Assessing the Role and Future of 2-Chloro-1-Propanol: From Foundation to Frontier
A Look Back at Development
Long before 2-Chloro-1-Propanol found a firm place in the toolbox of chemists, researchers struggled with the lack of stable, versatile intermediates in synthesis. This molecule rose out of early 20th-century labs aiming to balance reactivity and control. Chemists searched for chlorinated alcohols that could serve as bridges between basic building blocks and specialty chemicals. The emergence of 2-Chloro-1-Propanol reflected that spirit of experimentation, showing how a shift in functional groups—adding a chlorine atom to propanol—could open up a new class of accessible intermediates. Over time, the compound became a staple in both academic studies and large-scale industrial settings, building on a history stretching back more than 50 years.
Understanding What It Offers
2-Chloro-1-Propanol stands out for being both a primary alcohol and a halide. This dual character attracts interest because the alcohol enables straightforward oxidation, esterification, and substitution, while the chlorine position serves as a strong leaving group in many organic reactions. Through this flexibility, it anchors processes in both fine chemical and bulk production pipelines. Its moderate boiling point, water miscibility, and known reactivity make it a reliable candidate for reactions under common laboratory conditions. I have seen this compound appear in synthesis plans for everything from pharmaceutical ingredients to modified surfaces in materials science.
Properties That Matter in Real-World Use
With a clear, colorless appearance and a distinct, sharp odor, 2-Chloro-1-Propanol carves out a recognizable identity on the bench. It sits near water in terms of density and shows good solubility in polar solvents, which streamlines clean-up and mixing. Heating it above 100°C can drive decomposition, and it shows steady reactivity in both base- and acid-catalyzed reactions. Its specific gravity, refractive index, and volatility all factor into safety and process control. Thorough knowledge of these attributes lets chemists design safer equipment setups and anticipate bottlenecks, especially as temperatures or solvent choices shift the dynamic.
No-Nonsense Specs and Correct Labeling
Any bottle labeled 2-Chloro-1-Propanol needs to display unambiguous chemical names like 1-chloro-2-propanol or 2-chloropropan-1-ol, reflecting synonyms used across different standards and catalogs. Product purity, water content, and storage conditions should always appear up front, since impurities can trigger unintended side reactions. Proper hazard pictograms and regulatory notes for poisons or irritants belong on the label and safety documentation. Leaving out this information risks exposing researchers and workers to unnecessary harm, as I’ve learned in labs where loose labeling led to confusion.
How It’s Made: Behind the Scenes
Production of 2-Chloro-1-Propanol typically starts with direct chlorination of propylene followed by hydrolysis, or by treating 1,2-propylene oxide with hydrochloric acid. Each route brings balancing acts between yield, selectivity, and waste. Industrial processes lean heavily on continuous flow techniques, where precise control of reagent addition and reaction temperature keeps unwanted byproducts at bay. Batch processes find occasional use in specialty synthesis where scale permits. Choice of synthesis directly impacts cost, purity, and environmental footprint—an ongoing theme in every modern discussion about sustainable chemistry.
Key Reactions and Modifications
Thanks to its design, 2-Chloro-1-Propanol serves as a launching pad for broader chemical architectures. The primary alcohol enables quick conversion to aldehyde or carboxylic acid groups under standard oxidation conditions, supporting access to a wide variety of molecules. Nucleophiles, such as amines or thiols, readily displace the chlorine atom, forming substituted derivatives prized in the creation of pharmaceuticals, crop protection agents, and advanced materials. Researchers commonly transform it into epoxides, glycols, or complex ethers. Personal experience shows that its reactivity saves time compared to less accessible halogenated alcohols; its manageable profile sidesteps the more aggressive handling demanded by higher molecular weight chloro-alcohols.
Names in the Chemical Lexicon
Ask for 2-Chloro-1-Propanol in a European catalog and you might see it listed as 1-chloro-2-propanol; in North America, the name 2-chloropropan-1-ol appears more frequently. The synonyms echo a longstanding tradition in chemical naming conventions. CAS numbers and standardized identifiers help break through language barriers in global trade and research. Its inclusion in reference handbooks ensures that scientists and engineers tap into shared knowledge, limiting errors from nomenclature drift or regional differences.
Standards and Staying Safe
As a volatile liquid with moderate toxicity, 2-Chloro-1-Propanol requires real respect in handling. Good ventilation, chemical fume hoods, gloves, and eye protection all matter; accidental spills or inhalation episodes can bring on eye and respiratory irritation. Strict storage in tight containers away from direct sunlight keeps both people and product safe. Facilities that process large quantities design storage and disposal plans based on current occupational and environmental regulations. Over the years, I’ve seen accidents where careless transfer or inadequate labeling put people at risk. Routine training and clear protocols reduce these chances dramatically.
Bridges Across Industries
2-Chloro-1-Propanol supports a range of industries seeking reactive intermediates for advanced syntheses. In pharmaceuticals, it lays the groundwork for APIs. Agrochemical producers rely on it as a precursor to fungicides and herbicides. Materials science firms adapt it for resins, coatings, and speciality polymers. In laboratories, its straightforward chemistry enables hundreds of research projects each year, showing up in reaction optimization screens, synthesis planning, and method development. Through every application, the compound’s core appeal comes from its reliability and well-understood performance in diverse reaction settings.
Shaping Tomorrow: R&D in Focus
Chemists and engineers continue to push what’s possible with 2-Chloro-1-Propanol. Universities and R&D labs seek greener synthesis routes and lower-impact waste profiles by tweaking raw materials and process conditions. Some teams audit every stage for energy use, others pursue catalysts that cut batch times or boost selectivity. Applications research keeps branching out, with interest rising in biomedical and smart material domains. This commitment to refinement echoes patterns seen with other workhorse chemicals, where small tweaks yield big advances over a decade or more.
Toxicity and Our Responsibility
When discussing any halogenated alcohol, the subject of toxicity comes up as a central concern. Animal data and cell studies help researchers pin down safe handling limits, clarify which metabolic pathways break down 2-Chloro-1-Propanol, and flag persistence in biological systems. Acute exposure can produce mild to moderate irritation, and every update in the toxicology literature informs how regulatory agencies write exposure limits. Keeping up with current research matters, not just to stay in legal compliance but out of respect for the safety of everyone in the supply chain. Comprehensive risk assessments help highlight concerns and shape smarter facility policies to limit unnecessary exposure.
Outlook: Meeting New Demands
The future of 2-Chloro-1-Propanol hinges on evolving regulatory pressure, demand from emerging technologies, and progress in green chemistry. Interest grows in swapping fossil feedstocks for renewable precursors. Pressure mounts to recycle waste and find ways to reuse halogen-containing solvents and reagents. Clever process redesign and new reaction engineering concepts could help keep the compound relevant even as sustainability goals reshape chemical industry priorities. Collaboration between academia and manufacturers, coupled with careful stewardship, can turn 2-Chloro-1-Propanol into not just a tool of today, but a sustainable partner for tomorrow’s core reactions.
What are the main uses of 2-Chloro-1-Propanol?
Why 2-Chloro-1-Propanol Matters in Industry
2-Chloro-1-propanol isn’t exactly a household name, but its influence reaches further than most realize. As someone keeping an eye on chemical processes in industrial settings, I’ve spent time with compounds like this one—working alongside folks in labs, seeing first-hand what it brings to the table in manufacturing and beyond. This compound has a basic-looking structure but packs enough punch to shape a whole range of products.
Building Blocks for Bigger Things
One of the first places where this compound shows up is in the synthesis of other chemicals. It provides an essential link for building block molecules, especially for pharmaceuticals and specialty chemicals. Chemists turn to it when they need a simple, manageable starting point to make more complex products. In one of the plants I toured, they used 2-chloro-1-propanol when creating intermediates for drugs, especially those involving antihistamines and anti-infective agents. Its reactive chloro group combines easily with other chemicals, letting chemists steer reactions more predictably.
Stepping Into the World of Surfactants and Solvents
Down the production chain, surfactant-making often relies on compounds with both polar and nonpolar ends. 2-Chloro-1-propanol fits that bill thanks to its alcohol and chlorine portions, which makes it attractive to formulators looking to bridge gaps between water and oil. I watched engineers use it in the manufacture of custom surfactants that ended up in cleaning products and certain lubricating materials. It’s not the most common surfactant, but it definitely plays a role when companies want to adjust cleaning strength or rheology in specialty blends.
Specialty Polymer Manufacturing
Polymer chemists sometimes seek monomers with specific reactive groups. 2-Chloro-1-propanol delivers a handy point of entry for adding new branches to polymer chains. This opens doors for specialty plastics and resins, especially those needing precise flame retardancy or chemical resistance. The last time I visited a facility focused on custom resins, the technicians showed me how adding this compound helped the finished plastic resist solvents better and hold color longer. Its flexible reactivity lets them tweak the end properties without compromising processing ease.
Pharmaceuticals and Agrochemicals
Medicines and pesticides both demand a range of synthetic steps, and here 2-chloro-1-propanol delivers again. In pharmaceutical labs, people use it to make active pharmaceutical ingredients that combat infection, reduce inflammation, or block allergies. In the field of agrochemicals, researchers rely on it to build up herbicides that must survive weather while breaking down safely after doing their job. Years ago, I observed a pilot production run for an anthelmintic compound (used in veterinary medicine) that started with this building block.
Handling and Safety Concerns
Anyone who has worked with chlorinated solvents knows the potential risk involved. Proper ventilation, personal protective gear, and carefully labeled storage remain crucial. I’ve seen accidents with leaks and spills, and their impacts stick with you. Regulatory oversight continues to play a big part, with organizations like OSHA and the EPA watching over its handling and use. Training new technicians to respect its hazards, from skin irritation to potential byproducts, never feels like wasted time.
Room for Safer Alternatives
Green chemistry holds promise for the future. Researchers look for ways to swap out hazardous substances for more benign ones, or at least recycle and neutralize them more effectively. As industries update their equipment and develop stronger methods for containment and waste treatment, the reliance on traditional chlorinated compounds like this one could slowly drop. Collaboration between chemists, engineers, and environmental professionals pushes these changes forward in real time.
What is the chemical formula and structure of 2-Chloro-1-Propanol?
Breaking Down the Basics
2-Chloro-1-propanol isn’t a household name for most folks, yet its chemistry pops up in a broad range of lab and industrial settings. Stripping it down to its core, the molecule’s formula is C3H7ClO. On paper, it seems simple enough—a three-carbon backbone, a chlorine atom, and a hydroxyl group. That arrangement leads to some specific quirks and uses chemists find valuable, but it also raises a few flags in terms of handling and safety.
Visualizing Its Structure
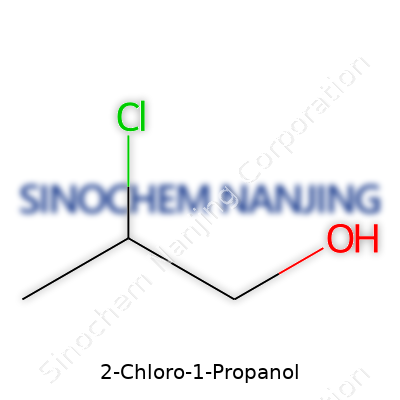
Peeking at its structure, you’ll see a three-carbon chain. The chlorine atom attaches to the second carbon, while the hydroxyl group—an alcohol—hangs off the first. That gives you CH2Cl–CH2–CH2OH as its condensed formula. This isn’t just textbook jargon; the specific placement of atoms shapes how the compound interacts with other molecules and how it’ll react under various conditions.
I can recall days in the lab where even a minor switch in atom location flipped the entire game in organic syntheses. 2-Chloro-1-propanol stands out because its dual functional groups—alcohol and halide—open doors for complicated reactions. It doesn’t act like just another solvent; it’s a building block for quite a few pharmaceuticals and intermediates.
Why Structure Matters Beyond the Textbook
As someone who’s handled both alcohols and chlorinated compounds, mixing the two isn’t just for chemist showmanship. The hydroxyl group primes the molecule for nucleophilic substitution. Meanwhile, the chlorine offers a reactive site for introducing new groups, making it ideal for custom molecule assembly in the lab. Chemists designing new drugs pay attention to compounds with split personalities like this one, since reactivity cuts both ways—sometimes the same property that helps it build complex products brings toxicological headaches or environmental risks.
Real-World Implications and Responsible Handling
Outside textbooks, 2-chloro-1-propanol draws concern over its health and safety profile. That chlorine-holding carbon wants to react, and it doesn’t always care what it reacts with. Breathing in vapors or skin contact could spell trouble, so anyone in a research or production setting needs solid protective gear and ventilation. Mistakes happen when you think “small molecule, small risk.” That attitude never pays off.
I have seen firsthand that underestimating hazardous intermediates only takes one careless afternoon to turn into an accident. Strong protocols, frequent training, and proper labeling go a long way. In places where chemical waste can enter water or soil, even seemingly minor compounds demand sharp attention. Regulations haven’t always kept pace with the chemical industry, so it falls to practitioners to fill those gaps with responsible stewardship and up-to-date knowledge.
Looking Ahead: Facts and Solutions
2-chloro-1-propanol matters in the grand scheme because its chemistry crosses boundaries—basic research, medicine, environmental science. To keep the benefits and avoid pitfalls, industry and academia have to keep talking, sharing research on safer alternatives and greener synthesis. Teaching chemists to think two steps ahead, both in reaction design and waste management, protects more than just profit margins—it protects everyone who depends on a clean environment and a safe workplace.
Understanding molecules like this gets easier when they’re seen as more than numbers on a page. These small details add up. Each time protocols get stronger or labs shift to less hazardous analogs, the whole process becomes safer. The chemical formula (C3H7ClO) and its structure lay the groundwork, but real value comes from thinking through the entire chain—from synthesis to use to disposal. That’s chemistry doing its job right.
What safety precautions should be taken when handling 2-Chloro-1-Propanol?
Understanding the Basics
Working around chemicals isn’t just about following rules, it’s about going home safe every day. 2-Chloro-1-Propanol, a clear liquid used in labs and industry, doesn’t look threatening. But just because it’s not bubbling or changing colors doesn’t mean it won’t cause harm. Skin burns, irritated lungs, and lasting health effects can slip up on you before you know it. Folks who have handled a chemical spill know—the scent, the sting, the regret of not reaching for gloves sooner. That lesson sticks with you.
Using the Right Gear Matters
No lab coat or glove is a fashion statement. Goggles with good splash coverage and chemical-resistant gloves aren’t extra protection; they’re a must. Regular latex gloves soak up many solvents, and 2-Chloro-1-Propanol is one of those chemicals that finds skin fast. Nitrile gloves, chemical aprons, and solid face shields keep splashes out of your eyes and off your skin. I remember someone once thinking their regular glasses were enough, until a single missed splash changed that idea forever. Eyes heal slowly—if at all.
Good Air Means Fewer Headaches
Breathing in fumes never feels right. 2-Chloro-1-Propanol puts out vapors that irritate your nose, throat, and lungs. Good airflow in the workspace blocks those headaches and coughs before they can start. If you work inside, running a fume hood keeps those vapors away from your face. Sometimes a fume hood isn’t around, and in those cases, a well-fitted respirator makes the difference between a clear head and dizziness.
No Shortcuts with Storage
Sealed containers, tightly closed and labeled, stop leaks and mix-ups. Stashing that bottle with water-reactive or oxidizing chemicals leads to trouble. Labeling might sound basic, but it prevents the kind of late-night spill that no one wants to clean up. I once found an unlabeled bottle left in a hot corner of the storeroom, sweating more by the hour. That kind of carelessness can clear a building.
Clean-Up Is Part of the Job
Anytime something spills, the reaction can be panic. A well-stocked spill kit close by—absorbent pads, chemical neutralizers, and plain baking soda—takes the panic out. A quick mop, a change of gloves, and a phone call to hazardous waste pick-up keeps the situation under control. Sure, it slows down the work but you only need to skip cleanup once to realize nothing is faster than stopping an injury before it starts.
Help When Things Go Wrong
Standing showers and eye wash stations are not just for show. In an accident involving 2-Chloro-1-Propanol, every second counts. Wash the chemical off at once. Emergency contacts and clear exit routes give everyone a way out if things go sideways. Every person on site benefits from short, hands-on safety sessions, not some tired slideshow. Confidence with an eyewash or spill kit beats reading directions when someone is hurt.
Learning and Improving
The best labs and factories listen to the people working on the ground. Reporting near misses, updating procedures, and having real talks about safety all matter. Changes often come from the person who sees things at eye-level, not from an office a hundred miles away. Respecting the risks of 2-Chloro-1-Propanol means staying alert, looking out for your coworkers, and knowing that good habits save people, not just paperwork.
How should 2-Chloro-1-Propanol be stored and transported?
Practical Realities of Handling 2-Chloro-1-Propanol
2-Chloro-1-Propanol sits among those chemicals that demand respect. Stored wrong, it gets hazardous quickly. Anyone working around this compound knows how much care keeps the workplace and supply chain protected.
Understanding the Risk
Even a minor spill could create health or fire risk. This liquid often gives off fumes that shouldn't get ignored. Breathing in those vapors or touching skin by accident leaves people at risk for irritation, chemical burns, or more severe outcomes over time. I’ve seen labs and warehouses wrestle with fumes that haunted them for weeks when caps weren’t tight or containers broke. That sort of incident stays with a crew for years.
Containers Matter More Than People Realize
Glass or high-quality plastic containers—think HDPE—work best here. Metal containers don’t hold up when this solvent starts to corrode. People sometimes overlook container compatibility and reach for the nearest drum, but the wrong choice could mean leaks or reactions. Labels fade, hand-written warnings get ignored; heavy-duty, chemical-resistant labeling never gets skipped. It seems like overkill, but I’ve watched people blame faded warnings for accidents more than once.
Keeping It Cool and Dry
Storing this chlorinated alcohol away from heat and sunlight helps lessen the risk. Flames or heat sources lying nearby make everything riskier. Moisture has a way of sneaking into unsealed drums or damaged bottles, kicking off reactions no one needs. It makes sense to lean on dedicated storage cabinets with proper ventilation. Facilities should keep it away from acids, bases, and oxidizing chemicals. One careless shelf location turns into a headache or a real emergency if bottles tip over or break.
Moving It With Common Sense
Transport doesn’t look like an afterthought. Trucks carrying dangerous goods have to signal what’s inside, both for legal reasons and for basic safety. Drivers trained in emergency response give everyone a layer of protection. I’ve seen dispatchers enforce checklists with a level of discipline that saves lives—inventory checks, sealed containers, spill kits loaded. Regulations are there for a reason. Regulations lean on the lessons written in accident reports, and old-timers in the field treat those stories as gospel.
Packing and Documentation: No Shortcuts
Sealed, upright containers avoid the mess. Double containment in some cases does not strike me as excessive—secondary tubs or bins catch what the first barrier misses. Shipping papers keep everyone in the loop, from receiving crews to first responders. Sometimes paperwork feels tedious, but I’ve never regretted chasing a missing MSDS or product code before anything moves.
Prevention Comes Down to Training
It all comes down to people knowing what’s at stake. New staff walk through the process before they go anywhere near this chemical. “Read the label, respect the fume hood, and don’t improvise” got drilled into me early on, and I push the same afterwards. Policies change after every serious scare, but training keeps risks manageable even on busy days.
The Value of Vigilance
Mistakes around chemicals like 2-Chloro-1-Propanol tend not to stay small. Everyone on the team needs to know the hazards, know how to spot small leaks before they turn into panic, and recognize how to label and store for the next shift. Good practices aren’t just rules—they’re the habits that keep a workplace running and people out of harm's way.
What are the potential health hazards and first aid measures for 2-Chloro-1-Propanol exposure?
Why 2-Chloro-1-Propanol Deserves Respect in the Workplace
People come across all kinds of chemicals in labs, factories, and even college classrooms. Some of them only bring minor risks. 2-Chloro-1-Propanol is not in that easy category. This chemical can go straight through the skin or hang in the air as a vapor. It smells faintly like alcohol, but the trouble with it often hides behind that ordinary scent. Workers who use it every day know the headaches and occasional skin tingling. Even quick tasks without gloves, like cleaning up a drop or breaking open a poorly labeled bottle, can turn into a lesson about chemical safety.
Main Health Hazards
The first trouble spot is the skin. Direct contact doesn’t just leave a rash—it can cause burns, severe redness, and blistering. Eyes react badly too. Splashes quickly mean pain, tearing, or swelling. Inhaling the vapors makes the nose burn, causes coughing, and for some people, triggers nausea or dizziness. Even low exposures for a few days might set off headaches or irritation in those with asthma or allergies.
The bigger problem, proven by health studies over the past several decades, lies in the way this chemical moves into the bloodstream. People who have worked with solvents for a long time already know the risk; some report trouble with memory or fatigue after years on the job. 2-Chloro-1-Propanol belongs to a group of compounds linked to possible nervous system effects and, in large doses, liver or kidney harm. Though data on this specific chemical isn’t as complete as with bigger industrial solvents, the risk isn’t worth gambling with.
First Aid Measures People Need to Know
Accidents happen in seconds. The faster someone moves, the less chance they have of short- or long-term problems. If 2-Chloro-1-Propanol touches skin, it’s important to flush the area with lots of water—not just a little splash—for at least 15 minutes, pulling off contaminated clothing right away. For eye exposure, get under a safety shower or eye wash station and hold the eyelids apart while rinsing, again for a full fifteen minutes. Don’t rush, even if the discomfort calms down early.
If someone breathes the vapors, bring them outside or into clean air. It pays to loosen tight clothing and keep the person calm. If the exposure brings trouble breathing, blue lips, or unconsciousness, emergency medical help counts more than anything else. Swallowing the chemical is rare, but if that ever happens, don’t trigger vomiting; call poison control or a doctor at once.
Solutions for Safer Handling
There are no shortcuts for working with chemicals. Proper gloves, splash-proof goggles, and lab coats belong on every checklist. A good exhaust system or fume hood in the workspace does more than meet safety codes—it keeps vapor out of lungs. Clear labeling and careful storage on lower shelves help cut down on spills. Training all staff, even if they only use the product once a month, keeps safety at the front of everyone’s mind. I’ve seen workplaces where the team looks out for each other, pointing out missing face shields or reminding folks to wash up after a spill. That makes a bigger difference, over time, than any one piece of equipment.
Exposure to 2-Chloro-1-Propanol isn’t something you want to brush off. A little preparation goes a long way, and with the right respect and action, most risks shrink fast.
| Names | |
| Preferred IUPAC name | 2-chloropropan-1-ol |
| Other names |
1-Chloro-2-propanol 1-Propanol, 2-chloro- 2-Chloropropanol 2-Chloropropan-1-ol 2-Hydroxypropyl chloride |
| Pronunciation | /tuː-ˈklɔːr.oʊ-wʌn-proʊˌæn.ɒl/ |
| Identifiers | |
| CAS Number | 627-30-5 |
| Beilstein Reference | 1718731 |
| ChEBI | CHEBI:51720 |
| ChEMBL | CHEMBL3185026 |
| ChemSpider | 52934 |
| DrugBank | DB08230 |
| ECHA InfoCard | 06b7d110-a0e1-442b-b0a1-282f25d4e9ec |
| EC Number | 200-959-6 |
| Gmelin Reference | 8546 |
| KEGG | C02704 |
| MeSH | D014336 |
| PubChem CID | 79118 |
| RTECS number | TX9625000 |
| UNII | 3A9ZZ6QF0X |
| UN number | UN3276 |
| Properties | |
| Chemical formula | C3H7ClO |
| Molar mass | 94.52 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | sweet odor |
| Density | 1.099 g/mL at 25 °C(lit.) |
| Solubility in water | soluble |
| log P | 0.1 |
| Vapor pressure | 0.7 mmHg (25°C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 15.44 |
| Magnetic susceptibility (χ) | -65.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.434 |
| Viscosity | 6.261 cP at 25 °C |
| Dipole moment | 2.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –(192.2) kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -627.2 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 74 °C (165 °F; 347 K) |
| Autoignition temperature | 490°C |
| Explosive limits | Lower: 2.7%, Upper: 13.2% |
| Lethal dose or concentration | LD50 oral rat 1100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 823 mg/kg (rat, oral) |
| NIOSH | CN9650000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Chloro-1-Propanol: Not established |
| REL (Recommended) | REL (Recommended): 9 mg/m3 (2 ppm) TWA |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1,3-Dichloro-2-propanol 1-Chloro-2-propanol 2-Bromo-1-propanol 3-Chloro-1-propanol |

