2-Butenoic Acid: A Commentary on Its Progress, Potential, and Pitfalls
Historical Pathways and the Essential Face of 2-Butenoic Acid
Walking through the evolution of 2-butenoic acid—sometimes better known as crotonic acid—brings up stories from classic organic chemistry laboratories and the older days of chemical discovery. Crotonic acid carried promise as soon as researchers isolated it from croton oil. Chemists have worked with it for generations; it’s interesting to see how a small, simple unsaturated acid carved out surprising uses and challenges. Early work on crotonic acid expanded the understanding of double bond chemistry, serving as a real-life model for conjugated systems. Over the decades, its clear presence in research and industry has tracked shifting priorities in science, whether that meant cleaner manufacturing, greener methods, or new bio-based applications.
Chemistry in Daily Life: Characteristics and Technical Demands
People who’ve handled 2-butenoic acid can describe its sharp smell—a pungent, almost vinegary vapor that lingers. It melts at relatively low temperatures, revealing its crystal structure, and dissolves in water with moderate ease. With a formula of C4H6O2, it acts both as a carboxylic acid and, through its double bond, an entry point to more elaborate molecules. The molecule’s reactivity isn’t just a theory for graduate exams; it plays out in the lab when crotonic acid takes on additions or polymerizes. Its labels often refer to “cis” and “trans” forms, but the trans variant sees most practical use. Every bottle or drum shares that recognizable hazard labeling—flasks marked for both irritant and environmental concern.
Preparation on an Industrial Scale: From Simplicity to Utility
For those watching the manufacturing side, sorbic acid and aldehyde chemistry often bring crotonic acid into play. Traditional methods use the oxidation of crotonaldehyde or dehydration steps with various catalysts. Refinement matters, and older methods led to inconsistent yields, but improvements have come from careful process control and better chemical engineering. Reaction conditions—temperatures, solvent choices, even the order of ingredient mixing—decide how clean and pure the final acid turns out. When suppliers claim “high purity,” the chemists behind them know the effort poured into separating byproducts, scrubbing impurities, and testing batches for stability.
Reactivity and Technical Transformations
If you’ve sat through advanced organic synthesis lectures or spent nights mapping out new compound syntheses on paper, the double bond in 2-butenoic acid grabs attention for its openness to modification. The molecule responds to additions of hydrogen, halogenation, and Diels-Alder reactions with striking versatility. In practice, these reactivities let chemists design everything from pharmaceutical intermediates to custom polymers. Dehydration, esterification, and salt formation round out a range of chemical moves, each with its own story of practical hurdles and optimizations. It’s hard not to appreciate the acid’s adaptability in creating block copolymers or chiral auxiliaries for laboratories exploring new reaction pathways.
Synonyms, Nomenclature, Hidden in Plain Sight
The list of names for 2-butenoic acid can feel like a primer in chemical language. Crotonic acid pops up most often in industrial and lab settings, with the IUPAC system pointing to (E)-2-butenoic acid. Trade names sometimes sneak into supplier listings, particularly where patents or proprietary polymer blends matter. Reading labels and safety forms requires a kind of fluency in synonyms to avoid confusion; no one wants to discover mid-experiment that they’ve mistaken a cis isomer for the needed trans. These naming quirks link back to traditions in chemistry as much as they reflect the acid's applications.
Safety, Handling, and Standardization: Quiet but Critical Details
The reality of crotonic acid’s hazards becomes obvious on the bench. Direct exposure can result in respiratory irritation; skin contact leave burns or rashes. Safe storage means rigorous respect for chemical compatibility and vented containers. Those who work in warehouse or production settings lean on up-to-date standards for labeling and material transfer, following local and global regulations like REACH or OSHA. There’s no shortcut around clear training—both for safety eyewash use and for proper air extraction in lab hoods. Small mistakes, even a loose cap, can spell headaches—literally and legally. Consistent application of safety protocols keeps both the workforce and community protected.
Applications That Touch Multiple Industries
People outside the chemical industry might never see crotonic acid in daily life, but its fingerprints show up in everything from adhesives to specialty polymers. Paints and resins, for example, rely on 2-butenoic acid’s ability to deliver functional groups for polymer backbones, improving adhesion and flexibility. In pharmaceuticals, researchers employ it as a building block for synthesizing complex molecules—medicines and intermediates both. The flavor and fragrance trade taps into crotonate esters for their distinct notes, sometimes using them to tweak aroma profiles in foods and cosmetics. Even agriculture finds utility in certain derivatives, tweaking plant growth or pest resistance. The spread of uses keeps creating fresh demand for both technical insight and responsible supply.
Research, Development, and the Race to Improve
From my own experience, projects involving crotonic acid often start with promise and sometimes end tangled in unexpected complications. Researchers keep searching for more sustainable routes to make and use this acid. Recent interest hovers around biocatalysis and green chemistry approaches, looking to cut down the environmental footprint and sidestep traditional petrochemical methods. Focused teams run kinetic studies, tweak catalysts, try to optimize reaction media, and test product purity at every turn. Industrial partners often push for better scalability while labs chase novel transformations. Every incremental improvement opens up new application territories—the chase for biomedical-grade purity, more robust film coatings, or smarter ways to graft the acid onto existing polymers.
Toxicity Conversations and Environmental Concerns
Toxicity isn’t an afterthought. Chronic exposure can sensitize the skin or respiratory tract, so studies drill down into dose-response relationships. Researchers review impacts on aquatic life, persistence in soil, and possible byproduct accumulation in supply chains. Environmental audits often flag improper disposal or accidental spillage, so modern protocols favor secondary containment, regulated emissions, and solvent recycling. Ongoing toxicology studies guide both legal frameworks and company best practices. It’s not just a lab lesson, but an ethical obligation to handle crotonic acid with foresight, aiming for zero incident workplaces and minimal byproduct waste.
Future Prospects and the Road Forward
Looking into the future, there’s no question that crotonic acid stands poised to gain ground from new markets that value specialty chemicals and sustainable sourcing. Advances in bio-manufacturing could make production both more affordable and less polluting. As researchers design more exotic polymers or therapeutic intermediates, demand will rise for chemical feedstocks combining high performance with certified safety. Regulation continues tightening, so only those producers who keep pushing clean methods will stay relevant. At the same time, curiosity-driven science will keep experimenting with crotonic acid’s reactivity, stretching out its range—from self-healing materials to smarter prodrugs. That’s the kind of ongoing development that keeps organic chemistry lively and essential in the real world.
What is 2-Butenoic Acid used for?
Where 2-Butenoic Acid Shows Up
Every time I walk through a lab or read about new materials, certain names pop up—2-Butenoic acid is one of those. Its molecular setup makes it a handy building block in chemical production. Some people know it by its more common name, crotonic acid. Its claim to fame really comes from its double bond; chemists are all about those reactive spots because they open the door for so many chemical reactions.
Shaping the World of Plastics
By far, the biggest use for 2-Butenoic acid shows up in plastics. Nobody picks up an acrylic paint or a tough, clear plastic bottle and thinks about what’s inside at the atomic level. Acrylic polymers often rely on monomers made using crotonic acid. These polymers end up in paints, coatings, adhesives, and lots of household plastics. The acid’s ability to join up with other molecules helps make the plastic harder or stickier, depending what the manufacturer needs.
Adhesives and Paints—Quiet Helpers
The world of glues and paints leans on crotonic acid more than most people ever notice. Adding this acid to copolymers changes the way adhesives bond, making the glue stronger or more flexible. When I help kids with craft projects, I fish out glues that depend on polymers built from tiny starting blocks like crotonic acid. The same acid lets paints spread thinly and stick to surfaces without cracking over time. That often spells fewer touch-ups on jobs around the house.
Pharmaceuticals and Specialty Chemicals
Chemists turn to crotonic acid as a starting point for more complex molecules. Anyone working in drug development knows how a small tweak in a molecule can lead to something that saves lives. This acid helps make intermediates for active drug ingredients, sometimes as a direct building block, other times to introduce a double bond in a compound. Some simple antiviral and anesthetic agents rely on related derivatives.
Food Industry and Preservation
Natural and synthetic sources of crotonic acid pop up during food processing. In small amounts, it shows up during the heating or fermenting of certain foods like cheese. Scientists use this fact to monitor spoilage or freshness; crotonic acid can serve as a marker compound for changes in stored food. Strict rules keep levels safe because the pure acid in large amounts causes irritation.
Safety Concerns and Environmental Care
No one should handle crotonic acid carelessly. Skin contact burns, and inhaling too much can cause lung irritation—lab classes always stress using gloves and keeping good ventilation. Environmental scientists point out that crotonic acid breaks down quickly in soil and water, reducing risk over time. Still, every chemical that ends up in the water supply needs careful tracking. Manufacturers keep improving filtration and cleanup as the science develops further.
Looking for Alternatives
Some companies explore greener building blocks for plastics and adhesives. Plant-based acids, like lactic acid, serve as options in certain contexts. Crotonic acid’s unique double bond still makes it valuable for products needing specific flexibility or hardness. Any switch to new chemicals in manufacturing brings cost and machinery changes, plus careful study so performance stays up to par. Advances in biotechnology promise more sustainable ways to keep the benefits, with less impact on health and nature.
What is the chemical structure of 2-Butenoic Acid?
What Makes 2-Butenoic Acid Distinct?
2-Butenoic acid stands out as a simple unsaturated carboxylic acid. Its structure, CH3CH=CHCOOH, draws a lot of attention from anyone interested in organic chemistry, not just for its straightforward composition but also for the way its double bond changes everything about how it behaves. That double bond links the second and third carbon atoms, leaving the carboxylic group hanging off the chain’s end, ready to take part in chemical reactions.
Why Structure Shapes Behavior
Organic acid chemistry taught me early on that where you place a double bond in a molecule, you change its reactivity and even the way it looks and smells. In 2-butenoic acid, this unsaturation right after a methyl group creates two geometric forms: the cis (Z) and trans (E) isomers. The way these atoms are arranged affects its melting point, boiling point, and solubility. In the lab, these differences pop up with smell and how the compound reacts with things like bromine water—a crucial test for double bonds.
Functional Groups at Work
The carboxylic group (COOH) on one end delivers the acidity. If you’ve ever worked with acids, you know how a group like that loves to donate its hydrogen ion in water, making the solution sour and reactive. The double bond adds another active site, showing that a small molecule can do a lot. This is the cornerstone in building polymers or running addition reactions, things every chemist enjoys testing out.
Relevance in Real World Chemistry
You’ll find 2-butenoic acid mentioned often as crotonic acid when it takes the trans form. Crotonic acid crops up in the creation of polymers, plasticizers, and even flavor agents. Its ability to polymerize is huge for manufacturing and synthetic materials science. Having tested reactions like these, I’ve seen how easily that double bond triggers chain reactions, forming long strings essential for practical plastics and resins. The physical properties—like solubility in water and ethanol—make it easy to blend with other chemicals, which matters in industrial applications.
Addressing Hazards and Safety
Working with unsaturated acids asked for some care. Exposure causes irritation, and inhalation of vapors isn’t pleasant. Storage calls for sealed containers and well-ventilated labs. Clear labeling and lab protocols help everyone avoid accidents and confusion. Safety data sheets call out these risks, which serve as constant reminders of the responsibility chemists hold.
Moving Forward with Sustainable Chemical Use
Sourcing 2-butenoic acid sustainably presents challenges, mostly due to reliance on petrochemicals. Green chemistry seeks alternatives, hoping to derive such molecules from plant-based sources. I’ve looked into fermentation methods that could yield similar acids with lower environmental impacts. More research and pressure from consumers can push companies toward these paths, reducing the chemical industry’s footprint and still delivering useful products.
Summary
2-Butenoic acid isn’t flashy, yet its structure shapes how it interacts with other compounds and determines its practical uses. Anyone who’s mixed chemicals or reviewed molecular models sees why these details matter. Getting a grip on its structure means unlocking a wider appreciation for chemistry’s ability to solve problems, create new materials, and keep safety at the forefront.
Is 2-Butenoic Acid safe to handle?
What is 2-Butenoic Acid?
2-Butenoic acid shows up in research labs and some niche manufacturing settings. More commonly, you might hear it called crotonic acid. The chemical formula is C4H6O2. It doesn’t show up in medicines or food additives you’d find on shelves, but chemists rely on it as a building block for certain polymers and specialty plastics.
Safety Concerns in the Workplace
I’ve worked in labs where personal protective equipment becomes second nature, and it’s for good reason. 2-Butenoic acid is a colorless solid with a distinct, sharp odor. Direct contact stings. The two main risks are skin and eye irritation. Safety data from established sources like the European Chemicals Agency and the U.S. National Institutes of Health agree on this. Handling the solid or the vapor without gloves or goggles means risking chemical burns and painful eye irritation.
It gets into the air easily during heating or aggressive mixing. This irritates airways, leading to coughing and possible shortness of breath if you’re not wearing a mask or using proper ventilation. On spilled liquid or dust, skin absorbs low levels, which can still lead to rashes or chemical dermatitis over time. Getting the substance in your eyes can mean burning, redness, and long-term damage if you don’t flush the area right away.
Environmental Impact and Challenges
Any time a lab chemical spills, people think about safety for both themselves and the area around them. 2-Butenoic acid breaks down fairly quickly when exposed to sunlight and air, but runoff can add acidity to water bodies and hurt local wildlife. Research papers cite its quick natural breakdown, but they also note the need to avoid pouring leftover chemicals into the drain. Good practice means gathering up waste and sending it for hazardous materials processing instead of letting it mix into the water supply.
Why Precautions Make All the Difference
PPE matters most with 2-butenoic acid. Thick nitrile or neoprene gloves, quality safety goggles, and a lab coat keep your skin barrier protected. I remember during my grad school days, a careless glove change by a lab partner ended with a trip to the campus medical center because of a minor but nasty burn. The fix is easy: change gloves after any spill and avoid touching your face during handling. Even outside the lab, anyone working in production lines with this chemical must follow the same guidelines. Labeling and storing this acid away from strong bases and oxidizing agents helps prevent explosive reactions. Since the vapors can bother your lungs, a properly vented fume hood or extractor fan belongs in any workspace using it.
Pushing for Safe Practices
Training makes the biggest difference. I’ve seen people dismiss a sharp smell or minor spill as nothing. Truth is, repeated exposure—just like sunburns—adds up. Employers should walk new staff through hands-on safety drills, post clear instructions around the work area, and run mock spill responses to build habits. Waste should always go in marked containers for hazardous collection, never in the trash. Safety gear shouldn’t collect dust in the corner. Checking for holes in gloves and cleaning eyewash stations make a real impact over time. Finally, anyone with underlying respiratory issues like asthma should alert their supervisors and the safety officer for extra protection.
Knowledge and Preparedness Go Hand-In-Hand
2-Butenoic acid stays safely in the lab as long as attention remains steady. Forgetting to follow basic protocols leads to preventable accidents. There’s no room for shortcuts when it comes to chemical safety. Ongoing education and good habits create environments where innovation thrives without health risks lurking in the background.
What are the storage conditions for 2-Butenoic Acid?
Why Caution Matters With 2-Butenoic Acid
People handle chemicals every day, often taking for granted how quickly things can go sideways when storage slips up. 2-Butenoic acid, better known as crotonic acid, stands out as one of those chemicals that reward respect and careful storage. Ask anyone in a university lab or a chemical storeroom; a leaky bottle or a whiff of fumes reminds you right away this stuff isn’t vinegar.
What Keeps 2-Butenoic Acid Stable?
2-Butenoic acid breaks down in heat, light, or around the wrong materials. Most guides point out that it needs a tight, well-sealed glass or high-density polyethylene container, not metal or low-quality plastic. Acids corrode certain plastics and metals, and crotonic's sharp odor tends to escape loose lids fast.
Temperature plays the biggest part. Crotonic acid likes consistency, best found at room temperature—20 to 25°C works well. Cold slows down reactions but pushing it into a fridge isn’t always smart unless the lab’s got real control over humidity and airflow. I’ve seen containers sweat and form crusts around the seal if left too cold, which can ruin the chemical and create cleanup headaches.
Environmental Factors and Safety
Some think any old shelf will do, but you can’t store crotonic acid next to oxidizers, alkalis, or strong bases. If it spills, it reacts, and you risk a lot more than a mess. Strong odors and fumes always mean you want decent ventilation—nobody wants to open a storeroom and get hit with eye-watering vapor before their shift’s even started.
Moisture causes as much trouble as heat or light. Keep dryness in mind: desiccators or cabinets with low humidity work best, especially in damp areas or rainy seasons. I’ve had bottles get sticky on humid days, which signals that the acid is drawing in water or even reacting, making every opening riskier.
Labeling, Segregation, and Routine Checks
A sharp label matters more than most realize. I worked with a team that nearly poured crotonic acid into a project meant for a benign buffer—just because the bottle sat among a mess of unmarked or poorly marked containers. Labels with full chemical names, hazard symbols, and open/expiry dates take seconds and prevent endless grief.
Segregating strong acids from bases or oxidizers isn’t just protocol—shelf organization saves lives. Many storage incidents boil down to a rushed moment or lack of awareness about what sits beside what. It stings to realize a stack of flammables right near acids just because nobody bothered to reshuffle after a delivery.
Regular checks might sound tedious, but expired or degraded acid gets risky fast. Vinegar-like smell, yellow tinge, or separation inside the container means it's time to ditch the batch and clean surfaces. Personal experience taught me to keep a checklist on paper or digitally, logging what chemicals are checked and when.
How Practice and Design Beat Theory
Solid storage means more than just following a book. It calls for real-world habits and designs—clear sections, solid shelving, and a “no shortcuts” attitude. Crotonic acid might not grab headlines, but a cracked seal or wrong shelf turns it into a reminder of why storage rules exist in the first place.
What is the purity specification of your 2-Butenoic Acid product?
Knowing the Purity Level Matters
Working in chemical manufacturing has taught me how often scientists and end users ask direct questions about purity. Few things affect formula outcomes more than impurity levels in an ingredient. 2-Butenoic Acid, also known as crotonic acid, finds its way into plastics, cosmetic intermediates, and pharmaceutical building blocks. Each industry stakes its reputation on product consistency and compliance.
Purity Specification Basics
Manufacturers typically deliver 2-Butenoic Acid with a purity of 99% or better. Analytical tests like gas chromatography or titration eliminate guesswork. You want to spot the numbers: a main assay of 99.5% or above, moisture below 0.5%, and residue on ignition under 0.1%. Each part per million of unknown can make a difference, especially if you work on high-sensitivity pharmaceutical reactions or food contact polymers.
International markets like Europe and North America expect batch certificates confirming purity. One slip on that sheet, and regulatory submissions fall apart, not just for drug makers but for anyone exporting finished goods. I once joined a team running a small batch extruder, and a three-day delay hit us because a drum of crotonic acid showed slightly high water content. Minor numbers stall big machines.
Impurities and Their Impact
No one wants acetaldehyde or butyric acid spoilage hiding in their material. For specialty applications, that matters even more. Chromatographic fingerprinting lets users check for shadowy byproducts and confirm the absence of excessive unknowns. Crude forms, with visible color or odd smells, usually come cheaper; premium supply lines work hard to exclude trace contamination that could become a persistent odor or discoloration.
Handling and Testing Routines
Warehouses rarely get a pass on quality checks. Opening a container and noticing off-scent often signals something missed in pre-shipment quality control. If someone maintains a lab, they run independent checks—a sensible move since small mistakes travel far across a supply chain. Using titration or GC, confirmation of expected composition stops trouble before it reaches the reactor.
Looking at trends in the industry, good suppliers share Certificates of Analysis and batch traceability details without a fight. In-house testing always backs up confidence, but supplier transparency creates trust fast.
The Value of Consistency and Traceability
Every conversation with R&D colleagues circles back to one thing: predictability. Unpredictable ingredient performance can sink experiments, wreck months of planning, and burn through budgets. Consistent purity means one less variable in a complex puzzle. I remember a chemist who tracked failed reactions to a tiny butyl impurity—barely a blip on the supplier sheet and yet the root cause.
Good practice also means documenting each lot and storing records longer than usual. Questions on headspace analysis, volatility data, or contamination now turn up long after an initial order. Having fast access to purity records beats the headaches of guesswork and paperwork scramble.
Next Steps for Responsible Sourcing
Buyers who ask for third-party lab verification increase their chances of catching outliers early. Partnering with suppliers open about analytical methods and willing to provide samples can save resources and keep downstream quality issues at bay. Error-free 2-Butenoic Acid extends well past a single shipment—consistent checks, open communication, and documented results keep recipes and reputations continually strong.
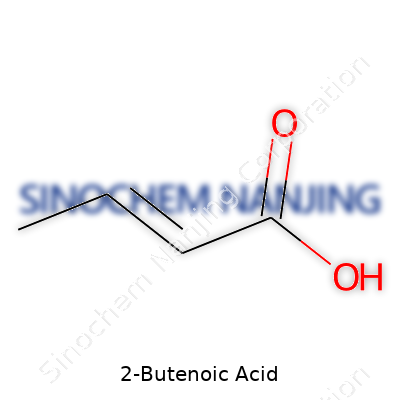
| Names | |
| Preferred IUPAC name | (E)-But-2-enoic acid |
| Other names |
Crotonic acid trans-2-Butenoic acid β-Methylacrylic acid |
| Pronunciation | /buːˈtiː.noʊ.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 107-93-7 |
| Beilstein Reference | 1209249 |
| ChEBI | CHEBI:35618 |
| ChEMBL | CHEMBL504 |
| ChemSpider | 14904 |
| DrugBank | DB04248 |
| ECHA InfoCard | The ECHA InfoCard of product '2-Butenoic Acid' is: **03a5677b-615b-49c7-a1d8-90cda1005f7d** |
| EC Number | EC Number: 203-477-8 |
| Gmelin Reference | 63052 |
| KEGG | C01010 |
| MeSH | D001561 |
| PubChem CID | 7909 |
| RTECS number | GZ0450000 |
| UNII | 5BVG0JLZ5N |
| UN number | UN2529 |
| Properties | |
| Chemical formula | C4H6O2 |
| Molar mass | 86.09 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Unpleasant, rancid |
| Density | 1.018 g/cm3 |
| Solubility in water | soluble |
| log P | 0.38 |
| Vapor pressure | 0.1 mmHg (20°C) |
| Acidity (pKa) | 4.41 |
| Basicity (pKb) | 10.25 |
| Magnetic susceptibility (χ) | -47.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.431 |
| Viscosity | 1.24 mPa·s (25 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -351.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -610.3 kJ/mol |
| Pharmacology | |
| ATC code | A16AX13 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P330, P337+P313, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 52 °C |
| Autoignition temperature | 440°C |
| Explosive limits | Explosive limits: 2.6–10.3% |
| Lethal dose or concentration | LD50 oral rat 500 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 2-Butenoic Acid: 177 mg/kg (rat, oral) |
| NIOSH | RW5086500 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | 200-621-7 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Crotonaldehyde Crotyl alcohol Crotonate ester Butyric acid Methacrylic acid |

