2-Butoxyethanol: More Than Just a Solvent
Historical Development
Chemistry classrooms used to buzz about ethers, but the world didn’t pay much attention until certain molecules stepped into the limelight. 2-Butoxyethanol is one of those chemicals. People first cooked it up back in the early 20th century, thanks to advances in glycol ether chemistry. Industrial demand for new cleaning agents and solvents in the 1920s and 1930s gave this compound room to grow. By the mid-1900s, with painters and custodians relying on it daily, its commercial footprint stretched across continents. This wasn’t brute chemical progress for the sake of making numbers; it was a chemist’s answer to stained clothes, stubborn grease, and the new machines that kept society humming.
Product Overview
2-Butoxyethanol sits at the intersection of affordability and functionality, making it a go-to option for companies rolling out everything from paints to glass cleaners. As a colorless liquid with a slight sweet odor, it often slips under the radar, even if people use it every day at work or at home. Its unique balance of being able to dissolve both oily and water-soluble substances gives it a leg up over older, more hazardous products. This balance has helped it become a workhorse in both industrial and household products, something I’ve seen time and again working alongside manufacturing teams tasked with keeping costs low and performance high.
Physical and Chemical Properties
Pulling out a jug of 2-butoxyethanol on a lab bench, you’d see a liquid that mixes with water, ethanol, and most organic solvents with barely any fuss. Its boiling point settles comfortably around 171 degrees Celsius, much higher than the stuff you pour in your car or on your countertop. This property lets it hang around long enough to actually do its job, whether cleaning, thinning, or facilitating chemical reactions. What always impressed me is the way it bridges polar and nonpolar worlds, dissolving greasy films on windows just as well as helping disperse pigments in latex paints.
Technical Specifications and Labeling
Chemicals don’t always come with plain language, but regulations demand clarity. Bottles and drums of 2-butoxyethanol usually carry hazard statements, warnings about respiratory irritation, and instructions that stress ventilation. Chemists and workers depend on the CAS number 111-76-2 to avoid confusion. No one wants mix-ups with other glycol ethers or solvents, especially since these compounds vary in toxicity. Clear technical data—density, vapor pressure, and flash point—all land on technical sheets that keep users in the know. It’s not just about hitting industry targets. Those labels translate into safer workdays and less guesswork.
Preparation Method
Industry usually leans on an etherification reaction. Ethylene glycol reacts with butanol using acid catalysis, a standard trick from the synthetic playbook. What matters here isn’t just the chemical magic—it’s the immense scale. Factories crank out gallons under high temperature and precise control, striving for purity since impurities bring headaches. Monitoring the process for byproducts that interfere with its use in paints or cleaners helps guarantee that finished products will work without surprises.
Chemical Reactions and Modifications
This molecule does more than dissolve. Chemists have taken its brush and repainted it into new turf. Under acidic or basic conditions, it undergoes hydrolysis, yielding butanol and ethylene glycol. That’s handy for managing waste or breaking it down after use. Its reactivity opens the door to the production of other glycol ethers. On a more practical note, I’ve seen it act as a starting point for surfactants and emulsifiers, making it a launching pad for things that stabilize foggy paints and boost detergency in soaps.
Synonyms and Product Names
Ask a chemist about 2-butoxyethanol, and you’ll get an earful of synonyms: ethylene glycol monobutyl ether, butyl cellosolve, or EGBE. Each name depends on whether you’re in a paint factory, a chemical supply warehouse, or a hospital. What matters isn’t the name—it’s knowing what you’re actually working with. In my own work, requests always came with “cellosolve” attached, thanks to years of commercial branding that stuck long after original patents expired.
Safety and Operational Standards
Long sleeves, gloves, and good ventilation aren't just suggestions—they’re essential when handling this solvent. Studies and regulatory bodies agree: absorb enough 2-butoxyethanol through skin or lungs, and various symptoms start stacking up, from headaches to blood chemistry changes. Workplace exposure limits from groups like OSHA and the European Agency for Safety and Health protect workers on shop floors, and for good reason. Sensible handling practices, routine air monitoring, and keeping MSDS sheets within arm’s reach don’t just check compliance boxes—they help people go home as healthy as they arrived.
Application Area
Ink manufacturers depend on its ability to keep pigments flowing smoothly without clogging print heads. Households use it in window cleaners for streak-free shine. Industrial paint operations cherish its contribution to quick leveling and reduced brush marks. Oil and gas sectors put its solvency to work cleaning up drilling fluids. Even textiles benefit when it acts as a dye carrier. This kind of versatility brings a chemical like 2-butoxyethanol into contact with more people, more often than many would guess. Awareness of where and how it pops up isn’t just a technical curiosity—it's a matter of everyday safety.
Research and Development
Public concern over chemical safety has given researchers fresh incentive to study glycol ethers. Recent years brought deeper dives into air quality exposure, routes of absorption, and alternatives to standard solvents. Developers look at more benign options for cleaning agents, knowing that regulatory winds keep changing. My own experience following lab teams shows a constant push-and-pull between performance and health: can formulations lose 2-butoxyethanol without giving up cleaning power or reliability? Teams in green chemistry think so, testing derivatives and blends that cut down on exposure risks while still kicking grease to the curb.
Toxicity Research
Animal studies highlight risks from both inhalation and skin contact. Red blood cell damage and organ toxicity top the list of concerns, particularly for those handling it in high concentrations. Workers exposed over time can develop symptoms ranging from mild irritation to more severe hematologic effects. Regulatory agencies haven’t listed it as a carcinogen at normal usage levels, but the margin for error feels razor-thin, especially for vulnerable populations. Research continues into long-term, low-level exposure. This isn’t academic guesswork; real families depend on whether factories and cleaning companies choose safer paths.
Future Prospects
Every time new environmental or workplace standards roll in, 2-butoxyethanol stands on shifting ground. Paint makers and cleaning product giants explore ingredients with fewer health concerns, anticipating future restrictions. Solvent chemists look toward green replacements, seeking lower toxicity, higher biodegradability, and fewer regulatory headaches. Still, its winning mix of solvency and price keeps it hanging on. I expect research into exposure reduction to trend upward, with new ventilation technologies, personal protective equipment, and less hazardous alternatives eventually edging it aside in some sectors. The goal remains the same as it did when chemists first blended up this ether: get the job done, but do it better—and safer—than the generation before.
What is 2-Butoxyethanol used for?
Why 2-Butoxyethanol Matters in Daily Life
Walking down the cleaning aisle, most people don’t think twice about the ingredients in glass cleaners or degreasers. There’s a good chance that many of these bottles contain 2-butoxyethanol. This chemical works as a powerful solvent, breaking down grease, dirt, and oily marks. Its ability to cut through grime has kept it popular among manufacturers for decades.
Used in both household and industrial cleaners, 2-butoxyethanol helps products spread evenly and wipe off without leaving streaks. It’s common in window sprays, all-purpose solutions, carpet cleaners, and some paint removers. Next time you wipe down a counter and notice a streak-free shine, there’s a decent chance this compound played a part.
Behind the Suds: Strong Solvent, Real Concerns
People who’ve worked in cleaning, painting, or printing often recognize the distinct odor of 2-butoxyethanol. It’s not just the smell—prolonged exposure, especially in unventilated areas, has led to headaches, nausea, and eye irritation for many. Some research from the U.S. Environmental Protection Agency shows that large doses can cause red blood cell damage and other serious health problems, especially if someone breathes it in or absorbs it through the skin.
I remember my first job stripping paint from old window sills. My gloves barely held out against the cleaner’s strength. A couple hours in, my hands turned dry and itchy. At the time, I thought it was just from working hard. Only later did I connect that experience to the chemical ingredients in the stripping solution. That direct contact with 2-butoxyethanol stays with me whenever I see it on a label.
Regulation and Safer Alternatives
The health concerns around 2-butoxyethanol haven’t gone unnoticed. Several countries and states—including California—set strict limits on how much can be used in cleaning products. Employers in many industries need to provide protective equipment and ensure good airflow when workers use products containing it.
Science keeps searching for safer cleaning agents. New “green” cleaners trade out harsh solvents for plant-based ingredients or safer synthetic alternatives. Some companies have reformulated products but still deliver results customers expect. While not every alternative outperforms 2-butoxyethanol for stubborn grease, the gap keeps shrinking.
Practical Steps for Everyday Users
Most people just want products that work. For folks using cleaners at home, reading labels and airing out rooms makes a big difference. In my house, I switched to less toxic cleaners for daily use, but I keep stronger products for tough jobs. Gloves, fans, and a bit of caution cut down risk for anyone still relying on traditional solvents.
Companies should continue looking for new formulas that protect workers and families. Sharing real-world stories helps—instead of just listing data on a label, clear warnings or online resources let customers weigh the risks for themselves. With awareness, everyone has a better shot at safety without sacrificing clean windows and counters.
Is 2-Butoxyethanol hazardous to health?
Everyday Uses with Hidden Risks
Almost anyone who has cleaned a kitchen, mopped a floor, or spent time scrubbing walls has likely encountered 2-butoxyethanol without realizing it. This solvent shows up in countless household cleaners, window sprays, and industrial products. Thanks to its ability to dissolve grease and dirt, companies add it to many formulas meant to tackle tough messes. Still, regular folks rarely stop to think about what contact with these chemicals means for their health.
What Science Tells Us About Exposure
Over the years, researchers have dug into this solvent and flagged several issues. Inhaling fumes or letting it touch naked skin in large amounts can irritate eyes, nose, and throat. Persistent exposure sometimes leads to headaches, nausea, or feelings of dizziness. Studies in animals suggest that breathing high levels may affect blood cells and possibly trigger more serious long-term harm. Still, evidence in people mostly points toward short-term symptoms, especially with jobs that rely on industrial-grade cleaners.
The U.S. Environmental Protection Agency recognizes 2-butoxyethanol as a chemical that can cause health problems, largely based on what happens in workplaces. It absorbs easily through skin or by breathing in vapor, making everyday exposure in poorly ventilated spaces a decent concern. For most cleaners at home, the stuff appears at low concentrations. That does not mean the risk disappears, especially if products get mixed or the room stays closed off.
Worker Safety at the Forefront
People who make, bottle, or use products with high concentrations of this chemical for hours every day face the brunt of risk. Safety rules recommend gloves, goggles, and plenty of fresh air. Some states have set their own workplace exposure limits. For example, California lists 2-butoxyethanol on their Prop 65 roster. Workers need to know what’s in their cleaning fluids and how long they spend around fumes, or they end up with more than just watery eyes.
Everyday Users: Simple Steps Matter
Home use usually brings lower levels, but habits can still push risk higher. Spraying glass cleaner in a closed bathroom, skipping gloves, or mixing cleaning agents ‘just to see what happens’ can ramp up skin and lung irritation. Reading the small print and paying attention to labels brings surprising value. Running a fan, popping open a window, or swapping gloves once they wear out helps.
New Ingredients and Cleaner Alternatives
Brands now offer sprays and solutions that skip or sharply reduce risky solvents. Plant-based cleaning agents have gained traction, with big-box stores stocking eco-labeled products. While nothing’s perfect, these alternatives cut down on doubt and limit exposure for both workers and families. Real change comes when companies invest in research, and shoppers stay curious about what’s inside each bottle. My own experience swapping harsher sprays for those with simple ingredient lists left my apartment smelling less intense—and the headaches faded, too.
Transparency Moving Forward
Staying safer starts with information. Companies owe clear, honest ingredient lists. Regulators sharpen their focus on workplace air quality and training. Shoppers can ask questions and choose brands that actually listen. No single step solves the problem overnight, but small shifts close that gap between convenience and wellbeing.
How should 2-Butoxyethanol be stored?
Looking Past the Label
2-Butoxyethanol sounds like something you’ll only find in a lab, but it pops up almost everywhere—from cleaners to paints. Even if it doesn’t catch headlines, this liquid deserves respect. It’s not the kind of stuff you just toss onto a shelf beside the family vinegar. Experience shows short cuts in storage bring big problems, especially with chemicals that pack a toxic punch and easily catch fire.
Practical Hazards, Real Solutions
Chemicals with strong smells, like 2-butoxyethanol, often warn us before the label does. In small workshops and big warehouses, breathing in fumes can lead to headaches or worse. The body absorbs it through skin, too, so nobody should treat storage like an afterthought. Data from the CDC lists symptoms that stretch from short-term eye irritation to more serious nervous system hits. Storing this liquid alongside food or in rooms where folks eat lunch raises the risk of dangerous mistakes.
Defeating Evaporation and Spills: Smart Storage Choices
Experience in safety tells me that the best line of defense starts with the container. 2-butoxyethanol doesn’t play well with every plastic, so a solid metal drum or high-density polyethylene container seals away fumes and shrugs off leaks. No need to get fancy—just keep that lid screwed tight between uses. Leave a container partially open and the stuff will soon fill the room with odor and vapor, which can set off fire alarms or worse.
Store the container in a cool, dry place, well ventilated. Temperature swings make vapors more aggressive. Don’t park these next to furnaces, radiators, or in the path of sunlight. Old habits in cramped storage rooms—like wedging bottles too close or stacking cardboard on top—usually end in tears, especially if a slipped hand cracks the seal.
Keep It Simple, Keep It Separate
Mishaps never announce themselves. Tight spaces crammed with everything from corrosives to cleaning fluids become a chemistry experiment nobody asked for. Grouping similar chemicals together makes sense—don’t slot 2-butoxyethanol next to acids or oxidizers. At home or work, storing chemicals in original containers with legible labels keeps guesswork out of the equation, and stops the all-too-common habit of pouring leftovers into mystery bottles.
I’ve seen more than a few places with chemical cabinets hidden under sinks or in dark corners. Problem is, if nobody can see what’s inside, water leaks or an unmarked spill go unnoticed. Good lighting and easy access aren’t just for inspectors—any worker or family member needs to know what’s where. Keep an eyewash station or a sink nearby, just in case someone slips up.
Clear Communication, Ongoing Care
Rules alone won’t save anyone if folks ignore them. Training people to spot warning signs and use gloves or goggles protects the team. Emergency plans, even simple ones taped to the cabinet door, make all the difference in the heat of the moment.
Listening to old-timers who’ve handled these liquids helps more than any manual—a quick story about a minor spill turning major gets the message across. Cleaning products have a place in daily life, but respect means storing them right, asking questions, and sharing what works. That way, 2-butoxyethanol stays useful, not dangerous.
What are the safety precautions when handling 2-Butoxyethanol?
Why Some Chemicals Demand Extra Respect
Growing up around industrial work, I saw a lot of folks get comfortable with strong cleaners and solvents. “Just a bit on your hands won’t hurt,” they’d say. Then, months down the line, a few people started coughing more, complaining about headaches, or noticing rough skin that just never healed right. Hearing these real stories, it becomes clear that certain chemicals, like 2-Butoxyethanol, deserve more caution than some give.
The Hidden Hazards of Everyday Use
2-Butoxyethanol shows up in all kinds of places — degreasers, window cleaners, paints, and countless janitorial supplies. It smells sweet, like an easy day’s work, but that scent masks a danger. Breathing it in or allowing it to soak into skin isn’t a minor deal; the chemical passes right through gloves or bare hands and sneaks into the bloodstream. Symptoms can show up quick: irritation to eyes, nose, and throat, headaches, even nausea. Prolonged exposure hits kidneys, liver, and even blood itself.
Gloves, Goggles, and More: Taking No Chances
Anyone who’s ever gotten chemical burns or felt the sting in their eyes knows the value of good gear. When working with 2-Butoxyethanol, plain latex gloves just don’t cut it. Nitrile or neoprene stands up far better. Eye protection with side shields matters, because even a single accidental splash causes damage in seconds. Lab coats and long sleeves cover skin, turning habits into safety.
Ventilation: Fresh Air Beats Regret
There's no substitute for enough airflow. Solvents like this don’t stay put — their vapors spread fast and linger. Without fans or open windows, fumes build up, especially in confined spots. Simple acts like opening a door or wearing a tight-fitting respirator keep lungs clear. More companies have moved to local exhaust systems above mixing stations, which cut down vapor exposure for everyone nearby.
Clean Habits and Emergency Readiness
It’s tempting to wipe hands with a rag and move on, but chemical-resistant soap and warm water make the real difference after a job. Changing out of work clothes before driving home stops invisible residue from following families into kitchens and bedrooms. Anyone working with 2-Butoxyethanol should know the overflow plan: where the eyewash is, which shower nearby works, and the steps for calling help if someone gets dizzy.
Safer Substitutes and Better Training
It’s not always necessary to use 2-Butoxyethanol for every cleaning or stripping job. Water-based cleaners with milder profiles now fill shelves, offering a way to get the work done without the health risks. Management and crew both deserve regular safety training with honest talks about what can go wrong and how people can protect themselves. Including 2-Butoxyethanol in hazard communication programs ensures everyone understands its risks and reads the Safety Data Sheet each time a new shipment comes in.
Real Protection Means Action, Not Just Rules
What keeps workers and families safe goes further than warning labels and lists of rules. Shared experience, straight talk, and attention to what we’re handling every day do more than posters in break rooms. 2-Butoxyethanol reminds us all that caution at work pays off far beyond the job site — long after the shift ends.
Can 2-Butoxyethanol be mixed with other chemicals?
Understanding What’s Really in the Bottle
Walk into any hardware store, grab a typical cleaner, and chances are, the ingredients include 2-butoxyethanol. You’ll find it in window sprays, degreasers, and paint thinners. It’s there for its powerful grease-cutting ability and because it dissolves both oily and water-soluble grime. Manufacturers often mix it with other chemicals like ethanol, surfactants, and ammonia. Most folks have no clue they’re working with a cocktail of chemicals that interact in ways neither obvious nor always safe.
Health Risks, Often Overlooked
My years washing restaurant floors and cleaning up after painting jobs taught me that nobody’s reading the fine print at the back of the label. Skin burns, stinging eyes, and headaches showed up plenty after long hours with those products. Science backs that up. According to the U.S. National Institute for Occupational Safety and Health, 2-butoxyethanol vapors can irritate lungs and eyes. It even gets absorbed through the skin and can mess with red blood cells and cause kidney problems if used without enough ventilation or gloves.
Some Mixtures Shouldn’t Happen
Let’s be real, not all mixing is equal. 2-butoxyethanol reacts poorly with strong acids or oxidizers. In my experience at a print shop, someone mixed a degreaser containing it with a bleach-based cleaner. The fumes sent everyone coughing and out the door. Official data warns of dangerous byproducts like peroxides that might form if the wrong substances meet. It’s easy to tell people to “just follow the instructions,” but most folks improvise or combine leftovers for convenience. That’s where harm creeps in.
Why Tighter Controls Matter
Product manufacturers add ingredients for results, not always for safety. They rarely tell you how those substances interact under different conditions. Some workplaces run regular training on chemical hazards, but many small businesses don’t. According to the CDC, accidental exposure spikes during home renovations and cleaning seasons. The lack of strong labeling laws and education means ordinary people can stumble into toxic fume territory without warning.
Safer Practices Need More Than Just Labels
Fixing these blind spots takes more than adding another warning sticker. The real shift starts with better information—making ingredient lists accessible, plain, and accurate. Retailers could play a major part by handing out fact sheets at the point of sale or training their staff to answer safety questions. On job sites, managers could hold quick daily toolbox talks about what not to mix. At home, folks could start storing separate chemicals apart and resisting that urge to whip up homemade “super solvants.” Investments in public awareness campaigns, like those for paint lead safety, pay off in fewer poison control calls and visits to the ER.
The Bottom Line
Few people ever think about the risks hiding inside that spray bottle under the kitchen sink. Mixing chemicals like 2-butoxyethanol without knowledge of what’s inside can cause lasting harm that goes beyond a sneeze or a headache. Taking a couple extra minutes to check the label and read up on what not to combine doesn’t just keep you safe; it protects whole households and workplaces, too.
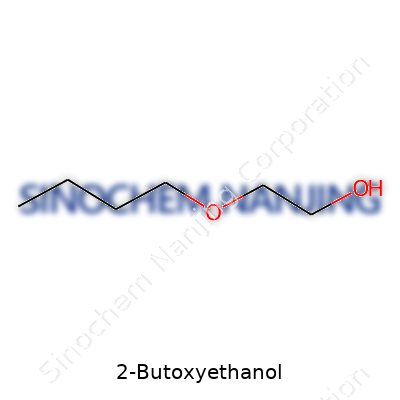
| Names | |
| Preferred IUPAC name | 2-butoxyethan-1-ol |
| Other names |
Ethylene glycol monobutyl ether Butyl cellosolve Butoxyethanol Glycol ether EB EGBE 2-Butoxy-1-ethanol |
| Pronunciation | /tuː-bjuːˌtɒk.siˈɛθ.ə.nɒl/ |
| Identifiers | |
| CAS Number | 111-76-2 |
| Beilstein Reference | 0274802 |
| ChEBI | CHEBI:35807 |
| ChEMBL | CHEMBL1357 |
| ChemSpider | 5170 |
| DrugBank | DB00138 |
| ECHA InfoCard | 200-598-5 |
| EC Number | 203-905-0 |
| Gmelin Reference | 84134 |
| KEGG | C06506 |
| MeSH | D001994 |
| PubChem CID | 8133 |
| RTECS number | KJ8575000 |
| UNII | 6T4CY0VG2K |
| UN number | UN2816 |
| Properties | |
| Chemical formula | C6H14O2 |
| Molar mass | 118.17 g/mol |
| Appearance | Colorless liquid with a mild, sweet, ether-like odor |
| Odor | Sweet odor |
| Density | Butoxyethanol has a density of 0.902 g/cm³ at 20 °C |
| Solubility in water | miscible |
| log P | 0.83 |
| Vapor pressure | 0.8 mmHg (20°C) |
| Acidity (pKa) | 14.8 |
| Basicity (pKb) | pKb ≈ 5.1 |
| Magnetic susceptibility (χ) | -6.59×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.419 |
| Viscosity | 2.9 mPa·s (at 20 °C) |
| Dipole moment | 3.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -460.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3199 kJ/mol |
| Pharmacology | |
| ATC code | D7AX02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H319, H332 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-Butoxyethanol NFPA 704: 2-2-0 |
| Flash point | 60°C (140°F) |
| Autoignition temperature | 245 °C (473 °F; 518 K) |
| Explosive limits | 1.1% - 10.6% |
| Lethal dose or concentration | LD50 oral rat 470 mg/kg |
| LD50 (median dose) | LD50 (median dose): 470 mg/kg (oral, rat) |
| NIOSH | K792 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 700 ppm |
| Related compounds | |
| Related compounds |
Ethylene glycol Diethylene glycol 2-Butoxyacetic acid Butyl cellosolve acetate Propylene glycol monobutyl ether |

