2-Butenal: An Underrated Workhorse in Modern Chemistry
Historical Development
Long before 2-Butenal found its place on benches and in journals, chemists stumbled upon it as a fleeting player in the dance of carbonyl chemistry. Aldehydes have shaped both small-scale lab feats and industrial design since the 19th century, and few structures pack as much reactivity per atom as this little molecule. Chemically known as crotonaldehyde, 2-Butenal grew out of early explorations in fermentation and organic synthesis. Early advances in distillation and purification allowed researchers to isolate this pungent, yellowish liquid and start probing its behavior. Decades passed, labs grew more advanced, but the curiosity about unsaturated aldehydes never faded. Today, 2-Butenal serves as both a relic from organic chemistry's colorful past and a relevant building block for present-day innovation.
Product Overview
People ask what makes 2-Butenal stand out among all the other aldehydes and organic intermediates. Its direct structure speaks for itself—an alpha, beta-unsaturated aldehyde, four carbons in length with a carbonyl at one end, a double bond just nearby. That conjugation between the carbonyl and the double bond isn't just a piece of trivia; it stirs up the reactivity, giving the molecule its edge both in aroma and chemical usefulness. In the chemical industry, crotonaldehyde shows up in batches ranging from a few grams in the lab to multi-ton synthesis in plants. It draws attention for both its boiling point, sitting just under 105°C, and its broad scope, touching everything from solvents to pharmaceuticals to food flavorings. In my own experience, opening a bottle releases an unmistakable, biting odor—hard to forget once it hits your nose.
Physical & Chemical Properties
2-Butenal pours as a clear to pale-yellow flammable liquid, packing a sharp, acrid odor that hints at its reactivity. Its molecular formula, C4H6O, doesn't tell the whole story. This molecule boils at about 104°C, melting near -68°C, and dissolves in alcohols, ethers, and water, although only to a limited extent. Conjugation between the alkene and aldehyde gives 2-Butenal its reactive streak. Exposed to light or air, it can polymerize or oxidize. If left open in lab air, it can yellow or even degrade. Reactivity comes quickly as it forms Schiff bases with amines and clicks readily into Michael additions. The volatility and low flash point call for caution, since vapors can travel and ignite from distant sparks.
Technical Specifications & Labeling
Labels matter, given that 2-Butenal deserves respect for both its value and hazard profile. A bottle in the storeroom likely carries multiple warning symbols—flammable, irritant, acutely toxic. Safety data sheets spell out the flash point (around 15°C), vapor pressure, and limit values for workplace environments. Regulations around the world put tight controls on how much can linger in air, not only for worker health but for fire prevention. Scientists and production managers have learned to keep storage cool, containers sealed, and handling within a fume hood, all because of the compound’s low threshold for irritation and potential for harm.
Preparation Method
Ask synthetic chemists, and many will point to aldol condensation for preparing 2-Butenal. The most widely-used path starts with crotonization of acetaldehyde, using base or acid catalysis. When two molecules of acetaldehyde meet, one forms an enolate, attaches to the carbonyl carbon of another, and after a quick dehydration, the double bond slides into place. On the industrial side, this reaction runs in continuous reactors at carefully controlled temperatures and pressures to maximize yield while minimizing tar and side reactions. From a home chemist’s perspective, even a small-batch preparation needs care to avoid splashing or vapor leaks, as the pungency and volatility don't leave much room for error. Purification by distillation ensures a cleaner product, but safety shields and well-ventilated setups become non-negotiable.
Chemical Reactions & Modifications
What makes 2-Butenal especially interesting is not just its reactive aldehyde but the conjugated double bond. Chemists use it as a starting point for Michael addition reactions, attaching nucleophiles on the beta-carbon and opening pathways to more complex molecules. Hydrogenation across the double bond yields butyraldehyde, another critical platform molecule. Oxidation bumps it up to crotonic acid, revealing new uses in polymers and resins. In the world of organic synthesis, crotonaldehyde can chain together with amino groups to form imines, link to alcohols in acetal formation, or serve as a dienophile in Diels-Alder reactions. The list of modifications stretches long, and each step builds value and function into end products ranging from pharmaceuticals to agricultural chemicals.
Synonyms & Product Names
2-Butenal parade aliases in chemical catalogs and research papers—crotonaldehyde being the most familiar, but others like alpha, beta-unsaturated butyraldehyde or trans-2-butenal sometimes crop up. Reading across product documentation, those in the know recognize the structure, no matter the label. These alternate names don't just fill lines on a safety sheet; they often reflect the context, whether food chemistry, analytical labs, or polymer science. Recognizing these synonyms keeps procurement and compliance teams from costly mix-ups.
Safety & Operational Standards
People who have handled crotonaldehyde usually remember their first spill—nose tingling, eyes watering, skin itching. Even exposure at fraction-of-a-percent concentrations can irritate mucous membranes. Regulations from agencies like OSHA in the US and the EU's REACH list show strong agreement: keep exposure low, handle with engineering controls, and don’t store near strong bases or oxidizers. Fume hoods, gloves, goggles, and protective lab coats become daily gear, not optional extras. Emergency showers sit nearby because of the compound’s ability to burn skin. Proper air monitoring and ventilation go beyond best practice; they form the backbone of worker protection where crotonaldehyde flows through production lines or research setups.
Application Area
Industry never wastes a chance to transform crotonaldehyde into profit. Companies pull it into the synthesis of sorbic acid (used as a preservative), stretch it into pharmaceutical intermediates, or react it to build up polymers, pesticides, and fragrances. Some flavor chemists work it into nutty or smoky notes, though food-grade applications must tiptoe past its hazardous properties. Agriculture and coatings companies add demand, using 2-Butenal as a bridge to resins, plasticizers, and surfactants. In my discussions with industrial colleagues, manufacturing teams often revisit this molecule when seeking alternatives to more complex, pricier aldehydes. The ability to modify its backbone with a wide range of nucleophiles gives operators flexibility in what final products emerge.
Research & Development
In the academic world, research trends follow applications and societal needs, so studies on 2-Butenal move with the times. Environmental chemists have tracked its role as a volatile organic compound (VOC), raising flags about emission controls and atmospheric chemistry. Biochemists dive into the interactions between crotonaldehyde and living tissues, noting its inclusion in cigarette smoke and the resulting adducts with DNA. Analytical chemists probe detection limits in air and water using gas chromatography and spectrophotometry. Each year, researchers tweak catalytic conditions, search out greener solvents, or test out microwave-assisted reactions in hopes of pushing preparation and modification into safer, more sustainable territory.
Toxicity Research
No discussion of 2-Butenal feels honest without a hard look at its toxicity. Studies point out that exposure via inhalation or skin contact causes irritation, and there’s enough data to classify it as harmful in concentrated forms. Laboratory animal studies, coupled with occupational health investigations, reveal that crotonaldehyde causes respiratory discomfort, skin burns, and potential long-term organ effects. Evidence links chronic exposure with DNA adducts that pose cancer risks, especially in smokers and workers without proper protection. Regulatory agencies set workplace exposure limits for a reason. Even experienced chemists keep bottles under strict inventory and disposal rules, since improper handling makes quick work of eyes, lungs, and skin.
Future Prospects
Looking down the road, the outlook for 2-Butenal will rest on better balancing utility and risk. The molecule capitalizes on its double-bonded functional group, carving out niches in organic synthesis, polymer development, and flavors. Researchers keep hunting for more sustainable preparation routes using bio-based acetaldehyde or milder catalysts to reduce hazardous waste. Safer operational protocols and smarter monitoring systems will shrink health risks. Chemists keep imagining next-generation uses, such as specialty active pharmaceutical ingredients or advanced coatings, while environmental scientists track every molecule in air and waste streams. With tighter regulations and greener practices gaining ground, those working with crotonaldehyde accept that new technologies and vigilance must go hand-in-hand for its continued role in chemistry and industry.
What is 2-Butenal and what are its common uses?
What Is 2-Butenal?
2-Butenal, known in labs as crotonaldehyde, comes across as a clear or yellowish liquid with a strong, pungent odor—something you won’t easily forget if you ever catch a whiff in a chemistry lab. Chemists put it into the “unsaturated aldehyde” category because of its specific structure, giving it traits that make it quite reactive. With a boiling point close to the temperature of boiling water, this stuff can vaporize easily, so folks who work with it take real precautions.
Where 2-Butenal Shows Up
Factories and labs don’t pick 2-butenal at random. Companies rely on it as a middleman on the road to making bigger and more important chemicals. Most of the world’s supply helps create n-butanol and butyric acid, both of which pop up in paints, plastics, flavors, fragrances, and even some pharmaceuticals. That sharp flavor found in artificial butter? 2-butenal plays a role there.
Manufacturers also blend it into special resins that end up in the coatings of metal cans or as part of ink formulas. Not a household item, but we all use things made in part from this chemical, whether we realize it or not. For instance, it helps prepare rubber accelerators, which get mixed into the tires on cars and bikes. Tires last longer thanks to help from chemicals like this one.
Why Chemists Value 2-Butenal
The reactive nature of 2-butenal can’t be overstated. Chemists use it for making more complex compounds in the lab. It acts as a building block, letting other groups latch on, which leads to new flavors, scents, or drugs. Sometimes it helps make vitamins like B1 (thiamine) and even dyes that wind up in common products.
This molecule even shows up in research focused on fighting bacteria or developing new pesticides. Its double bond and the aldehyde group make it suitable for these tricky reactions. I remember a project years ago where a small bottle of crotonaldehyde, tightly sealed under the hood, ended up being the crucial step in creating a flavoring that mimicked toasted bread. A stubborn smell, but a key to unlocking a new product.
Concerns and Safety Measures
With benefits come risks. 2-Butenal can irritate the skin, eyes, and lungs. Factories where it gets used keep strong ventilation and personal protective gear as the rule, not the exception. The U.S. National Institute for Occupational Safety and Health (NIOSH) recommends exposure limits because breathing in the vapors leaves real risks for workers.
Communities living near chemical plants sometimes worry about leaks or accidents. Strict rules exist for storage and handling, mostly to prevent fires or health problems. It doesn’t travel far outside a controlled setting, so regular folks don’t run into it much, but the industries using it take its hazards seriously.
New Ways to Handle Challenges
The push now aims at safer production methods. Companies look for ways to capture any 2-butenal released, neutralize it quickly, or recycle it into other, less dangerous chemicals. Cleaner processing cuts down on waste and keeps plant workers safer. The chemical industry, including regulators, keeps updating protective measures as more data comes in from ongoing research.
People outside chemistry may never see 2-butenal on a label, yet it plays a quiet and crucial role in making paints, flavors, plastics, and more. The more we pay attention to the safety, the better for everyone who ends up depending on these vital chemical links in daily life.
What are the safety precautions for handling 2-Butenal?
Understanding the Chemical
2-Butenal, also known as crotonaldehyde, pops up in industrial labs, chemical manufacturing, and sometimes in research settings. This clear, pungent liquid finds its use in making other chemicals and as a solvent. Even the most experienced lab hands need to give it respect, because 2-Butenal comes with some proven health risks. The Centers for Disease Control (CDC) and the Occupational Safety and Health Administration (OSHA) both list it as hazardous. Inhaling the fumes, touching the liquid, or simply spending time in a poorly ventilated workspace can set off headaches, eye irritation, and even breathing problems. The substance can catch fire easily, so safety measures aren’t optional—they’re the basic price of entry.
Personal Protective Equipment Is Non-Negotiable
Let’s get real: regular clothes and thin gloves won’t cut it. I’ve seen folks burned by chemical carelessness, and no software can run a lab when someone’s out with an injury. 2-Butenal will burn skin, so sturdy chemical-resistant gloves like nitrile or butyl rubber make all the difference. Eye protection calls for safety goggles—regular glasses never make a good backup. A lab coat and face shield add another protective layer, especially during transfer or mixing. For some, the fumes alone will trigger coughing and watery eyes, so a suitable respirator (approved by NIOSH) protects your lungs in areas where good ventilation doesn’t reach.
Ventilation Sucks the Danger Out of the Room
Small spaces amplify the risk. I once watched colleagues move a drum of 2-Butenal in a cramped storeroom, only to find the smell everywhere minutes later. Fume hoods aren’t just laboratory window dressing; they pull vapors away from your face and the workspace. Make it a rule: never open or transfer this compound without a working fume hood or an exhaust system. To guard against hidden vapor leaks, keep an air monitor or sensor running in areas where large containers are used regularly, and test it often so no one gets blindsided by a malfunction.
Careful Storage Prevents Disasters
Common sense rules the day here. Store 2-Butenal in tightly closed containers, preferably glass or metal bottles rated for corrosive or flammable liquids. Any shelf holding these containers should sit in a cool, shaded part of the lab, with flammable storage cabinets adding an extra buffer. Mixing up incompatible chemicals in one storage space risks fires and toxic gas releases. Read the MSDS—don’t assume two clear liquids can sit together safely. Chemical spill kits, absorbent pads, and fire extinguishers need to be somewhere you can grab them, not boxed up in a distant storeroom.
Training and Emergency Preparation Save Lives
Even the safest setup falls apart if nobody knows what to do in a crisis. New team members should get hands-on training about 2-Butenal hazards, not just read printed guidelines. Walk through spill cleanup together. Run a fire drill with the chemicals in the spot they actually get used. A posted emergency phone number, first aid gear like eyewash stations, and up-to-date safety data keep panic from turning small mistakes into large tragedies. Don’t forget to report near-misses—a close call today teaches a crucial lesson for tomorrow.
Environmental Caution Matters
Pouring 2-Butenal down the drain or tossing used gloves in regular garbage doesn’t just break lab policy—these shortcuts hit local water sources and soil. Place all waste in properly labeled hazmat containers, and use professional disposal services trusted by your facility or lab group. Every step protects not only your workspace but the greater community outside those walls.
Staying alert, double-checking safety, and taking real precautions keep everyone healthy and productive. Good chemical handling isn’t an option; it’s the foundation for any serious lab or production floor.What is the chemical structure and molecular formula of 2-Butenal?
Getting to Know 2-Butenal
If you’ve ever smelled aging butter or certain fruits, you’ve probably come across a molecule like 2-Butenal—also called crotonaldehyde. The chemistry here isn’t about memorizing formulas just for a textbook answer; it’s worth understanding because these small molecules pop up in industrial practices, food science, and inside living systems.
Structure and Molecular Formula
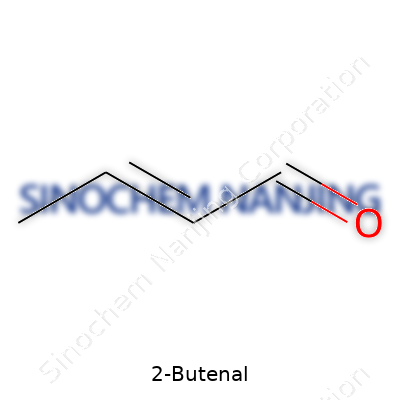
2-Butenal’s molecular formula is C4H6O. In plain talk, the compound holds four carbon atoms, six hydrogens, and a single oxygen. It’s an unsaturated aldehyde, which means there’s a double bond in the carbon backbone. The chemical structure looks like this:
CH3–CH=CH–CHO
There’s a straight chain of four carbons, with a double bond linking the second and third carbons. At the end, there’s an aldehyde group (–CHO), which gives the molecule its reactivity and scent. The positioning matters: the double bond and the aldehyde are right next to each other, setting up both for fast reactions in real-world environments.
Why 2-Butenal Shows Up All Over
I’ve seen 2-Butenal come up in conversations about flavor chemistry, where it turns up as part of the natural breakdown of fats and oils, especially as food starts to age. Some folks may think of chemical names as distant or academic, but the stuff you smell in stale bread or heated dairy owes a lot to little molecules like this one. Toxicologists care about it, too—high concentrations in the air mean trouble for the lungs and eyes, so it isn’t just about food or fragrance.
Industrially speaking, 2-Butenal is part of the process for making things like sorbic acid, which keeps food shelf-stable. The reactivity of that double-bonded aldehyde matters for building bigger molecules. In the paint and polymer world, this kind of structure is a building block. Factories don’t always talk about these background chemicals, but they’re workhorses in synthetic routes.
The Importance of Responsible Handling
With compounds like 2-Butenal, reading the label is only the start. Studies from the International Agency for Research on Cancer show that crotonaldehyde’s reactivity means it can act as a mutagen—though you’d need significant exposure in uncontrolled workspaces to see real risk. OSHA has set permissible exposure limits, so wearing protective gear and ensuring good ventilation are essential steps. If you’re working in a lab or a facility where you handle raw chemicals, you get an upfront sense of respect for these tiny, potent molecules.
Looking Ahead: Practices and Alternatives
There’s no way to avoid chemicals like 2-Butenal in certain industries. Instead of searching for total elimination, companies invest in better containment methods, air monitoring, and developing alternatives where possible. Synthetic biology teams come up with enzyme-driven ways to make similar molecules with fewer side reactions, which reduces byproducts and, hopefully, occupational risk.
Understanding the backbone of 2-Butenal—structure, formula, and behavior—brings home just how many everyday aromas, food preservatives, and industrial processes rest on tiny changes at the molecular level. Chemistry here connects the science classroom with the workplace, the kitchen, and the manufacturing floor.
How should 2-Butenal be stored and transported?
Why Paying Attention to 2-Butenal Makes Sense
2-Butenal shows up often in chemical production, food technology, and research. Step into a facility handling this compound and you’ll notice the sharp, penetrating smell right away. Fact is, it’s not just a noticeable substance—it’s highly flammable and volatile. In ambient air, vapors can spark fires without much warning. On top of that, direct exposure can irritate the skin, eyes, throat, and lungs. So, treating 2-Butenal with care isn’t just Red Tape. It’s about keeping everyone out of harm’s way and preserving property.
How Proper Storage Stops Problems Before They Start
A worker handling a drum of 2-Butenal probably isn’t thinking about storage specs—until something goes wrong. Leaks and fumes build up fast, and even a small spill can force an evacuation. Practical experience, not just guidelines, makes it clear: use tight-sealing, corrosion-resistant containers. Stainless steel or aluminum drums work well. Forget plastics or rusty legacy tanks; these break down under chemical stress. Store them in a cool, shaded, well-ventilated area, away from sunlight, open flames, or anything sparking. The reason is simple—the lower the temperature and the less light, the less 2-Butenal wants to vaporize and fill the room with flammable gas.
Chemical compatibility makes a huge difference. I once saw a batch react with an old cleaning solvent, turning a routine cleanup into a small scare. Don’t stow 2-Butenal near oxidizers, acids, or bases. Segregation in a chemical storage room is more than just a rule; it’s a clear break between a smooth shift and a dangerous one.
Transport Rules Go Beyond Paperwork
Anyone who's spent time on a loading dock knows that moving barrels of hazardous chemicals calls for sharp focus. DOT and international rules label 2-Butenal as a flammable liquid (Hazard Class 3) for good reason. A single missed seal or a cracked container in transit spells trouble for everyone on the road. It always helps to double-check for leaks, dents, or corrosion before sending anything out. Using approved metal drums with pressure-relief mechanisms beats trying to rig up outdated containers. Labels—clear, weatherproof, chemical-resistant—keep emergency crews and drivers in the know in case of accident.
Temperature control matters just as much on the road as it does in storage. Heated trucks have led to pressure buildup in containers, with some tragic consequences. The best outfits set up shipments to avoid extreme weather and use secondary containment pallets to catch drips and spills.
Building a Culture of Responsibility
Plenty of accidents start with small shortcuts. Training everyone—not just “the safety folks”—builds awareness and buy-in. Workers who understand what 2-Butenal can do recognize warning signs sooner and treat incidents with the urgency they deserve. Regular safety drills, quick access to neutralizers and absorbent pads, and sharing real stories of near misses all chip in to raise standards.
Digital inventory tracking also keeps small storage issues from growing. It’s easy to lose track of what sits in the back corner without a good log. A simple barcode scan system flags old stock before it degrades or creates risk.
Better Rules Lead to Fewer Regrets
A few big lessons stand out: use the right containers, keep chemicals apart, keep everything cool and ventilated, and don’t fudge paperwork or training. Sticking to these basics not only dodges regulatory fines but keeps people safe. A well-run operation stands out less for its emergencies and more for its record of nothing happening at all.
What are the health effects and toxicity concerns associated with 2-Butenal exposure?
Understanding What 2-Butenal Brings to the Table
2-Butenal, better known by some as crotonaldehyde, doesn’t make headlines like asbestos or lead, but anyone working in industries where it pops up—plastics, leather, or the making of certain chemicals—has probably caught a whiff of its sharp, irritating odor. I’ve walked through factories where the chemical smell clings to your skin long after you’ve left, and you just know you’re not breathing fresh air. That sort of experience sticks with you and triggers questions about long-term safety.
Short-Term Exposure: What Folks Experience
Just spending a short time around 2-Butenal can lead to nose and throat irritation. Eyes burn and water. Some start coughing or feel tightness in the chest. Breathing high levels even for a little while can bring on headaches, nausea, and a bad taste in the mouth. The body sends warning signals when exposure hits, and ignoring them has never done anyone a favor. Workers on the factory floor, especially those handling containers or cleaning spills, feel the direct hit. Even at low concentrations, folks with asthma or certain sensitivities complain about worsening symptoms.
Long-Term and Repeated Exposure: Where Risks Rise
Repeated exposure to 2-Butenal throws up the stakes. Some studies found that the chemical can inflame airways, harm liver and kidney health, and possibly play a role in nerve disorders. Chronic exposure sometimes results in damage that sticks around even when people step away from their work environment. The International Agency for Research on Cancer (IARC) categorized crotonaldehyde as "possibly carcinogenic," meaning enough questions hang in the air to demand caution.
Real-world evidence comes from workers reporting declining lung function and breathing trouble after months or years around this chemical. Animal research shows clear evidence for organ toxicity and increased cancer rates in those dosed regularly, which lines up with what medical surveillance sometimes uncovers in human cases. With that in mind, preventing repeat contact looks a lot like common sense.
How 2-Butenal Enters the Body and What the Data Says
Most exposure happens through breathing foul air, but skin absorption isn’t off the table. Spills or splashes find their way onto the skin, causing burns or rashes. In busy workspaces, splashing can go unnoticed during rushes, so accidents happen. The U.S. Environmental Protection Agency and OSHA both monitor workplace air for 2-Butenal and set exposure limits because research points to harm at higher concentrations.
Reducing Risks: Focusing on Practical Solutions
Ventilation, personal protective equipment, and close tracking of air quality matter—not just for compliance, but for peace of mind. I’ve seen the difference when a shop brings in local exhaust systems: headaches and complaints drop, and folks stop worrying so much about bringing problems home. Basic training about spill management lowers the odds of skin contact or accidental splashing.
Medical monitoring also plays a role. Periodic health checks catch early warning signs, and that gives workers options before things get serious. Substituting less toxic chemicals isn’t available in every case, but exploring alternatives sometimes shields people from unnecessary health risks.
Final Thoughts on Staying Safe
Paying attention to chemicals like 2-Butenal means valuing the well-being of folks on the job and in neighboring communities. Common sense solutions—ventilation, protective gear, training—work when backed up by strong company culture and ongoing vigilance. What matters most is putting long-term health above quick fixes or shortcuts.
| Names | |
| Preferred IUPAC name | but-2-enal |
| Other names |
crotonaldehyde crotonal β-methylacrolein |
| Pronunciation | /tuː.bjuːˈtiː.næl/ |
| Identifiers | |
| CAS Number | 4170-30-3 |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:78422 |
| ChEMBL | CHEMBL169784 |
| ChemSpider | 12051 |
| DrugBank | DB04211 |
| ECHA InfoCard | 100.003.414 |
| EC Number | 200-612-6 |
| Gmelin Reference | 775 |
| KEGG | C00447 |
| MeSH | D001070 |
| PubChem CID | 7880 |
| RTECS number | EL8575000 |
| UNII | 8D58795H6H |
| UN number | UN1165 |
| Properties | |
| Chemical formula | C4H6O |
| Molar mass | 70.09 g/mol |
| Appearance | Colorless to yellow liquid. |
| Odor | penetrating; pungent |
| Density | 0.853 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.38 |
| Vapor pressure | 3.29 kPa (at 25 °C) |
| Acidity (pKa) | 13.5 |
| Basicity (pKb) | 13.23 |
| Magnetic susceptibility (χ) | -32.7e-6 cm^3/mol |
| Refractive index (nD) | 1.4344 |
| Viscosity | 1.544 mPa·s (at 20 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −130.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1268.4 kJ/mol |
| Pharmacology | |
| ATC code | A16AX15 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H301, H311, H315, H317, H319, H331, H335, H341, H351 |
| Precautionary statements | P210, P264, P280, P301+P312, P305+P351+P338, P330, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 30 °C (86 °F; 303 K) - closed cup |
| Autoignition temperature | 179 °C |
| Explosive limits | Upper: 15%, Lower: 2.1% |
| Lethal dose or concentration | LD50 oral rat 55 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 2-Butenal: 55 mg/kg (Rat, oral) |
| NIOSH | AV9275000 |
| PEL (Permissible) | PEL = 2 ppm (parts per million) |
| REL (Recommended) | 0.02 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Crotyl alcohol Butyric acid Butyraldehyde |

