summer outbreaks because the alternative sometimes means even nastier chemicals that linger in the environment for years.
Structural Pest Control
Walk into an older building, especially in places with moisture issues, and odds are someone’s had to deal with termites, ants, or silverfish. Pest control pros often reach for Propoxur as a crack-and-crevice specialist because of its rapid action and ability to tackle a broad spectrum of crawling insects. As someone who spent a few summer jobs patching up entry points in basements, this compound showed up in many commercial baits or gels. The quick results—dead insects in hours, not days—made it a favorite for rental agencies preparing apartments for new tenants.
Challenges and Looking Ahead
There’s a trade-off in every tool. While Propoxur’s speed impresses, its toxicity to non-target organisms, including birds and some aquatic life, raises concerns. Cases of accidental poisoning in households where proper instructions weren’t followed stick in my mind. I’ve seen extension agents warn against unrestricted residential use, and some countries have restricted availability or pulled it from home and garden shelves. The European Food Safety Authority and US Environmental Protection Agency review safety data to set maximum residue limits in food and monitor environmental impact. These ongoing checks help, but the real solution comes from smarter use—training, better equipment, and ongoing research into less hazardous alternatives. Bioinsecticides and genetic controls move ahead only with informed standards and plenty of field trial evidence.
The Big Picture
Propoxur, or 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate, still holds a place in modern pest control strategies where other tools fall short. It answers the call for both food production and public health. Keeping its use responsible, grounded in clear science and field experience, points agriculture, cities, and homeowners toward safer, more sustainable pest management in the years ahead.
Is 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate toxic to humans or animals?
What Is 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate?
Anyone who spends time on a farm or tends a home garden can tell you about the huge range of chemicals used to keep pests away from crops and ornamental plants. 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate, better known in some regions under trade names like Propoxur, stands among the older, widely-applied carbamate pesticides. People often turn to carbamates because they knock down insects quickly, targeting their nervous systems. Problem is, these same chemicals can have effects that spill far beyond the bugs.
How This Compound Harms Humans and Animals
This carbamate controls pests by blocking acetylcholinesterase — an essential enzyme for the healthy function of nerves. Humans and pets rely on the same enzyme for their bodies to work right. Blocking it scrambles nerve signals, leading to symptoms like nausea, muscle twitching, confusion, and even seizures in bad cases. Larger exposures, like through an accident or poor handling, can bring on more serious effects: trouble breathing, loss of consciousness, sometimes death. Pets get into trouble fast because they sniff or lick treated areas. Birds and small mammals prove even more sensitive than people to nerve agents in this class.
Real-World Exposure: What Are The Risks?
Anyone mixing or spraying carbamates gets exposed most often through skin contact or by breathing tiny droplets. People eating vegetables straight from the garden after a recent spray pick up small amounts too. According to the World Health Organization, Propoxur ranks among moderately hazardous pesticides. Food safety authorities worldwide track residue levels to keep them below set limits. I’ve seen how improper storage or disposal, especially in rural settings, leads to poisonings, not just for farmers but for local wildlife and children. Accidential exposures from unlabeled containers or spilled product also make the news more often than many city folks realize. Dogs and cats walk through recently treated areas and then lick their paws — a direct ticket for poisoning which shows up as twitching, vomiting, or worse.
Environmental Impact Adds Extra Weight
Besides immediate toxicity, carbamates don’t stay put. Heavy rain can wash them into streams or groundwater. Even at low concentrations, fish and amphibians stir up trouble; frogs and tadpoles can’t dodge nerve agents like adult animals. Insects meant to be left unharmed, including important pollinators like bees, wind up as collateral damage. It’s sobering to watch bee populations slump after a big spray day, and anyone who has worked the land knows fewer pollinators means less fruit for the next season.
Building Safer Practices
On small farms, simple steps save lives: keep chemicals locked up, train workers, use gloves and masks, and always follow label instructions. Public agencies need strong rules and enough resources for regular residue checks and emergency responses. Integrated pest management — rotating crops, encouraging beneficial insects, taking advantage of less hazardous options — lets farmers use less chemical in the first place. In my own experience, partnerships with extension agents make a real difference because they link science with what works in the field. Home gardeners should choose alternatives if possible and keep kids and pets far away. No one solution fits every community, but awareness and simple actions stop tragedies before they can start.
What are the safety precautions for handling this product?
Recognizing Hazards in the Products We Use
Even something as normal-looking as a cleaning product or garden chemical can be risky if ignored. The hazards often aren’t hiding. Labels shout warnings for a reason. A strong smell, a list of tongue-twisting ingredients, or a “flammable” symbol should signal our brains to take this stuff seriously. People get used to reaching for things under the sink or pulling out a bottle in the garage. Familiar containers can promote a false comfort. That’s how accidents happen, and most of us have seen or heard the stories — a burn, a nasty rash, someone hacking after using cleaner in a closed space. It pays to pause, size up the product, and treat each one with a bit of healthy suspicion.
Personal Protection: Mask, Gloves, and Clothing
A barrier between skin and chemical is never wasted effort. Even dish soap can dry out hands, but things like industrial solvents or pesticides often burn or irritate. Thick rubber gloves beat latex ones when facing strong chemicals. Eyes need just as much help; pouring bleach or mixing garden sprays sends up little splashes and fumes. Good goggles wrap around the side for full coverage. Long pants and sleeves turn a minor spill into a nuisance, not an emergency. Airborne particles in powders, paints, or sprays find their way into lungs quickly. A simple mask or respirator makes a big difference, especially in closed spaces.
Ventilation Beats Regret
Too many folks underestimate how fast fumes can build. Bathrooms, basements, sheds—places without open windows or fans can get toxic soon after you pop a cap. A gust of outside air, an open window, or a small fan can prevent headaches, dizziness, and more serious reactions. I’ve seen what happens in homes where people mix chlorine and ammonia in a stuffy room. It’s not pretty, and it always ends with a frantic rush for air. If a breeze isn’t possible, step out often for fresh air. Good habits around ventilation protect far better than any warning label.
Storage: Out of Sight, Secure from Accidents
Leaving strong products where curious kids or pets can reach them doesn’t just lead to close calls; it can cause long-term health trouble. High shelves and locked cabinets slow down accidents. Lids need to go back on tight, bottles out of reach and away from snacks or pantry food. Mixing up containers—pouring a cleaner into an old soda bottle—has led to tragedies more than once. The original container always reports danger in language or symbols that anyone can recognize. Take old or leftover material to the local hazardous waste center. I dropped off unused paint thinner last year and learned from the staff how many fires and poisonings start with leftovers kicked under a workbench.
Information and Smart Decisions
I check the product label or manufacturer’s website before trying unfamiliar stuff. Most brands offer a safety data sheet that spells out problems and first-aid measures in plain terms. That’s not information for scientists alone. Reading these sheets taught me that products with “quaternary ammonium” in the name demand serious respect and quick cleanup. If something spills or splashes, a rinse with clean water works best for skin or eyes while calling for help. Emergency numbers from poison control or a label belong in any workshop or laundry room. Instead of just squinting at symbols or trusting luck, preparing and reading up keeps everyone safe without overthinking the task.
How should 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate be stored and disposed of?
Recognizing Why Safe Handling Matters
Anyone who’s spent time near industrial chemicals or on a farm knows that carbamates aren’t just another item on the shelf. They protect crops, but exposure brings risks: headaches, dizziness, and worse. 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate falls right into that category. Stories of careless storage leading to chemical leaks or accidental poisoning are not unusual, and those don’t just make for bad headlines—the consequences last long after news fades. Good habits make the difference between safety and stories you never want to tell your neighbors.
Taking Storage Seriously
Sealed drums in a locked, ventilated shed kept my crew out of trouble on tough summer days. Humidity always lurks as an enemy, breaking down containers and sending fumes where they’re not wanted. Any chemical labeled with “carbamate” in the name belongs far away from feed, seed, and water sources—one spilled bag and you’ve got a problem for both wildlife and families. Keeping it under lock and labeled in plain language cuts down confusion for visiting workers or young hands eager to help out.
If you’ve ever seen corrosion on a drum’s edge, you know air finds its way in. Plastic containers crack with heat. Stick with original packaging when possible, and avoid transferring contents if you can. Storage temperature shapes outcomes, too—warmer spots raise the chances of breakdowns and toxic fumes, so cool, shaded areas work better. Nobody wants to find a leaking container because the storeroom baked in the sun all summer.
Protecting People and the Environment
Runoff after heavy rain doesn’t just stick to drainage systems—it travels, and what heads into streams or groundwater won’t disappear on its own. Studies from the EPA and CDC point out how even low levels of some carbamates can mess with aquatic life for months. Old, unlabeled bottles can turn into environmental headaches if they’re tossed with other trash or dumped out “just to get rid of it.” The urge to take shortcuts has never ended well for communities downstream.
Disposal That Doesn’t Cut Corners
Calling your local hazardous waste line isn’t a silly step, and city or county governments often run collection events for chemicals like this one. Incineration at licensed facilities (with scrubber systems that capture toxic byproducts) stands out as the main route for leftover carbamates—landfilling or burning on-site risks too much. Empty containers need triple rinsing, and the rinse water counts as hazardous too; don’t pour it into the soil or a regular drain. These are the sort of details those who work with pesticides or carbamates keep in the back of their minds, knowing fines and environmental trouble lurk behind every lazy move.
Solutions for Safer Communities
Training goes further than the required safety meetings. Sharing what happens after accidents helps the next group avoid the same fate. Modern tracking systems, like barcoded warnings and automatic inventory checks, mean fewer surprises when it’s time for inspections. Community outreach isn’t just for outsiders; local talks and on-the-job mentoring help anchor these routines in the real world. We’ve seen it make the difference between “just another job” and a workplace where people look out for each other.
Careful storage and disposal practices respect more than rules—they protect health, water, and the land itself. Every community benefits when old lessons meet new tools and shared responsibility stays the center of the story.
What is the chemical structure and molecular weight of this compound?
Getting Familiar with Chemical Structure
Anyone digging into chemicals runs into the puzzle of structure right away. Every molecule has its own blueprint—a map showing how atoms stick together and what kind of bonds tie them up. People often imagine it’s just about formulas, but there’s a lot to learn from these little diagrams. Chemistry classes drill this point home: swap a single bond for a double, toss in a ring, or remove one hydrogen, and the whole property set can flip. Acetaminophen and phenacetin look pretty close on paper, but that little tweak in arrangement changes how the body deals with them. That’s why you see scientists studying how the atoms are arranged, not just the names or numbers.
Molecular Weight: Not Just Number Crunching
People often breeze past molecular weight, thinking it’s just another figure in a textbook. This number—measured in daltons or g/mol—shows up everywhere, from drug approvals to manufacturing choices. In the lab, molecular weight shows how much of a compound fits in a beaker, and helps chemists pocket out dose sizes with real accuracy. Back in college, a lab partner and I once doubled a reagent’s weight, not realizing the compound’s structure carried extra heavy atoms. The reaction fizzled. Since then, I check my numbers every single step—one wrong value, and nothing works right.
Why Structural Details Shape Performance
The structure of a molecule decides things: how it mixes with water, whether it breaks down in sunlight, even how it tastes. Vanillin and ethyl vanillin share the same family, but that extra ethoxy group turns ethyl vanillin into a flavor powerhouse, about four times as strong as regular vanilla. Chemists pay attention to such details for a reason. In medicine, these structural differences draw a line between a life-saving drug and a dangerous lookalike. For example, thalidomide—one structure helps treat cancer, but its mirror image led to disaster in the 1960s.
Fact Checks and Reliability
Accuracy in reporting structure and weight helps everyone, from pharmacists to farmers. A single digit slip on a datasheet ruins batches or could even spell danger. Regulatory agencies like the FDA lean on precise chemical data to greenlight new products, because public safety depends on it. In my own experience, requesting a certificate of analysis for every new shipment became a habit, after once seeing a lab shut down over mislabeled containers. Businesses that value transparency and use independent testing labs build stronger trust.
How to Get the Right Details
Instead of trying to sketch out molecules by hand or trusting the first online listing, folks should rely on trusted, peer-reviewed literature or company-provided material safety data sheets. Reputable databases like PubChem or ChemSpider offer thorough breakdowns, including three-dimensional structures and every atom’s role. In-house labs double-check laboratory results against these, not just out of habit, but because the cost of skipping this step could be much higher.
Solutions and Practical Steps
Consistency in describing and handling chemicals starts with education. Sharing guides and checklists for reviewing chemical data helps new technicians pick up good habits fast. Companies investing a bit more in quality assurance see fewer mistakes and avoid expensive recalls. Instead of tossing information into a binder, organizations can use simple digital systems to flag inconsistencies early. Holding workshops with experts from regulatory or academic circles also fills gaps in local knowledge, reducing trial-and-error.
Safe science grows from details. Structure and molecular weight aren’t just facts on a page—they shape the way the world uses chemicals every day.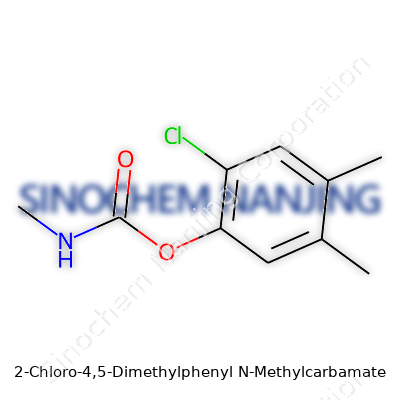
| Names | |
| Preferred IUPAC name | 2-chloro-4,5-dimethylphenyl N-methylcarbamate |
| Other names |
Propoxur Baygon Blattanex Bolfo Unden OMS 33 Prentox Sendran |
| Pronunciation | /tuː-ˈklɔːr.oʊ-ˈfɔːr-ˈfaɪv-ˈdaɪˌmɛθ.əl-ˈfiː.nəl ɛn-ˈmɛθ.əlˈkɑːr.bəˌmeɪt/ |
| Identifiers | |
| CAS Number | 2387-70-8 |
| Beilstein Reference | 3526423 |
| ChEBI | CHEBI:39700 |
| ChEMBL | CHEMBL2106887 |
| ChemSpider | 167361 |
| DrugBank | DB11438 |
| ECHA InfoCard | 03a58d2d-0639-4bb8-8b11-13058fa966ae |
| EC Number | 255-355-7 |
| Gmelin Reference | Gmelin Reference: 83458 |
| KEGG | C18437 |
| MeSH | Dichlorvos |
| PubChem CID | 151426 |
| RTECS number | GG3675000 |
| UNII | W8M27IRM23 |
| UN number | UN2757 |
| CompTox Dashboard (EPA) | 2-chloro-4,5-dimethylphenyl N-methylcarbamate |
| Properties | |
| Chemical formula | C10H12ClNO2 |
| Molar mass | 193.67 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.16 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.96 |
| Vapor pressure | 0.000017 mmHg (25°C) |
| Acidity (pKa) | 13.2 |
| Basicity (pKb) | 8.88 |
| Magnetic susceptibility (χ) | -54.3 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.588 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.94 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 317.8 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -293.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5535.7 kJ/mol |
| Pharmacology | |
| ATC code | N01BX03 |
| Hazards | |
| Main hazards | Harmful if swallowed, inhaled, or absorbed through skin. Causes eye and skin irritation. May cause respiratory tract irritation. Toxic to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H332, H315, H319, H410 |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-1-0 |
| Flash point | Flash point: 160 °C |
| Autoignition temperature | 335 °C |
| Lethal dose or concentration | LD50 oral (rat): 41 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 100 mg/kg |
| NIOSH | SY2950000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Chloro-4,5-Dimethylphenyl N-Methylcarbamate: Not established |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Carbaryl Carbofuran Methomyl Propoxur Aldicarb Fenobucarb Bendiocarb Promecarb |

